Abstract
Alzheimer’s disease (AD) is the most common cause of dementia. Epileptic seizures or epileptic activity have been detected in AD, and people with epilepsy have a higher risk of dementia compared to the general population. This suggests that seizures or epileptic activity might often coexist with AD. It is increasingly evident that epileptic activity or seizures are common but often overlooked comorbidities of AD. However, the causal relationship between seizures, epileptic activity and cognitive decline remains uncertain. Experimental data show that amyloid-β (Aβ) and Tau protein can cause neuronal hyperexcitability and has epileptogenic effects. Neural network hyperexcitation regulates the ratio of Aβ isoforms and is linked to the initiation of AD, indicating a shared mechanism. Clinical studies suggest that cognitive impairment accelerates in AD patients with seizures or epileptic activity. This review discusses the relationship between epileptic seizures and AD, the impact of epileptic activity in AD, and potential treatments.
Introduction
The global prevalence of Alzheimer’s disease (AD) is estimated at 32 million people, including 6.7 million Americans over 65 years of age living with AD.1,2 Between 1990 and 2019, the incidence and prevalence of AD and other dementias increased by approximately 150% and 160%, respectively, leading to a significant rise in the global burden measured in death and disability-adjusted life years (DALYs). 3 Despite the approval of immunotherapy therapy such as Lecanemab (Lequembi) by the FDA, 4 which aims to clear beta-amyloid protein in the brain, the treatment’s effectiveness requires further observation and studies to confirm its long-term benefits. The high cost of this therapy, estimated at $26,500 annually per patient, 5 raises concerns as well. There is still a need for effective treatments to reduce the disease’s global burden. Current strategies focus on minimizing modifiable risk factors such as mid-life obesity, hypertension, diabetes, hyperlipidemia, smoking, depression, and sleep disturbances.6,7
An under-recognized risk factor in AD is epileptic activity. Thirty-nine studies conducted in multi-nations have shown that the prevalence of seizures among patients clinically diagnosed AD ranged from 1.5% to 12.7%. 8 In addition, patients with AD have a higher incidence of seizure than controls, and some behavioral changes in AD may be associated with epileptiform activities observed in electroencephalography (EEG). 9 In a study of 1617 dementia patients undergoing routine EEG recordings, epileptiform discharge were noted in 42 (3%) subjects, 60% of whom did not exhibit any clinical seizures. 10 Moreover, the group of AD with epileptiform activity was younger than those without such activity, which is consistent with previous reports indicating an increased risk of epilepsy with early onset of cognitive decline. 11
Studies using 24-hour continuous EEG recordings found that 22% to 53% of patients with AD had epileptiform discharges without clinical seizure, which is significantly higher than in healthy subjects. These discharges occur without clinically overt epileptic seizures, but may lead to transient cognitive impairment.12-14 It is becoming increasingly evident that these epileptic activity are common but often overlooked comorbidities of AD.15-17 Notably, the sensitivity of detecting epileptiform discharges increases with magnetoencephalography, indicating that some discharges might originate from deeper cortical layers. 12 Other specialized tools, such as foramen ovale electrodes, have been used to detect spikes or sharp waves in the deep hippocampus or mesial temporal lobe.18,19
An experimental mouse model of AD exhibited an increase in the frequency of spontaneous Ca2+ transients near amyloid beta plaques. The aberrant hyperactive neurons appeared to be due to insufficient synaptic inhibition.11,19,20 This model provides a basic mechanism for the disturbed cortical function observed in AD. 21 It is clear that patients with AD or mild cognitive impairment have a higher risk of epileptic seizures, which subsequently worsen cognitive functions and disease progression.12,17 This review will highlight the significance of epileptic activity, the correlation between epileptic activity and seizures, and their impact on cognitive changes in patients with AD.
Epileptiform Discharge: Definition and Clinical Aspect
Regarding the initial definition of epileptiform discharges, the International Federation of Societies for Electroencephalography and Clinical Neurophysiology emphasized that they are distinct waves or complexes distinguished from background activity, and resemble those recorded in a proportion of human subjects with epileptic disorder. 22 The International Federation of Clinical Neurophysiology (IFCN) revised the glossary of terms in EEG for reporting EEG findings in 2017. 23
The operational definition of epileptiform patterns (synonyms: interictal epileptiform discharge (IED), epileptiform activity) must fulfill at least 4 of the following 6 criteria: (1) Biphasic or tri-phasic waves with sharp or spiky morphology. (2) Wave-duration that differs from the ongoing background activity. (3) Asymmetry waveform, with either a sharply rising phase and a more slowly descending phase, or vice versa. (4) A sharp transient followed by a slow after-wave. (5) Background activity disrupted by the presence of the epileptiform discharges. (6) Distribution of the negative and positive potentials suggesting a source of the signal in the brain, corresponding to a radial, oblique or tangential orientation of the source, best assessed by inspecting voltage maps constructed using common-average reference (Table 1). Epileptiform Patterns: Criteria and Descriptions. IFCN: International Federation of Clinical Neurophysiology.

The revised definition of epileptiform discharge has significantly improved the diagnostic performance and inter-rater agreement in identifying IED by the electroencephalographers (Table 1).24-26 Notably, with increasing age, focal IEDs are less sharp, have lower amplitudes, and less prominent slow-waves and they become more lateralized. 27
In practice, temporal intermittent rhythmic delta activity (TIRDA) had a localizing value of epileptogenic zone in mesial temporal lobe and small sharp spikes is considered a benign EEG waveform variant. Both of them have been reported to correlate with mesial temporal lobe epilepsy.28-30 A cross-sectional study using 24-hour ambulatory scalp EEG showed a significantly higher appearance of TIRDA in patients compared to those without epilepsy and healthy controls, with TIRDA having a high positive predictive value for epileptiform activity. 13 Additionally, frequent small sharp spikes are associated with epileptiform discharges and are identified as potential biomarkers of hyperexcitability in AD. These EEG features are important in the care of dementia, guiding the identification of AD patients at high risk for clinical seizures.
Mechanism of Epileptiform Discharge in AD
Dysfunction of synaptic homeostasis and network hyperexcitability is related to the impairment of balance between neuronal excitation/inhibition (E/I).31,32 Impaired synaptic long-term plasticity, which modifies both behavior and neural function, disrupts the ability to adjust the E/I balance.33,34 Animal models of AD show a higher rate of epileptic seizures and IEDs.21,35 In these models, the deposition of amyloid-β and tau proteins enhances network excitability, and this hyperexcitability can further promote the deposition of these abnormal proteins.36,37 An abnormal increase in excitatory neuronal activity and compensatory remodeling of the inhibitory hippocampal circuit have been detected.
19
These models have also shown increased neuronal network excitability, epileptiform activity in EEG, nonconvulsive seizures, and a higher proportion of abnormally hyperactive neurons in cortical circuits (Figure 1).
20
Dysfunction of synaptic homeostasis and network hyperexcitability in Alzheimer’s disease (AD) is linked to an impaired balance between neuronal excitation and inhibition. Impaired synaptic long-term plasticity disrupts this balance, contributing to increased rates of epileptic seizures and interictal epileptiform discharges in AD animal models. The deposition of amyloid-β and tau proteins enhances network excitability, which in turn promotes further protein deposition. This cycle is associated with increased neuronal network excitability, epileptiform activity, and nonconvulsive seizures. Hyperexcitability also promotes amyloid precursor protein (APP) deposition and spike bursts, influencing the ratio of Aβ isoforms and potentially contributing to sporadic AD. Unprovoked seizures and epileptic activity can occur at early stages of AD, suggesting a role in its pathogenesis. Brain hyperexcitability may promote AD neuropathology independently of Aβ but related to tau protein. Reducing tau has shown benefits in cognitive function and seizure prevention in AD models. Overall, there is a complex interplay between AD pathology and brain hyperexcitability, with each influencing the progression of the other.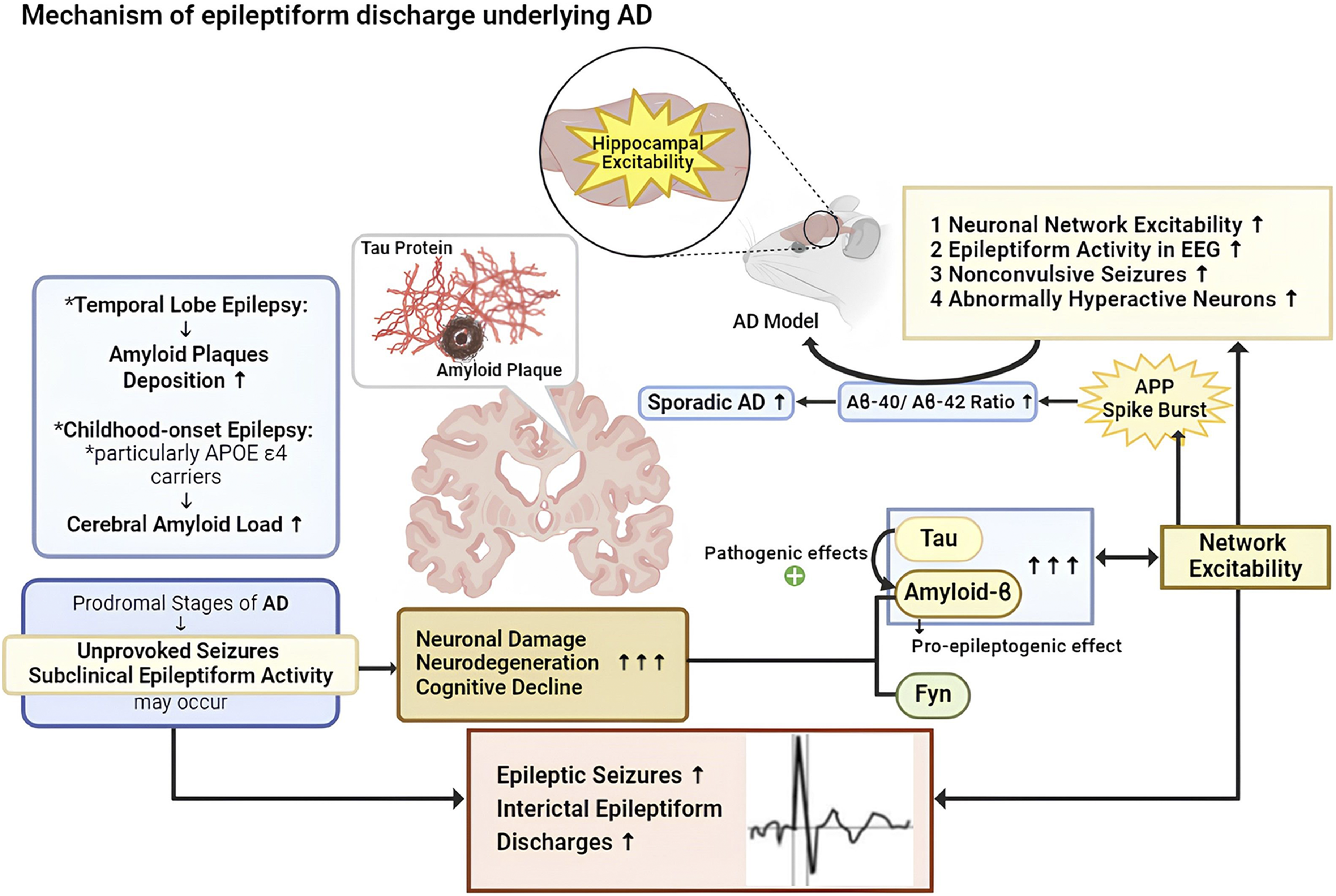
In temporal lobe epilepsy, amyloid plaques deposition is significantly higher.38,39 The hippocampus, a main target of AD, frequently showing increased excitability, 40 indicating a common neural mechanism underlying both diseases. Hyperexcitation in neural networks promotes amyloid precursor protein (APP) deposition and spike burst formation in rodents. These spike bursts facilitate the conformational change of presenilin 1, the catalytic subunit of γ-secretase, subsequently increasing the Aβ-40/Aβ-42 ratio.41,42 Thus, spike bursts related to neural network hyperexcitation regulate the ratio of Aβ isoforms and contribute to the initiation of sporadic AD (Figure 1).
Unprovoked seizures and epileptic activity can occur at the prodromal stages of AD when cognitive functions and brain imaging appear normal or show subtle changes, suggesting that network hyperexcitability plays a role in the early pathogenesis of AD. 43
Brain hyperexcitability may promote AD neuropathology independent of the Aβ metabolic pathway but related to microtubule-associated tau protein. Mounting evidence suggests that tau protein mediates or enhances the pathogenic effects of Aβ.44-47 Tau reduction alleviates cognitive decline induced by the synergistic effects of Aβ and the nonreceptor tyrosine kinase Fyn. Tau reduction also prevents spontaneous epileptiform activity and reduces the severity of both spontaneous and chemically induced seizures in human APP transgenic mice. These results suggest that Aβ, tau, and Fyn together influence synaptic, network, and cognitive functions in both Aβ and Fyn overexpressing mice. 48 Epileptic seizures may foster neuronal damage and neurodegeneration, accelerating cognitive decline.49,50 Experimental data revealed that Aβ has a pro-epileptogenic effect early at the oligomeric soluble state, long before plaque deposition can be observed. 51 A Finnish population-based case-control study, which prospectively followed adults with childhood-onset epilepsy for over 5 decades and compared them with matched controls, showed that individuals with childhood-onset epilepsy, particularly APOE ε4 carriers, had an increased cerebral amyloid load as measured by positron emission tomography. 52 In summary, there are bidirectional link and complex interplay between AD pathology and brain hyperexcitability (Figure 1).
Implication of Epileptic Activity in AD
Increased excitability associated with hypersynchronous neural networks is essential for epileptiform discharge. The greatest synchronization among neuron populations occurs during slow-wave sleep (SWS). 53 Plaques and tangles are associated with disruptions in the sleep-wake cycle and in the SWS phase of non-rapid eye movement (NREM) sleep. The proportion of NREM sleep and slow-wave activity is reduced in AD patients. Since SWS plays a critical role in memory consolidation, reduced slow-wave activity is associated with memory impairment in AD patients.54-56 Improving slow-wave sleep may thus be a promising therapeutic target for AD. 57 Epileptiform discharges are maximally activated during SWS, 58 but decrease during rapid eye movement (REM) sleep. 59 Most epileptic activity is detected during NREM sleep, particularly deeper SWS.12-14 During NREM sleep, the continuous alternation between neuronal depolarization and hyperpolarization creates cellular instability that enables the epileptogenic cortical substrate to produce IEDs. 60 It is likely that both pathological and neurophysiological abnormalities—specifically, amyloid-β and tau deposition along with increased epileptic activity during SWS—impact memory performance.
Soula M et al investigated how IEDs affect memory in an animal model of AD. 61 They implanted multilayer probes in the hippocampus of AD transgenic mice, which exhibited local field potential features (theta oscillations and sharp-wave ripples) and firing patterns of pyramidal cells similar to those in humans. Under physiological conditions, the discharge of CA3 pyramidal cells yields local field potential sharp waves in CA1 neurons, leading to brief and fast oscillations known as ripples. These sharp-wave ripples (SPW-Rs) control learning neuron discharge patterns, either aborting or prolonging them to reduce or enhance memory, respectively. IEDs suppressed and altered the physiological properties of SPW-Rs, disturbing hippocampal–cortical communication and interfering with memory.
Ictal activity can be subclinical or go unnoticed. It is well established that seizure activity, whether clinical or subclinical, can cause multifaceted cognitive impairment.62,63 In contrast, IEDs have a chronic negative impact on cognitive functions.12-14,64-66 Balanced excitatory and inhibitory synaptic currents facilitate coding and energy efficiency, promoting information processing in cortical neurons, which is crucial for maintaining cognition. 33 The brain responds to seizures or frequent epileptiform discharges by remodeling neural circuits, such as axonal mossy fiber sprouting.67,68 Local inhibitory neurons’ axonal sprouting restricts the spread of epileptic activity to distant areas. 69 Meanwhile, interictal epileptiform activity spreads within the same pathological network, but the underlying aberrant activity does not reach the seizure threshold. 70 This theory explains the clinical observations of no clinical seizures or subtle transient cognitive impairments, such as brief deficits in attention, memory, and speech function. 71
Numerous clinical studies highlight the deterioration of cognitive functions in AD patients with epileptic activity. Vossel KA et al compared AD patients with and without epileptic activity, finding no initial difference in cognition. However, patients with epileptic activity showed faster declines in global cognition (3.9 points/year), assessed by the Mini-Mental State Examination (MMSE), and in executive function after an average of 3.3 years of follow-up. 12 Horvath AA et al investigated 38 AD patients over a 3-year prospective follow-up in cognitive evaluation. Epileptiform discharges were associated with lower memory performance scores. 14 AD patients with spikes experienced a 1.5-times accelerated decline in global cognitive scores compared to those without spikes, with spike frequency showing a significant positive correlation with cognitive performance decline.
Unfortunately, these studies did not compare biomarker changes between epileptic activity and control groups, such as blood and CSF amyloid, phosphorylated tau levels, brain MRI, and PET scan findings. Epileptic activity in AD is associated with neuronal synchrony abnormalities that attenuate the alpha band and increase theta-delta band coherence, corresponding to an accelerated decline in MMSE scores. 72 A Taiwanese study enrolled 288 AD patients in 1 hospital, finding that 20% had epileptic activity detected by conventional EEG. Patients with epileptic activity showed significantly greater declines in Cognitive Abilities Screening Instrument (CASI) and MMSE scores over 1 year. 73 The same research team also revealed worsening behavioral and psychological symptoms in patients with epileptic activity. There was no significant difference in the prevalence of behavioral and psychological symptoms of dementia between patients with and without epileptic activity. However, the Neuropsychiatry Inventory score for the irritability subsyndrome was significantly higher in patients with epileptic activity. 74
Role of Antiseizure Medication Therapy
Although we discuss epileptic activity, it is uncertain if these abnormal discharges are truly “silent” or “subclinical.” Some subtle focal seizure symptoms, such as very brief loss of attention, consciousness, or movement standstill, may be overlooked by caregivers. Monitoring whether some of the epileptic activities evolve into electrographic and clinical seizures is challenging in clinical practice. Selvitelli et al. found the longer the duration of epilepsy, the higher incidence the incidence of IEDs. However, they did not find an association between IED frequency and the number of days since the last seizure or seizure frequency. 75 Whether IED density can serve as a potential surrogate marker for seizure frequency remains a topic of debate. In genetic generalized epilepsy, there is a strong correlation between IEDs and seizures, whereas this relationship is more controversial in focal epilepsy.76-78
Antiseizure medications (ASM)s may have effects on suppressing IEDs. Laboratory researches demonstrated that carbamazepine, topiramate, and valproic acid could suppress prolonged (>3 seconds), ictal-like epileptiform activities induced by spreading depolarization (SD) and generate paroxysmal depolarization shifts (PDS) in rat brain slices, but they did not affect shorter (<3 seconds) discharges.79,80 At the cellular level, carbamazepine, phenytoin, valproic acid, lamotrigine, and zonisamide were found to reduce the frequency of PDSs and firing bursts. Tiagabine showed a moderate effect in suppressing PDSs following SD. Ethosuximide, felbamate, vigabatrin, and second generation ASMs such as gabapentin and levetiracetam (LEV) did not significantly affect SD-induced epileptiform discharges in our previous study. 81 Lamotrigine (LTG) and topiramate have been shown to reduce IEDs, and valproic acid also demonstrated a similar trend, although conflicting results have been reported across studies.
Clinical observations have shown that LEV, oxcarbazepine, and vigabatrin can reduce IEDs in children. However, in adults, neither conventional ASMs like phenytoin and carbamazepine nor newer ASMs such as gabapentin and vigabatrin have demonstrated a consistent effect on IEDs over time. 82 A systematic review examining the effects of ASMs on epileptiform discharges (EDs) in genetic generalized epilepsy (GGE) indicated that some ASMs tend to reduce ED load, frequency, cumulative duration, and burst duration in GGE. This ASM-mediated reduction in ED burden was associated with improved seizure control and cognitive function. 83 However, the statistical significance of these associations remains unclear due to limited data, and the effects on generalized EDs may not be directly applicable to epileptic activity in patients with AD.
The potential impact of ASMs on IEDs in animal models or patients with epilepsy warrants further investigation to determine whether similar effects could be observed on epileptic activity in AD. Nonetheless, ASMs that target abnormal network hyperexcitability are considered a rational and promising approach to potentially mitigate mental deterioration in AD patients.
Among the ASMs, in terms of treating epileptic seizures, LEV and LTG have shown excellent efficacy in patients with epilepsy, achieving 60%–70% seizure reduction within 1 year. Both medications are well-tolerated and do not adversely affect cognitive function.84-86 Notably, LEV caused fewer adverse events than other ASMs and was associated with improved cognitive performance, specifically attention and oral fluency, while LTG improved mood as compared with carbamazepine and gabapentin.87,88 Three randomized control trials on elderly patients with new-onset seizures treated with gabapentin, LTG, or LEV compared to patients taking carbamazepine found no difference in seizure control rates among the treatment groups. Consequently, these ASMs can be used as initial treatment for newly diagnosed epilepsy in geriatric patients.89-91
A retrospective, uncontrolled trial analyzed the effectiveness and tolerability of 10 ASMs in 417 patients with epilepsy over 55 years old. LTG and LEV exhibited the highest tolerability with a 1-year retention rate over 70%. LTG displayed the highest 1-year seizure freedom rate (54.1%), followed by LEV (46.2%) and GBP (18.5%). Notably, LTG and LEV did not negatively impact cognitive function, supporting their use in the elderly. 90 A 2021 Cochrane systematic review, including 1 randomized control trial of medication treatment in 95 AD patients, offered no evident support for LEV, phenobarbital, or LTG in managing seizures in AD patients. For efficacy and tolerability, no significant differences were reported among these ASMs. 88
In terms of intervention of epileptic activity in clinical studies on patients with AD, in a randomized, double-blind, placebo-controlled trial using LEV 125 mg twice a day for 4 weeks to assess cognitive performance in AD patients with and without epileptic activity, LEV was found to be well tolerated and significantly improve performance on the Stroop naming task and spatial memory task via virtual route learning in AD patients with epileptic activity. LEV also non-significantly improved global cognitive performance in the epileptic activity. Positive group as assessed by ADAS-Cog and NIH EXAMINER scores. 89 According to current guidelines, the management of seizures in AD does not differ from that in patients with epilepsy.91,92 We reported a case in which epileptic activity. Appeared in the temporal areas, contributing to cognitive decline. Alongside epileptic activity, non-convulsive seizures were also clinically suspected. Treatment with ASM (oxcarbazepine and perampanel) improved the patient’s epileptic activity, nonconvulsive seizures and cognitive performance. 93
In addition to ASM trials to manage brain hyperexcitability-related cognitive deterioration, improving deep sleep is a feasible approach. Trazodone, a potent serotonin 5-HT2A antagonist, weak serotonin reuptake inhibitor, and antagonist of other monoaminergic receptors, including 5-HT2B, 5-HT2C, adrenergic (α1A and α2C) receptors and a partially at 5HT1A receptors, showed promise. 94 A retrospective study investigating 25 trazodone users and 25 propensity-matched non-users found that non-users had a 2.6-fold faster decline in MMSE compared to trazodone users. 95 Post-hoc analysis also showed subjective improvement in sleep complaints among trazodone users. This benefit may be due to an increase in slow-wave sleep. These studies suggest the need for more randomized control trials with larger participant groups and longer follow-up periods to obtain better evidence of ASM efficacy in treating AD patients and delaying cognitive decline.
Neuromodulation
Neuromodulation involves alternative treatment approaches for drug-resistant epilepsy, and is increasingly recognized for its potential to enhance cognition in dementia, including AD. Vagus nerve stimulation (VNS) is a relatively minimally invasive procedure that uses stimulation of the left vagus nerve to send electrical impulses to the brain, aiming to reduce seizure onset. 96 Studies have shown that VNS can positively affect basal synaptic function, enhance synaptic plasticity, and potentially modulate cognition. 97 Deep brain stimulation (DBS) targets key propagation points within the epileptogenic network to prevent the spread of seizure activity. 98 For instance, thalamic DBS works by promoting the release of glutamate and adenosine, which reduces thalamic oscillations,99,100 and may further help to desynchronize epileptogenic neural networks. 101 Additionally, DBS has shown potential therapeutic effects in AD. 102 Transcranial magnetic stimulation (TMS) has also demonstrated possible cognitive improvement. 103
While neuromodulation offers seizure reduction in drug-resistant epilepsy and potential cognitive benefits, its effects on epileptic activity remain unknown. Seizure activity in patients with AD is often subtle or lacks overt clinical symptoms, highlighting the need for large-scale clinical trials to evaluate the impact of neuromodulation on epileptic activity and cognition in AD.
Concluding remarks
Epileptic seizures are often under-recognized in AD patients, possibly due to their frequent atypical or subtle presentation and overlap with behavioral symptoms. Epileptic activity may cause more significant and permanent cognitive impairment, can be found in up to 50% of AD patients. However, EEG is not routinely or widely used in dementia assessment, making it difficult to detect clinical seizures or epileptic activity. EEG study is important in the routine exploration of patients with AD, especially sleep EEGs.
Hyperexcitability of brain networks may promote amyloid-beta deposition, a fundamental neuropathology of AD. The deposition of amyloid-β and tau proteins also enhances network excitability. These abnormal neurophysiological and neuropathological changes appear to have a bidirectional, mutually influential effect.
ASMs seem a promising option to slow down mental decline in AD patients with epileptic activity. However, no high-evidence-level studies have proven the efficacy of specific ASMs in improving cognitive outcomes to date. Further large-scale randomized control trials are needed to establish guidelines for treating this comorbidity of AD and cognitive problems.
Footnotes
Acknowledgments
The authors thank Yu-Tung Hsiao for the assistance with digital plotting.
Author Contributions
Tsang-Shan Chen: Conceptualized, designed, and coordinated the work, writing-original draft preparation; Ming-Chi Lai: Manuscript review, revision, and reference work; Wei-Ping Hong: Manuscript review and revision; Chin-Wei Huang: coordination work, figure generation, writing-review and editing, manuscript revision. All Authors read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from the National Science and Technology Council (112-2314-B-006 -047 -MY2), Taiwan.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
