Abstract
Recently, some researchers claimed neuropathological changes lead to Alzheimer’s-like brains after severe infection of SARS-CoV-2. Several mechanisms have been postulated on how SARS-CoV-2 neurological damage leads to Alzheimer's disease (AD) development. Neurobiochemical changes during infection may significantly induce Alzheimer's disease in severely COVID-19 infected people. The immune system is also compromised while infected by this novel coronavirus. However, recent studies are insufficient to conclude the relationship between Alzheimer's disease and COVID-19. This review demonstrates the possible pathways of neuropathological changes induced by the SARS-CoV-2 virus in AD patients or leading to AD in COVID-19 patients. Therefore, this study delineates the challenges for COVID-19 infected AD patients and the mechanism of actions of natural compounds and alternative treatments to overcome those. Furthermore, animal studies and a large cohort of COVID-19 survivors who showed neuroinflammation and neurological changes may augment the research to discover the relationship between Alzheimer's disease and COVID-19.
Significance Statement
• Neuropathological impact of SARS-COV-2 infection may lead to Alzheimer’s disease (AD) in long COVID-19 survivors. • SARS-COV-2 infection combat is highly challenging for patients with Alzheimer’s and dementia. • Alternative treatments provide better approaches for AD patients to fight COVID-19 by neurotherapeutics effects. • Selective natural compounds and vitamins are inhibitory to neuronal apoptosis and prevent AD pathophysiology.
Introduction
Alzheimer's disease is a neurodegenerative disease characterized by the deposition of amyloid-β protein plaques and tau neurofibrillary tangles in the brain. It has been proved that the deposit formed by the proteins is the leading cause of Alzheimer's disease. According to genetics, the amyloid-β and tau become dysfunctional during the disease progression.1-3 At the beginning of the disease, the mild memory difficulties turn into difficulties in daily activities, for example, planning, solving problems, difficulties in learning, and diminished mental condition. 4 A patient diagnosed with Alzheimer's disease showed cognitive decline and memory deficits. 5 But Alzheimer's disease pathogenesis is not limited to only neurological changes caused by proteins but also connected to immunological changes in the brain. Alzheimer's people have neuroinflammation in the brain because of an innate immune response characterized by the release of inflammatory mediators. 6 In the severe form of Alzheimer's disease, the patient goes to the terminal stage of their life and eventually dies. Alzheimer's disease is the fifth leading cause of death in the age category of 65 and older in the United States of America. 7
Since the emergence of SARS-Cov-2 in 2019, many people have died due to the severe effect of viral infection. Some people have long COVID-19, suffer from neurological symptoms, and die after recovery. 8 It is also found that the COVID-19 mortality is age-specific. The lowest fatality is shown among 5-9 years old, and the risk increases in the older age groups until 65 years of age. 9 The people sustained from exposure to COVID-19 may have delirium and cognitive impairment; thus, neuropsychological evaluations need to be conducted.10,11 A 6-month observational study has found neurological and psychiatric morbidity among COVID-19 survivors. 11 An immunohistochemical study showed β-amyloid deposits in the brain of a 58-year-old dead patient infected with COVID-19 before death. 12
It is also evident that angiotensin-converting enzyme-2 (ACE-2) binds with the spike protein of the coronavirus and enters the cell.13-15 ACE-2 receptors also exist in the brain's neuronal cells. 16 During severe SARS-COV-2 infection, the Blood-Brain Barrier (BBB) is compromised. Consequently, the virus spike protein can easily cross the BBB and enter the brain. 17
Several studies using postmortem brain samples showed that SARS-CoV-2 was detected in the brains of patients who died after SARS-CoV-2 infection. In one study, 22 autopsy tissue samples were collected who died due to COVID-19 and found that the highest number of SARS-CoV-2 present in the respiratory tract whereas lower numbers were detected in the kidney, liver, heart, blood and brain. 18 SARS-CoV-2 was detected at low levels in the brain samples in another study. 19 However, SARS-CoV-2 virus can be widely distributed in the brain found in the autopsies of patients who died from COVID-19. 20 These findings proved that SARS-CoV-2 can enter into the brain after infection.
A few studies reported the co-relationship between Alzheimer's disease and SARS-Cov-2. 21 In those studies, the number of patients is not enough to conclude. The mechanism by which the virus enters the brain is not yet elucidated. However, their investigation revealed that Alzheimer’s-like neuropathology was found in the brain sample. Further study with a large group of mouse models and people can confirm the implication of COVID-19 in Alzheimer's disease.
Factors for Alzheimer’s Disease Development
Alzheimer's disease causes the blood-brain barrier (BBB) dysfunction of the brain, 22 which is progressive, neurodegenerative 5 and incurable. 23 In this disease, the people suffer from cognitive decline, which results from the deposition of amyloid-beta and tau proteins in the brain region of the patient. In the early stage of the disease, the person's cognitive ability decreases, and they face numerous difficulties in their daily life. Their memory is diminished; they cannot think logically, feel mentally disturbed, and maintain daily chores. 24 With the severity of the disease, they need help from their caregivers to keep a quality life, further increasing the socio-economic burden. 25
Alzheimer's disease is not a normal part of aging. The older age group of 65 or older has a high probability of this disease. However, younger-onset Alzheimer's disease has prevailed. 26 After AD diagnosis, the average life span is between 4-8 years.27-29 It is the leading cause of dementia. The number of Alzheimer's diseases is increasing at an alarming rate, and in 2006, it was estimated that the number will be 3-fold in 2050. There is no cure for AD, and only symptomatic treatment is available. 30 This type of treatment is based on acetylcholinesterase inhibitors, including donepezil, rivastigmine, and galantamine. Another pharmacological drug is memantine; an (N-methyl-D-aspartate) NMDA channel blocker recommended. 31 It is also thought that antioxidants may delay the disease progression. An amyloid-β vaccine is also investigated for modifying the disease.32,33
COVID-19 Impacts on Alzheimer’s disease
Pathogens can induce AD pathology, including (Amyloid-β peptide) Aβ accumulation, phosphorylation of tau protein, neuronal injury, and apoptosis. Moreover, pathogens cause neuroinflammation and neurodegeneration, indications of AD.32-34 In Alzheimer’s patients, the BBB is vulnerable so that the virus can easily enter the brain, making the patient more susceptible to severe viral attacks and compromising their body organs. Studies have found that excessive ACE2 proteins are expressed in Alzheimer's patients’ brains. 35 And we know that coronavirus, SARS-CoV-2, enters the host cells by binding with the ACE2 receptor.34,36 Thus, COVID-19 may exacerbate the condition of Alzheimer's patients. In severe forms of Alzheimer's disease, the patient is entirely dependent on caregivers. As they lose their memory, they have no sense of wearing musk, sanitizing hands, and social distance from others. Their lifestyle could be more vulnerable if the caregivers do not take standard intervention to prevent COVID-19. 37 Moreover, at an advanced age, especially among 65, mortality rate is higher among this age group.38-40 Recently it is also anticipated that COVID-19 may cause Alzheimer's disease. 41 The probable mechanism by which COVID-19 patients may develop Alzheimer's disease is investigated. However, some characteristics, Aβ deposition, presence of Apolipoprotein E-4 (APOEε4) gene, neuroinflammation and microglia activation could be causative pathways responsible for linking COVID-19 and AD.42,43
Adverse Impacts of COVID-19 on the Brain and Cognition
Alzheimer's disease (AD) is a neurodegenerative disorder that affects a patient's memory and learning, behavior/attitude, and brain's cognitive functions. Accumulation of amyloid-beta (Aβ) or neurofibrillary tangles in the hippocampus of the brain results in AD.
44
Neurological and long-term cognitive complications are critical concerns for COVID-19 survivors.
45
A significant number of studies were conducted to determine neurological and cognitive function decline: In Copenhagen, Denmark 29 post-COVID patients participated in the cognitive assessment;
46
In South America, 45 post-COVID patients were evaluated in Shenzhen, China, 126 COVID-19 survivors participated in the study;
47
in the US, where the brain and cognition performance of 1517 participants were assessed;
48
In New York, America 156 post-COVID patients completed the survey.
49
A 6-month-long retrospective cohort study on 236379 post-COVID patients identified 14 different neurological and psychiatric consequences using patients' electronic health records. Among them, 33.62% of patients were diagnosed with the incidence of neurological or psychiatric issues, 12.84% received such diagnosis for the first time, and a higher percentage the patients (46.42%) were diagnosed with these consequences and were admitted to the intensive therapy unit (ITU) with a 25 .79% for a first-time diagnosis.
11
A systematic review reported a 43 to 66.8% prevalence of cognitive deficits in 644 hospitalized COVID-19 patients.
50
In the United Kingdom (UK), 39 cases out of 125 COVID-positive patients were identified with neurological manifestations, and 23 patients were diagnosed with the neuropsychiatric syndrome, vastly psychosis de-novo.
51
Prevalence of neurological or psychiatric manifestations was observed in 22.5% of 40,469 COVID-positive patients in a study that retrieved data from a global health collaborative platform.
51
All the conducted studies demonstrated the adverse impacts of COVID-19 infection on the brain and cognitive responses of the affected patients.45,46,52 Mild to severe neurological and cognitive impairment has been reported based on the extent and severity of COVID-19 infection in the patients.
52
Adverse consequences of post-COVID neurological and cognitive impairment have been evaluated using stringent assessment of the cognitive domains, including memory, attention, language, executive functions, and visuospatial skills, such as psychiatric symptomatology scales.
52
Although not on a large scale in some studies, promising biomarkers were used to determine the cognitive decline caused by COVID-19 infection. For example, NFL protein (neurofilament light chain protein), GFAP (Glial fibrillary acidic protein), S100b, astrocyte markers for neuronal damage/injury, total tau protein as a biomarker for neurodegeneration, and IL-6 (Interleukin-6), TNF (Tumor necrosis factor), CRP (C-reactive protein), complement system protein; C5, C6, and C8 as inflammation biomarkers for post-sepsis cognitive decline.
45
Figure 1 (Figure 1) showed the changes in the potential serum biomarkers responsible for adverse effects on brain and cognition. Irrespective of the assessment methods, all study results showed mild manifestation of the neurological complications, including loss of smell and taste, headache, dizziness, nausea, tingling sensations, severe fatigue, lack of motivation, anxiety, insomnia, post-traumatic stress disorder, depression, whereas the severe adverse manifestations are strokes, seizure, tremor, and aphasia.45,46,51-54 More importantly, COVID-19 induced neuronal damage is responsible for long-term cognitive impairments such as memory difficulties, inability to concentrate and focus, temporal orientation, task-set shifting, proactive control, and executive dysfunction.45-48,51-54 In addition to that, delirium or declined consciousness was reported in a higher percentage (14.8%) of severely COVID affected patients compared to those with less severity (2.4%).
37
The adverse effects of COVID-19 on the brain and cognition with the changes of related potential serum biomarkers. NFL: Neurofilament light chain, GFAP:Glial fibrillary acidic protein, TNF: Tumor Necrosis Factor, IL6: Interleukin 6, S100b: calcium-binding protein B.
The Underlying Mechanism of the Adverse Impact of SARS-CoV-2 on the Brain and Cognition
The underlying mechanism of the adverse impact of SARS-CoV-2 on the brain and cognition is not comprehensively precise. It may be multifactorial, involving direct invasion of the virus into the brain, indirect influence from oxygen starvation of the brain or extreme inflammatory response in the severely affected COVID-19 patients, leaky blood-brain barrier (BBB), intracerebral hemorrhagic lesions, white matter microhemorrhage, consequences of mechanical ventilation, long-term stay in the intensive care unit, the use of sedative medications and their secondary effects.45,51 The neurobiological model (Figure 2) below shows the etiology of the COVID-19 induced cognitive impairments. COVID-19 invasion into the body produces a cytokine storm and thus, elevates the levels of IL6, IL-1β, and TNF-α. In the Later phase, IL-6 and TNF-α can cross the BBB and activate microglia, which in turn release IL-1β. This IL-1β binds with receptors present in the hippocampal neurons and makes hippocampal neurons vulnerable, which is associated with long-term potentiation, attention, and memory disruption.
51
ACE-2 receptors stimulate BDNF (brain-derived neurotrophic factor) and regulate normal brain function. SARS-CoV-2 virus binds with these ACE-2 receptors reducing ACE-2-mediated BDNF function and thus impairing cognitive response. Furthermore, increased IL-6 level enhances permeability to BBB and activates astrocytes and microglia, which contributes to the inflammatory signals to the brain. High BBB permeability and (C-reactive protein) CRP/IL-6 levels are reported in the SARS-CoV-2 infected patients.50,51 Neurobiological model for the etiology of Alzheimer’s disease induced by SARS COV-2.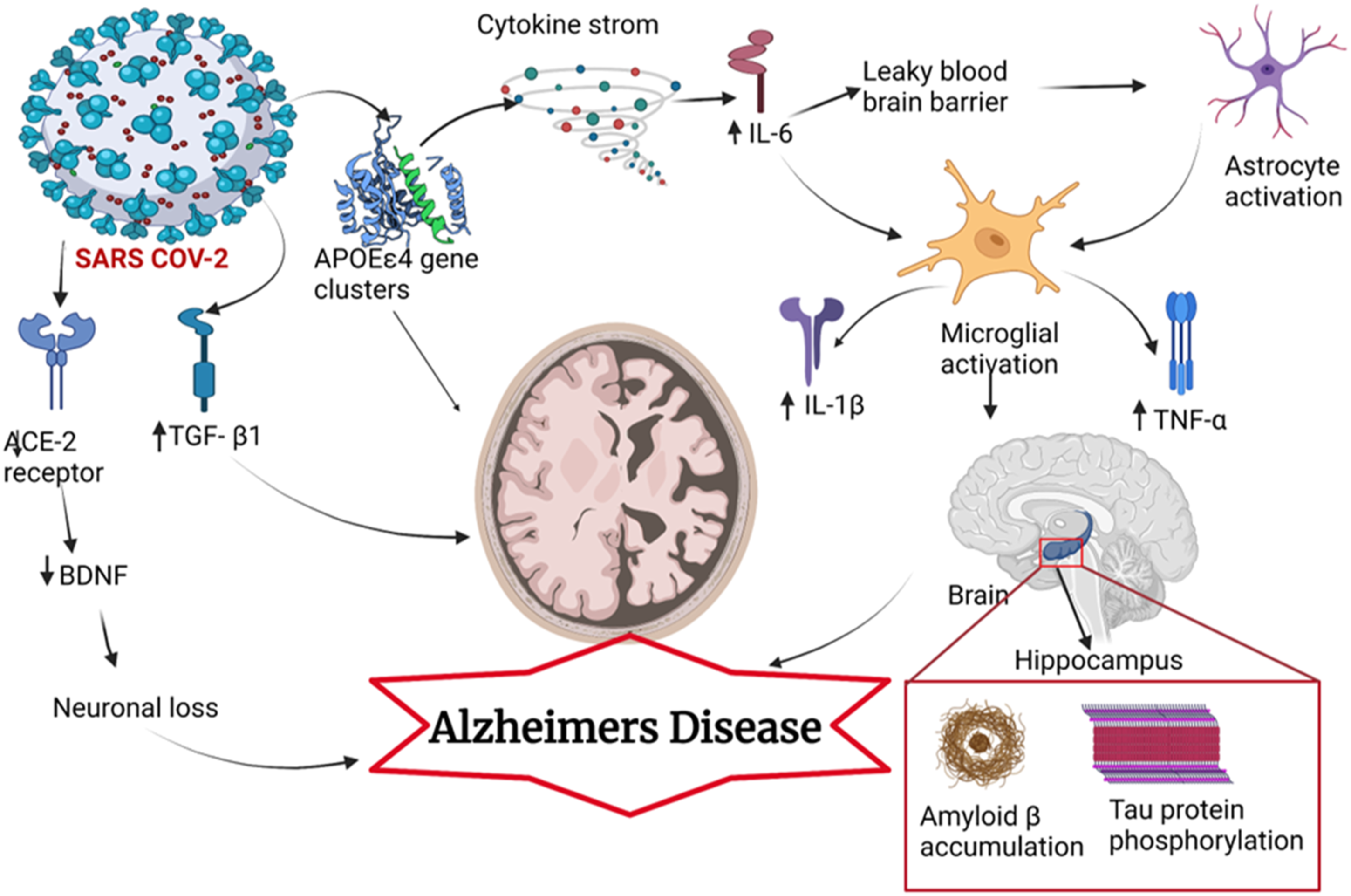
A study examined brain tissues of people with and without dementia who died of COVID-19 to understand the mechanism of SARS-CoV-2 in amplification of brain diseases in people with pre-existing dementia. The investigation revealed that SARS-CoV-2 virus altered the N-methyl-D-aspartate receptor 2 (NMDAR2), neuronal nitric oxides synthase (nNOS), SH-2 containing inositol 5’ polyphosphatase 1 (SHIP1) and major facilitator superfamily domain-containing protein 2 (MFSD2A) in the brain tissues of people who died of COVID-19 without dementia. It was concluded that endocytosis of SARS-CoV-2 spike protein may cause the CNS diseases in COVID-19 patients and pre-existing AD or dementia could be major risk factors for severe/fatal COVID-19 infection. 55 Then Idress et al investigated the interactions of SARS-CoV-2 spike proteins with heparin and amyloidogenic proteins which is potentially associated with the neurodegeneration in COVID-19 patients. They found that SARS-CoV-2 S1 has affinity for amyloid forming proteins eg, prion, tau, amyloid β (Aβ), α-synuclein, TDP-43 and heparin binding allows amyloid forming proteins to bind more strongly. The study also suggests that the heparin-binding site of the spike protein may act as a binding site for Aβ42 peptides, potentially leading to the aggregation of Aβ42 on the viral surface. 56 It was supported by another study investigating the binding of β-amyloid (Aβ) 1-42 to the spike protein S1 subunit (S1) of SARS-CoV-2 and the viral receptor ACE2, finding that Aβ 1-42 binds to these proteins with high affinity and enhances viral entry and IL-6 production in a SARS-CoV-2 infection model. 57 Another research group identifies the S2 subunit of the SARS-CoV-2 spike protein as a modulator of γ-secretase, a membrane complex involved in Aβ production, and suggests that the spike protein may contribute to neurological changes in COVID-19 patients. 43
Impact of COVID-19 on Alzheimer’s Disease Patients of Tomorrow
Epidemiological studies reported that 20 to 40% of the COVID-19 infected patients are over 60 years of age and the emerging data of the increasing SARS-CoV-2 cases in older people apprise extremely vulnerable situations for those who are already suffering from AD or other forms of dementia since several studies have confirmed the brain and cognitive decline in post-COVID patients such as memory difficulties, concentration/attention problems, distortion in temporal orientation, and executive function45-48,51-54 Escalation of neurodegenerative disorder, Alzheimer’s disease has been reported in post-COVID patients. AD patients’ brains and serums contain an extremely high concentration of immune mediators such as TNF-α, IL-6, and IL-1β. AD and COVID-19 induce inflammation through similar signaling pathways. Pre-existing neuroinflammation in AD patients is further aggravated by the SARS-CoV-2 induced pro-inflammatory cytokines which together cause a severe condition in patients, thus leading to death. 58 Genetically, people of sixty-year-old or over 60 who are carrying the APOEε4 allele are at higher risk of developing AD and at a greater risk of getting infected with the COVID-19 virus due to excessive production of cytokines and heightened inflammation.54,59
Furthermore, APOEε4 gene clusters-mediated protein products may serve as receptors for SARS-CoV-2 as these protein products have been reported to be used as receptors for other viruses such as hepatitis C virus and herpesvirus. In addition to that, due to memory and cognitive decline, AD patients forget to wear masks, maintain physical distancing, and other safeguarding material, which also aggravates the COVID spreads and are at greater risk of contracting the SARS-CoV-2 virus and its consequences. 54 Apart from the chronic neuroinflammation mentioned earlier, several independent researchers reported Guillain-Barré syndrome in post-COVID patients. And this is quite interesting that overexpression of TGF-β1(Transforming growth factor β1) was found in patients with Guillain-Barré syndrome and TGF- β1 over-expression was detected in AD patients, and TGF- β1 over-expressed mice developed AD-like symptoms, this observation confirms TGF-β1 as a contributing factor to AD. This probe implies the involvement of post-COVID Guillain-Barré syndrome with AD. 58 The rate of hospitalization of AD patients infected with AD compared to people without this disease is higher, according to the accumulating data. Several studies report that COVID-19 infected older adults with dementia are more likely to experience severe outcomes of it, including death, compared to COVID-infected patients with dementia. 59 AD has emerged as a critical contributing factor to morbidity and mortality of COVID-19 to overexpression of angiotensin-converting enzyme-2 (ACE-2) receptor and proinflammatory factors, AD-related complications such as diabetes, lifestyle changes in AD patients, and drug-drug interactions. AD and COVID virus infection worsen since AD patients have already been suffering from cognitive and other memory dysfunction, and the severe COVID viral infection has been proven to produce memory and cognitive decline. 58 It is noteworthy to mention that COVID-19 infected patients are required to go for quarantine or in isolation which poses an extra burden on AD patients since they are dependent on their caregivers or family members. 58 This contact restriction or isolation also poses a barrier to AD prevention since the physical activity of AD patients is required to improve cerebral perfusion, synaptogenesis, and neurogenesis. Physical activity is similarly essential in reducing neuronal loss, amyloid β accumulation, tau protein phosphorylation, and preserving brain volume in the specific region susceptible to AD.58,60 It has been reported that a 27.3% physical activity decrease in the senior community was evident 30 days after the pandemic was declared. 58
Association of Alzheimer-Like Brain With COVID-19 Survivors
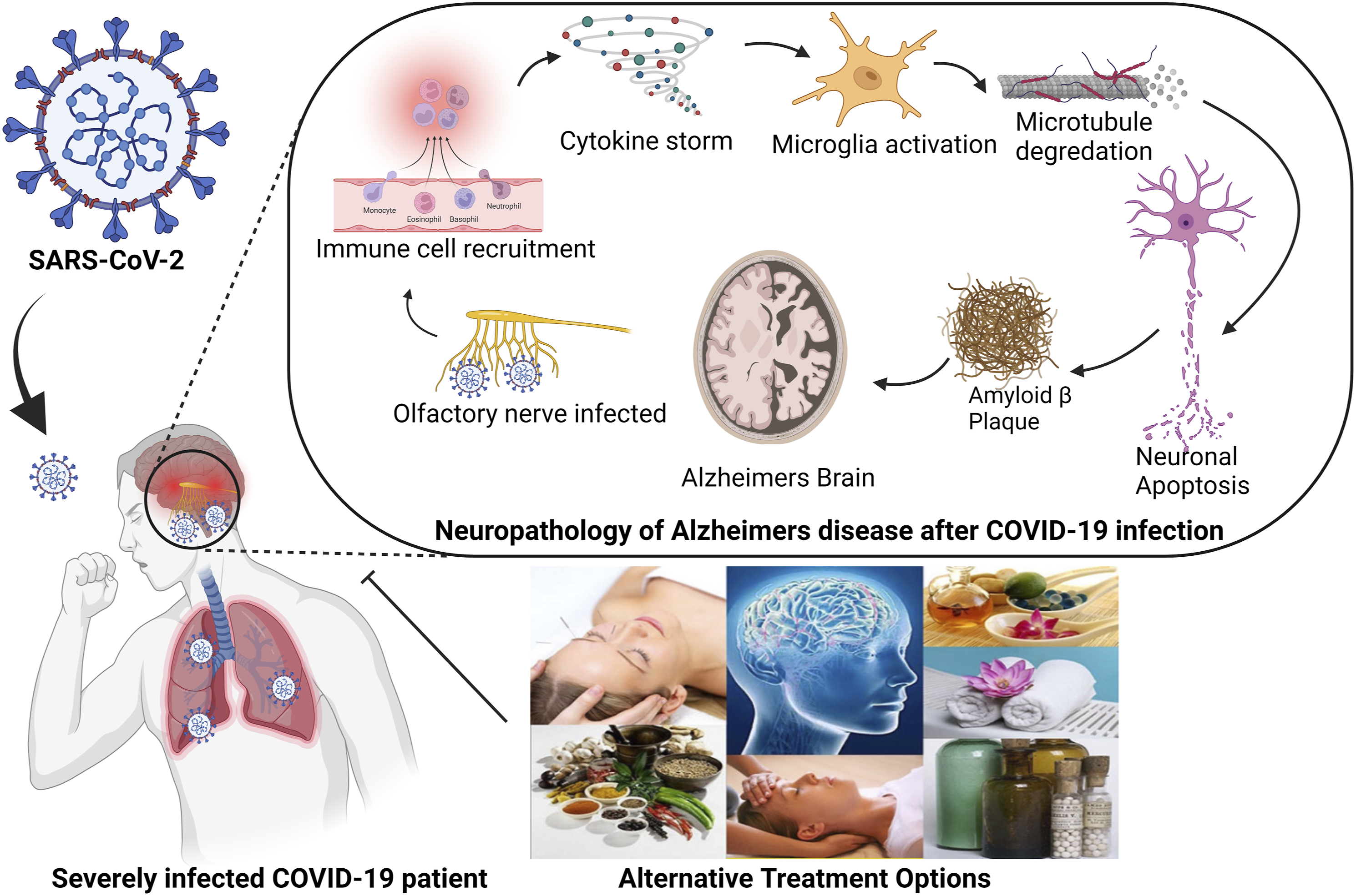
COVID-19 is initially thought to damage the respiratory system, but it can potentially harm the whole body, including the neural region. Headache, altered consciousness, and paresthesia are the common neurological symptoms among one-third of COVID-19 patients. Evidence of brain tissue edema, stroke, neuronal degeneration, and neuronal encephalitis have also been observed.61,62 A recent study discovered diffuse neural inflammatory markers in the brain of more than 80% of COVID-19 patients. It suggests that these processes may contribute to the reported neurological symptoms. 63 For example, hyposmia and hypogeusia (loss of ability to smell and taste) are common symptoms in SARS-CoV-2 infection and early-stage Alzheimer's disease (AD). Therefore, there is a possible link between COVID-19 and Alzheimer's disease. 64 Recently, it was discovered that the brain also expresses ACE2 receptors on glial cells and neurons, most notably in the brainstem, the paraventricular nucleus (PVN), the nucleus tractus solitarius (NTS), and the rostral ventrolateral medulla, all of which play a role in cardiovascular regulation. Moreover, chemokine can cause inflammation in the blood-brain barrier (BBB) by increasing its permeability since it is responsible for monocyte transmigration across the BBB. As a result, this allows more inflammatory cytokines and chemokines into the brain and exacerbates the patient's neurological symptoms via the ACE2 receptor due to invasion of SARS-CoV2. 65
It is also evident that the virus can cause central nervous system (CNS) and neurological damage without invading the brain. Cognitive dysfunction and neurodegeneration may be caused by cytokine storms which may develop Alzheimer's disease in COVID-19 survivors in the years to come. 65 In addition, SARS-CoV-2 infection most likely accelerates neuro inflammatory responses, synaptic pruning, and neuronal loss which are also the structural basis of Alzheimer disease; It is evident that glutamatergic-GABAergic imbalance caused by excitotoxicity reaction is a potential cause that promotes neuronal loss and further cerebral tissue damage. The mechanism of ACE2 involving both glutamatergic and GABAergic neurons suggests that invasion of SARS-CoV-2 infection can cause an imbalance of both signaling pathways in the CNS. Moreover, SARS-trans-synaptic CoV-2’s transfer toward the axon tips and toward the cell body allows the virus to infiltrate the entire brain slowly and diffusely. This may promote chronicity and neurodegenerative changes months and years after the acute infection.66-68
Interestingly, researchers found an association between Nicotinic acetylcholine receptors (nAChRs) and ACE2 receptor (viral target) localized in the brain. Since nicotine excitation of the nAChR increases ACE2 expression, it is thought that neural cells and astrocytes are more susceptible to SARS-CoV-2 infection in smokers. An ACE2 signaling pathway is thought to prevent oxidative stress and neuroinflammatory. 21 Long-term studies may be required to determine the links between SARS-CoV-2 infection, Alzheimer's disease, and other neurodegenerative sequelae. 69
Challenges and Alternative Treatment Options for COVID-19 Patients with Alzheimer’s Disease
Challenges
SARS-CoV-2 poses a considerable challenge to patients with Alzheimer's disease who have already suffered from brain and cognitive deficits. This sort of patient requires continuous support from their family members or caregivers for their daily life activities such as hygiene, medication, grocery shopping, and other day-to-day activities of life.58,60 SARS-CoV-2 infected AD patients are at a greater risk of worsening behavioral disturbance or psychiatric symptoms due to mandatory social isolation or self-quarantine. 70 Older adults with AD are especially susceptible to SARS-CoV-2 since they may not execute, comprehend, or recall any public health measures, including social distancing and wearing face masks. 37 In addition to that, there is a higher risk of spreading infection in COVID patients living with AD due to their memory deficit which ultimately results in not wearing or removing masks and not maintaining physical distancing while navigating public spaces or care homes.37,58,60,70 At the same time, they are also putting their caregivers or family members at a higher risk of contracting SARS-CoV-2. 60 According to the experts’ opinion, a long-term lockdown or movement restriction could result in chronic psychiatric complications in older patients with AD or other dementia, and the reported consequences are enhanced agitation and aggression, which can be challenging to manage for the family members, caregivers, or care home personnel.58,70 Furthermore, a considerable challenge is faced in the management of hospitalized COVID patients with existing AD or other dementia because isolation in a single room for a prolonged period in unfamiliar surroundings without support from their family members or relatives elevates the risk of delirium to a greater extent. 37 Agitation and wandering of the patients, such as entering other patients’ rooms, removing the face mask, reaching out and touching hospital staff, and contacting high-touch spaces and equipment, make it highly arduous to comply with infection control procedures. 37
Alternative Treatment Options
Currently, not many drugs are available that can completely cure dementia, and cognitive decline and most of the drugs can only alleviate the symptoms. The most prescribed medications used to treat mild to severe AD belong to cholinesterase inhibitors, including rivastigmine, galantamine, donepezil, and memantine. Other drugs treat aggression, depression, anxiety, and restlessness, such as citalopram, imipramine, sertraline, duloxetine, mirtazapine, and bupropion. The main drawback of all anti-AD drugs is they are inept at averting the disease progression further or reversing its effects. Additionally, anxiolytic and antidepressant drugs exhibit severe side effects, including dizziness, falls, confusion, and sleepiness. 71
Limited efficacy, significant side-effects of currently available AD treatments, poor patient compliance, socio-economic cost of long-term attendance, and hospital expenditure urge the discovery and development of more effective and safer alternative therapies to treat AD.
72
Many alternative treatments have been found beneficial in preserving or restoring cognition and synapses' function. Ayurveda, Traditional Chinese Medicines, Herbal medicines, Acupuncture, Aromatherapy, meditation, exercise, etc., have proven effective in slowing down AD progression and complementing the currently available mainstream AD treatments.
71
Figure 3 (Figure 3) shows the overview of the mechanism of action of the alternative treatment options for COVID-19 patients with AD. Table 1 and Table 2 have compiled the list of alternative treatment approaches for dementia, cognitive decline, and significant AD signs. Below is a detailed delineation of these approaches. Overview of the mechanism of action of the Alternative treatment options for COVID-19 patients with AD. Alternative Treatment Options for Dementia and Cognitive Decline. Herbal Medicines/Natural Supplements and Vitamins for Treatment and Prevention of Dementia and Cognitive Decline.


Coconut Oil
Coconut oil has been reported to enhance cognition due to lauric acid, medium-chain triglycerides, and ketone bodies. Research conducted on these constituents of coconut oil revealed that enhancement of cognitive ability involves several mechanisms such as autophagy, regulation of ketone body that the brain uses as energy instead of glucose, reduction of oxidative stress and antioxidant activity, reduction of Aβ peptide secretion and mRNA level of amyloid precursor protein (APP) expression through inhibition of ARF1 (ADP-ribosylation factor 1). The ARF1 is a transport protein critical for the secretory pathway to transport APP to the cell surface. 73
Omega-3 Fatty Acids
Omega-3 fatty acids are essential polyunsaturated fatty acids. DHA (Derivatives of docosahexaenoic acid) is a type of omega-3 fatty acid in seafood, including deep-water oily fish, seaweeds, mollusks, and crustaceans, and is crucial for synaptic function. A high concentration of docosahexaenoic acid (DHA) contributes to the plasma membrane's fluidity and thus facilitates neurotransmitters' transport. Omega-3 fatty acids have been reported to improve cognitive function by protecting oxidative stress and promoting neuronal maturation and development in the hippocampus. 74
Co-enzyme Q10
Coenzyme (Co-Q10) is a component of the electron transport chain that generates adenosine triphosphate (ATP) and participates in aerobic cellular respiration. Co-Q10 is predominantly present in meat, fish, and poultry and to a small extent in nuts, soybeans, cereals, and vegetables such as broccoli and spinach. Accumulating research data evidence that Co-Q10 improves cognitive functions, learning, and memory deficits through several mechanisms, including antioxidant/pro-oxidant activity, ATP synthesis, and brain atrophy attenuation. Most importantly, Co-Q10 partially reduces the overproduction of amyloid β.75,76
Acupuncture
Acupuncture originated in ancient China and has been primarily used in the clinic for a long time. According to TCM (Traditional Chinese Medicines) theory, inserting sharp and thin needles in specific acupoints generally dredge meridians and relieves blockage in Qi and blood flow which is the fundamental energy that activates and maintains the holistic body system. In addition, acupuncture can augment congenital and required Qi to promote health and produce a therapeutic effect by preventing invasive factors.71,77 In treating AD, acupuncture mitigates cognitive impairment by modulating synaptic plasticity since the insertion of needles stimulates nerve receptors at specific brain points linked to neuron synaptic plasticity. In the SAMP8 mice model of AD, manual acupuncture improved spatial learning, relearning, and memory.95,96 Vagus nerve stimulation (VNS) is one of the important physical therapy techniques that can improve cognitive function. Previous study has confirmed the positive effect of invasive VNS (iVNS) on cognition improvement in AD patients. It is noteworthy to mention that 70% of AD patients showed no further cognitive function impairment after application of iVNS to 17 AD patients for 6 months. 97 Potential of VNS in altering multiple AD progression has been demonstrated in the previous study where VNS provided anti-inflammatory signaling, increased metabolic support of neuronal function by dint of astrocytes, modified accumulation of Aβ pathology, and supported neuronal plasticity. 98 Activation of the locus coeruleus (LC) by VNS results in catecholamine release in the neocortex and hippocampus which ultimately causes reduction of inflammatory signals and augmentation of synaptic plasticity. Non-invasive transcutaneous vagus nerve stimulation (tVNS) has also been found effective in enhancing memory functions in 60 participants in a previous study. 98 It is important to note that acupuncture therapy for AD has recently gained increasing popularity worldwide, reinforcing its efficacy and safety.95,96
Herbal Medicines/Natural Supplements
Herbal medicines or natural supplements have been used in treating various diseases. Natural products have proven effective in reducing the progression and symptoms of AD. Accumulating research data revealed the benefits of using whole plant extracts or isolated compounds to treat AD. Different phytochemical classes of compounds, including polyphenols, flavonoids, lignans, sterols, triterpenes, tannins, and alkaloids, have produced many pharmacological effects such as antioxidant, anti-inflammatory, anticholinesterase, and anti-amyloidogenic which are crucial to generate Anti-AD effect. 85 The whole plant extracts of Ginkgo biloba, Centella asiatica, Convolvulus pluricaulis, Zingiber officinaleis, Bacopa monnieri, Panax ginseng, etc., have been reported to provide neuroprotection, and improve cognitive ability and memory deficit71,79,81-85,99,100
Ginkgo biloba
The mechanism of the anti-AD effect of Ginkgo biloba involves free radical scavenging, c-Jun N-terminal kinase (JNK) pathway and Extracellular signal-regulated kinase (ERK) pathways activation, prevention of neuronal apoptosis, and reduction of mitochondrial dysfunction. 79
Centella Asiatica
Centella asiatica extract provides neuroprotection and memory enhancement through decreasing oxidative stress response in the brain and β-amyloid pathology in vivo, inhibiting Aβ1-40- induced neurotoxicity; reducing ROS production, and modulating the antioxidative defense system81,82,99,100
Convolvulus Pluricaulis
Convolvulus pluricaulis extract exerts nootropic and memory enhancement action by calming nerves through modulation of the stress hormone, adrenaline, and cortisol levels, alleviating neurotoxicity by reducing the mRNA levels of tau and amyloid-β protein precursor (AβPP). 83
Zingiber Officinaleis
Zingiber officinaleis is widely used as food supplements as an extract or as an ingredient of ginger tea, and it provides protection against AD and enhances cognitive function by preventing AD by inhibiting lipid peroxidation, repressing NMDA receptor overstimulation, and increasing Ach level in the synapse by inhibiting the AChE enzyme. 84
Panax ginseng (Ginseng)
Panax ginseng (Ginseng) supplement improves thinking and working memory and cognition via Removal of Aβ, anti-inflammatory action, inhibition of apoptosis, stimulation of soluble AβPPα formation; apoptosis, ROS production; reduction of oxidative stress, and enhancement of CNS cholinergic activity. 71
A significant number of studies confirmed the anti-AD effect of natural compounds isolated from plant extracts.
GP-17 (A Novel Phytoestrogen of Ginseng)
GP-17 (a novel phytoestrogen of Ginseng) gives neuroprotection and reduces AD-related complications through inhibition of Aβ formation, autophagy-based elimination of Aβ through transcription factor EB activation, stimulation of soluble AβPPα formation, inhibition of inflammation, apoptosis, ROS production, reduction of oxidative stress and enhancement of CNS cholinergic activity. 71
Centella Asiatica
Asiatic acid and asiaticoside isolated from Centella asiatica provides neuroprotection by scavenging free radicals, reducing H2O2-induced cytotoxicity, and preventing β-amyloid-induced cell damage in vitro. 81
Huperzine A
Huperzine A is a sesquiterpene alkaloid present in Huperzia serrate and has demonstrated learning capacity. Memory enhancement and the mechanisms involved were inhibition of AChE enzyme and apoptotic factors including, p53, Bax, and Caspase 3, regulation of nerve growth factor expression and secretion, reduction of Aβ plaques and oligomeric A formation in the hippocampus and cortex, inhibition of BACE, γ-secretases, and PKC/MAPK pathways, and inhibition of NMDA receptor and K-channel in the brain. A polyphenolic compound named resveratrol gave neuroprotection and improved spatial memory via several mechanisms such as depletion of Amyloid β level through induction of non-amyloidogenic breakdown of APP, clearance of Amyloid β, inhibition of AChE activity in the neuronal cells. 85
Curcumin
Curcumin, a curcuminoid is the main active compound of Curcuma longa, which can protect neurons via reduction of plasma β-amyloid protein concentration, inhibition of β-amyloid accumulation, prevention of lipid peroxidation and oxidative damage in the brain, counteraction of β-amyloid pathology, scavenging ROS, and protection against β-amyloid-induced neurotoxicity through inhibition of τ-hyperphosphorylation and oxidative damage. 85
Quercetin
Quercetin, a flavonoid present in berries, apples, onions, green tea, and red wine, has improved cognition and memory impairment and provided neuroprotection. Mechanism of anti-AD action of quercetin involves inhibition of β-amyloid accumulation and reduction of Aβ-induced neuronal cell apoptosis. 85
Berberine
Berberine is an isoquinoline alkaloid isolated from Coptis chinensis which has demonstrated amelioration of memory impairment through inhibition of AChE and butyrylcholinesterase, monoamine oxidase activity, significant reduction of β-amyloid-induced IL6, Inducible nitric oxide synthase (iNOS), and COX-2 levels, attenuation of NF-κB expression via PI3K/protein kinase B and MAPK pathways. 85
Luteolin
Luteolin is a flavonoid present in many medicinal plants, including Pteridophyta, Magnoliophyta, Pinophyta, and Bryophyta. Studies reported that luteolin could improve cognitive function and memory by reducing zinc-induced τ-protein hyperphosphorylation and Aβ-formation by decreasing the amyloid precursor protein expression, depleting ROS formation, and improving the endogenous antioxidant system through augmentation of Superoxide Dismutase (SOD), Glutathione Peroxidase (GPx), and cat activities. 85
Rosmarinic Acid
Rosmarinic acid is a phenol-type carboxylic acid found in many Lamiaceae species. Several studies confirmed that it enhances cognition and prevents dementia through the reduction of τ-protein hyperphosphorylation, scavenging ROS, inhibition of caspase-3 activation and DNA fragmentation, and removal of NF-κB and TNF-α expressions.
85
Since the extracts of herbs, and medicinal plants and isolated pure compounds have been reported to exhibit significant and notable anti-AD effects in either in vitro or in vivo studies or very limited clinical studies more clinical studies are warranted to establish them as effective therapeutics for AD treatment. Additionally, more natural sources must be explored for novel, effective and safe drugs for the treatment of AD. Figure 4 (Figure 4) demonstrates the role of herbal medicines and natural supplements to inhibit the mechanism of action of Alzheimer's diseases progression. Mechanism of anti-AD action of herbal medicines/natural supplements/vitamins to inhibit the Alzheimer's disease progression due to SARS-COV-2 virus.
Vitamins
The deficiency of vitamins has a connection with decreasing cognition due to the increased level of homocysteine. Vitamin B6 (Niacin), B9 (Folate), and B12 (Cyanocobalamin) play a vital role in homocysteine metabolism, and thus, supplementation with these vitamins can enhance cognitive ability by decreasing the level of homocysteine. 71 It has been reported that vitamin A and C improve cognition and memory through their antioxidant activity. Furthermore, supplementation of vitamin A with β-carotene can improve cognitive ability by preventing amyloid-β-induced cell death because a poor level of retinol (Vitamin A) is detected in AD patients and the combination of vitamin A and β-carotene are essential for cognition, learning, and memory. 86 Previous studies revealed an association of lower levels of vitamin D with poor cognitive performance. It has also been reported that the dietary intake or supplementation of vitamin D can enhance cognitive power through suppression of APP transport and clearance of Aβ plaques.71,86 Reduced levels of vitamin E are also determined in AD patients, and it can provide neuroprotection and improve cognition by exerting antioxidant effects through scavenging free radicals and regeneration of SOD and anti-inflammatory effects.71,86
Probiotics and Prebiotics
Probiotics are living microorganisms that directly provide health benefits whereas prebiotics are oligosaccharides and polysaccharides that modulate/regulate the growth, survival, and functions of gut microbiome which in turn exert beneficial effects on human hosts. 92 Probiotics and prebiotics have been found beneficial in improving memory and cognitive functions in AD patients in a wide array of research conducted in animal models as well as in clinical trials (Table 2).87,89,91-94 Administration of probiotics and prebiotics are two vital approaches that modulate the gut microbiome toward functional and compositional states and thus confer improved cognitive outcomes in the AD patients. 92
Probiotics
Probiotics administration exerted a potential effect on cognitive and memory functions improvement in many studies (Table 2). Oral administration of Bifdobacterium breve strain A1 (B. breve A1) in AD model mice prevented cognitive dysfunction and demonstrated potential amelioration of cognitive decline and ultimately improved cognitive function in AD. Particularly B. breve A1 exerted this therapeutic effect in preventing cognitive impairment in AD through suppression of hippocampal expression of inflammation and immune-reactive genes induced by amyloid-β.87,88 Probiotic-induced microbiota have been found beneficial in preventing memory loss in AD and therapeutically effective in ameliorating cognitive decline and improving memory functions. Aged rats treated with bifidobacterium and lactobacillus mixture showed altered brain metabolites such as [glutamate and γ-aminobutyric acid (GABA) in the cortex and hippocampus and thus, demonstrated improved neuronal signal propagation and memory functions. 87 Memory functions of the Lactobacillus plantarum MTCC 1325 treated AD rats were improved via attenuation of Aβ plaque formation and restoration of acetylcholine levels. 87 In another study Bifidobacteria and lactic acid bacteria (SLAB51) was found to be effective in inhibition of AD onset and development in PD (Parkinson’s disease) induced-mice through abatement of Aβ plaque formation. 89 A previous study investigated the therapeutic effect of Lactobacillus acidophilus, Bifidobacterium bifidum and Bifidobacterium longum and their findings confirmed that these probiotics can improve spatial learning and memory in adult normal reared male Wistar rats via restoration of synaptic plasticity in the brain hippocampus, prevention of Aβ plaque formation; reduction of malondialdehyde level in brain, and regulation of brain metabolites. 90 Clostridium butyricum WZMC1016 was administered in AD induced 6-month old transgenic mice for 4 weeks and the spatial learning and memory functions of the mice were found to be improved via reduction of Aβ and Aβ42 formation in the brain; Depletion of IL-1β and α-TNF levels. 91 Lactobacillus rhamnosus UBLR-58 was found to improve memory via reduction of acetylcholinesterase activity, MDA level, and amyloid plaque deposition in the brain.
Prebiotics
Prebiotic-assisted gut microbiome modulation and improvement of cognitive and memory functions in AD have been reported in several studies conducted previously (Table 2). Therapeutic potential of Mannan-oligosaccharide in AD was tested in 5×Familial AD transgenic mice and the findings demonstrated decreased Aβ accumulation in the brain and neuroinflammation suppression which resulted in improved cognition and behavioral disorders. 92 Cognitive function was restored in rats exhibiting cognitive decline when treated with xylo-oligosaccharide supplementation for 14 weeks. Additionally, fructan and GOS (Galacto-oligosaccharides)-based prebiotics have been found consistently effective in improving cognitive and behavioral functioning in a clinical trial through enhancement of abundance in bifidobacteria. 92 Oligosaccharide-enriched inulin was tested in 47 participants in a double blind placebo (Maltodextrin) controlled study and improved recall performance and cognitive memory tasks have been identified in participants who consumed inulin compared to placebo. Mechanism involved was increasement of Coprococcus and Bifidobacterium abundance to exert such an effect. 93 In another double blind randomized, placebo-controlled crossover study in 18 healthy participants, polydextrose consumption resulted in modest cognition improvement via significant change in abundance of Ruminiclostridium 5. 94
Conclusion
SARS-CoV-2 infection in the last 3 years has had a huge impact on daily life globally, and the widespread infection has affected many peoples with a remarkable mortality rate. Those who survived experienced several health issues, including long COVID-19. Several research groups are following the updates on health conditions before and after SARS-CoV-2 infection. Recently, it has been claimed that post-COVID patients face numerous neurological changes that they never diagnosed before the infection. A few studies 21 were conducted to understand the brain inflammation and deposition of Aβ and neurofibrillary tangles. Few patients showed Alzheimer's brain changes after a long time of SARS-CoV-2 infection. Therefore, it has become a concern among the healthcare communities to reveal the mechanism behind the reasons for such changes and find out the relationship between COVID-19 and AD. The recent studies are not sufficient to conclude the relation. However, further animal studies 21 and a large cohort of COVID-19 survivors who showed neuroinflammation and neurological changes may augment the research to know the truth. Moreover, natural products with immunomodulatory and anti- inflammatory potential could be tested using current animal models of AD being challenged by SARS-CoV-2. 101 Additionally, many alternative treatments have been found beneficial in preserving or restoring cognition and synapses' function of AD patient discussed here. 8
Footnotes
Author Contribution
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
