Abstract
Objective
To investigate the incidence of delirium and its related risk factors in patients with senile dementia during hospitalization.
Methods
A retrospective analysis of clinical data of 157 patients over 65 with cognitive impairment who were hospitalized in the comprehensive ward from October 2019 to February 2023 was conducted. Patients were assigned into delirium and non-delirium groups according to whether they exhibited delirium during hospitalization. General information about the patients and Visual Analogue Scale (VAS) score, blood C-reactive protein level, and blood superoxide dismutase (SOD) level were recorded. Univariate analysis was used to identify potential risk factors for delirium, and factors with statistical significance were subjected to multivariate logistic regression analysis. A prediction line chart for delirium in elderly dementia patients was constructed using R 4.03 software, and the model was validated.
Results
Among the 157 patients with senile dementia, 42 patients exhibited delirium and 115 patients exhibited non-delirium. Multivariate logistic regression analysis showed that diabetes, cerebrovascular disease, VAS score ≥4 points, use of sedative drugs, and blood SOD <129 U/mL were independent risk factors for delirium during hospitalization in elderly dementia patients. A prediction nomogram was plotted based on the five risk factors, and receiver operating characteristic curve analysis presented an area under the curve of .875 (95% CI: .816-.934). The nomogram model was internally validated by the Bootstrap method, and the calibration curve showed good agreement between predicted and actual results. Hosmer-Lemeshow test demonstrated that the model had a good fit and high predictive ability.
Conclusion
Diabetes, cerebrovascular disease, VAS ≥4 points, use of sedative drugs, and blood SOD <129 U/mL were independent risk factors for delirium in patients with senile dementia during hospitalization. The nomogram model had good accuracy and clinical application value for predicting delirium in this study.
Introduction
Senile dementia is a clinical syndrome that occurs in older people, most commonly in the form of Alzheimer’s disease, which is characterized by memory and cognitive impairment. 1 Delirium occurring in dementia patients is known as delirium superimposed on dementia (DSD). Increasing evidence suggests that in elderly patients, dementia is an independent risk factor for delirium. 2 A meta-analysis of 81 studies (involving 81,536 dementia patients) found that the overall incidence of DSD during hospitalization was 48.9%, much higher than that of other hospitalized elderly patients. 3
Delirium is an acute neurocognitive disorder, featured by acute and sudden cognitive impairment, disorientation, decreased attention, decreased level of consciousness, and perceptual disturbance. 4 Evidence suggests that compared with dementia patients without delirium during hospitalization, DSD patients have longer hospital stays, greater functional and cognitive decline, more functional impairment, and higher risk of death. 5 It has been reported that DSD patients have a 2.6-fold increased risk of dying in hospitals compared to patients in the absence of cognitive impairment (neither delirium nor dementia), while there is no significant correlation between dementia alone and increased risk of in-hospital death. 6 In the United States, annual medical costs related to delirium range from 3.8 to 15.2 billion dollars, placing a heavy load on families and society. 7 Therefore, early identification of high-risk factors for delirium is particularly important. Inouye et al. 4 analyzed 11 studies and found that major risk factors for all patient populations include dementia or cognitive impairment, functional impairment, visual impairment, alcohol abuse history, advanced age (>70 years), and comorbidities or specific complications (such as stroke or depression). However, individual factors that affect delirium development in elderly patients with dementia have not yet been determined. Therefore, this study intended to determine the incidence of delirium during hospitalization in patients with senile dementia and its associated risk factors.
Materials and Methods
Study Participants
A total of 157 dementia patients aged 65 years and over, who were admitted to the comprehensive ward of China-Japan Friendship Hospital from October 2019 to February 2023, were included in this study.
Inclusion criteria: aged 65 years and over; met the diagnostic criteria for elderly dementia in the 2018 Chinese Guidelines for Diagnosis and Treatment of Dementia and Cognitive Impairment; 8 had complete clinical data; signed informed consent forms.
Exclusion criteria: patients with unstable vital signs; severe visual or auditory impairment; those considered unsuitable for the trial by the attending physician.
This study was approved by the ethical committee of China-Japan Friendship Hospital.
Methods
Data Collection
General information about each patient was obtained, including age, gender, body mass index (BMI), body temperature, biochemical indicators (C-reactive protein (CRP) and superoxide dismutase (SOD)), comorbidities (hypertension, diabetes, hyperlipidemia, cardiovascular disease, cerebrovascular disease, renal insufficiency, anemia, hypoproteinemia), smoking history, drinking history, whether accompanied by family members, surgical history during hospitalization, visual analog scale (VAS) score, use of sedative drugs, and whether a drainage tube was placed. The VAS uses a range of 11 numbers, from 0 to 10, to represent the intensity of pain. A higher number indicates a higher level of pain. The VAS scoring criteria were adopted from the study by Hawker et al. 9 .
Delirium Diagnosis
During hospitalization, delirium was diagnosed by the vice chief physician or above following the Confusion Assessment Method (CAM) standard 10 during daily rounds. Patients were assigned into delirium and non-delirium groups according to the presence or absence of delirium.
Statistical Methods
Data were processed on SPSS 26.0 statistical software. Count data were presented as n (%) and analyzed by chi-square test or Wilcoxon rank-sum test. Measurement data were presented as mean ± SD and analyzed by t-test. Factors with statistical significance (P < .05) were subjected to multivariate logistic regression analysis. A nomogram was established using R (4.0.3) software. The independent risk factors for DSD in elderly patients were evaluated by receiver operating characteristic (ROC) curve analysis.
Results
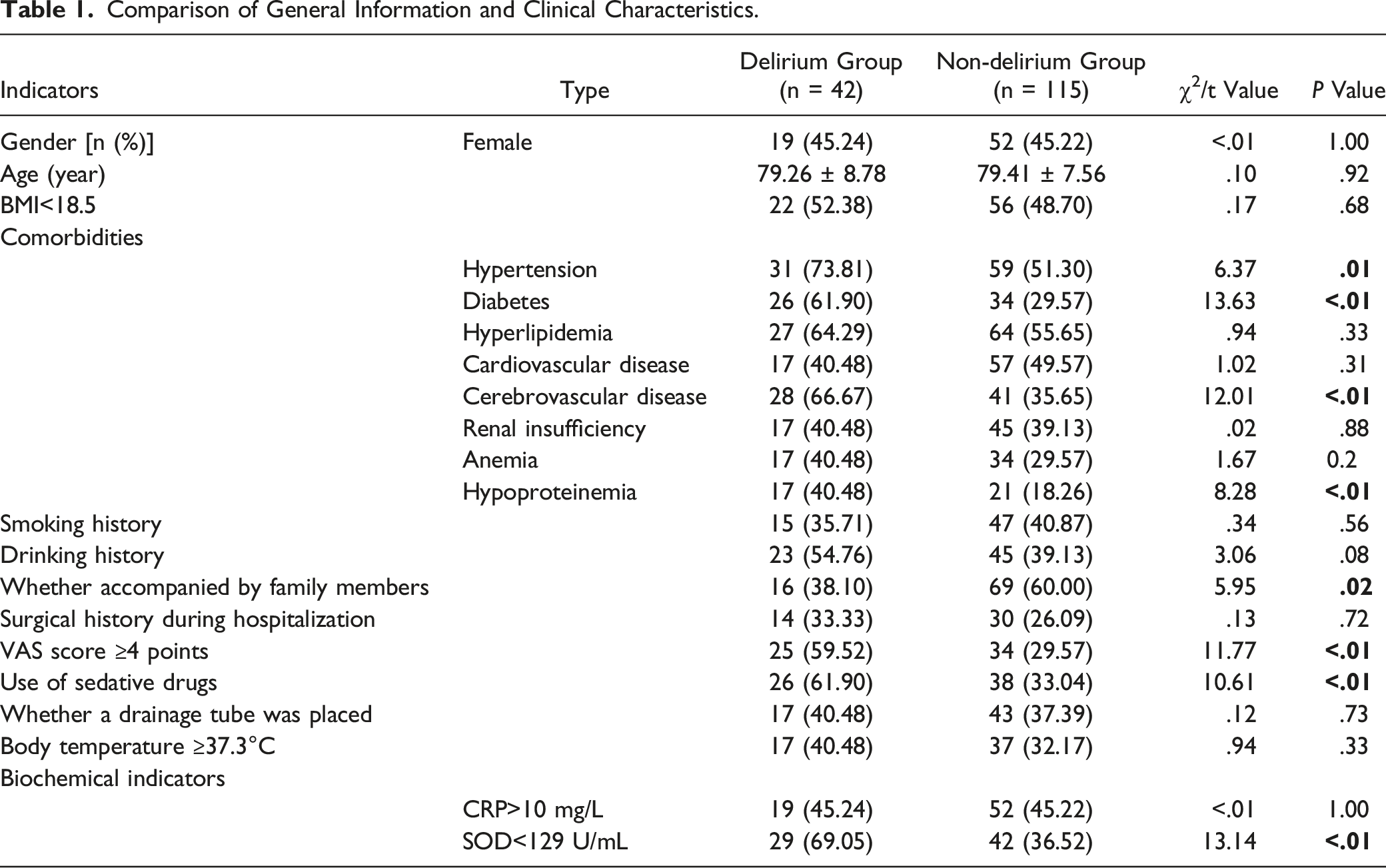
Comparison of General Information and Clinical Characteristics
Comparison of General Information and Clinical Characteristics.
Multivariate Logistic Regression Analysis of Delirium in Dementia Patients
Multivariate Logistic Regression Analysis of Delirium in Dementia Patients.

Construction of a Prediction Model
On the basis of multivariate logistic regression analysis, the five independent risk factors for DSD were included in the nomogram to build a prediction model for the risk of DSD. Each patient’s total score was determined by adding the results of the respective variables, and the probability of DSD was computed by converting the relationship between total score and probability of occurrence into a function (Figure 1(A)). Construction of the Prediction Model. (A) Nomogram for DSD in elderly patients; (B) Calibration curve for internal validation of the nomogram.
The calibration curve of the prediction model was drawn by Bootstrap validation, with the optimal model being the 45-degree line. The prediction results of the nomogram were highly consistent with actual results, and the Hosmer-Lemeshow test showed a good fit of the model (Figure 1B), indicating the model with favorable discrimination and accuracy.
ROC curve analysis presented that the optimal detection of DSD could be achieved at a cut-off point of .1459 (sensitivity 69.57%, specificity 90.48%) (Figure 2). Area under the curve (AUC) was .875, with a 95% CI of .816-.934. ROC curve for predicting DSD in elderly patients. The red dashed line represents the reference line.
Discussion
Patients with senile dementia are at higher risk of suffering delirium. According to statistics, DSD incidence in elderly inpatients and community populations aged 65 and above ranges from 22% to 89%. 11 In this study, 42 out of 157 patients with senile dementia admitted to the hospital developed delirium, with an incidence rate of 26.75%. Due to the potential fragility of the brain in elderly patients with dementia, it may be difficult to prevent delirium. 12 Therefore, evidence-based treatment strategies are needed to alleviate delirium severity in dementia patients. We attempted to explore the incidence of delirium during hospitalization in elderly patients with dementia and its related risk factors through univariate and multivariate logistic regression analyses. The results showed that diabetes, cerebrovascular disease, VAS score ≥4 points, use of sedative drugs, and blood SOD <129 U/mL were independent risk factors for DSD in elderly patients. Healthcare workers should pay close attention to these patients during diagnosis and treatment and improve treatment strategies in a timely manner to detect and treat delirium as early as possible, thereby improving the patient’s prognosis and quality of life.
The specific pathogenesis of delirium is undefined, and it is currently believed to be the consequence of multiple factors. 13 According to current research, the risk factors for delirium vary among different patient populations. For example, in ICU patients, advanced age, mechanical ventilation, and use of sedative drugs are common risk factors for delirium14,15; in elderly patients with gastric cancer after surgery, age, albumin/fibrinogen ratio, neutrophil/lymphocyte ratio, and use of sleeping pills increase the risk of delirium. 16 This study found that comorbid diabetes or cerebrovascular disease is an independent risk factor for DSD in elderly patients. Previous studies also consistently suggest that cerebrovascular disease is one of the delirium risk factors in hospitalized elderly patients, 17 possibly due to the disruption of structural connections between the brain interhemispheric and frontal-subcortical-cerebellar networks and microstructural changes in regions related to memory and executive function. 18 Diabetes has a potent predictive effect on both dementia and delirium in elderly patients.19-21 This study found that patients with senile dementia and comorbid diabetes had a nearly 5-fold increased risk of delirium (OR = 4.99). This may be related to brain insulin resistance, as animal model experiments have shown that brain insulin resistance promotes the generation of amyloid-beta and hyperphosphorylation of tau protein to drive Alzheimer’s disease.22,23
In addition, this study found that pain with VAS score ≥4 points increased the risk of DSD in elderly patients by four-fold (OR = 4.19). The likely reason is that dementia patients have a significantly reduced ability to express pain, which results in ineffective and delayed pain treatments. This leads to negative emotions such as anxiety, depression, and mania, which in turn affect physiological functions and disrupt the sleep-wake cycle,24,25 ultimately increasing the risk of delirium. With regard to the use of sedative drugs, antipsychotics and sedatives such as sleeping pills are commonly used for dementia patients as their clinical symptoms include sleep disturbances and behavioral and psychological symptoms. Studies have shown that antipsychotic drug use in dementia patients in long-term care facilities ranges from 12% in Sweden to 54% in Spain. 26 In this study, 61.9% of patients in the delirium group had used sedative drugs during hospitalization. Other research has also shown that benzodiazepine usage, particularly the use of lorazepam and midazolam, is independently linked to an elevated risk of delirium.27-29
Furthermore, we found that the blood marker SOD is associated with delirium in dementia patients. SOD is an internal antioxidant that induces superoxide ions conversion into hydrogen peroxide in cells. 30 In a double-blind, placebo-controlled study with 70 healthy participants (30-55 years) who experienced regular stress and tiredness, the intervention group received a daily supplement of 10 mg Extramel (each capsule containing 140 IU of SOD). Following 4 weeks of oral taking, the participants showed reduced stress and physical fatigue, and significant improvements in cognitive performance on psychological measurement scales. 31 This mechanism has been confirmed in animal models, where SOD treatment in mice prevented stress-induced cognitive impairment by lowering lipid peroxidation and keeping hippocampal neurogenesis. 32 This study indicated that a blood SOD level of less than 129 U/mL was an independent risk factor for DSD in elderly patients. This result suggested that healthcare workers need to keep a watchful eye on senile dementia patients with low blood SOD levels and take appropriate preventive measures, such as additional oral supplementation of SOD.
A nomogram is a graphic that integrates multiple predictive indicators and displays them on the same plane using scaled lines to illustrate the relationships between different variables. It is frequently applied to forecast the risk of diseases.33,34 This study constructed a nomogram for the DSD prediction model based on the five aforementioned risk factors. Each factor was assigned a corresponding score, and the total score was computed by adding scores of each risk factor according to the lines, to predict the incidence rate of DSD in elderly patients. In addition, this study evaluated model accuracy by calibration and ROC curves, presenting that this model had favorable accuracy and discriminative ability and was clinically effective. Currently, there is limited research on the prevention and management of DSD, with many clinical guidelines emphasizing the identification and treatment of potential triggers and advocating for multi-component interventions aimed at meeting the basic needs of patients. These interventions are typically interdisciplinary and non-pharmacological, such as cognitive stimulation, improving vision, providing hearing aids to enhance sensation, optimizing nutrition, and adequate but not excessive pain management.35,36 A meta-analysis found that multi-component non-pharmacological treatments for delirium prevention can effectively reduce the incidence rate. 37 However, studies have shown that evidence for multi-component treatments in changing the trajectory of delirium is not strong, 38 but such ways are fundamentally practical. The DSD high-risk factors selected in this study are beneficial for accurately identifying DSD high-risk patients in clinical practice, and early intervention to reduce controllable risk factors can help reduce the delirium incidence rate, providing a groundwork for clinical management of patients with senile dementia.
However, this study has some limitations. Firstly, the risk factors in this study are not comprehensive enough, and there are still many risk factors for DSD in elderly patients during hospitalization, such as education level, marital status, and dementia type. Secondly, this is a single-center study with a relatively limited sample size. Finally, this study failed to explore the correlation between risk factors and the severity and duration of delirium. Therefore, future studies should further expand the sample size, include more risk factors, and observe the impact of high-risk factors on the incidence and long-term outcome of DSD patients.
In summary, this study found that diabetes, cerebrovascular disease, VAS score ≥4 points, use of sedative drugs, and blood SOD level <129 U/mL were independent risk factors for DSD in elderly patients. A prediction model was constructed using a nomogram, which showed high accuracy and discriminative ability. For elderly dementia patients with these high-risk factors, it is essential to strengthen the prevention and treatment of DSD.
Footnotes
Author Contributions
Conceptualization: Qifan Xiao
Data collection: Suqiao Zhang
Data analysis: Chenlu Li
Draft: Qifan Xiao
Writing – review & editing: Yuqing Zhu
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National High Level Hospital Clinical Research Funding (2023-NHLHCRF-YYPPLC-ZR-23).
Availability of Data and Materials
All the data within this manuscript could be gotten from corresponding author at reasonable request.
