Abstract
Alzheimer’s disease (AD) is a progressive neurological disease. Gut microbial dysbiosis is associated with AD. This study involves the comparative assessment of spatial learning, β-amyloid peptide accumulation, and fecal microbiota alterations in 3×Tg-AD mice from 3 age groups: AD asymptomatic stage (3 m), presymptomatic stage (6 m), and the symptomatic stage of AD (9 m). We demonstrate that spatial memory deficits, brain Aβ accumulation, and weight gain in 3×Tg-AD mice gradually appear after 6 months of age. However, the total gut bacterial counts underwent changes from 3 to 6 months of age and were further altered at 9 months of age. Importantly, changes in gut bacteria abundance of Desulfobacterota and Actinobacteriota phyla in 6-month-old mice preceded apparent spatial memory deficits. In summary, Changes in the gut microbial community are one of the mechanisms of early AD pathology.
Keywords
Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder and affects 50 million elderly people around the globe.1,2 In addition to cognitive decline and memory loss continue to exacerbate, patients with AD also exhibit abnormal glucose and lipid metabolism and thus may exhibit obesity due to metabolic dysfunction. 3 Accumulation and deposition of amyloid β (Aβ) peptide and neurofibrillary tangles are key pathological hallmarks of AD. However, the mechanisms that trigger AD and their chronological apparitions remain poorly understood and unresolved. 4 There is increasing evidence that the gut microbiota is involved in the pathogenesis of neurodegenerative diseases such as AD. 5 Disturbances in the brain-gut-microbiota axis disrupt bidirectional communication pathways between the CNS and the gastrointestinal tract, which contribute significantly to cognitive decline. 6
APP/PS1/Tau triple-transgenic (3×Tg) mouse models are a valuable resources for studying AD pathogenesis. They carrying familial AD mutations in the amyloid-β precursor protein (APP Swedish), presenilin1 (PSEN1 M146V) and tau (MAPT P301L) genes have been used as a standard AD models. 3×Tg-AD mice show age-related spatial memory deficiency compared to wild-type controls. 7 Histopathological changes of cerebral Aβ accumulation occur between 3-6 months of age, and there are no obvious behavioral deficit at this time.8,9 Hyperphosphorylated tau generally appears by later than 12 months old. 10 Finding the diagnostic biomarkers of AD is beneficial for AD early intervention. Changes in gut bacteria may be associated with cognitive decline in 3×Tg-AD mice. 11 However, The dynamic changes of gut microbiota composition in the asymptomatic, pre-symptomatic and symptomatic stages of AD in 3×Tg-AD mice have not been reported.
Given the important role of gut microbiota composition in AD pathology, the observed perturbation in the microbiota composition and diversity may serve as the mechanisms underlying age-dependent 3×Tg-AD mice amyloid deposition and memory deficits. Herein, we intended to investigate the gut microbiota and assessed its relationship with the triggering and development of cognitive impairment of AD. Our findings suggest that aging of 3×Tg-AD mice is accompanied by alterations in the gut microbiome temporally comparable to behavioral abnormalities. These results suggest that the 3×Tg-AD mouse is a useful model to study the behavioural and gut microbiota changes in AD, and modulation of gut bacterial composition may be a target for early intervention for AD prevention.
Materials and Methods
Transgenic Mice
3×Tg-AD mice [129-Psen1tm1Mpm Tg (APPswe, tauP301L) 1Lfa/Mmjax] and wild-type (WT) mice (B6129SF2/J) were purchased from Jackson Laboratory (stock no.34830-JAX), produced in SPF-grade animal house at Key Laboratory of Basic Pharmacology of Ministry of Education of Zunyi Medical University (Certificate no.: SYXK 2021-0003) and maintained at 22-23°C and relative humidity (60-70%) on a 12-h light/dark cycle. Drinking water and food were available ad libitum. 3-month-oldmale 3×Tg-AD mice (n = 8 mice per group) and age- and sex-matched nontransgenic litter-mate WT mice (n = 8 mice per group) were used in the present study. Animal weight and healthy were monitored throughout. The experimental design is shown in Figure 1A, the behavior test was performed in the WT and 3×Tg-AD mice at 3, 6, or 9 months, and the fecal samples were collected from 3×Tg-AD mice at 3, 6 and 9 months (n = 3 or 4 per group). All animal procedures were in accordance with the principles of the Declaration of Helsinki and were approved by Laboratory Animal Welfare and Ethics Committee of Zunyi Medical University (ZMU11-2203-468). Schematic diagram of experimental design for the indicated time points. (A) Fecal samples were collected from 3×Tg-AD mice at 3, 6 and 9 months of age, and (B) the study design of novel object localization test. The left and middle are the environment habituation and the location habituation stage respectively. The test trial was conducted 24 hours after the location habituation stage.
Novel Object Location Test
Behavioral studies were applied to evaluate spatial memory using the novel object location test in 3×Tg-AD mice at 3, 6, and 9 months old. Novel object location test consists of 3 sessions (Figure 1B), the mice were habituated to an object-free arenas (50 cm3 open plastic container) for 5 min 24 h prior to the location habituation training. During the location habituation session, the mice reentered the arenas and were granted freely exploring both identical objects to familiarize themselves with locations A and B for 5 min. 24 hours after the habituation period, object B was moved to a novel localization C, and mice were tested to explore both position objects within 5 min. The exploration behavior of the mice in both locations was recorded by an overhead video camera system (Topscan, CleverSys, Inc, Reston, VA, USA). Arenas and objects were cleaned with 70% ethanol between each mouse entry into the arena. Total time spent exploring each object location was recorded. The recognition index was calculated by the novel object location exploration time (TC)/total exploration time (TA + TC). The discrimination index was calculated by the time spent exploring the novel object than old objects (TC-TA)/total exploration time (TA + TC), as described. 12 After behavioral testing, the mice were euthanized and brain tissues were collected for analysis.
Immunohistochemistry Analysis
Brain sections were processed as specified before. 13 Briefly, mice were deeply anesthetized with sodium pentobarbital (100 mg/kg, i.p.) and transcardially perfused with cold phosphate buffered saline. Following, mice were decapitated and the brains were rapidly extracted and fixed with 4% paraformaldehyde (P1110, Solarbio, China). Transverse hippocampal slices were cut with a Vibroslice oscillating microslicer (RM2245, Leica). The brain tissue section on a slide glass was deparaffinized and antigen retrieved in .01 M citric acid. Non-specific binding of antibodies was blocked using goat serum, then incubated overnight at 4°C with the primary antibody mouse anti-Amyloid Fibrils antibody (1:500) (Novus; NBP1-97929). The brain section was rinsed in PBS for 30 min and then incubated with biotinylated goat anti-mouse IgG (H + L) secondary antibody (1:1000) for 2 h at room temperature. Following a series of washes in PBS and 30 min incubation in avidin-biotin peroxidase complex. Afterwards, the brain slice was visualized using a DAB detection kit (Solarbio, China). Digital images of Aβ load were acquired by an Olympus microscope (Olympus, Japan) and was quantified by Image J software.
Fecal Microbiota Analysis
When 3×Tg mice were 3, 6, and 9 months old, we collected daily fresh fecal samples from these mice for gut microbiota analysis. 7-days fecal pellets were collected from each mouse into a sterile polypropylene tube and frozen immediately in dry ice. These fecal samples were delivered to Beijing Novogene Bioinformatics Technology Co (Beijing, China) for 16S rRNA sequencing.
Bacterial DNA Isolation and Sequencing
Total genome DNA from fecal samples was extracted using the cetyltrimethylammonium bromide (CTAB) method. DNA concentration and purity was monitored by a nanodrop. Genome DNA was amplified using primers targeting the 16S V3 and V4 regions under the following conditions: 98°C for 1 min, followed by 30 cycles of denaturation at 98°C for 10 s, annealing at 50°C for 30 s, and elongation at 72°C for 30 s. Finally, 72°C for 5 min. Sequencing libraries were generated with the commercial stool TruSeq® DNA PCR-Free Sample Preparation Kit (Illumina, USA) according to manufacture instruction. The library quality was assessed using the Qubit 2.0 Fluorometer (Invitrogen Ltd, Paisley, UK). At last, DNA was extracted from fecal samples and 16S amplicons of the V3 and V4 region underwent 250 bp paired-end sequencing via an Illumina NovaSeq platform. Sequences analysis were performed by Uparse software (Uparse v7.0.1001). Sequences with ≥97% similarity were assigned to the same operational taxonomic units (OTUs) for further annotation.
Statistical Analysis
All data were presented as mean ± standard error. Two groups were compared by t test. Multiple group comparisons were analyzed by one-way with Tukey’s post hoc test. A two-way ANOVA with Bonferroni multiple comparisons post hoc test was used in the comparison of the multiple groups at different time points. Graphpad Prism 8 statistical software (GraphPad Inc, La Jolla, CA, USA) was used for data analysis. P < .05 was set as statistically significant.
Results
Age-Related Cognitive Deficits in 3×Tg Mice
Novel object location tests were performed to evaluate the spatial memory ability of the mice. After the environment and object location habituation, the location of B was changed to C. WT mice at 3, 6, and 9 months old showed increased interest to the new objective location C with comparable recognition index in the test trial (Figure 2). However, the recognition index for new object location was significantly decreased in the 6 and 9 months old AD mice. Analysis of the discrimination index, it is shown that the 9-month-old mice have significantly lower discrimination index than the WT mice. There were no significant differences in the speed of WT and 3×Tg mice in the 3 groups. These results suggest that 6-month-old 3×Tg-AD mice are unable to judge the novel locations, and the spatial memory tends to decline. While at 9 months of age, the mice performed worse on visual episodic memory, indicating that memory deficits in 3×Tg mice gradually developed at 6 to 9 months of age. Novel object localization test. The recognition index, discrimination index and speed of object-location A and C in the test trial in 3 months (A) 6 months (B) and 9 months (C) 3×Tg-AD mice. (D) Representative trajectory of 3×Tg-AD mouse in the test trial. A: familiar objective-location; C: novel objective-location. *P < .05 novel object-location C vs familiar object-location A analyzed by t test (n = 8 mice per group). Data were expressed as mean ± SEM.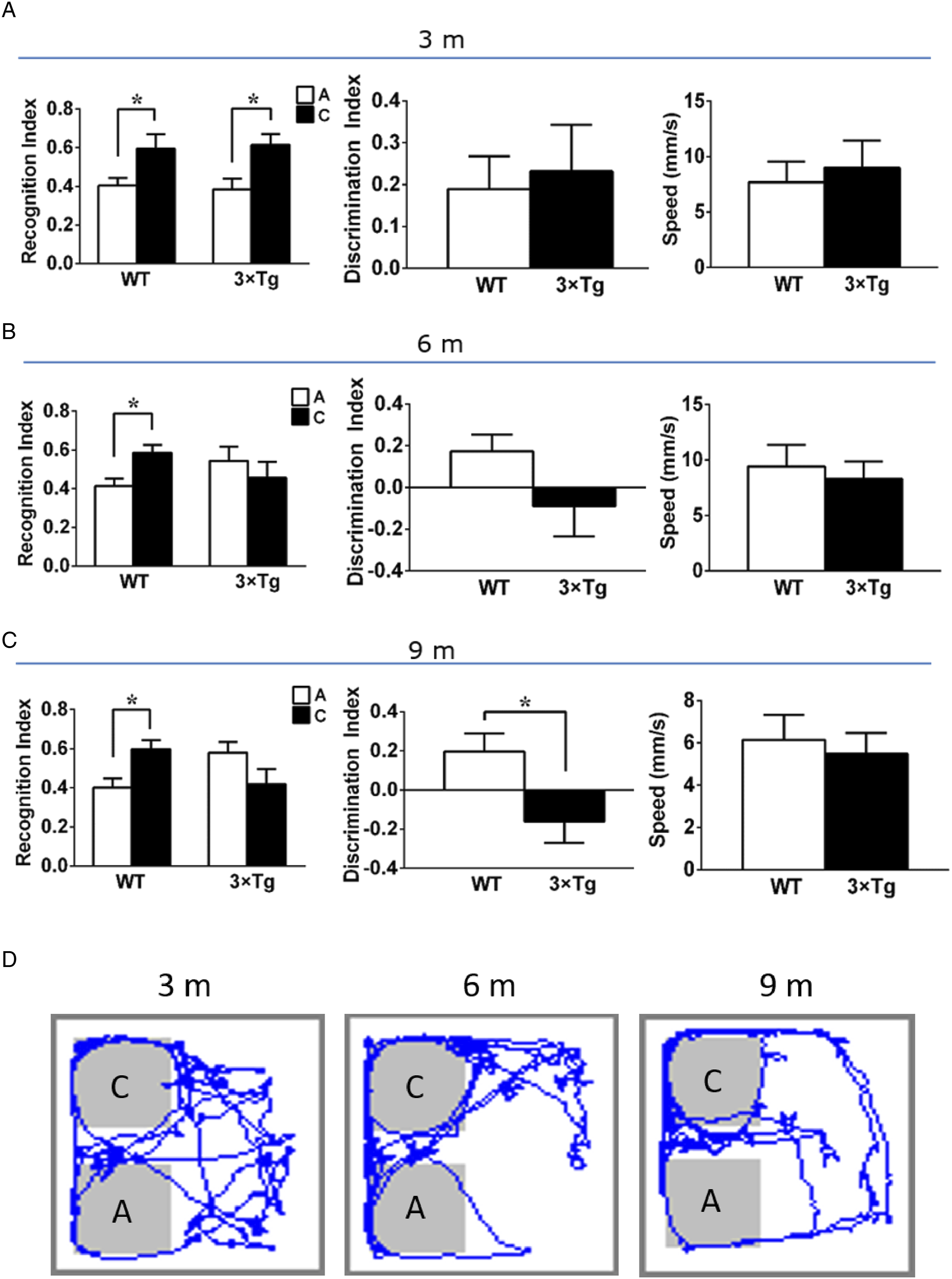
Formation of Amyloid Plaques in 3×Tg-AD Mice With Age
Amyloid-β toxicity is regarded as the initial and crucial event in AD development.
14
Amyloid plaques are formed mainly by aggregation of Aβ, which is one of the most characteristic pathological features of AD. Therefore, we evaluated the generation of Aβ deposition in brain regions implicated in memory formation, such as the hippocampus and cortex. Elevated Aβ fibril and fibrillar oligomer levels in 3×Tg-AD mice at 6 and 9 months were validated by specificity immunohistochemical (DAB) analysis (Figure 3). These results suggest that Aβ pathology emerges in the presymptomatic phase of AD in 3×Tg -AD mice, increasing with age. Representative images of Aβ deposition. (A) immunohistochemistry was obtained in the hippocampus and cortex of 3×Tg-AD mice at 3, 6 and 9 months old. (B) analysis of Aβ plaques in brain regions, *P < .05, analyzed by one-way ANOVA with Tukey’s post hoc test (n = 3 mice per group). Data were expressed as mean ± SEM.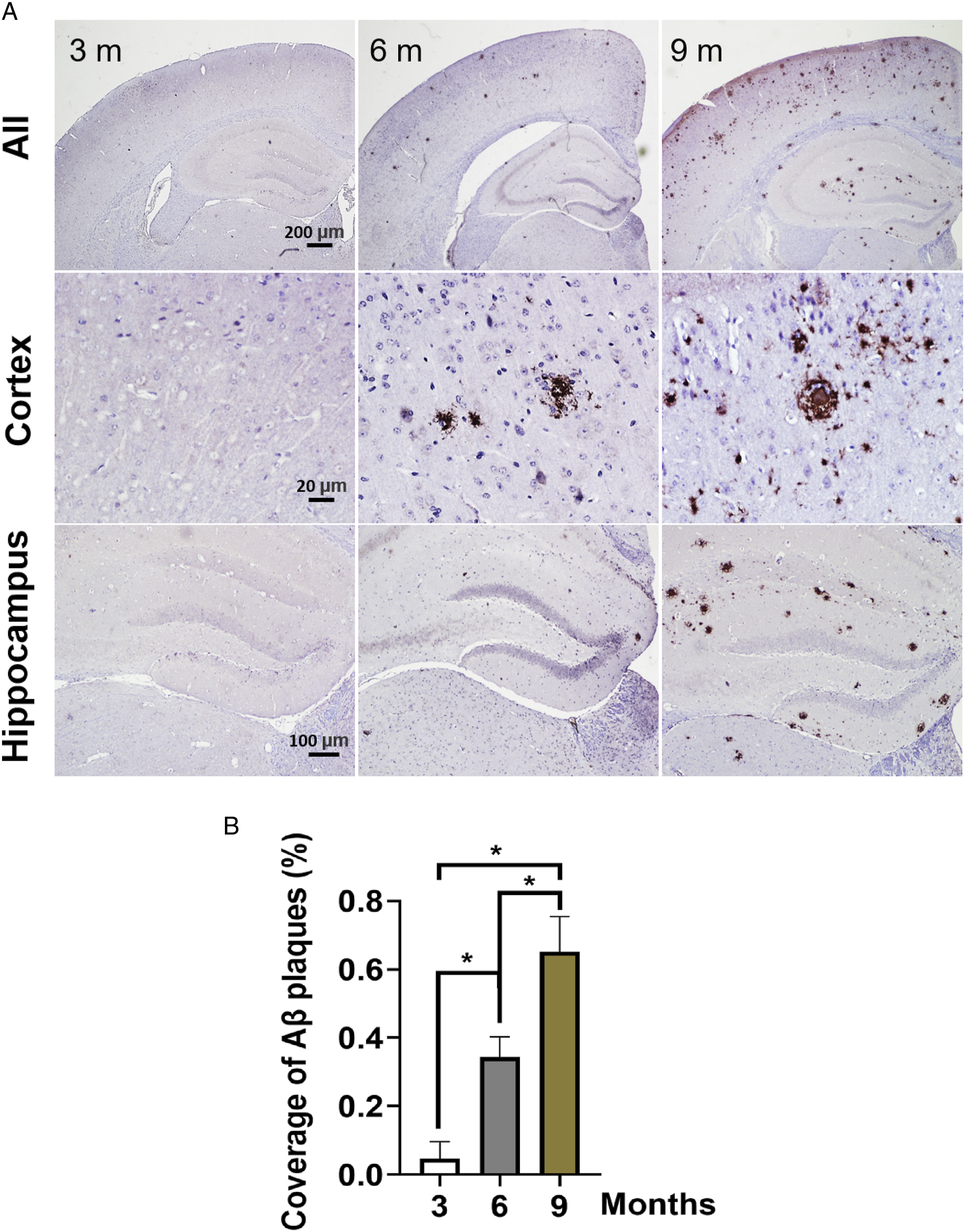
Body Weight in 3×Tg Mice of Different Months of Age
Microorganisms play important roles in host energy homeostasis through their symbiotic interactions. Gut microbiota dysbiosis causes imbalances in energy homeostasis that ultimately leads to obesity.
15
We monitored the body weight of the WT group and the 3×Tg-AD group over the whole course of this experiment. As shown in Figure 4, the values of body weight of 3×Tg mice from the mice were 5 to 9 months old were significantly higher than those in the WT group. Changes in body weight in 3×Tg-AD mice of 3 groups throughout the experiment. *P < .05 vs WT group, analyzed by two-way ANOVA with Bonferroni’s post hoc test (n = 8 mice per group).
Gut Microbiome Distribution Patterns Were Different in 3×Tg Mice of Different Months of Age
We first performed principal component analysis (PCoA) on all filtered genes from 16S rRNA sequencing to visualize distribution patterns. Figure 5 showed that 3 months (cyan), 6 months (blue), and 9 months (red) had little overlap in the distribution patterns. These results indicated that the fecal microbiota structure among the 3 groups of mice changed significantly with age. The distribution pattern analysis of fecal microbiota in 3×Tg-AD mice at 3, 6 and 9 months of age. M3. TG (green), M6. TG (blue), and M9. TG (red) indicate 3×Tg-AD mice at 3, 6, and 9 months of age, respectively. Each square representing a different individual mouse is colored according to the corresponding mouse age. An obvious clustering for each group was shown.
α-Diversity Analysis of Gut Microbiota
As shown in the Venn diagram of Figure 6A, 525 OTUs appeared in 3 groups. 884 OTUs overlapped in the 3 and 6 months groups, while 658 OTUs overlapped in the 3 and 9 months groups, suggesting that the diversity of bacteria decreased significantly with the age of 3×Tg AD mice from 6 to 9 months. Bacterial diversity in both 6- and 9-month-old was lower than that in 3-month-old mice. Furthermore, the observed bacterium species numbers, Chao1 richness, Ace, Simpson, PD-whole-tree, and Shannon, which are indicators of α-diversity, were estimated among the 3 groups, with the 3-month group being higher than that in either 6-month or 9-month group. As is shown in Figure 6B-G, the one-way ANOVA showed significant differences in the Shannon index (P = .043) and Simpson index (P = .013) for age factors in the 9-month group, which were decreased significantly compared with the 3-month group. These results mean that the microbial diversity decreases as AD disease progresses in 3×Tg mice.

Analysis diversity of fecal microbiota community in 3×Tg-AD mice at 3, 6 and 9 months of age. (A) Venn diagram. M3. TG (green), M6. TG (blue), and M9. TG (red) indicate 3×Tg-AD mice at 3, 6, and 9 months of age, respectively. (B-G) Analysis α-diversity of the diferential microbial community among the 3 groups. *P < .05, vs 3-month group, analyzed by one-way ANOVA with Tukey’s post hoc test. (n = 3 or 4 mice per group).
Relative Abundance of Gut Microbiota at the Phyla Level
We analyzed the fecal flora sequences of 3×Tg-AD mice at 3, 6, and 9 months of age at the phylum level. As shown in Figure 7, the characterization of the fecal bacterial diversity relative abundance of 10 major phyla among the 3 groups in individual samples. From 3 to 9 months of age, there was a decrease in the abundance of Actinobacteriota (.9%, .7%, .6% from 3 to 9 months), Verrucomicrobiota (.8%, .3%, .0%) and Acidobacteriota (.4%, .2%, .1%). A decrease was observed for Bacteroidota (36.4% to 34.7% from 3 to 6 months), Deferribacteres (.4% to .1% from 3 to 6 months), Campylobacterota (1.4% to .7% from 3 to 6 months), and Proteobacteria (.7% to .3% from 3 to 6 months) in 3×Tg-AD mice from 3 to 6 months of age, while at 9 months old, a highest abundance of these phyla were detected (51.6%, 6.6%, 3.2% and 1.8% for Bacteroidota, Deferribacteres, Campylobacterota and Proteobacteria, respectively). Firmicutes were more abundant in 6 months old 3×Tg- AD mice (52.2%) than 3 months (40.8%) or 9 months (32.6%). Similarly, Desulfobacterota were more abundant in 6 months (1.7%) vs 3 months (1.1%) or 9 months (.5%). Structural comparison of fecal microbiota at phyla levels in 3×Tg-AD mice at 3, 6 and 9 months old. Firmicutes, Bacteroidota, Deferribacteres, Campylobacterota, Desulfobacterota, Proteobacteria, Actinobacteriota, Verrucomicrobiota and Acidobacteriota are displayed in column charts by tag colors at the right side of the figure. The Y-axis indicates the relative abundances of each phylum.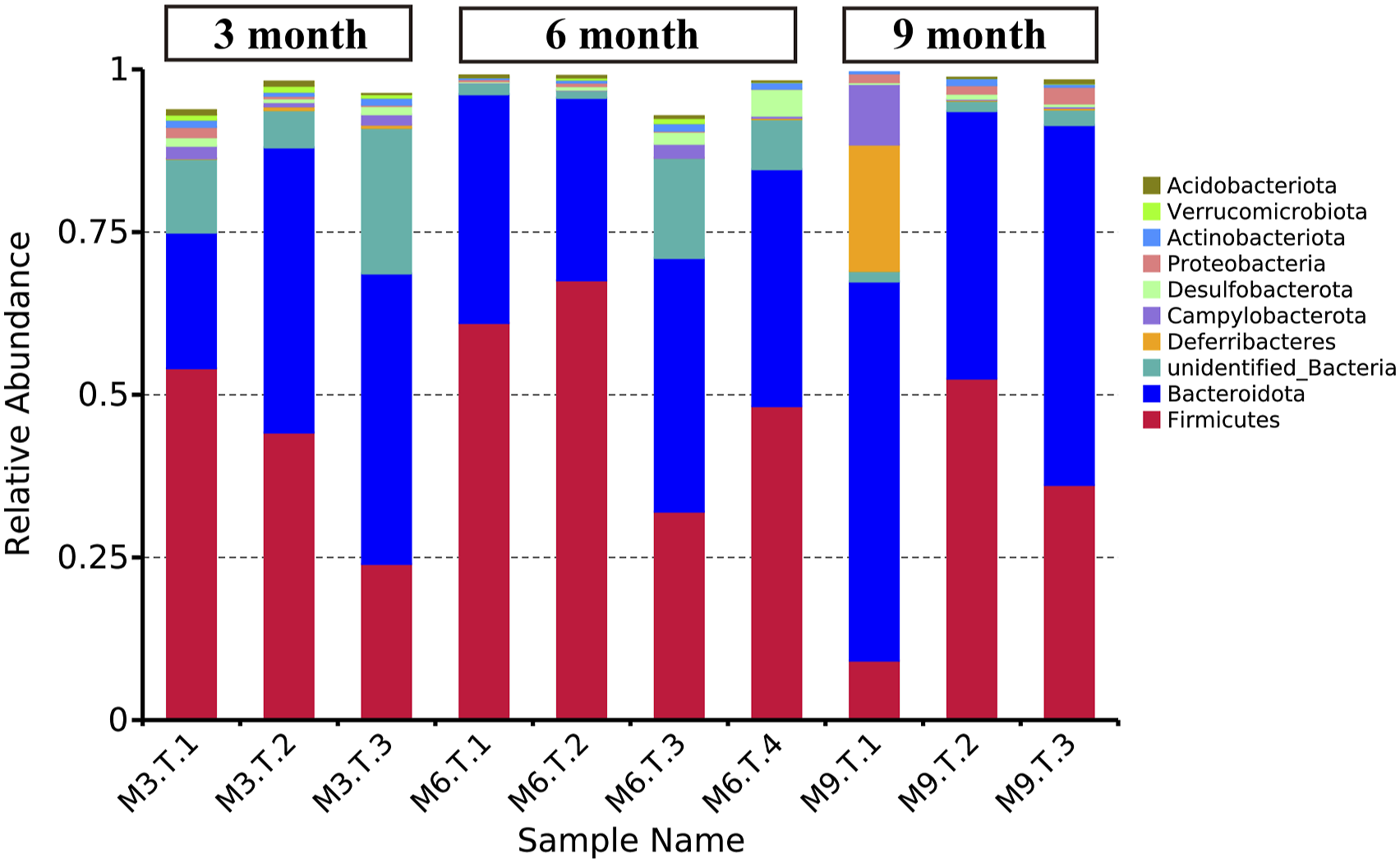
Bacteria With Statistically Significant Changes for Each Age Group by 16S DNA Sequencing Analysis
The cladogram generated from the linear discriminant analysis effect size (LEfSe) analysis, revealed distinct fecal microbiota compositions among mice from 3 groups in the order of phylum (p_), class (c_), order (o_), family (f_), genus (g_) (Figure 8A and B). For 3-month-old 3×Tg-AD mice (red), the abundance was in f_Planococcaceae, o_Acidobacteriales, c_Acidobacteriae. 6-month-old 3×Tg-AD mice (green) affected the abundance of o_unidentified_TK10. While 9-month-old 3×Tg-AD mice (blue) mainly affected the abundance of o_Corynebacteriales, f_Vagococcaceae, f_Enterococcaceae, f_Veillonellaceae, o_Acetobacterales, c_Gammaproteobacteria. These results mean that the relative bacterial abundances may have great changes in 3×Tg-AD mice of different months of age. Bacteria with a statistically significant change in 3×Tg-AD mice at 3, 6 and 9 months old. (A) Genus evolutionary tree. LEfSe analysis and cladogram of fecal fora among the 3 groups. (B) Linear discriminant analysis (LDA) coupled with effect size measurements identifies the most differentially abundant taxons in each age group. Only taxa meeting an LDA significant threshold >2 are shown.
Discussion
This longitudinal study examines the gut microbiota changes related to spatial memory in APP/PS1/Tau (3×Tg) AD transgenic mouse models at 3, 6, and 9 months of age. In our results, 3×Tg-AD mice promoted the pathogenesis of AD with age, as evidenced by memory deficits at 6 and 9 months. Furthermore, deposition of Aβ, a typical AD pathologic hallmark, was observed in the brain of 3×Tg-AD mice at 6 and 9 months old. Importantly, we showed that gut microbiota changes in 3×Tg-AD mice at 3, 6, and 9 months of age have been proposed to facilitate behavioural alterations. And this change precedes obvious behavioral and pathological changes. That is, the imbalance of gut bacteria homeostasis may be an early change in AD symptoms.
In the behavior test, we evaluated the spatial memory of mice by recognizing that objects in familiar locations are replaced by new locations and found that a progressive memory impairment increases with age in 3×Tg-AD mice. Compared with WT mice in the 6 and 9 months groups, 3×Tg-AD mice of the same age did not show increased interest in the new object location with comparable recognition index in familiar or novel object location. When the mice at 9 months old, the discrimination index of the 3×Tg-AD mice to the novel object location was significantly reduced. In addition, there was no difference in the speed of the mice in each group, indicating that the exploratory behavior of mice was not affected by motor activity. These results could be explained by previous reports that 3×Tg-AD mice at 3 months and 6 months were considered in pre-symptomatic stage and early symptomatic stage of AD pathology. During this time, cognitive deficit have not yet manifested. The present study demonstrated that 9-month- old 3×Tg-AD mice showed AD symptoms and developed significant spatial memory deficits.
It has been elucidated that the perirhinal cortex and dorsal hippocampus are critical brain areas for spatial recognition memory.16-18 All AD patients undergo progressive Aβ deposition in brain regions serving cognition and memory. Aβ overproduction in the cortex and hippocampus area triggers a range of AD neuropathological cascades, including tau filament formation, neuroinflammation, oxidative stress and ultimately the causation of neural death. Aβ peptide is a series of products cleaved from the amyloid precursor protein (APP) via the amyloidogenic pathway. A bit of aggregation-prone, insoluble Aβ aggregates into a core and spreads, serving as a template to facilitate oligomerization and fibration of other soluble Aβ, eventually leading to the deposition of senile plaques. 19 In the present study, the area of Aβ-containing plaques was significantly increased in 6- and 9- month-old 3×Tg-AD mice brains, which could be related to the memory impairment observed in 6- and 9-month-old mice. Recent studies have proposed that Aβ has been regarded as the immune protective response of the body. It is believed that Aβ production also occurs in the intestine, 20 and modulation of gut microbiota could affect cerebral amyloid deposition.21,22 Gut microbiota may play an important roles in the immune homeostasis of the brain, and dysbiosis or its products, such as amyloid and bacterial endotoxin LPS disrupt intestinal epithelial barrier integrity and blood-brain barrier, as well as cause intestinal immune system activation. 23 On the one hand, this process leads to metabolic disorders.24,25 Obesity caused by insulin resistance or dyslipidemia could increase the risk of AD. 26 On the other hand, gut microbiota composition dysbiosis have been variously associated with neurocognitive disorders such as AD, schizophrenia and depression. 27 Additionally, gut dysbiosis could also accelerates learning and memory impairments due to the transfer of toxic substances of intestinal origin from the gastrointestinal tract to the CNS.28,29 In the present study, 3×Tg-AD mice gained weight faster than age-matched WT mice, and accompanied by accumulation of β-amyloid peptides in the hippocampus and cerebral cortex (Figures 3 and 4). In summary, 3-month-old 3×Tg-AD mice were in a normal state, whereas 6-month-old mice were in the presymptomatic stage of AD onset, and 9-month-old mice showed obvious AD symptoms, which is consistent with previous literature. 30 Gut microbiota dysbiosis and its actual mechanism and association with the onset and development of AD is still elusive. 3 However, previous studies have demonstrated that 3 major divisions, the Firmicute, Bacteroides and Actinobacteria make up the major bacteria of the feces in adult humans, 31 as observed in our results. At the phyla level, aging has been reported to alter the gut microbiota composition, such as increasing the abundance of Bacteroides, Bifidobacterium and Proteobacteria, while Firmicutes is decreased. 32 Shen et al revealed 33 that APP/PS1 transgenic murine model showed increased Desulfovibrionaceae, Odoribacter and Helicobacter in comparison to wild-type mice. Additionally, proinflammatory bacteria, such as Proteobacteria, were found to be more abundant in AD and MCI (mild cognitive impairment) patients and several AD animal models.34,35 These studies agree with our found, where we documented that Bacteroidota and Firmicutes represent the major bacteria in the gut of 3×Tg-AD mice, while Bacteroidota increased and Firmicutes decreased with the progression of AD. Meanwhile, the proportion of Proteobacteria also exhibits a significant growth in 9-month-old 3×Tg-AD mice. Desulfobacterota level progressive enhancement was observed in 6-month-old 3×Tg-AD mice compared to 3- months-old mice. We found that the abundance of Actinobacteriota decreased gradually from .9%, .7%, and .6% in 3×Tg-AD mice at 3, 6, and 9 months old. Noticeably, the phylum Actinobacteriota has been used as an anti-inflammatory indicator and is highly related to AD. 36 These results suggest that these changes in the gut microbiota composition may already be signs of triggering symptoms of AD before the apparent spatial memory deficits in AD mice.
A reduction in microbial diversity has been reported in a microbiome study of human patients with AD. 37 In the present study, by comparing the species of microorganisms present in the fecal samples, age-related species diversity profiles in the 3×Tg-AD mice were revealed. We analyzed α-diversity in the gut microbiota and found that there were no significant differences between 3×Tg-AD mice at 3 and 6 months old. This could be because α-diversity may not be a sensitive marker of early AD development. However, α-Diversity analysis showed an overall decrease in bacterial diversity in 9-month-old 3×Tg-AD mice. Shannon and Simpson indexes in 9-month-old mice could significantly decrease in comparison with 3×Tg-AD mice at 9 months old. It is worth mentioning that the results of PCoA indicated a variation tendency to change the structures of the main microbial composition (Figure 5). However, the total bacterial count was found to be lower in 6- and 9-month-old mice than in 3-month-old mice (Figure 6A).
In summary, the present study demonstrated that AD pathology might not only affect brain function and exacerbate spatial memory deficits in the 3×Tg-AD mouse model, but also produced age-related alterations in the gut microbiota community. Alteration of the gut microbial community is at least one of the mechanisms by which early AD pathology occurs, and modulation of gut bacterial composition could be considered and used as a target for early intervention for AD prevention. However, the causal association between neurodegeneration and gut microbial community requires further elaboration.
Supplemental Material
Supplemental Material - Alterations of Spatial Memory and Gut Microbiota Composition in Alzheimer’s Disease Triple-Transgenic Mice at 3, 6, and 9 Months of Age
Supplemental Material for Alterations of Spatial Memory and Gut Microbiota Composition in Alzheimer’s Disease Triple-Transgenic Mice at 3, 6, and 9 Months of Age by Zhang Wei, Daidi Li, and Jingshan Shi in American Journal of Alzheimer’s Disease & Other Dementias®.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Construction of National First Class Pharmacy Discipline, No. GESR-2017-85 (to JSS); National Natural Science Foundation of China, No: 81773739 and No. 82060728, Shijingshan’s Tutor Studio of Pharmacology, No. GZS-2016-07 and The Medical Scientiic Research Foundation of Guangdong Province (A2020228).
Data Availability
Data is available from corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
