Abstract
Dementia with Lewy bodies (DLB) is a major neurocognitive disorder (MNCD) that is primarily characterized by motor, cognitive, and behavioral symptoms. Although not dominant in the clinical portrait of DLB, impairments affecting language processing have been reported. It is sometimes challenging to differentiate DLB from Alzheimer’s disease and dementia associated with Parkinson’s disease in clinical practice. Therefore, a better comprehension of the typical clinical presentation of DLB may be useful to ease the medical diagnosis. In this article, current data on cognitive and language disorders in DLB are reported, with special attention paid to their primary or secondary functional origin. The main elements that should be considered for the neuropsychological and speech-language assessment of individuals with possible or proven DLB are also highlighted. Additional studies are needed, especially for language impairment, to obtain a more accurate portrait of the clinical presentation of DLB and characterize its progression.
Significance Statement
• Dementia with Lewy bodies (DLB) is primarily characterized by motor, cognitive, and behavioral symptoms. • The cognitive profile of DLB is mainly characterized by impairments affecting attention, executive functions, visuoperceptual abilities, and episodic memory. • Language impairments affecting word and sentence comprehension, word production, spontaneous speech and reading have also been reported. • Most of cognitive impairments in DLB have a primary origin, while language impairments mainly result from deficits of semantic memory, visuoperceptual abilities and executive functions.
Introduction
In industrialized countries, such as France and Canada, low birth rates and increasing life expectancy are leading to the general aging of the population. For example, according to Statistics Canada, 1 the number of people aged 65 and over has increased significantly in recent years, and this group represented about 18.5% of the Canadian population in 2021. Due to the increase in life expectancy, the number of centenarians more than tripled from 2001 to 2021, increasing from 3522 to 12 822. 1 This demographic phenomenon has been accompanied by major changes in the workforce, the economy, and the management of health care systems. Moreover, the aging of the population has induced a drastic increase in the incidence of age-related diseases, including major neurocognitive disorders (MNCD). 2 This new terminology of dementia, derived from the DSM-V, 3 refers to the acquired, significant, and progressive reduction of abilities in one or more cognitive domains that are significant enough to affect activities of daily living.
Major neurocognitive disorder typically progresses through 3 distinct phases: (1) a pre-clinical phase in which individuals can be placed on a continuum from “completely asymptomatic” to a very subtle decline that is known as “subjective cognitive decline” or “subjective cognitive impairment”; (2) mild cognitive impairment (MCI), which is the symptomatic pre-MNCD phase and is characterized by impairment in memory or other areas of cognition; and (3) MNCD.4,5 Furthermore, MNCD is a common condition that occurs primarily in older people. It is characterized by a significant decline in cognitive functioning that is severe enough to impact activities of daily living and social functioning. Cognitive impairment associated with MNCD can affect long- and short-term memory, attention, visual perception, executive functions, motor planning and execution, problem solving, and language.
Major neurocognitive disorder can be caused by a wide range of disease entities, of which Alzheimer’s disease is the most common. Less common neurological diseases, including vascular dementia; dementia associated with alpha-synucleinopathies, such as dementia with Lewy bodies (DLB); tauopathies, such as corticobasal degeneration; multiple sclerosis; and Huntington’s disease, can also cause MNCD. Even if language disorders are not among the dominant clinical manifestations of these diseases, several studies have shown that language abilities are far from fully preserved and therefore deserve special attention in clinical practice. 6 The initial assessment is the first significant step toward the clinical management of MNCD and is based on consensus diagnostic criteria.
The main objective of this article was to make an update on the cognitive and language disorders associated with DLB, with a particular focus on their functional origin. A more general objective was to address the potential contribution of the neuropsychological and speech-language assessment to the differential diagnosis of DLB. To enables readers to establish links between the medical and the cognitive/linguistic symptoms, the diagnostic criteria for DLB will be first presented.
Clinical Characteristics of DLB
Neuropathologically, DLB is characterized by inclusions of ubiquitinated, misfolded α-synuclein in neurons in the neocortex, the midbrain, and the limbic system. These inclusions, which are called Lewy bodies, are also typical features of Parkinson’s disease and Parkinson’s disease with dementia. 7
Lewy body dementias consist of DLB and Parkinson’s disease dementia. Dementia with Lewy bodies is second most prevalent primary dementia syndrome in older people after Alzheimer’s disease. Its incidence is approximately 3% to 7% of all dementia cases. 8 Dementia with Lewy bodies differs from dementia associated with Parkinson’s disease in the sequence of onset of cognitive and motor symptoms. 9 Parkinson’s disease is typically characterized by a period of quasi exclusive motor symptoms before significant cognitive decline and the emergence of dementia in up to 80% of patients. By contrast, the onset of DLB is typically characterized by cognitive impairment, accompanied, or followed by motor symptoms. 10
In 2017, McKeith et al proposed a major revision of the diagnostic criteria for DLB, which had been established in 2005.11,12 They specified that the essential feature for the diagnosis of DLB was the presence of dementia, which they defined as a progressive cognitive decline of sufficient magnitude to interfere with normal social or occupational functions; the presence of memory impairment; and cognitive deficits in attentional, executive, and visuoperceptual abilities.
McKeith et al.’s criteria also included core features of DLB (ie, fluctuating cognitive functioning, recurrent visual hallucinations, REM sleep behavior disorder, parkinsonism symptoms), features supporting the diagnosis (eg, severe sensitivity to antipsychotic agents, postural instability, repeated falls), biomarkers indicative of the disease (eg, abnormal myocardial scintigraphy, polysomnographic confirmation of REM sleep without atonia), and biomarkers congruent with DLB (eg, relative preservation of medial temporal lobe structures on MRI or CT). 11
In their diagnostic guide, McKeith et al differentiated criteria for possible DLB and probable DLB. According to them, a diagnosis of DLB is probable if: (a) two or more core clinical features of DLB are present, with or without the presence of indicative biomarkers, or (b) only one core clinical feature is present, but with one or more indicative biomarkers. A diagnosis of DLB is however less likely (possible DLB): (a) if any other physical disease or brain disorder, including cerebrovascular disease, that could explain all or part of the clinical picture is present or (b) if parkinsonian features are central to the clinical picture and first appear in a severe dementia stage. 11
Neuroanatomically, neuroimaging studies have revealed abnormalities or atrophy in the basal nuclei and the occipital lobe with relative preservation of the posterior cingulate cortex, while medial temporal structures, including the hippocampus, are generally better preserved.13-15 However, other studies have found pathology in the hippocampus in DLB, although affecting different subfields than in Alzheimer’s disease. 16
Cognitive Impairments in DLB
Main Characteristics of Cognitive Disorders in DLB.

The involvement of the basal nuclei and their connective circuits with the cortex and the thalamus in attentional processes is well demonstrated. 22 The disruption of attentional processes has been proposed to underlie the cognitive fluctuations frequently found in DLB. 23 These subcortical nuclei, which are affected in DLB, also play an important role in executive functions, particularly for decision making, planning, mental flexibility, and working memory. 24 It is therefore not surprising that disorders affecting attentional and executive abilities are central to the cognitive symptomatology of DLB. 17 Attentional disorders can affect sustained attention and selective attention skills. 25 Executive disorders, which may appear several years before a diagnosis of DLB is made, can affect divided attention as well as inhibition and mental flexibility abilities.19,26,27
The occipital lobe is functionally devoted to visual object recognition, and its pathology in DLB is responsible for predominant visuoperceptual impairment.28,29 Basic visual-perceptual abilities (ie, analyzing visual features of objects, such as their orientation and light contrasts) are usually preserved in the early stage of DLB, whereas intermediate (eg, motion processing, color processing) and high (ie, integration of visual information into a coherent whole, object recognition) abilities are affected early in the disease, and even in its prodromic stage.30-33 Difficulty performing more complex aspects of visual search, such as feature binding is also compromised early in the course of DLB. 34 Finally, studies also showed the presence of impairment of visuoconstructive abilities in DLB, even in the prodromal DLB stage, likely due to perceptual difficulties.35,36 The contribution of visuoperceptual impairment to the development of visual hallucinations in DLB has also been proposed. 29 In a recent study, Pezzoli et al. showed that visual hallucinations were associated not only with visuoperceptual impairment in DLB but also with attention and memory deficits. 37 Hypometabolism in the orbitofrontal cortex on 18F-FDG PET in DLB was also reported. 38 This cortical structure is known to be involved in the human olfactory system. 39 Disorders of odor perception are also part of the clinical features of DLB. These impairments may manifest as difficulties in identifying or remembering odors. 40
Many studies have found episodic deficits in DLB, although they have been found to be less severe than in Alzheimer’s disease. 41 A relationship between these deficits and the tau pathology found in specific hippocampal subfields of patients with DLB has been suggested. 15 Moreover, the sensitivity to cueing in free recall tasks suggests that the functional origin of the memory impairments in DLB is also tied to executive dysfunction. 9 Other comparative studies have shown that people with DLB generally performed better than those with Alzheimer’s disease in verbal episodic memory tests, while their results were comparable in tests administered with visual material due to their visual-perceptual impairment.42,43
Finally, semantic memory seems to be just as affected in DLB as in Alzheimer’s disease when the tests are administered in the verbal modality, whereas performance is more affected in DLB in the visual modality because of the associated visual-perceptual impairments.44,45
Language Impairments in DLB
Main Characteristics of Language disorders in DLB.
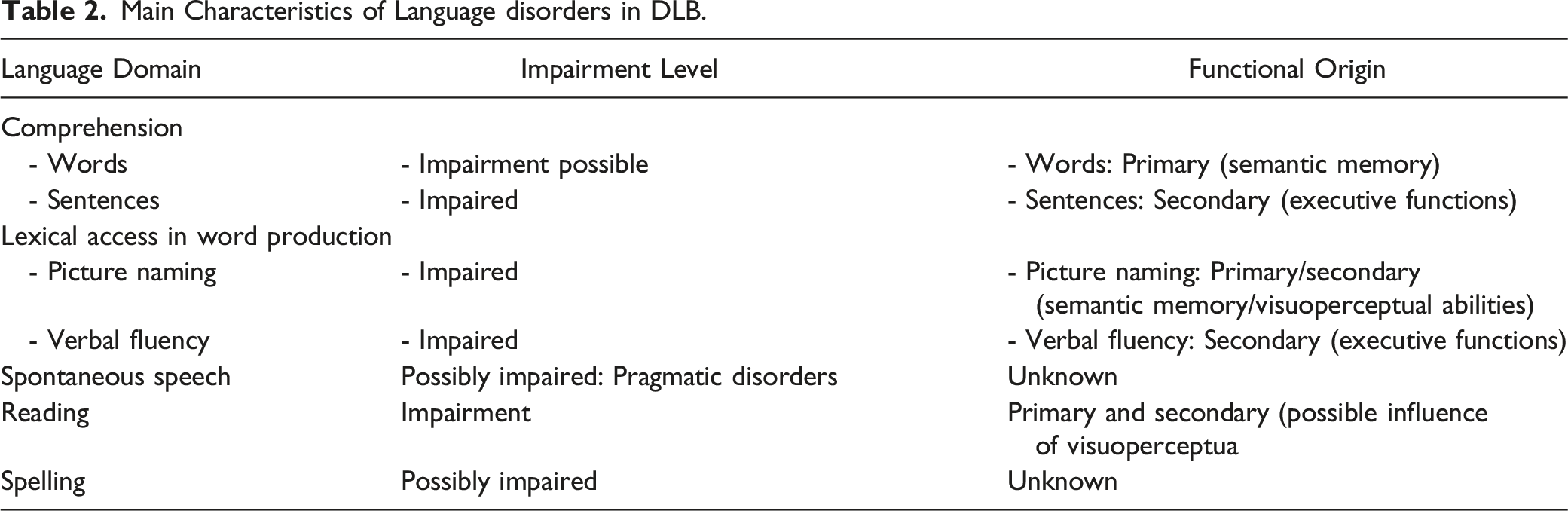
Individuals with DLB may also exhibit syntactic impairments in sentence comprehension tasks, which is a deficit typically attributed to associated disorders affecting their working memory and executive functions.48,49 A similar executive origin has been proposed to explain their difficulty in organizing narrative discourse in both language production and comprehension.50,51
Furthermore, to the best of our knowledge, no studies have specifically examined reading abilities in DLB. However, when reading tasks are included in the baseline cognitive assessment of DLB, peripheral impairments resulting from visual-perceptual impairment have been noted. 52 There is also very limited knowledge about written production. The authors of a few studies have observed more significant writing disorders in DLB than in Alzheimer’s disease but did not describe their nature.52,53 In a recent study of 17 people with prodromal to mild DLB, the authors showed the very early presence of spelling disorders, mainly affecting the written production of non-words and sentences. 54 According to the authors of this study, the errors found in DLB are attributable to the attentional and executive deficits associated with the disease.
Clinical Assessment of Cognitive and Language Functions in DLB
Recommendations for Assessment of Cognitive and Language Functions in DLB.

Given the nature of the disorders observed, the contribution of speech-language pathology assessment to the differential diagnosis of DLB is very limited. Medically, this diagnosis is primarily focused on differentiating DLB from Alzheimer’s disease. However, there is a significant overlap in the linguistic symptoms seen in the 2 neurodegenerative syndromes. Therefore, speech-language pathologists involved in the diagnostic process will have to pay more attention to the clinical signs that are specific to them, such as the early semantic and transmodal impairment in Alzheimer’s disease; the important executive contribution to expressive and receptive language disorders in DLB; and the important impacts of visual-perceptual disorders in the performance of tasks involving pictographic material in DLB. This assessment must be based on data from the scientific literature and focus on language disorders secondary to the impairment of attentional and executive capacities. In this respect, tasks involving the comprehension and production of syntactically complex sentences, verbal fluency by semantic or orthographic criteria, and narrative or descriptive discourse should be favored. Comparing performance on naming tasks in visual (pictures) and verbal (definition-based naming) modalities could also help identify the primary or secondary (executive, perceptual) origin of a deficit affecting lexical-semantic access.
Conclusions
In this article, the cognitive and language impairments accompanying DLB have been briefly reviewed. Some of these impairments have been repeatedly reported, while others have been studied only in few articles in which a comprehensive and specific approach was used. That doesn’t necessarily mean that these symptoms are less frequent, but certainly that they are less studied. The novelty of this article lies in the more systematic specification of the functional origin of cognitive and language impairments in DLB, as well as in the recommendations for their clinical assessment.
These impairments may occur early in the disease process and can have a primary or a secondary origin. As presented in this article (see Table 2 for a summary), it remains difficult to rule on the primary or secondary origin of language deficits in DLB based on the existing literature. However, there can be no doubt that language and cognitive impairments are linked in DLB. 58 The nature and characteristics of the impairments affecting cognitive functions are relatively well established in the scientific literature. In contrast, the data on the deficits affecting the production and comprehension of language is less well developed. For example, the impact of language impairments on spontaneous speech or narrative discourse still needs to be better explored and described. As reported in this article, specific linguistic abilities, such as reading and writing, are almost completely ignored in studies on DLB.
The publication of consensus diagnostic criteria for the major forms of MNCD increasingly includes typical characteristics of cognitive and language impairments. Going a step further in the identification of cognitive and language markers of MNCD will require studies in which more specific subcomponents of the different cognitive and linguistic domains are explored. Further research is also needed to better characterize the effects of the progression of DLB on cognitive and language abilities. Overall, a better characterization of cognitive and language deficits in DLB would be very useful for developing behavioral treatment approaches as well as for counseling and educating patients, families, and caregivers.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board Statement
Not applicable
