Abstract
Lifestyle factors may individually protect against the development of mild cognitive impairment. We investigate the relationships between both self-reported physical activity and measured physical function with cognition in a population of elderly adults, more than half of whom follow vegetarian dietary patterns. Otherwise healthy adults (n = 127, mean age 74.9 ± 7.9 years, 61.3% current vegetarians) were assessed using a comprehensive neuropsychological battery. A principal components analysis derived processing speed, executive function, and memory/language factors. Participants reported current levels of vigorous physical activity on questionnaires, and physical function and mobility were measured with the Physical Performance Test (PPT) and Timed Up and Go (TUG) Test. Generalized linear models estimated β coefficients for cross-sectional associations between cognitive factors and indicators of physical abilities and self-reported physical activity. Better physical function indicated by PPT was associated with higher scores on the processing speed factor (β = 0.21 SDs for each 4.4-point increase in PPT score; p = 0.02). Faster TUG times were also associated with higher processing speed factor scores (β = 0.21 SDs increase for each 2.8 second less TUG time; p = 0.02). Self-reported levels of vigorous physical activity were not associated with any area of cognitive function; the association between PPT, TUG and processing speed was independent of physical activity. Associations between PPT and TUG and processing speed were stronger among participants who followed vegetarian dietary patterns. Better physical function may have an effect on cognition in a context of healthy lifestyles.
Significance
Among elderly adults who adhere to vegetarian diets, better physical functioning is related to better cognitive function. The notion that a “comprehensive” healthy lifestyle, encompassing more than one domain of health-promoting behaviors, should be further investigated.
Introduction
Mild cognitive impairment (MCI), a heterogeneous intermediary spectrum of cognitive dysfunction between normal cognition and dementia, 1,2 affects 16-20% of US adults aged 60 and older, with prevalence estimates varying based on diagnostic criteria used and how criteria are operationalized. 1,3 -6 Individuals with MCI experience deficits in memory and or other cognitive domains beyond normal age-associated cognitive changes. While those deficits are not severe enough to significantly impact their ability to function independently as with dementia, 2,7,8 individuals with MCI are at a higher risk for Alzheimer’s disease (AD), 9 and progress to dementia at a rate of 10-15% per year. 1,4,10 As such, cognitively impaired persons require nursing home care at twice the rate of cognitively intact persons and incur significantly greater healthcare costs. 11,12 Despite the fact that age increases the risk of MCI, substantial cognitive decline is not believed to be an inevitable consequence of aging, but instead reflects underlying diseases or conditions. 4,13 While substantial investment in drug development has been made, no medication is currently available to effectively treat the pathogenic substrates thought to underlie MCI. Thus, identifying alternative approaches to prevent or delay the onset of cognitive impairment (and dementia) is of significant interest.
A number of population-based observational and experimental studies suggest that in addition to greater education, lifestyle factors may individually protect against the development of MCI, including participating in cognitively stimulating activities, being physically active, consuming a diet high in mono- and poly-unsaturated fatty acids and following the Mediterranean diet. 14 -23 Diet is strongly associated with cardiovascular disease (CVD), 24 -27 and CVD in turn is associated with cognitive dysfunction. 28 Having a physically active lifestyle which could lead to greater physical fitness, may protect the aging brain by improving cerebral blood flow, increasing cortical and hippocampal volumes, and improving synaptic plasticity and neurogenesis. 29 -31 Few studies have examined the effect of more than one “healthy” lifestyle factors on cognitive function in the elderly, such as the combination of a healthy diet and physical activity. 32,33 Furthermore, studies have utilized self-reports of physical activity and exercise, 17 -19,34 and others have included evaluations of physical function, 35,36 attributes that indicate the ability to perform everyday physical tasks. 37 While these two factors (frequency of activity/exercise and functional abilities) are related, they nevertheless represent distinct constructs that may be associated with cognitive function through different mechanisms. Physical activity has been shown to improve cerebrovascular health which, in turn, can lead to improved cognitive function, 29 -31 whereas performance on tasks of physical function may, at least in part, reflect efficiency of central nervous system networks involved in completing activities of daily living. 38 Thus, we examine cross-sectional associations between both self-reported physical activity and measured physical function and different areas of cognition and memory impairment in a population of elderly adults for whom more than half follow vegetarian dietary patterns.
Materials and Methods
Study Population: Adventist Health Study-2 (AHS-2) Cohort
The AHS-2 is a prospective cohort study of over 96,000 Seventh-day Adventists in the US and Canada, which was originally established in 2002 to investigate the role of various foods and nutrients, lifestyle factors and metabolic risk indicators in cancer causation. During 2002-2007, Caucasian (65.3%) and African-American (26.9%) adult men and women with a mean age of 59 years (range 30-110 years) were enrolled in the AHS-2 cohort, and completed a 50-page baseline questionnaire which included sections on demographic, dietary, anthropometric and lifestyle factors. 39 Participants are mailed annual study newsletters and followed-up with biennial questionnaires. The cohort is healthy: at baseline, high proportions reported being in excellent health; 45% of cohort members follow vegetarian diets, 40 the non-vegetarians consume relatively lower amounts of meat compared to the general population, 40,41 only 1.1% are current smokers and 6.6% currently drink alcohol. 39 Since the cohort has aged and the majority of the cohort is elderly, the AHS-2 presents an opportunity to study additional age-related (non-cancer) chronic diseases.
In 2016, we identified 2,685 members of the cohort for whom study records indicated were 60 years or older, community-dwelling and living within 75 miles of Loma Linda University (LLU). During 2016-2018, 199 were reached by telephone and invited to participate in the study. Of those, 168 (84%) agreed to participate and were screened for eligibility. Two did not meet inclusion criteria for being proficient in writing, speaking and understanding English. Twelve changed their mind, one could not be scheduled, 12 could not be reached again, and 5 postponed participation due to travel or health-related reasons. One hundred and thirty-five otherwise healthy adults were enrolled in the study and attended an in-person visit at our study clinic. Four were later excluded for having a medical condition that could adversely impact cognitive function. One hundred and twenty-seven participants had complete data on cognitive and physical abilities and were included in analyses (Figure 1).
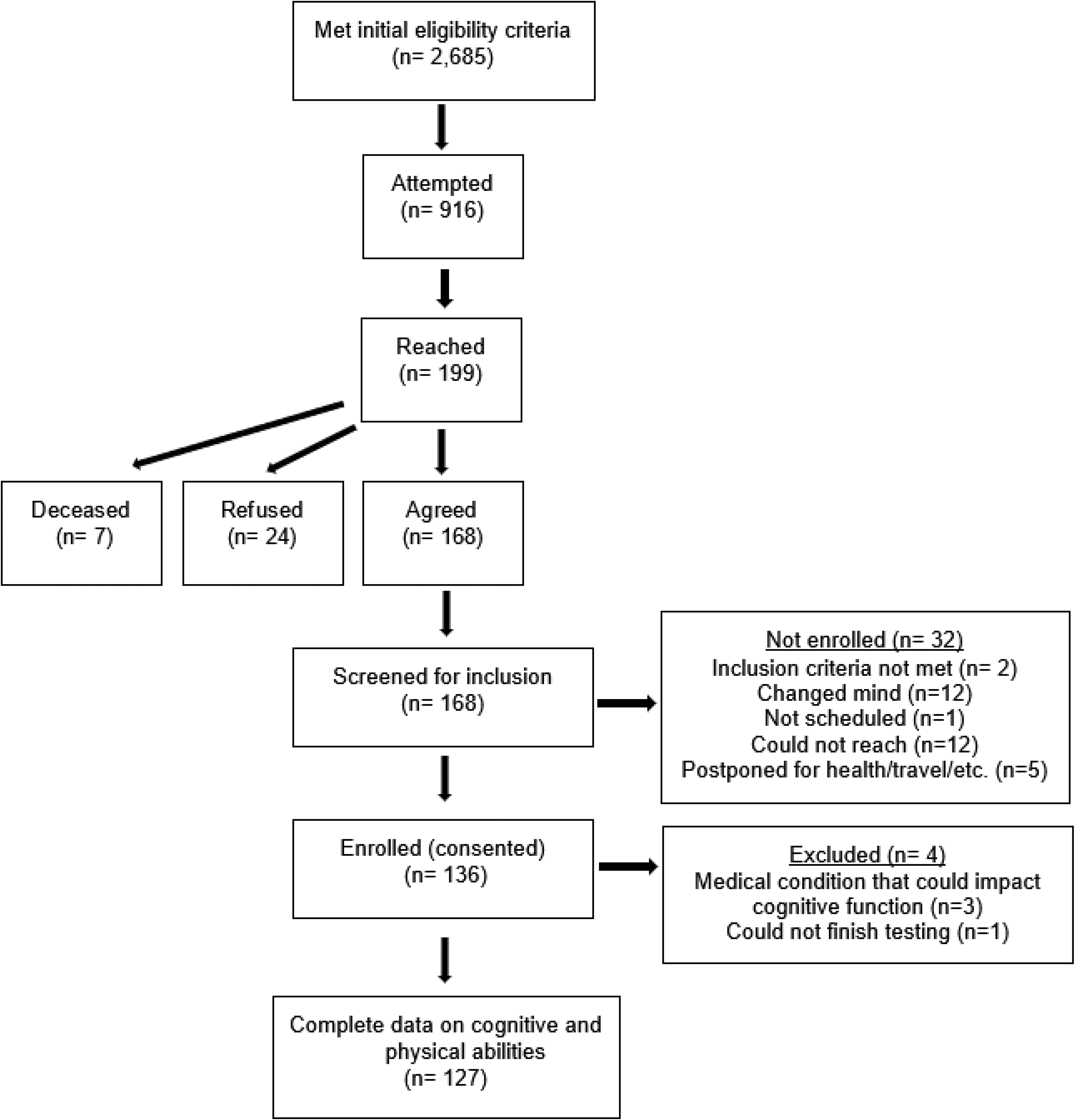
Flow of participants in the AHS-2 CAN GRASP study.
Data Collection
Participants completed a brief questionnaire designed to be consistent with previous AHS-2 follow-up surveys which included items on current medications, recent hospitalizations and regular levels of vigorous physical activity 42 over the past 12 months (“self-reported physical activity”). Physical activity items asked about the number of times per week and average minutes per time engaged in activities such as brisk walking, jogging and bicycling long enough or with enough intensity to work up a sweat, get the heart thumping or get out of breath, and were adopted from Paffenbarger Physical Activity Questionnaire (PPAQ) which has been previously validated in the AHS-2 and other populations. 43 -45 Usual levels of vigorous physical activity in metabolic equivalents (METs)-hours per week (continuous) were calculating multiplying the self-reported frequency and duration in these activities by intensity, following previous work. 46,47
Current level of physical function and mobility were measured during the clinic visit using the 9-item version of the Physical Performance Test (PPT) and Timed Up and Go (TUG) Test. The PPT assesses multiple domains of physical function including upper fine motor function, upper coarse motor function, balance, mobility, coordination and endurance using observed performance of tasks that simulate activities of daily living of various degrees of difficulty in elderly persons. 48 Participants are instructed to perform various tasks including writing a sentence, simulated eating, lifting a book onto a shelf above shoulder height, simulated dressing, picking up a penny from the floor, walking 50 feet, turning in a circle, climbing one flight of stairs and number of flights of stairs climbed. The time to complete each task is measured in seconds, and scores ranging from 0-4 are assigned based on completion times. A total score (maximum possible of 36) is calculated as the sum of individual scores from each task, with a higher score indicating better physical performance. Participants have 2 trials to complete each task, if needed, in which case the better of the 2 scores is used to calculate the total score. Assistive devices are permitted for the last 5 tasks. The Timed Up and Go (TUG) Test assesses a patient’s ability to rise from a seated position, walk 10 feet, turn and return to their seat. 49 Participants are allowed 2 trials, with only the second trial time recorded (if used). For the TUG, scoring is based on the time in seconds to complete the task, with a higher score indicative of poorer physical performance.
Baseline dietary pattern was determined using responses to a validated food frequency questionnaire (FFQ) that was self-administered at cohort enrollment.
50
Dietary habits were queried at the time of cognitive assessment through recall of frequency of consumption of 5 foods (meat, poultry, fish, eggs and dairy) for each decade from 10 years of age to present. Using participant’s age, current dietary habit (vegetarian or non-vegetarian) was determined from questionnaire responses. Vegetarians were defined as either (a) consuming meats, fish, and dairy <1 time/month (vegans), (b) consuming dairy ≥1 time/month
At the in-person visit, participants were administered a 2-hour comprehensive neuropsychological battery, which included tests of verbal learning and memory [Rey Auditory Verbal Learning Test (RAVLT), and Logical Memory subtest of the Wechsler Memory Scale, 4th edition], attention (Digit Span subtest of the Wechsler Adult Intelligence Scale, 4th edition, WAIS-IV), processing speed (Coding subtest of the WAIS-IV, Stroop, Trail-Making Test, Cogstate), executive function (Stroop, Trail-Making Test, FAS), visuospatial abilities (Rey-O Complex Figure), language (Boston Naming Test, FAS, Animals), and global cognitive functioning [Montreal Cognitive Assessment (MoCA), Mini-Mental State Examination (MMSE) 51 ] by trained psychometrists. The American National Adult Reading Test (AMNART) 52 was administered as a measure of estimated premorbid verbal intelligence (VIQ), and the Geriatric Depression Scale (GDS) 53 to assess mood. Height was measured with a portable stadiometer and weight with a Tanita® scale. Body mass index (BMI) was calculated [weight (kg)/height (m2)] and categorized into normal or underweight (BMI ≤ 24.9), overweight (BMI = 25-29.9) or obese (BMI > 30).
Biospecimen Collection and Genotyping for ApoE
Approximately 8 ml of peripheral blood from each participant was drawn into BD Vacutainer® Mononuclear Cell Preparation Tubes (CPTs) with sodium citrate (REF 362761; Becton Dickinson, Franklin Lakes, NJ) by a phlebotomist following standard venipuncture procedures and manufacturer’s instruction. CPTs were delivered to and processed within 2 hours of the blood draw at the Center for Genomics at LLU following manufacturer’s protocols. Briefly, the CPTs were centrifuged at 1,300 RCF for 30 minutes at room temperature in a swing-bucket centrifuge. The bottom layer containing white blood cells (WBCs) was transferred into 15 ml conical tubes and washed with PBS by centrifugation at 300 RCF for 15 minutes. Cell pellets were treated with RBC Lysis Solution (Cat. 158902; Qiagen, Hilden, Germany) and washed again with PBS. WBCs were collected and examined on a TC10 automated cell counter (BioRad, Hercules, CA) for cell numbers and viabilities. WBC pellets were stored in −80°C freezers before DNA extraction.
Genomic DNA were extracted using the Qiagen All prep DNA/RNA/miRNA Universal kit (Qiagen, Hilden, Germany) from frozen human WBCs. DNA was quantified using Qubit 3.0 with dsDNA high sensitivity reagent (Life Technologies, Pleasanton, CA). Genotyping was carried out in 96-well plates using TaqMan Genotyping Master Mix and SNP probes (Life Technologies, Pleasanton, CA). qPCR was performed on the Applied Biosystems QuantStudio 7 Real Time PCR System according to manufacturer’s specifications and data was analyzed using the SDS2.4 software. Genotyping for 2 single nucleotide polymorphisms (rs429358 and rs7412) was used to determine the apolipoprotein E (ApoE) genotype (2/3; 3/3; 3/4; 4/4). For analyses, ApoE genotype was collapsed into 3 categories (2/3; 3/3; 2/4, 3/4 or 4/4).
The study was approved by the Institutional Review Board at Loma Linda University (Protocol# 5150240) and each participant provided written informed consent.
Statistical Analysis
For data reduction purposes, a principal component analysis was performed on the individual neuropsychological test scores using ones as prior communality estimates. The principal axis method was used to extract components employing a varimax (orthogonal) rotation. Nine eigenvalues were >1 which would have resulted in more components than was desirable in relation to our sample size. Visual examination of a scree plot led to a decision to retain the first 3 components, which accounted for 49% of the total variance. An item was considered to have loaded on a given component if the factor loading was greater than |0.40|. Using these criteria, 13 items loaded on the first factor which was named “processing speed”; 8 items loaded on the second factor “executive function” and 10 items loaded on the third factor “memory/language” (Table 1).
Rotated Factor Pattern From Principal Components Analysis.

* Indicates a factor loading >|0.40|.
In order to examine diagnostic risk associated with physical function, participants were classified as having mild memory impairment if at least one of their scores on tests of memory (RAVLT and Logical Memory immediate and long delayed recalls) were less than 1.5 standard deviations below population-based normative means (n = 25, 19.7%). 54,55 We focused specifically on memory impairment, and did not attempt to classify participants as impaired along other cognitive domains because amnestic MCI in particular (as opposed to non-amnestic forms of MCI) has been identified as a precursor to AD. 5
Descriptive statistics for participants were summarized and correlation coefficients were calculated to assess correlations between variables. Generalized linear models were used to estimate β coefficients for cross-sectional associations between cognitive factors and measures of physical function and mobility (PPT and TUG), and of self-reported vigorous physical activity (questionnaire-based). For models of cognitive factors, β coefficients were expressed in standard deviation (SD) units above or below the mean score. If we found an association with a cognitive factor, we further examined whether individual tests that contributed to the factor were driving the association. For models of individual cognitive tests, β coefficients were unit increases or decreases in test scores. Logistic regression models were used to estimate odds ratios (ORs) and 95% confidence intervals (95% CI) for mild memory impairment. We considered whether covariates including age (years), sex, race (white, non-white), education (years), BMI, mood (GDS score), and ApoE genotype were confounders of these associations, initially by examining associations of these variables with the cognitive factors and then by comparing β coefficients from models of cognition and physical functioning and activity with and without the inclusion of these variables. We retained those variables in multivariable models that were either univariately associated with cognition and or that led to changes in β coefficient by > 10%. We also examined whether the association between physical function and cognition was independent of physical activity by comparing models with and without adjustment for self-reported vigorous physical activity. If there was an association between individual cognitive factors and physical function and activity, we further investigated effect modification by age, sex, physical activity (for models with physical function) and dietary pattern testing interaction terms in regression models. If there was evidence of a statistically significant interaction (p < 0.10), we conducted stratified analyses by categories of the effect modifying variable. For interpretation, continuous variables of BMI, GDS, PPT, TUG, and self-reported vigorous physical activity in MET-hours per week were centered on their standard deviation.
All analyses used SAS version 9.4 (SAS Institute Inc., Cary, N.C., USA).
Results
Study participants ranged in age from 60-92 years (mean age 74.8 ± 7.9 years), were predominantly white and the majority were female (Table 2). At baseline/enrollment in the cohort, 53.5% of participants followed vegetarian dietary patterns; of those who completed questionnaire items on dietary habits, 60.9% were vegetarian at the time of cognitive assessment. Vegetarians were less likely to be overweight or obese and had higher scores on the GDS than non-vegetarians, otherwise were comparable in characteristics (Supplemental Table 1). The average BMI was 27.1 (± 5.5), and 40.2% of participants were of normal weight or were underweight. Mean scores on the PPT and TUG were comparable with published values for populations of other community-dwelling adults. 56 -58 Participants were well-educated and their estimated VIQ indicated high level of premorbid intelligence; 19.7% were categorized as having mild memory impairment, based on impairment in at least 1 of 4 memory scores. Average GDS scores (3.0 ± 3.1) indicated that participants did not exhibit significant depressive symptoms. 53,59
Characteristics of Study Participants (n = 127).

† n = 113.
* n = 100.
‡Metabolic equivalents.
Scores on the PPT were strongly inversely correlated with TUG time (r = −0.82; p < 0.0001). Self-reported levels of vigorous physical activity were weakly correlated with PPT scores (r = 0.14; p = 0.11) and modestly with TUG time (r = −0.29; p = 0.001). Age, BMI and GDS fit criteria as confounding variables and were therefore retained in multivariable models of measured physical abilities.
Better physical performance indicated by the PPT was associated with higher scores on the cognitive processing speed factor (β = 0.21 SDs for each 4.4-point increase in PPT; adjusted for age, BMI and GDS); p = 0.02) (Model 2, Table 3A). This association appeared to be driven by individual tests that loaded heavily on this factor, with better scores observed for Stroop Word (β = 0.64, [SE(β) = 0.33]; p = 0.05), and Coding (β = 0.65, [SE(β) = 0.31]; p = 0.04). Higher PPT scores were also associated with faster reaction times on the Cogstate Identification task (IDN) (β = 6.56, [SE(β) = 2.36]; p = 0.006). The relationship between PPT and processing speed was independent of self-reported level of vigorous physical activity (Model 3, Table 3A). Tests for interaction terms for PPT by age, sex and self-reported vigorous physical activity were not statistically significant (all p-values > 0.10), however the interaction with vegetarian dietary pattern was (p-value = 0.03). Models stratified by dietary pattern suggested that the association between PPT and cognitive processing speed was stronger among vegetarians (β = 0.38 [SE(β) = 0.14]; p = 0.008) than non-vegetarians (β = 0.10 [SE(β) = 0.12]; p = 0.44) with adjustment for age, BMI, GDS and self-reported vigorous physical activity (Model 4; Table 3A; Figure 2). Compared to non-vegetarians with PPT scores at or below the mean, vegetarians with PPT scores above the mean did better on cognitive processing speed tasks but these results did not achieve statistical significance (β = 0.18 [SE(β) = 0.26]; p = 0.49).
A and B, Results From Regression Models of Associations (β [SE (β)]; p-Value) Between Measured Physical Abilities, Self-Reported Vigorous Physical Activity, and Processing Speed Factor.

* Metabolic equivalents; †GDS: Geriatric Depression Scale; ‡n = 100.
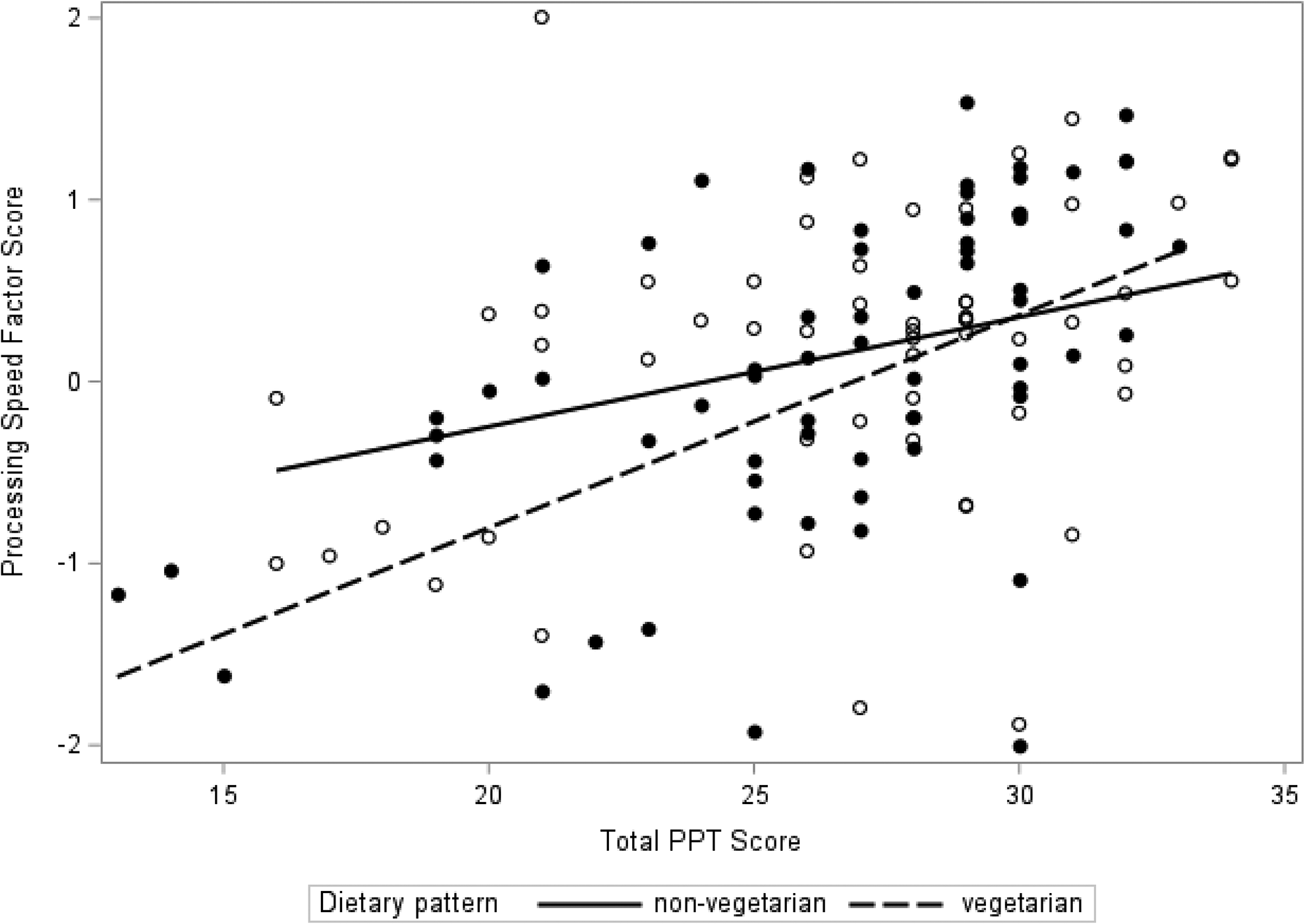
Association between PPT and processing speed abilities by dietary pattern.
Faster times on the TUG test were also associated with higher scores on the cognitive processing speed factor (β = 0.21 SDs increase for each 2.8 seconds less time on the TUG; p = 0.02 adjusted for age, BMI and GDS) (Model 2, Table 3B). Faster reaction times on the Cogstate IDN (β = 26.8; [SE(β) = 10.2]; p = 0.01) appeared to be driving the association. Similar to PPT, the relationship between TUG and cognitive processing speed was independent of self-reported level of vigorous physical activity (Model 3, Table 3B). Tests for interaction terms for TUG by age, sex and self-reported vigorous physical activity were not statistically significant (all p-values > 0.10), however the p-value for interaction with vegetarian dietary pattern was 0.08. Stratified models also suggested that the association between TUG and cognitive processing speed was stronger among vegetarians (Table 3; Model 4). Compared to non-vegetarians with TUG times above the mean, vegetarians with TUG times at or below the mean did better on processing speed tasks but these results did not achieve statistical significance (β = 0.24 [SE(β) = 0.24]; p = 0.33).
Better physical functioning measured by the PPT and TUG was not associated with the memory/language or executive function factors (Supplementary Table 2).
Self-reported levels of vigorous physical activity were not associated with cognitive processing speed (β = −0.04 [SE(β) = 0.09] per SD MET-hours per week; p = 0.63) or the other 2 cognitive factors (Supplementary Table 2). Neither measure of physical function or self-reported physical activity was associated with a statistically significant higher odds of mild memory impairment (Supplementary Table 2).
Discussion
In this study of elderly community-dwelling adults who predominantly follow vegetarian dietary patterns, greater observed physical abilities, as measured by the PPT and TUG, were associated with better performance on cognitive tasks associated with cognitive processing speed. Self-reported vigorous physical activity, on the other hand, was not individually associated with cognitive function, and the relationship with measured physical function and processing speed was independent of physical activity. Our results also suggest that a vegetarian diet modified the association between physical functioning and better cognitive processing speed abilities.
Prior research supports relationships between measured physical abilities and cognitive function 14,60 and results from some studies are similar to our own. A study of 125 predominantly white non-cognitively impaired community-dwelling adults aged 75 years and older who did not engage in regular exercise, found that a cognitive speed factor derived from a factor analysis was independently associated with total modified PPT score. 36 The 6 individual tests that loaded on the cognitive speed factor were measures involving cognitive processing and psychomotor speed but requiring visual scanning, attention and learning. In that study, performance on memory tasks was not associated with PPT score. A large multi-center study of Canadian adults with a mean age of 63 years and negative histories of neurological diseases reported that 3 measures of mobility (walking speed, balance and chair stands) were each associated with processing speed, verbal learning and executive function. 61 In that study, age modified the mobility-cognition relationships, with associations generally being stronger with increasing age. Performance on the TUG was cross-sectionally associated with Stroop scores among community-dwelling middle-aged and older adults in China. 62 A small cross-sectional study of community-dwelling adults 55 years and older also found higher physical activity, measured with a wrist-worn accelerometer, was associated with better global cognitive function among individuals without MCI but not among participants with probable MCI. 15
Our study was small, thus not detecting associations with some of the cognitive measures, self-reported physical activity, or with mild memory impairment may have been a result of being underpowered. Moreover, our cohort was exceptionally healthy with a relatively low rate of overall cognitive impairment; even among those with mild memory impairment, more than half had an impaired score on only 1 of the 4 memory scores. Thus, a more heterogeneous sample with a wider range of impairments would be helpful for future studies. With cross-sectional data, we cannot infer the directionality or temporality of the associations that we detected. It could be that declines in cognitive function affect or precede physical declines, but we did not have baseline cognitive function with which to compare. We examined demographic, metabolic, psychological, genetic and lifestyle factors as potential confounders and adjusted for age, BMI and mood in our analyses. Other factors which we did not take into consideration such as comorbidities or medication use could confound the associations. Our sample size limited the number of variables we could include in models as confounders as well as our power to test for interactions. Our small sample size could have limited the detection of differences in associations by sex, since biological sex has been suggested as a possible mediator of relationships between exercise and brain health in aging. 60
Physical activity improves both cardiovascular and metabolic function, 35 and could potentially improve cognitive function through mechanistic changes at molecular, cellular and systemic levels. 30 Increases in cerebral blood flow and metabolism as would result from physical activity, could lead to the formation of neural networks 16 as well as more efficient neural activity 14 associated with frontal-executive abilities such as processing speed, verbal encoding and retrieval which we observed in our study. Physical activity may also increase brain neurotrophins and growth factors which could lead to greater synaptic plasticity and neurogenesis. 14,29 Physical activity may reduce against oxidative stress by upregulating endogenous antioxidant enzymes. 63,64 Physical activity may also help maintain brain volume and reduce atrophy. 65 In retrospect, given that our study population consisted of elderly adults, it may not have been appropriate to query vigorous physical activity, and we may have been better able to examine study hypotheses had we collected data on the frequency and duration of moderate activities as well.
The PPT is scored using timed measurements so that it may be administered with minimal interpretation or judgment by the observer. 48 Many of the cognitive measures that loaded heavily on the processing speed factor in our study are also timed measures. It is possible that the correlation between PPT and cognitive processing relates to the pace with which participants are able to complete both physical and cognitive tasks. However, despite the timed basis for scoring, the PPT was designed to measure several domains of functioning including upper body strength and dexterity and mobility so as to capture a range of abilities and abilities associated with activities of daily living. Moreover, some of the cognitive processing speed tasks did not involve motor, i.e. physical speed, but relied on mental speed, mainly perceptual and verbal speed. Thus, we would argue that performance on the cognitive processing speed tasks represents a different construct from the physical abilities measured by PPT, and that our findings demonstrate that better physical functioning is associated with faster cognitive processing speed.
The PPT and TUG are direct observations of physical function, and therefore an advantage of our study was that we were able to objectively quantify functional capabilities of our participants. While we asked participants about their physical activity, we did not solely rely on self-reports in our analyses. 48 Self-reports of physical activity in older adults may have low to moderate validity compared with objective assessments, 15,66 with studies suggesting that older adults may overestimate higher activity levels 67 and underestimate lower activity levels. 68 Furthermore, physical function represents another aspect of health distinct from level of physical activity, as it reflects the efficiency with which one can complete simple everyday tasks, and may be more closely tied to preservation of functional independence in older adults. 69 Among elderly, the PPT has been shown to have excellent reliability 48,70 and good to excellent validity with other tests of physical performance 71 but moderate correlations with self-reports of physical abilities on questionnaires. 72
Several studies generally find some protective effect of a Mediterranean diet on MCI incidence and prevalence, and on MCI conversion to AD. 72 -79 Similar to the “Mediterranean diet,” vegetarian dietary patterns are plant-based, yet typically incorporate even larger quantities of fruits and vegetables, and exclude consumption of some or all animal products. 80,81 As such, vegetarian diets are rich in phytochemicals and antioxidant micronutrients, 82 many of which may exert anti-inflammatory and antioxidant effects. 83,84 At enrollment in the AHS-2 cohort and 10 years prior to this study, 53.9% of study participants followed vegetarian dietary patterns. At the time of cognitive assessment, 61.3% of study participants were vegetarian. Our results thus support associations between physical function and cognition with additional modification by vegetarian dietary pattern as a lifestyle factor. The notion that a “comprehensive” healthy lifestyle, encompassing more than one domain of health-promoting behaviors, may prevent cognitive decline has been proposed. 32 The joint effect of diet and physical function and activity on preventing the risk of cognitive decline has received some support. 33,85 Additional studies should investigate potential common mechanisms underlying the relationship of different lifestyle-related behaviors with cognition. 86 The results of this study with others could support recommendations of behaviors which clinicians could encourage in their elderly patients.
Supplemental Material
Supplemental Material, Figure_Legends - Observed Physical Function Is Associated With Better Cognition Among Elderly Adults: The Adventist Health Study-2
Supplemental Material, Figure_Legends for Observed Physical Function Is Associated With Better Cognition Among Elderly Adults: The Adventist Health Study-2 by Nicole M. Gatto, Jennifer Garcia-Cano, Crissy Irani, Tiantian Liu, Cameron Arakaki, Gary Fraser, Charles Wang and Grace J. Lee in American Journal of Alzheimer"s Disease & Other Dementias
Supplemental Material
Supplemental Material, Supplemental_Material - Observed Physical Function Is Associated With Better Cognition Among Elderly Adults: The Adventist Health Study-2
Supplemental Material, Supplemental_Material for Observed Physical Function Is Associated With Better Cognition Among Elderly Adults: The Adventist Health Study-2 by Nicole M. Gatto, Jennifer Garcia-Cano, Crissy Irani, Tiantian Liu, Cameron Arakaki, Gary Fraser, Charles Wang and Grace J. Lee in American Journal of Alzheimer"s Disease & Other Dementias
Footnotes
Abbreviations
Authors’ Note
Authors contributed to this work as follows: formulating the research question(s): NG & GJL; designing the study: NMG, GJL, GF, CW; carrying it out: NMG, GJL, GF, CW, CA, CI, TL; analyzing the data: NMG, JGC, GJL; writing the article: NMG, GJL, GF, CW, JGC, CI, TL, CA. This study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving research study participants were approved by the Institutional Review Board at Loma Linda University. Written informed consent was obtained from all subjects.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by GRASP Award 2140309 from Loma Linda University, Office of Research Affairs.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
