Abstract
Dementia with Lewy bodies is a progressive neurodegenerative disorder, clinically characterized by gradual cognitive impairment and fluctuating cognition, behavioral changes and recurrent visual hallucinations, and autonomic function and movement symptoms in the type of parkinsonism. It is the second most common type of dementia in the Western world after Alzheimer disease. Over the last 20 years, many neurophysiological, neuroimaging, and cerebrospinal fluid (CSF) biomarkers have been described toward a better discrimination between dementia with Lewy bodies, Alzheimer disease, and other neurodegenerative conditions.In the present review, we aim to describe the neurophysiological, imaging, and CSF biomarkers in dementia with Lewy bodies and to question whether they could be reliable tools for the clinical practice.
Introduction
Dementia with Lewy bodies (DLB) is a progressive neurodegenerative condition clinically characterized by gradual cognitive impairment and fluctuating cognition, behavioral changes and recurrent visual hallucinations, and autonomic function and movement symptoms in the type of parkinsonism. Dementia with Lewy bodies accounts for the 15% of dementia cases at autopsy and is the second most common type of neurodegenerative dementia after Alzheimer disease (AD). 1 Day-to-day clinical practice is not always easy to differentiate between the different types of dementia based only on clinical features, especially when the characteristic symptoms are not present, and the definite diagnosis can only be confirmed by postmortem findings; however, several neurophysiological, neuroimaging, and cerebrospinal fluid (CSF) markers can increase the accuracy of the diagnosis and to organize the best possible treatment plan for the patients and their carriers. In the present review, we aim to describe the neurophysiological, imaging, and CSF biomarkers in DLB.
Clinical and Pathological Hallmarks
Dementia with Lewy bodies is clinically characterized by cognitive impairment with fluctuations of cognition, extrapyramidal features, visual hallucinations, and/or rapid eye movement sleep behavior disorder. 2 Fluctuating cognition with pronounced variations in attention and alertness, recurrent well-formed and detailed visual hallucinations, rapid eye movement sleep behavior disorder, and one or more spontaneous cardinal features of parkinsonism are the core clinical features of the disease. The current diagnostic criteria for DLB require the presence of 2 or more core clinical features with or without indicative biomarkers for probable DLB, and only 1 core clinical feature with no indicative biomarkers or one or more indicative biomarkers with no core clinical features for possible DLB. 2
The pathological hallmark of the disease includes aggregation of α-synuclein in cellular inclusions and dystrophic neurites throughout the brain 3 , and furthermore, AD-like pathology can be found in up to 89% of patients with DLB in autopsy studies. 4,5
Cerebrospinal Fluid Biomarkers
α-Synuclein
α-Synuclein is one of the potential CSF biomarkers for DLB; however, the outcomes of different studies are controversial. Spies et al, analyzing the levels of α-synuclein in the CSF of 40 patients with DLB, 131 patients with AD, 28 patients with vascular dementia, and 39 patients with frontotemporal dementia found no significant differences in CSF α-synuclein levels between different groups. 6 Two additional studies reported significant differences in CSF α-synuclein levels between DLB, Parkinson disease (PD), non-DLB dementias, and control patients. 7,8 Mollenhauer et al, using enzyme-linked immunosorbent assay to measure total α-synuclein level in CSF in 57 patients with DLB, 35 patients with AD, and control cases, found lower mean α-synuclein concentrations in DLB 7 , while Kasuga et al examined the total and α-synuclein levels in 34 patients with DLB, 31 patients with AD, and 21 other dementia cases and found significantly lower α-synuclein levels in DLB compared to AD and other dementias. 8 A total of 15 studies were found for the role of α-synuclein in the discrimination between DLB, PD, and AD. The outcomes, although controversial, showed that α-synuclein in the CSF is a reliable biomarker for the differential diagnosis between DLB and AD but cannot be used for the discrimination between DLB and PD 9 –20 (Table 1).
Details of the Findings on the Studies on the Use of α-Synuclein for the Diagnosis of Dementia With Lewy Bodies.
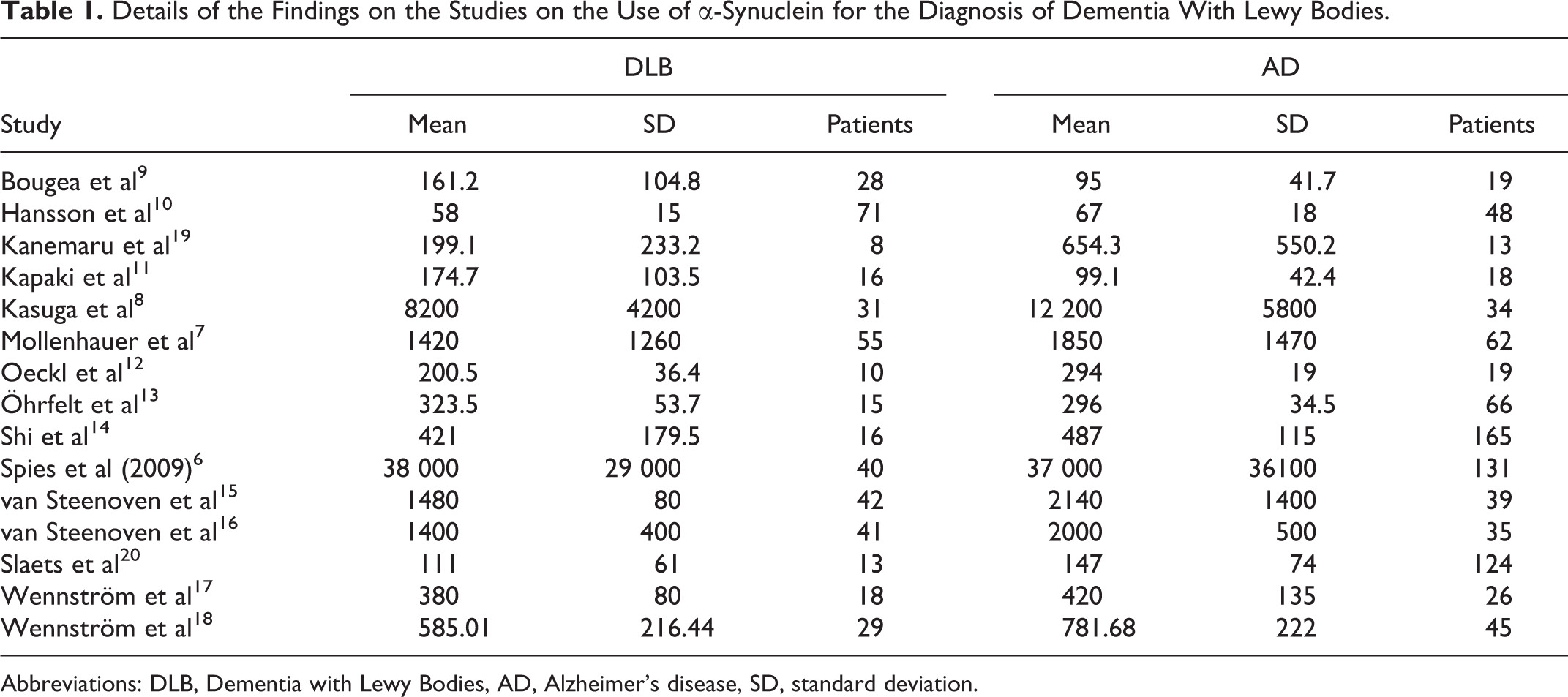
Abbreviations: DLB, Dementia with Lewy Bodies, AD, Alzheimer’s disease, SD, standard deviation.
Tau Protein
Although CSF phosphorylated tau 181 (p-tau 181) is present in several neurodegenerative disorders, it seems to be often implicated in DLB. 21 Patients with AD have significantly higher CSF levels of p-tau 181 compared to controls and patients with DLB 22 and can be used for the differentiation between DLB and AD with a sensitivity of 91% and specificity of 94%. 23 In another study in autopsy-confirmed cases, CSF p-tau 181 exhibited a sensitivity of 75%, specificity of 61%, and a diagnostic accuracy of 73% in discrimination between DLB and AD. 24 Hamper et al reported similar results while they found that increased CSF levels of p-tau 181 have a sensitivity of 94% and a specificity of 64% for the differentiation of DLB against AD. 25 Studies on whether the total CSF tau protein can be used for the discrimination between DLB and AD are controversial, while both nonsignificant and significant differences have been reported. 26,27 An additional study reported significantly lower levels of total tau in DLB than in AD and substantially higher levels than PD and PD dementia. 28
Analysis of 1221 CSF samples from different memory centers in France, of which 57 with pro-DLB, 154 with DLB, 132 with prodromal AD, 783 with AD, and 95 controls, for phospho-Tau181, total-Tau, amyloid β (Aβ) 43, and Aβ40, showed reduced levels of Aβ43 in late stages of DLB, compared to AD cases, where Aβ42 levels were decreased even from early stages. The Aβ42/Aβ40 ratio in pro-DLB was similar to controls, and total Tau protein (t-Tau) and phospho-Tau181 were unchanged in DLB cases, independent of the stage. The study concludes that CSF t-Tau and phospho-Tau181 are potential biomarkers for the differential diagnosis between DLB and AD 29 (Table 2).
Summary of the Most Important Findings on Cerebrospinal Fluid Biomarkers for the Diagnosis of Dementia With Lewy Bodies.

Abbreviations: Aβ, amyloid β; DLB, dementia with Lewy bodies, AD, Alzheimer disease, SD, standard deviation; PD, Parkinson disease; CSF, cerebrospinal fluid; t-Tau, total tau protein.
Amyloid β Peptide
Amyloid β peptide 1-42 is characteristic for AD and however can also be found in DLB, in combination with lower levels of cystatin C, a peptide known to inhibit fibril formation and oligomerization of the amyloid peptide. 32 Furthermore, Aβ peptide 1-40 is significantly decreased in DLB compared to AD. 6 Although CSF levels of Aβ42 on their own cannot be used to differentiate DLB from AD, they have been reported to be significantly lower than controls in both conditions but with no significant difference between them 33,34 ; the ratio between Aβ1-42 and Aβ1-40 is considered a reliable biomarker for the discrimination between AD and other dementias including DLB. 30 An oxidized α-helical form of Aβ peptide, referred as Aβ1-40*, was found to be significantly increased in patients with DLB in comparison to PD dementia, having a sensitivity of 81% and specificity of 71% at DLB and PD dementia differentiation 31 (Table 2). Furthermore, reduced glutathione, but not the oxidized form, was significantly lower in patients with DLB than in controls; however, the total glutathione which has been studied in several neurodegenerative diseases including AD did not show any significant difference. 35
Other CSF Biomarkers
A study on 26 patients with AD, 18 patients with DLB, and 24 controls reported significantly decreased CSF orexins levels in patients with DLB, being more prominent in female patients, and concluding that CSF orexins are specific to DLB versus AD but can be related to gender that warrants further investigation. 18
Older studies on inflammatory markers in the CSF showed that none of the interleukins 1b and 6, the precursor enkephalins, and substance P peptides could be used to differentiate DLB from other dementias. 36,37 However, Wennstrom et al in a more recent study found significantly lower CSF levels of interleukin 6 (IL-6) in patients with DLB compared to patients with AD. 17
The cocaine- and amphetamine-regulated transcript neuropeptides were found to be significantly reduced in the CSF of patients with DLB in comparison to patients with AD and controls. 38
Asparagine, glycine, and nitric oxide are also raised in the CSF of patients with DLB compared to controls and can be used as supportive biomarkers for the diagnosis of DLB 39,40 ; however, they have not been tested against other dementias.
Calcium and magnesium levels were elevated in DLB compared to AD and controls and using combined measurements of both had a sensitivity of 93% and specificity of 85% to differentiate DLB from AD 41 (Table 2).
Neurophysiological Biomarkers
Electroencephalography
The presence of posterior temporal transient slow or sharp waves is included in the diagnostic criteria as a supportive feature for the diagnosis of DLB. 42 –44 Certain electroencephalography (EEG) topographical differences between DLB and AD have also been described, while in DLB, abnormalities are found in posterior regions and in AD in temporal lobes 45,46 ; in addition, patients with DLB exhibit increased posterior slow-wave activity compared to controls and patients with AD. 43,45,47 –49
Patients who were not taking donepezil showed increased spectral power density in δ and θ bands in comparison to controls, while patients with AD did not exhibit the same changes. 50 Andersson et al described a greater degree of parietal delta power band variability in DLB against AD and controls 50 ; nevertheless, similar findings have been described by different studies in patients with AD, making this feature nonspecific for DLB. 51
Electroencephalography coherence between left anterior, right anterior, left posterior, and right posterior has been found to be increased in the delta band but decreased in the alpha band in patients with DLB versus controls. 50 Further differences in intrahemispheric and temporo-centro-parieto-occipital beta band coherence values between patients with DLB and AD have been reported by Kai et al. 52
Patients with DLB exhibited greater fluctuations in frequencies on a second-by-second basis compared to patients with AD and controls. 45,53
A longitudinal study on 47 individuals with mild cognitive impairment, 50 individuals with DLB, 50 patients with AD, and 50 controls revealed typical abnormalities in patients with mild cognitive impairment who converted to probable DLB, including a reduced dominant frequency and increased dominant frequency variability. 54 Londos et al analyzing retrospectively resting EEG recordings from 45 AD and 48 DLB cases reported no significant differences between the 2 groups 55 (Table 3).
Summary of the Most Important Findings on Neurophysiological Biomarkers for the Diagnosis of Dementia With Lewy Bodies.

Abbreviations: AD, Alzheimer’s disease; CSF, cerebrospinal fluid; DLB, dementia with Lewy bodies, EEG, electroencephalogram; MSA, Multiple System Atrophy; SD, standard deviation; PD, Parkinson disease.
Event-Related Potential Studies
Bonanni et al reported a lower amplitude and greater latency of auditory P300 in patients with DLB compared to AD and an overall inversion of the pattern of P300 amplitude gradients. 57 The presence of inverted amplitude differentiated DLB from AD with a sensitivity of 70% and a specificity of 97%. 46 Perriol et al examined the prepulse inhibition of the N1/N2 component of auditory-evoked potentials in 10 DLB, 10 AD, and 10 PD dementia cases, and found a reduced prepulse inhibition in DLB compared to AD and control individuals 56 (Table 3).
Blink Reflex Studies
Blink reflex studies have also shown worth-mentioned findings. Bonanni et al studying patients with DLB, AD, and multiple system atrophy against controls demonstrated a significantly delayed blink reflex response with increased latency in the DLB group 57 (Table 3).
Additional Physiological Biomarkers
Auditory startle reaction is another neurophysiological index that has been investigated in DLB. Kofler et al examined the auditory startle reaction in DLB cases, patients with other parkinsonian syndromes, and control individuals and found an abnormal startle reaction profile for patients with DLB. 58
Additional physiological markers that have been investigated include autonomic response markers. Sympathetic skin response and heart rate variability were reported to be significantly different in DLB, with lower sympathetic response and a lower ratio of low-frequency to high-frequency power with the sensitivities being 85% and 90%, respectively, and the specificity of each method being 85% and 85%, respectively. 59
In another study on autonomic responses in DLB, Akaogi et al found reduced sweat response amplitudes in patients with DLB and PD compared to controls, lower vasomotor reflex amplitudes in patients with DLB but not patients with PD, and significantly lower coefficient variation in R-R intervals value in patients with DLB 60 (Table 3).
Transcranial Magnetic Stimulation
The short-latency afferent inhibition, a corticomotor parameter, was reduced in comparison with controls 61,62 and in fact was correlated with visual hallucinations severity 62 ; however, whether this could be a reliable biomarker remains controversial, while Nardone et al reported no significant difference between patients with DLB and controls. 63
Neuroimaging Biomarkers
Structural MRI
Compared to AD, DLB brains exhibit relatively preserved volumes of hippocampus and medial temporal lobes in structural magnetic resonance imaging (MRI) 64 and additional studies that described mild hippocampal atrophy in patients with DLB compared to normal controls. 65 Lebedev et al, using a multivariate classification of cortical thickness, have shown that this has a sensitivity of 82.1% and a specificity of 85.7% in the differentiation between DLB and AD. 66 Dementia with Lewy bodies showed atrophy predominantly to the superior temporo-occipital and lateral orbitofrontal regions, and the middle and posterior cingulate cortices, while AD brains are characterized by cortical thinning within the temporal pole, subgenual cingulate regions, and the parahippocampal areas. 66 The rate of cerebral atrophy in DLB is higher than in normal controls but significantly lower than in AD. 67 Another study revealed elective atrophy of different hippocampal areas in DLB compared to AD. In DLB, the perirhinal cortex and the parahippocampus are mainly affected, whereas in AD, the cornu ammonis and the subiculum undergo significant atrophy. 68 Reduced amygdala volume and atrophy in other cortical and subcortical areas such as the striatum, substantia innominata, hypothalamus, and dorsal midbrain have been described in DLB cases. 69,70
Cortical thinning in DLB is more significant than in PD as Burton et al described using voxel-based morphometry, with reductions being more significant in the temporal, parietal, and occipital lobes 69,70 ; however, another study was unable to identify different patterns of cortical atrophy between DLB and PD. 71 Further studies have described an additional occipital and striatal cortical thinning in DLB, compared to PD, and a positive relationship between anterior cingulate, right hippocampus, and amygdala gray matter reduction and cognitive performance as well as loss of cortical thickness in the left precuneus and inferior frontal lobe and visual hallucinations have also been reported. 73,74
Apart from different patterns of cortical loss between patients with DLB, AD, and PD, a greater burden at baseline white matter hyperintensities has been described in patients with AD compared to healthy controls, patients with DLB, and patients with PD, but the importance of this finding remains poorly understood 75 (Table 4).
Summary of the Most Important Findings on Neuroimaging Biomarkers for the Diagnosis of Dementia With Lewy Bodies.

Abbreviations: DLB, Dementia with Lewy bodies, AD, Alzheimer disease, PD, Parkinson disease; SWT, susceptibility-weighted imaging; MRI, magnetic resonance imaging.
Diffusion Tensor Imaging
Dementia with Lewy bodies brains show increased diffusion and decreased fractional anisotropy in corpus callosum and pericallosal areas and in frontal, parietal, and occipital areas in comparison to normal controls. 76 Furthermore, patients with DLB show reduced fractional anisotropy in the precuneus area compared to both normal controls and patients with AD 77 and in parieto-occipital white matter tracts compared to normal controls, which seems to be an early phenomenon. 78 The same study also showed that in AD there is no evidence of longitudinal changes in mean diffusivity or fractional anisotropy, but there is a greater longitudinal increase in mean diffusivity in parietal and temporal regions. 78 Additional reduced fractional anisotropy in the pons and left thalamus in DLB did not seem to be reproducible 78 (Table 4).
Susceptibility-Weighted Imaging
Kamagata et al reported that measuring nigrosome 1 hypointensity with susceptibility-weighted imaging achieved 90% diagnostic accuracy with 93% sensitivity and 87% specificity, and Shams et al showed that the caudal and posterolateral part of the substantia nigra is hypointense in patients with DLB compared to patients with AD and other dementias. 79,86
Functional MRI
Functional MRI studies suggest increased functional connectivity between the right posterior cingulate gyrus and the inferior parietal cortex and putamen and decreased connectivity in the frontoparietal operculum, medial prefrontal cortex, and the primary visual cortex when compared to AD, while a reversal of connectivity was observed in the right hippocampus. 80,81 Dementia with Lewy bodies has been shown to display increased connectivity in the default mode network in comparison to AD. 87 Memory function is relatively preserved in DLB, and the nonsignificant difference in hippocampal connectivity between DLB cases and normal controls is in keeping with this. 82 Schumacher et al investigating functional connectivity changes in 31 patients with DLB compared to 31 healthy controls and 29 patients with AD using dual regression and FSLNets found decreased within-network connectivity in patients with DLB compared to controls, mainly in motor, temporal, and frontal networks. Moreover, they described intact between-network connectivity and increased frontal and temporal network connectivity in patients with DLB, and subtle differences between patients with DLB and patients with AD. They concluded that patients with DLB and AD may show more similarities than differences in mild disease cases. 83 Patients with DLB also demonstrated greater activation in the superior temporal sulcus compared to AD, especially during motion tasks, 84 but those findings were not reproduced in another study which examined the response of visual and parieto-occipital cortex of 17 DLB cases and 19 controls to visual stimuli, and no significant differences were found in functional response to objects, motion stimuli, or checkerboard in V1, V2, and V3 occipital areas. 85 The same study also reported a reduction in V5/MT brain areas activation in response to motion stimuli, 85 concluding that the visual system in DLB is dysfunctional. Patients with DLB also exhibit greater alterations in functional connectivity at the precentral and postcentral gyri, cerebellar, occipital, and temporal regions compared to controls, whereas in PD functional connectivity changes are mainly limited to frontal cortex and the precuneus 88 ; however, no significant difference was found on DLB versus PD comparison (Table 4).
Positron Emission Tomography and Single-Photon Emission Computed Tomography
Positron emission tomography (PET) and single-photon emission computed tomography (SPECT) studies have shown decreased glucose metabolism and deficits in perfusion in parietotemporal and occipital areas in DLB, while AD is characterized by more generalized decreased cerebral metabolism. 89 –93 Decreased 18 F-fluorodopa reuptake in the striatum can be used to differentiate DLB from AD having high sensitivity and specificity according to a recent study. 94 Liu et al using Pittsburgh compound B (C-PIB) PET scans for Aβ deposition and 18F-FDG PET scans for regional cerebral metabolism in 37 patients with DLB reported Aβ deposition in the cortex and hypometabolism in the temporo-parieta-ooccipital regions bilaterally as well as the frontal lobe, the insular lobe, the posterior cingulate, the precuneus, and caudate nuclei. 95
A combined study of 18F-FDG PET scan and C11 Pittsburgh compound B PET in 39 DLB cases, 39 AD, 78 controls, and 10 autopsies examined for Braak neurofibrillary tangle staging revealed a higher ratio of posterior cingulate to precuneus plus cuneus metabolism, cingulate island sign than AD, and this was independent of Aβ load. The same study also describes an inverse relationship between higher cingulate island sign ratio and Braak staging and cognitive symptoms. 96,97
Reduced dopamine transporter binding in the caudate and posterior putamen have also been described by different studies 98,99 in DLB compared to AD but with no difference from PD. Dopamine transporter imaging with SPECT yields a sensitivity of 88% and a specificity of 100% in the differential diagnosis between patients with DLB and AD 100 but cannot be used to differentiate between patients with DLB and PD, where there is a profound loss of dopamine transporter binding in the striatum. 101 A phase III multicenter study on 326 patients with probable (n = 94), possible (n = 57) DLB, or non-DLB dementia (n = 147) reported a mean sensitivity of 77.7% of Dopamine Transporters (DAT) scan for detecting probable DLB, with a specificity of 90.4% for excluding non-DLB dementia, concluding that low DAT uptake in the basal ganglia can be a suggestive feature for diagnosis of DLB over non-DLB dementia.
Thoracic SPECT studies have also shown reduced cardiac reuptake in DLB, significantly different from AD but with no difference from PD. 102 A number of studies have shown diminished cardiac (123) I-metaiodobenzylguanidine uptake in PD and DLB, and in 2005, the Dementia With Lewy Bodies Consortium considered (123) I-metaiodobenzylguanidine cardiac scintigraphy a “supportive” diagnostic feature. A meta-analysis based on 46 studies demonstrated that (123) I-metaiodobenzylguanidine cardiac scintigraphy can accurately distinguish DLB and AD but cannot be used to discriminate DLB from PD. 103 Pittsburgh compound B, an in vivo indicator of Aβ, has shown an 80% increased amyloid load in DLB with a respective 20% in PD. 104
Amyloid β deposition cannot distinguish DLB from AD where Aβ retention is comparable, but it can be used to differentiate DLB where cortical Aβ burden is significantly high from PD where Aβ is scarce 105 , and it seems that Aβ deposits are associated with cognitive impairment in DLB and not PD, dementia in PD, or PD with mild cognitive impairment. 106 Furthermore, amyloid deposition is positively related to cortical thinning and ventricular expansion in DLB. 107
A study using the radioligand fluorine 18-labeled AV-1451, which has been proven suitable to assess tau deposition, has shown variable and higher cortical tau uptake in the inferior temporal gyrus and precuneus compared to controls, and this was associated with cognitive impairment, indicating a role of tau pathology in DLB pathophysiology. 108 Another study using the same ligand demonstrated significantly more extensive tau uptake within the medial temporal lobe in AD than in DLB. 109
Further differences in the spatial distribution of cholinergic deficiencies between DLB and AD have been reported, while in DLB there is a widespread reduction predominantly at the neocortex, the posterior regions, and in the thalamus, whereas in AD the deficits are more prominent in the temporal lobes. 110
Nedelska et al in a recent study on 33 patients with DLB, 18 patients with posterior cortical atrophy, and 100 controls, using tau PET with the 18F-AV-1451 tracer, revealed a significant difference in the AV-1451 uptake, with patients with posterior cortical atrophy showing the greatest uptake throughout all the gray matter, and they conclude that this AV-1451 uptake has a sensitivity of 88% and specificity of 100% in the discrimination between DLB and posterior cortical atrophy which usually share similar clinical features 111 (Table 5).
Summary of the Most Important Findings on PET and SPECT Biomarkers for the Diagnosis of Dementia With Lewy Bodies.

Abbreviations: AD, Alzheimer disease; DLB, dementia with Lewy bodies, PD, Parkinson disease; PET, positron emission tomography; SPECT, single-photon emission computed tomography.
Magnetic Resonance Spectroscopy
Magnetic resonance spectroscopy studies revealed reduced N-acetylaspartate in the temporal lobes and increased myo-inositol, while in DLB N-acetylaspartate–creatine ratio and myo-inositol levels are relatively normal. 112,113
Discussion
Numerous studies have been carried out in order to identify reliable biomarkers for the differential diagnosis of DLB from AD, dementia in PD, and other dementias. Although most of the results are controversial, few CSF, imaging, and physiological biomarkers have been reported to be significant enough and can be used as a supportive to clinical suspicion for increased accuracy in the diagnosis of the probable DLB. α-Synuclein, tau protein, Aβ peptides, and other miscellaneous biomarkers have been extensively studied in DLB and other dementias, and although the data are either controversial or not enough, most of them when combined could be critical for the differential diagnosis of DLB.
Total α-synuclein, combined with Aβ peptide 1-42 and either total of phosphorylated tau, improves the differential diagnosis between different dementia types, including DLB, AD, frontotemporal dementia, and other neurological conditions.
The ratios Aβ 40/42 and tau/ Aβ 42 are also potentially significant biomarkers and when combined with α-synuclein can severely increase both the sensitivity and the specificity of the test. The ratio of Aβ1-42 to Aβ1-38 and Aβ1-42 to Aβ1-37, when combined with t-tau levels, has 100% sensitivity and 92% specificity in differentiating AD from DLB and controls. 114 The oxidized α-helical form of Aβ peptide, known as Aβ 1-40*, has also a sensitivity of 81% and specificity of 71% at DLB and PD dementia differentiation. 114
Total tau was found to be of no significance in the discrimination between DLB and other dementia types, and mainly AD; however, p-tau 181 is considered as a reliable biomarker.
Orexins, IL-6, cocaine, and amphetamine-regulated transcript neuropeptide, asparagine, glycine, and nitric oxide, and the combination of calcium and magnesium are also potentially significant biomarkers 38,41 ; however, further studies are needed in order for their significance to be confirmed.
Among physiological biomarkers, the most important ones are the topographical differences between DLB and AD, with DLB exhibiting mainly increased posterior slow-wave activity. Indeed, the presence of posterior transient slow or sharp waves are included in the diagnostic criteria as a supportive feature for the diagnosis of DLB. 42 –44
Auditory-evoked potential also showed significant evidence in differentiation between DLB and AD, 54,55 and another significant physiological parameter seems to be blink reflex which showed substantial differences between patients with DLB and AD, multiple system atrophy, and controls; however, further studies are needed to confirm whether this marker is sufficiently robust to diagnostically separate DLB from other dementias. Abnormal startle reaction and differences in autonomic nervous system response have also been shown to be significant biomarkers and could be used in the differential diagnosis between DLB and other dementias. While overall the findings for autonomic nervous system responses could also be particularly salient to the detection of early DLB, 115 further investigation is necessary before they could be used in the clinical practice. Magnetic transcranial stimulation although promising has not shown any noteworthy biomarkers so far.
In addition to CSF and physiological biomarkers, DLB brains exhibit certain neuroimaging differences in comparison to PD, AD, and other neurodegenerative disorders. The main structural differences can be summarized in topographical dissimilarities, whereas DLB shows atrophy mainly in the perirhinal cortex, the parahippocampus, the amygdala, the striatum, the substantia innominata, the hypothalamus, and the dorsal midbrain in cortical thickness and the rate of cerebral atrophy which is higher than in controls but significantly lower than in AD, although the latter remains debated.
Diffusion tensor imaging and susceptibility-weighted imaging can also be potential tools for the discrimination between patients with DLB, controls, patients with AD, and patients with other dementias.
Functional MRI, PET, and SPECT also provide potentially momentous biomarkers. Definite differences in functional connectivity and different cortical activation patterns in response to external stimuli can be used to discriminate between DLB and AD and/or PD. Dissimilarities in brain glucose metabolism and perfusion and differences in tracers’ uptake can also make a significant difference in the differential diagnosis between the aforementioned disorders, and other neurodegenerative diseases, such as frontotemporal dementia, multiple system atrophy, and posterior cortical atrophy which may share a few common clinical similarities especially in the initial stages. The great heterogeneity in many studies on biomarkers in DLB and other dementias could be explained by differences in methodology, in clinical design of the studies, and in relation to technical aspects. The sample sizes, inclusion criteria, clinical assessment methods, the number of dementia types analyzed, and the definition of control groups are comparatively clearly different.
To conclude, many biomarkers are currently available for the discrimination between DLB and other dementia types; however, a detailed history and well-organized, detailed clinical and psychological examinations remain the golden standard for the initial clinical suspicion and probable diagnosis, with biomarkers being clearly supportive for an improved diagnostic accuracy. Although many biomarkers have been proven significant, the combination of more than one, and more specific the combination of different biomarkers including at least CSF and/or neuroimaging, can reduce the limitations of their application and provide reliable support in the diagnosis of DLB.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
