Abstract
This study was designed to evaluate the spatial working memory (as studied in Y-maze) or short-term and long-term spatial memory (assessed in radial 8 arms-maze task), in a scopolamine-induced memory deficits model in mice, by the oral administration of 2 angiotensin-converting enzyme inhibitors—captopril and ramipril and also the effects of the AT1 receptor antagonist, losartan. The present article was initiated as a reaction to the clinical setting of hypertensive disease, which involves lifelong administration of antihypertensive drugs, dietary or lifestyle constraints, and aging, which all take a toll on the higher functions of the nervous system. Most of the patients with cognitive decline suffer of various metabolic imbalances, hypertension, cardiac and kidney disease, many of them which are treated with oral administration of Renin–angiotensin aldosterone system-altering agents like those presented above. Our results showed a protective effect of captopril, ramipril, and losartan prescopolamine administration on spontaneous alternation in Y-maze task, as compared to scopolamine-alone treated mice, as well as decreased number of working memory errors and reference memory errors in radial-arm maze for both losartan + scopolamine and ramipril + scopolamine groups versus scopolamine alone. This could have a therapeutical relevance, especially since oral administration was preferred in our report, as it is used in the therapeutic procedures in humans, further enhancing the similarities with the clinical conditions.
Introduction
The renin–angiotensin aldosterone system (RAAS) has been a major target for antihypertensive medications, including angiotensin-converting enzyme (ACE) inhibitors (ACEi), angiotensin (Ang) receptor blockers, aldosterone antagonists, and renin inhibitors. Among these, the most commonly used are ACEi and Ang II receptor blockers (ARB), considering their ease of administration (oral dosage, good pharmacodynamical characteristics), reproductible results, and a good tolerance. There are no exhaustive data concerning the number of patients receiving RAAS-altering drugs, but given the fact they are used in arterial, hypertension, acute myocardial infarction, heart failure, diabetic nephropathy, stroke, and other conditions, the number of patients has reached hundreds of millions throughout the world. 1 On the other hand, the RAAS exists in all mammalian brain, 2 with the Ang II playing an important neuromodulator role in the central nervous system where it interacts with different neurotransmitters such as acetylcholine, catecholamines, serotonin, and other peptides. 3 This relationship has been studied in detail, using various types of behavioral and biochemical techniques, demonstrating that these alterations in the RAAS can modulate various memory and behavioral responses. 4,5
Some reports showed that centrally administered Ang II disrupted memory in various working and spatial memory tasks in rodents, 6 –8 while other authors showed some positive effects of Ang II administration in this matter. 9,10 On the other hand, Ang II blockers (both ACE blockers or AT receptors antagonists) were shown to enhance retention performance in a variety of short-term and long-term memory behavioral tasks. 7,8,11,12 However, even in this case there are studies showing controversial effects. 13,14
Still, based on their reported protective effects, the Ang II blockers are proposed as a possible mechanistical or even therapeutical avenue in an increased number of neuropsychiatric deficiencies, including Alzheimer’s disease (AD), 15,16 Parkinson disease (PD), 17 anxiety, 18,19 schizophrenia, 20,21 or autism. 22
Bearing this in mind, this experiment was designed to evaluate the spatial working memory (as studied in Y-maze) or both short-term and long-term spatial memory (assed in the radial 8 arms-maze task), in a scopolamine-induced memory deficits model in mice, by the oral administration of 2 ACEi—captopril and ramipril and also the effect of the AT1 receptor antagonist, losartan. In fact, the present article was initiated as a reaction to the clinical setting of hypertensive disease, which involves lifelong administration of antihypertensive drugs, dietary, lifestyle constraints, and aging, which all take a toll on the higher functions of the nervous system. In this way, most of the patients with cognitive decline suffer of various metabolic imbalances, hypertension, cardiac, and kidney disease, many of them which are treated with oral administration of RAS-altering agents like those presented above.
Material and Methods
Animals
The experimental protocol involved the use of male adult Swiss mice (n = 128), from Cantacuzino Institute in Bucharest, weighing 20 to 30 grams, randomly distributed throughout the study groups described below (separate groups were used for the 2 separate behavioral tasks described below). Habitation and transit were performed according to the protocols for each test, in the vivarium of the Department of Pharmacodynamics from the Faculty of Pharmacy in Iasi, Romania, being maintained in a constant temperature (21°C) and a 12-hour light/dark cycle. The animals were placed in special mice cages (n = 8 per cage), with food and water ad libitum. We performed the habitation behavior 15 days before the experiments, observing appetite, water intake, digestive transit, and neurologic signs and behavior (eg, socialization, group behavior).
The animals were divided in groups of 8 to 16 subjects, as follows: for the Y-maze task we randomly divided 78 mice into 5 groups as following: 16 animals were designated to be in the control group, 16 animals in the scopolamine group, 15 animals in the captopril + scopolamine group, 16 animals in the losartan + scopolamine group, and 15 animals in the ramipril + scopolamine group. For the radial maze task, 46 mice were randomly divided into 6 groups as it follows: 8 mice were selected for the control group, 8 mice in the scopolamine group, 8 mice in the losartan group, 8 mice in the losartan + scopolamine group, 7 mice in the ramipril group, and 8 mice in the ramipril + scopolamine group. The exact dosage administered and administration time for the Y-maze and radial maze tasks can be also seen in Table 1.
The Drugs Dosage Used for the Y-Maze Task and Radial-Arm Maze Task.
Abbreviations: IP, intraperitoneally; PO, orally.
Drugs
Drugs were administrated only for the duration of the behavioral tasks (different animals for the 2 different behavioral tests used—8 days for radial-8-arms maze and 1 day for Y-maze task), as described in Table 1. Ramipril (Zhejiang Huahai Pharmaceutical Co, Ltd, Zhejiang, China), captopril, and losartan (Sigma-Aldrich, Missouri) were homogenized in a suspension of 0.1% NaCMC (sodium carboxymethylcellulose), prepared extemporaneously daily and administered by gavage using a gastric probe, connected to a single-use 1 mL syringe. The procedure was nontraumatic for the mouse, which was gently held in the hand of the experimenter. The administered volume did not exceed a hydration amount of 0.5 mL/25 grams of animal. Scopolamine (Sigma-Aldrich) was dissolved in 0.9% saline and administered intraperitoneally.
Oral administration of the drugs was preferred because they have a good bioavailability, and the parenteral administration would have introduced a supplemental stress and pain, which might have altered the results. Also, oral administration is used in the therapeutic procedures in humans, further enhancing the similarities with the clinical conditions. Captopril was used as being the golden standard for oral ACEi and it is the only one which is not used as a prodrug. Ramipril, a prodrug or precursor drug, is converted to the active metabolite ramiprilat by carboxylesterase 1, 23 which further encourages its use orally. Losartan is administered orally and suffers a first-pass metabolization for producing its active metabolite, marked EXP3174, which is much more potent than losartan. 24
The behavioral tasks we used were represented by the Y-maze and 8-arm radial maze tasks.
The Y-maze used in the present study consisted of 3 arms (35 cm long, 25 cm high, and 10 cm wide) and an equilateral triangular central area. The rat was placed at the end of one arm and allowed to move freely through the maze for 8 minutes. An arm entry was counted when the hind paws of the rat were completely within the arm. Spontaneous alternation behavior was defined as entry into all 3 arms on consecutive choices. The number of maximum spontaneous alternation behaviors was calculated as total number of arms entered minus 2, and percent spontaneous alternation was calculated as (actual alternations/maximum alternations) × 100. 25
Regarding the radial-8-arm maze, it consisted of 8 arms, numbered from 1 to 8 (48 × 12 cm), extending radially from a central area (32 cm in diameter). The apparatus was placed 40 cm above the floor and surrounded by various extramaze visual cues placed at the same position during the study. At the end of each arm, there was a food cup that had a single 50-mg food pellet. Prior to the performance of the maze task, the animals were kept on restricted diet, and body weight was maintained at 85% of their prefeeding weight over a week period, with water being available ad libitum. Before the actual training began, 3 or 4 rats were simultaneously pretrained in the radial maze and allowed to explore for 5 minutes and take food freely. The food was initially available throughout the maze but was gradually restricted to the food cup. The animals were pretrained for 3 days to run to the end of the arms and consume the baits. The pretraining trial continued until all the baits had been consumed or until 5 minutes had elapsed. After pretraining, all rats were trained with one trial per day, for 8 days. Briefly, each animal was placed individually in the center of the maze and subjected to working and reference memory tasks, in which same 5 arms (nos. 1, 2, 4, 5, and 7) were baited for each daily training trial. The other 3 arms (nos. 3, 6, and 8) were never baited. An arm entry was counted when all 4 limbs of the rat were within an arm. Measures were made of the number of working memory errors (entering an arm containing food but previously entered) and reference memory errors (entering an arm that was not baited). Also, the time taken to consume all 5 baits was recorded. Reference memory is regarded as a long-term memory for information that remains constant over repeated trials (memory for the positions of baited arms), whereas working memory is considered a short time memory in which the information to be remembered changes in every trial (memory for the positions of arms that had already been visited in each trial). 26
The aforementioned dosage and the duration of treatment were selected using our pilot studies and previously published reports regarding RAS behavioral effects. 27,28 The animals were treated in accordance with the guidelines of animal bioethics from the Act on Animal Experimentation and Animal Health and Welfare Act from Romania and local ethics decisions (decision from 06/10/2016), and efforts were made to minimize animal suffering and to reduce the number of animals used.
Data Analysis
The animal’s behavior in Y-maze was analyzed by using Student t test (2 tailed, unpaired). The 8-arm radial was analyzed by 2-way analysis of variance (ANOVA) repeated measures for treatment effect (control versus groups), time effect (days of training), and treatment × time interaction. We used the Tukey-Kramer post hoc test for our unequal sample sizes. All results are expressed as mean ± standard error of the mean. P < .05 was regarded as statistically significant. The data were analyzed with parametric tests as they are expected to fall within a well-defined interval of the results. The skewness cannot be altered significantly by outliers in the results. Also, all over the literature, the results of the behavioral tests are analyzed by using tests such as Student t test (2 tailed, unpaired) or 2-way ANOVA repeated measures, which should point toward its appropriateness. Still, regarding the sample characteristics, a Shapiro-Wilk test (P = .766), a Kolmogorov-Smirnov (P = .200) and a visual inspection of their histograms, normal Q-Q plots, and box plots showed that the number of spontaneous alternations were approximately normally distributed in our experimental groups with a skewness of 0.037 (standard error [SE] = 0.272) and a kurtosis of −0.476 (SE = 0.538). Furthermore, a Shapiro-Wilk test (P = .357), a Kolmogorov-Smirnov test (P = .200) and a visual inspection of their histograms, normal Q-Q plots, and box plots also showed that the number of entries were approximately normally distributed in our experimental groups with a skewness of −0.349 (SE = 0.272) and a kurtosis of −0.276 (SE = 0.538).
Results
Y-Maze Task
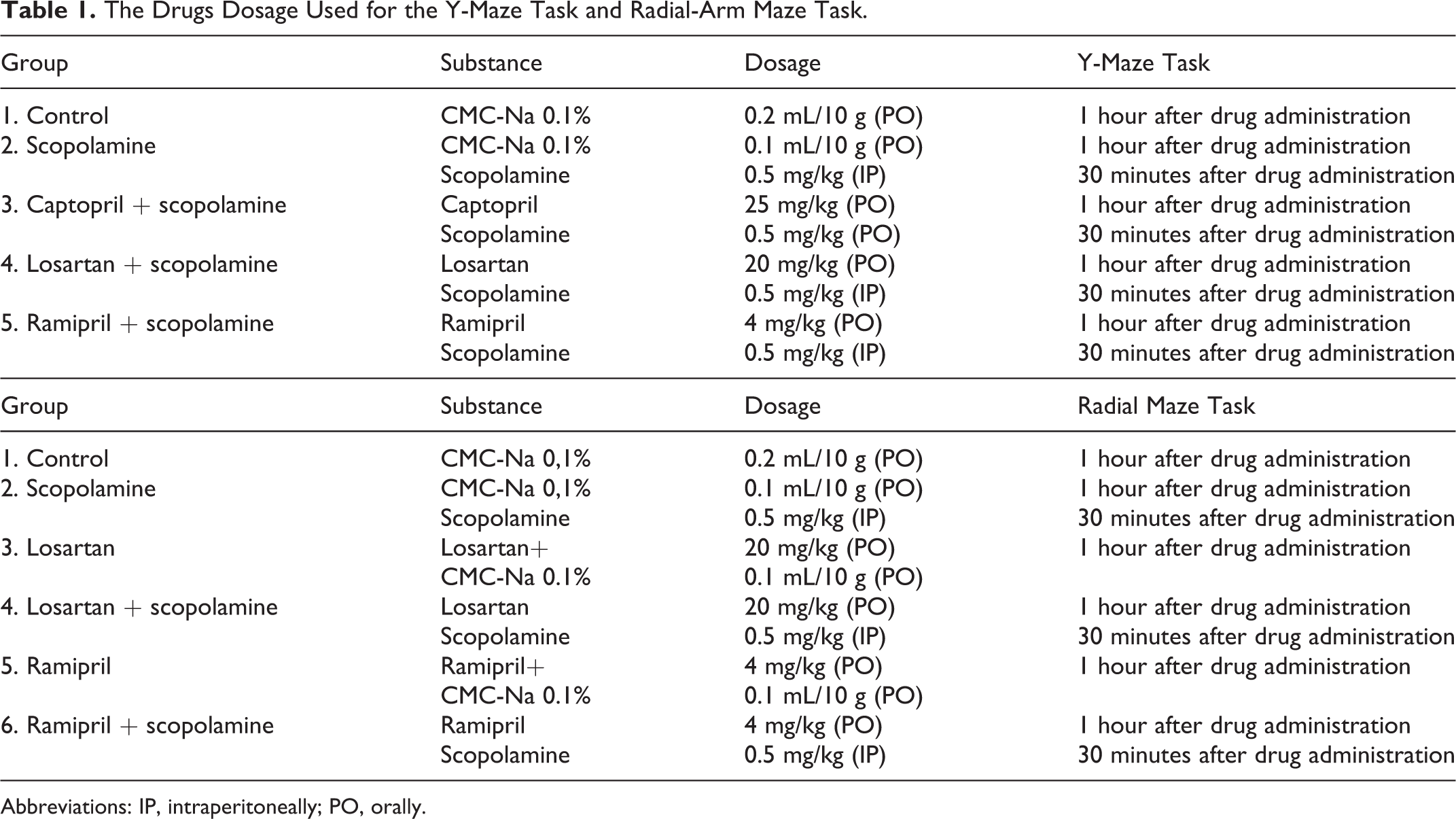
Regarding the spatial working memory in the Y-maze task, our ANOVA analysis showed significant differences (P < .001) between the 5 groups of mice (Figure 1A), suggesting that the treatment received by the mice in any of the experimental group had significant effects on spatial working memory. Additionally, post hoc comparisons showed a significant decrease in the spontaneous alternations percentage in the scopolamine group, as compared to the control group (P = .041). Furthermore, all 3 experimental group receiving Ang II blockers showed a significant increase in the spontaneous alternation percentage, as compared to scopolamine alone group: P = .017 when compared with the captopril + scopolamine group, P = .005 when compared to the losartan + scopolamine and P < .001 when compared to the group which received ramipril + scopolamine. Still, no significant differences were found between any other possible pair of groups remained (eg, Ang II blockers between them or versus control mice; Figure 1A).
The effects of captopril, losartan, and ramipril administration on the spontaneous alternation percentage in the Y-maze task. The values are mean ± standard error of the mean (SEM). A, Ang II blockers vs. control mice. B, Comparison between the five groups of mice.
Furthermore, the locomotor activity in the Y-maze task, as expressed through the number of arm entries, was not significantly different (P = .12) when we compared the 5 groups of mice (Figure 1B). This suggests that the aforementioned effects of Ang II blockers on memory were not related to some motor deficiencies in these mice.
Eight-Arm Radial Maze Task
Working memory errors
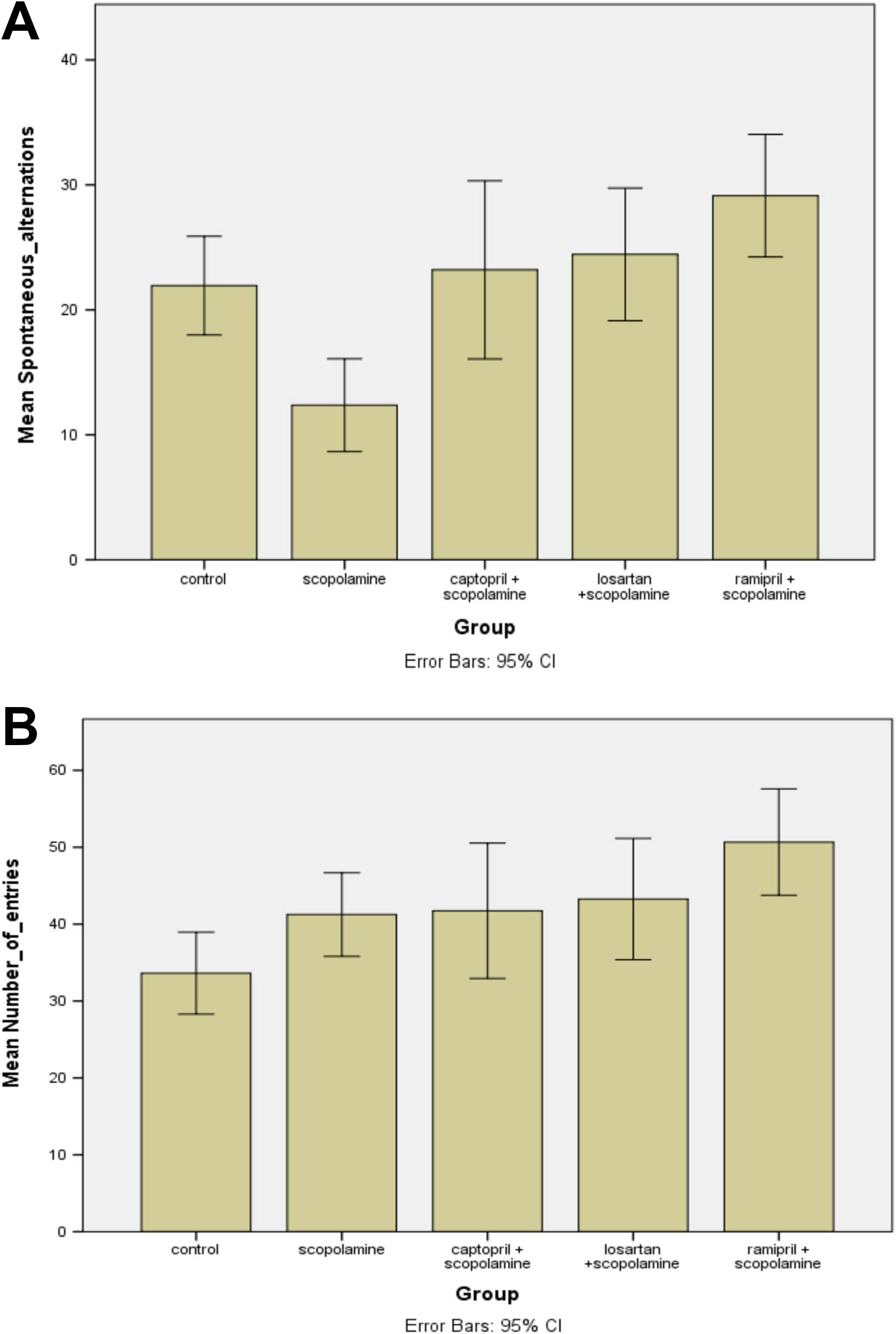
Regarding the number of working memory errors in the radial arm maze (Figure 2A), the interaction effect of time × group was significant, Wilks’ λ = 0.121, F 35,149 = 2.78; P < .001. Furthermore, a significant main effect of time was also observed, Wilks’ λ = 0.523, F 7,3 = 4.563; P = .001.
The effects of captopril, losartan, and ramipril administration in the 8-arm radial maze task. A, Working memory errors. B, Reference memory errors. C, Time taken to consume all 5 baits. The values are mean ± standard error of the mean (SEM).
When we compared all the 6 groups, there was also a significant difference between them regarding the working memory errors, F 1,5 = 6.96; P = .001.
Also, post hoc comparisons between groups showed a significant increase in the working memory errors in the scopolamine group versus controls (P < .001), and in the losartan-treated mice versus scopolamine group (P = .014; Figure 2A). Still, no significant difference was found between control versus losartan + scopolamine, control versus ramipril or between control group versus ramipril + scopolamine group. Scopolamine group showed also an increased number of errors as compared to losartan + scopolamine mice (P = .022), to ramipril group (P = .017) and to ramipril + scopolamine group (P = .006).
No significant difference was found between the control group and losartan group. Furthermore, the losartan group presented no significant differences when compared to any of the groups, except the control group as mentioned above. In addition, the losartan + scopolamine group, the ramipril group and the ramipril + scopolamine group were significant different only from the scopolamine group, as also mentioned above. In rest, no significant differences were found when we paired any of these 3 groups with any other possible experimental group (Figure 2A).
Reference memory errors
Regarding the number of reference memory errors in the radial arm maze, the interaction effect of time × group was significant, Wilks’ λ = 0.175, F 35,149 = 2.1; P = .001. Furthermore, there was a significant main effect of time, Wilks’ λ = 0.383, F 7,35 = 8.04; P < .001.
When we compared all the 6 groups, there was significant difference between them regarding the reference memory errors, F 1,5 = 8.094; P < .001 (Figure 2B).
Regarding the post hoc comparisons between groups, we showed a significant decrease in the reference memory errors in the controls versus scopolamine group (P < .001) and a significant decrease in the reference memory errors in the losartan group versus controls (P = .001). In addition, no significant difference was found between control versus losartan + scopolamine, control versus ramipril or between control versus ramipril + scopolamine group.
The scopolamine group showed also a significant increase in the number of reference memory errors as compared to losartan + scopolamine group (P = .004) and to ramipril + scopolamine-treated mice (P = .006; Figure 2B).
The time necessary to finish the 8-arm radial maze
Regarding the time necessary to finish the 8-arm radial maze, the interaction effect of time × group was significant, Wilks’ λ = 0.272, F 35,149 = 1.553; P = .037. There was also a significant main effect of time, Wilks’ λ = 0.256, F 7,35 = 14.496; P < .001.
In addition, when we compared all the 6 groups, there was a significant difference between them regarding affecting time necessary to finish the 8-arm radial maze, F 1,5 = 7.616; P < .001 (Figure 2C).
Also, post hoc comparisons between groups showed a significantly increased time (expressed in seconds) in the scopolamine group as compared to control mice (P = .001), to scopolamine + losartan group (P < .001), to ramipril group (P = .001), and also to ramipril + scopolamine group (P = .003), but not between control and losartan group (P = .270). Still, no other significant difference was observed between when we paired any other remaining treatment groups (Figure 2C).
Discussion
Our results showed a protective effect of captopril, ramipril, and losartan prescopolamine administration on spontaneous alternation in Y-maze task, as compared to scopolamine-alone treated mice, as well as decreased number of working memory errors and reference memory errors in radial-arm maze for both losartan + scopolamine and ramipril + scopolamine groups versus scopolamine alone.
This study was designed to evaluate the spatial working memory (as studied in Y-maze) or both short-term and long-term spatial memory (assed in radial 8 arms-maze task), in a scopolamine-induced memory deficits model in mice, by the oral administration of 2 ACEi—captopril and ramipril and an AT1 receptor antagonist, losartan.
Although it is now clear that besides blood pressure, the separate RAAS in the brain exerts also a variety of superior functions related to memory, anxiety, pain perception (as our group previously demonstrated for all these aforementioned aspects 23,27,28 ), there are still some controversies regarding the functions of Ang II in the memory processes (eg, the Braszko group stated clear facilitatory on memory effects for Ang II administration in the passive avoidance task, possibly through a transformation in Ang IV, 7,8 while other groups showed negative effects of Ang II on cognitive processes 4 –6 or no effects at all, 11,12 as well as for some of its blockers (eg, which are mainly cited for their positive effects in the memory functions for example in light-dark test, 9 in water maze, 10 1-way avoidance including genetic and morphological deficiencies 6 or shuttle-box, 5 while other groups clearly stated that in some experimental situations, such as the one described by Sheperd et al, 14 selective AT 1 and AT 2 receptor blockers exhibited no significant effects on the working memory.
It seems that these differences could be explained by the variety of behavioral tests used, the various dosages and experimental situations or the animal models implied.
Also the mechanistic behind these effects could include oxidative stress modification, for example, as our group also previously demonstrated, with increased levels of malondialdehyde, as a lipid peroxidation marker and decreased levels of antioxidant enzymes in the hippocampus of mice, as a result of Ang II intracerebroventricular (ICV) administration, as well as by the antioxidant effects of ICV captopril administration. 25
In addition, in other very recent papers, it was showed that Ang II blockers are attenuating lipopolysaccharide-induced memory deficits through some complex effects that include anti-inflammatory actions mainly exerted on markers such as TNF-α, GFAP, COX-2, and NF-κB, which could be mediated by the BDNF/CREB expression and apoptosis. 29
In addition, recent review works are stressing out the correlations between ACE, β-amyloid accumulation and inflammation, which could establish some promising therapeutical avenues for the aforementioned ACE blockers in AD. 15
In fact, lately there is an increased interest in the connections that might exist between RAAS and some neuropsychiatric disorders, starting of course with AD, where some authors are proposing even a link between mild cognitive impairment (a nosological entity proposed as an intermediate state between normal aging and dementia, where our group also showed among the first a similar pattern of decrease for antioxidant enzymes as in AD 30 —AD and RAAS (as reviewed by O’Caoimh et al 16 ).
Same goes with the PD where AT1 receptor levels were found to be decreased by 90% in the substantia nigra, 15 as well as in anxiety and depression, 16,17 but also autism where some data previously suggested the involvement of RAAS genetic diversity in increasing the risk of autism 20 or other psychiatric deficiencies where the memory processes are also affected, such as schizophrenia where data suggest that the elevated cerebrospinal fluid ACE found in schizophrenic patients may imply an activated RAAS. 18,19
Regarding the mechanisms through which these effects might occur, it is known that Ang II can be metabolized into several peptides with biological activity, among which Ang 1 to 7, Ang IV, Ang 5 to 8, and others. 31,32
In this way, several authors found a direct connection between the administration of ACEi or ARB’s and the increase in the cerebral levels of Ang IV or in the expression of its receptors. 5,33 However, without any investigation toward the amounts of circulating or local Ang fragments, it would be useless to insist in this direction, except as a hypothesis.
We should also mention in this context the new intranasal approach administration for some peptides including those from RAAS, as well as the existence of some scarce preliminary results in this area of research, where the group of Danielyian, which demonstrated protective effects for losartan intranasal administration in an APP/PS1 transgenic mouse model of AD. 34 Also, our group is currently working on a complex experimental design implying the administration of Ang II blockers intranasally and their cognitive effects in this context.
Regarding the limitations of our report, we could mention here the lack of an Ang II-treated group, but as the purpose of this article was to keep as close to the clinical settings as possible, and there are no clinical situations in which Ang II is used, either as internal secretion or as external source, we decided to design the study in this direction.
Conclusions
We report here a protective effect of captopril, ramipril, and losartan prescopolamine administration on spontaneous alternation in Y-maze task, as compared to scopolamine-alone treated mice, as well as decreased number of working memory errors and reference memory errors in radial-arm maze for both losartan + scopolamine and ramipril + scopolamine groups versus scopolamine alone. These aspects could show some therapeutical relevance, especially since oral administration was preferred in our design, since it is used in the therapeutic procedures in humans, further enhancing the similarities with the clinical conditions.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are stating that there is no conflict of interest to disclose, except for Ciobica Alin who is supported by a Young Teams grant offered by UEFISCDI Romania, no. PN-III-P1-1.1-TE-2016-1210, contract no. 58 from 02/05/2018.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
