Abstract
There has been no comparison of excessive daytime sleepiness (EDS) in patients with Alzheimer’s disease dementia (AD), dementia with Lewy bodies (DLB), and behavioral variant frontotemporal dementia (bvFTD). We identified patients with mild dementia who met criteria for these disorders who also had the Epworth Sleepiness Scale (ESS) completed. The sample included 17 bvFTD, 111 AD, and 31 DLB. An ESS score ≥10 was considered abnormal and consistent with EDS. Analyses with age and sex as covariates revealed higher mean ESS scores for DLB compared to the other groups (DLB 13.9 [5], bvFTD 9.6 [8], AD 8.8 [5],
Keywords
Introduction
Excessive daytime sleepiness (EDS) is a common feature among aging individuals, especially in those with dementia. The EDS has been described in dementia with Lewy bodies (DLB), 1 –4 but has not been well characterized in terms of its frequency and severity. Less is known regarding EDS in patients with Alzheimer’s disease dementia (AD) and behavioral variant frontotemporal dementia (bvFTD). 1 –8 The Epworth Sleepiness Scale (ESS) is a measure that assesses subjective sleepiness over the prior 2 weeks. 9 In most settings, the patient completes the measure. In dementia populations, this measure is typically completed by informants. 1 We sought to compare ESS scores between patients with mild DLB, AD, and bvFTD as rated by their informants, and hypothesized that patients with mild DLB would have greater degrees of severity and frequency of EDS compared to those with mild bvFTD and AD.
Material and Methods
The Mayo Clinic Alzheimer’s Disease Research Center (ADRC) database was queried to identify patients evaluated over a 10-year period who (
The Mayo Sleep Questionnaire (MSQ) 14 is always completed concurrently with the ESS at our center. An affirmative response to question 1 on the MSQ (Have you ever seen the patient appear to “‘act out his/her dreams”’ while sleeping? [punched or flailed arms in the air, shouted, or screamed]) was considered consistent with probable rapid eye movement sleep behavior disorder (RBD). The MSQ has been validated and is 98% to 100% sensitive and 74% to 95% specific for polysomnography-confirmed RBD. 14,15
Descriptive statistics were used to compare demographic data, total ESS scores, and the frequency of ESS score ≥10 among the 3 dementia syndrome groups. Analysis of covariance (ANCOVA) was used to compare ESS scores in AD versus DLB versus bvFTD. A
Results
There were 1044 patients with the qualifying diagnoses with the following breakdown: 641 AD, 250 DLB, and 153 FTD. Among these, 159 had the ESS completed and had a CDR = 1, resulting in the sample (total = 159) being 111 AD, 31 DLB, and 17 bvFTD. All patients were Caucasian.
There were no group differences in education (mean 14.9 [3]). Male representation was lowest in AD (52%) compared to bvFTD (77%) and DLB (88%;
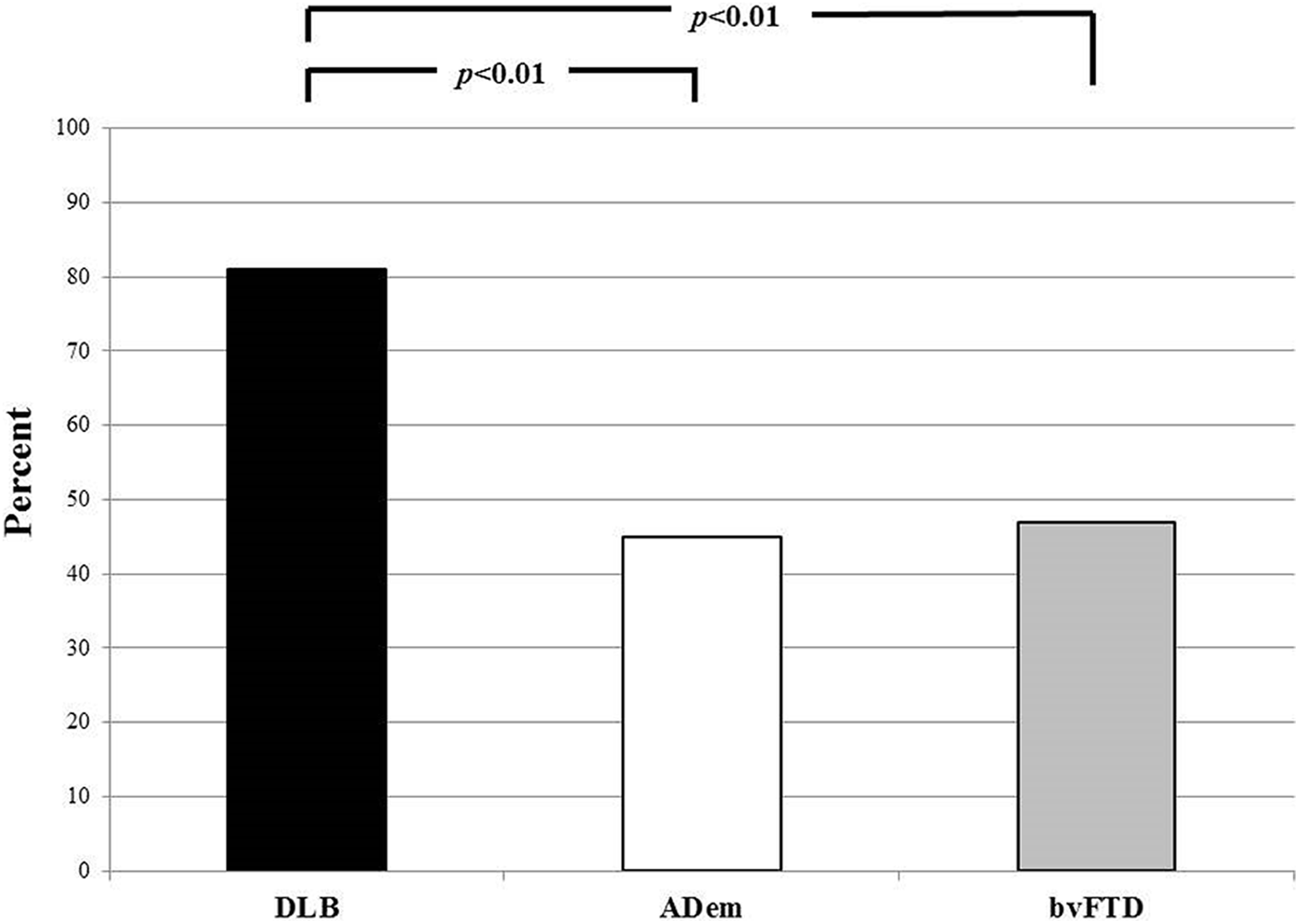
Frequency of excessive daytime sleepiness (EDS) in patients with dementia with Lewy bodies (DLB) versus Alzheimer’s disease (AD) dementia versus behavioral variant frontotemporal dementia (bvFTD).
Discussion
Patients with mild DLB had a higher frequency and greater severity of EDS compared to those with AD and bvFTD who also had a mild dementia. Excessive daytime sleepiness (EDS) has been described in patients with AD and bvFTD, but the degree and frequency was similar for those 2 groups and is not as prominent as in patients with DLB. 8,16
Our data are similar to that reported by others on DLB, in which hypersomnia has been shown based on subjective (ie, the ESS) and objective (ie, the multiple sleep latency test and maintenance of wakefulness test) measures in DLB. 1 –4,8,17
The underlying cause(s) for EDS being more frequent and severe in DLB compared to AD and bvFTD is likely multifactorial. The EDS can be due to obstructive or central sleep apnea, sleep fragmentation for many reasons, restless legs syndrome, effect of medications, depression, and so on as well as being intrinsic aspects of the underlying disease processes themselves. 18 Our analyses focused on those with mild dementia, and our data underscore the potential discriminative value of EDS in differentiating DLB versus AD versus bvFTD early in the dementia course. The EDS is also now considered a supportive clinical feature for the diagnosis of DLB. 12 Whether EDS has any prognostic implications in terms of rate of progression for any of the dementia subtypes will require further study.
In the setting of dementia, EDS as well as RBD can be viewed as risk factors, prodromal markers, or parts of the symptom complex. Our data on EDS pertain to the symptom complex issue. Recent findings suggest an association between β-amyloid deposition (as reflected by amyloid positron emission tomography imaging) and EDS in the cognitively normal elderly patients. 19 Future analyses are necessary to determine whether EDS is a risk factor or prodromal marker of 1 or more of the major dementia subtypes.
There is compelling evidence that RBD is a prodromal feature of the synucleinopathies, including DLB. 20,21 While RBD is far more common in DLB than in AD and bvFTD, 20,21 which is also supported by our data, EDS is not usually associated with RBD. 22,23
We acknowledge the limitations of this study. This is a retrospective study using a measure of subjective sleepiness as reported by an informant. We did not control for other sleep comorbidities such as obstructive sleep apnea since most patients had not undergone a polysomnogram. However, in contrast to the other studies described above that included patients with mild-to-severe dementia, we purposefully focused on those with mild dementia (as reflected by the global CDR score being 1) and adjusted comparisons based on age and sex, which strengthen the finding of EDS being more significant in DLB compared to AD and bvFTD. The large differences in age and sex led us to control for these in our analyses. The small number of females (N = 4 DLB and N = 4 bvFTD) did not allow us to stratify our analyses by sex.
Future directions should include determining whether EDS in the prodromal phase of each dementia syndrome (ie, mild cognitive impairment [MCI]) may be predictive of eventual phenoconversion from normal to MCI or MCI to dementia. In other words, one could hypothesize that EDS in the setting of MCI may predict phenoconversion to DLB versus AD or bvFTD. Another key point is that EDS is a problematic symptom/feature in DLB which is worthy of treatment trials; while one open-label pilot study in DLB has suggested improvement in EDS with armodafinil, 17 double-blind placebo-controlled studies with wake-promoting agents are worthy of further investigation.
Footnotes
Authors’ note
Angelica Boeve is now affiliated with Department of Psychology, University of Maine, Orono, ME.
Acknowledgments
We extend our appreciation to the patients and their caregivers for participating in aging/dementia research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Neill Graff-Radford, MD, receives research support from NIH, has received publishing royalties from UpToDate, Inc, and receives research support from Biogen, Lilly, Axovant, and Novartis. He has consulted for Cytox. Michelle Mielke, PhD, served as a consultant to Eli Lilly and Lysosomal Therapeutics, Inc. She receives research support from the NIH and unrestricted research grants from Biogen, Lundbeck, and Roche. David Knopman, MD, serves on a Data Safety Monitoring Board for Lundbeck Pharmaceuticals and for the DIAN study; is an investigator in clinical trials sponsored by TauRX Pharmaceuticals, Lilly Pharmaceuticals, Biogen and the Alzheimer’s Disease Cooperative Study; and receives research support from the NIH. Ronald Petersen, MD, PhD, serves on data monitoring committees for Pfizer, Inc, Janssen Alzheimer Immunotherapy; is a consultant for Biogen, Roche, Inc, Merck, Inc, and Genentech, Inc; receives publishing royalties from Mild Cognitive Impairment (Oxford University Press, 2003); and receives research support from the NIH. Brad Boeve, MD, is an investigator for clinical trials sponsored by Axovant and Biogen; receives royalties from the publication of a book entitled
