Abstract
The spectrum of cognitive decline in the elderly ranges from what can be classified as normal cognitive decline with aging to subjective cognitive impairment to mild cognitive impairment (MCI) to dementia. This article reviewed the up-to-date evidence of MCI including the diagnostic criteria of MCI due to Alzheimer’s disease, vascular cognitive impairment and MCI due to Parkinson disease, management and preventive intervention of MCI. There are various etiologies of MCI, and a large number of studies have been conducted to ascertain the practical modalities of preserving cognition in predementia stages. Lifestyle modification, such as aerobic exercise, is an approved modality to preserve cognitive ability and decrease the rate of progression to dementia, as well as being recommended for frailty prevention.
Keywords
Introduction
The size of the elderly population has been dramatically increasing worldwide. In 2017, people aged 60 or older accounted for 13% of the global population at about 962 million people. The size of this population is predicted to rise to 1.4 billion, 2.1 billion, and eventually 3.1 billion people by 2030, 2050, and 2100, respectively. 1 Furthermore, this population accounts for a higher proportion of total medical expenses than do younger age groups; one important factor is due to frailty. 2 Frailty is one of the geriatric syndromes caused by declining body reserve in multiple vital systems, characterized by decreased ability to tolerate acute stress and increased vulnerability of unfavorable clinical outcomes such as falls, disabilities, hospitalization, and death. 3 -5 The interrelationship between physical frailty and cognitive impairment is apparent. It leads to worsening physical and cognitive function and poor quality of life. 6 Cognitive frailty is defined as the co-occurrence of physical frailty and cognitive decline in older people without dementia. It is associated with more adverse health outcomes than patients with prefrailty and frailty without cognitive impairment, according to the population-based cohort in Singapore with the prevalence of 10.7%. 7 The China Cognitive Frailty, a study of 5708 community-dwelling elderly people without dementia, found that the prevalence of cognitive frailty was 2.7% and increases with age. 5 To maintain independency in older adults, focusing on cognitive function is the novel target concern since some causes of cognitive decline might be reversible or potentially reversible/treatable. Therefore, understanding cognitive decline in older adults is one of the important issues.
Normal aging can cause psychomotor slowing, decreased visual and auditory acuity, decreased vibratory sensation, smaller pupil size, upward gaze paresis, decreased muscle bulk, decreased Achilles tendon reflex, minimal swaying as measured by the Romberg test, mild lordosis, and limitation of movement in the neck and back. Additionally, while some cognitive functions are preserved, others tend to decline. 8 In normal aging, sustained attention, simple copy, remote, and procedural memory are preserved while divided attention, learning new information, verbal fluency, and reaction time tend to deteriorate. 8 The spectrum of cognitive decline in older adults ranges from what can be classified as normal cognitive decline with aging to subjective cognitive impairment (cognitive complaint with normal cognitive screening test) to mild cognitive impairment (MCI) to dementia. This review focused on finding up-to-date information with regard to prevalence, diagnosis, pathogenesis, outcomes, subtypes, and management of MCI due to Alzheimer’s disease, vascular cognitive impairment (VCI), and Parkinson disease by classifying as pharmacological and nonpharmacological management and prevention of MCI.
Prevalence of MCI
Mild cognitive impairment or mild neurocognitive disorder is an intermediate state 9 -15 between normal aging and dementia. This state can progress to dementia, mostly in the form of Alzheimer’s disease. 16,17 The prevalence of MCI in adults older than 60 is approximately 6.7% to 25.2%. It increases with age and lower level of education and is more prevalent in men. 9 -12 The prevalence is varied due to differences in definitions of MCI used in most studies. Formerly, MCI was defined by focusing mainly on amnesia, but it later includes a wider definition that covers either impairment in single-domain nonamnestic or several cognitive domains with or without memory deficit. 9 The annual rate of progression to dementia is approximately 5% to 17%. 13 -15,18 Some established biomarkers associated with the progression from MCI to Alzheimer’s disease are a positive amyloid positron emission tomography (PET) scan, apolipoprotein E4 genotype, abnormal cerebrospinal fluid (CSF) tau levels, a positive PET scan due to tau deposition into the lateral temporal lobe structures. 11 -13,15,19,20
Diagnosis of MCI
The diagnostic criteria for MCI include concern regarding a change in cognition, abnormal cognitive function in one or more domains, normal daily activity, and absence of dementia. 13,21 A thorough interview regarding the patient’s history from knowledgeable informants in order to detect the clinical clues is fundamental in making diagnosis. Adding appropriate cognitive screening tests is another crucial part for clinical evaluation of patients with MCI. The Montreal Cognitive Assessment (MoCA) with a cutoff point of 24/25 is the recommended cognitive screening tool for MCI. The sensitivity and specificity of the test have been found to be 80.48% and 81.19%, respectively. 22 At the cutoff point of 25/26, it had sensitivity of 80% and 100% and specificity of 50% to 76%. 12 However, it does get affected by educational level, lifestyle factors, and ethnic diversities. 23,24 For example, the Cantonese Chinese version with a cutoff point of 22/23 showed a sensitivity of 78% and specificity of 73% in detecting amnestic type of MCI. 25 A study in Canada showed that adjusting the MoCA total score for education decreased its overall sensitivity from 80% to 69% and small increment of specificity from 89% to 92%, and at the best cutoff point of 24/25, it provided sensitivity and specificity of 61% and 97%, respectively. 23
The Mini-Mental Status Examination (MMSE) and the Dementia Rating Scale (DRS) are not recommended as screening tools for MCI due to their limitations with regard to detecting abnormal cognitive function. 22,26 The potential explanations regarding the limitation of the MMSE performances in detecting MCI are the cultural factors, educational level, factors related to favored language use, and correlation of cognitive domains in early cognitive deficits. For example, the level of education did affect the MMSE score, particularly in no-memory scores (orientation, attention, and language) among older Mexican Americans. 22,26,27 Furthermore, a study comparing the characteristic differences in the MMSE used across Asian countries found that it was an unstandardized cognitive tool due to a variety in administration and contents. 28 For the usefulness of the DRS in identifying persons with MCI, though it could predict declined function and incident of dementia in some studies, 29,30 Clinical dementia rating (CDR) scores did not have a good correlation with MCI. 31 An overall score of 0.5 veiled the diverse functional status of individuals. Some persons with MCI based on the DRS had extensive brain pathology and poorer episodic memory and executive functions and greater risk of developing dementia; therefore, using global CDR alone appears to be an imperfect tool for detecting MCI. 32 The Cochrane Database of Systematic Reviews found that the sensitivities of the MMSE in detecting the progression from MCI to dementia ranged from 23% to 76%, 27% to 89% for MCI to Alzheimer, and 36% for MCI to vascular dementia, and the corresponding specificities were 40% to 94%, 32% to 90%, and 80%, respectively. 33 At the cut points of 27 or 28 of the MMSE in detecting MCI, the sensitivities were varied from 45% to 60% and the specificities were 65% to 90%. 12
One systematic review showed that the area under the receiver operating characteristic (ROC) curve of the MoCA in detecting MCI at the cutoff point of 24/25 was 0.846 (95% confidence interval [CI]: 0.823-0.868) with a sensitivity of 80.48% and specificity of 81.19%. For the MMSE, the area under the ROC curve at the cutoff point of 27/28 was 0.736 (95% CI: 0.718-0.767) with a sensitivity of 66.34% and specificity of 72.94%. 27 A direct comparison of the MoCA and the MMSE also reported that MoCA was more sensitive for precisely differentiating persons with MCI from those with normal cognitive function. 12 Although MoCA is recommended primarily in MCI screening in several studies, 9,16,27 there are some limitations as described above; clinical judgment including premorbid functioning such as intellectual function and occupational status remains the essential elements in diagnosing MCI.
Pathogenesis and Outcomes of MCI
Dementia resulting from decades of proteinopathy, such as Alzheimer’s disease, is associated with amyloid-β deposition: extraneuronally neuritic plaques and intracellularly neurofibrillary tangles. For progressive supranuclear palsy, corticobasal degeneration, and frontotemporal lobar degeneration, they are classified as tauopathy, and for Lewy body dementia and Parkinson disease dementia, they are synucleinopathy. 34 However, there are other important factors in the pathogenesis of dementia such as sedentary lifestyle, poor nutritional status, social or environmental factors, and genetic factors that could be modifiable. 34
The various etiologies of MCI such as systemic diseases, neurological diseases, medications, and psychiatric disorders lead to heterogeneous outcomes. 13 There are few outcomes of MCI: reversion to normal aging, stability, or progression to dementia, which can be explained by its pathogenesis. 14,18,35,36 Basic investigations for metabolic conditions are recommended because of the atypical presentations of these conditions in older adults. Neuroimaging should be performed selectively as clinically indicated. Totally reversible causes are rare and mostly occur in surgical and depressive patients.
Subtypes and Their Management of MCI
Many studies have been carried out in order to better understand the pathogenesis of dementia, especially Alzheimer’s disease. More recent studies have shown more insight in pathogenesis such as the fact that autophagy and microglia function have important roles in cognitive ability. 37,38 Although there have been innovative knowledge about the pathogenesis of dementia (especially Alzheimer’s disease), there are currently only 2 strategies to deal with dementia: symptomatic relief and behavioral intervention, 39,40 without promising curative treatment. If the preclinical stage of dementia can be identified and potential interventions to prevent or delay its onset can be developed, the progress of MCI to dementia may be alleviated, and successful aging may finally be achieved. 41 This review highlights some interesting issues about 3 types of MCI: MCI due to Alzheimer’s disease, VCI, and MCI due to Parkinson disease and their management.
Mild Cognitive Impairment Due to Alzheimer’s Disease
There are many terms used to refer to MCI such as benign senescent forgetfulness, age-associated memory impairment, late-life forgetfulness, mild cognitive decline, age-associated cognitive decline, age-related cognitive decline, mild neurocognitive decline, cognitive impairment no dementia, and MCI due to Alzheimer’s disease. 16,42
Cognitive impairment can be divided into 4 categories depending on the domains of the deficit (Table 1). 16,42
Core Clinical Criteria of MCI Due to Alzheimer’s Disease.

Abbreviation: MCI, mild cognitive impairment.
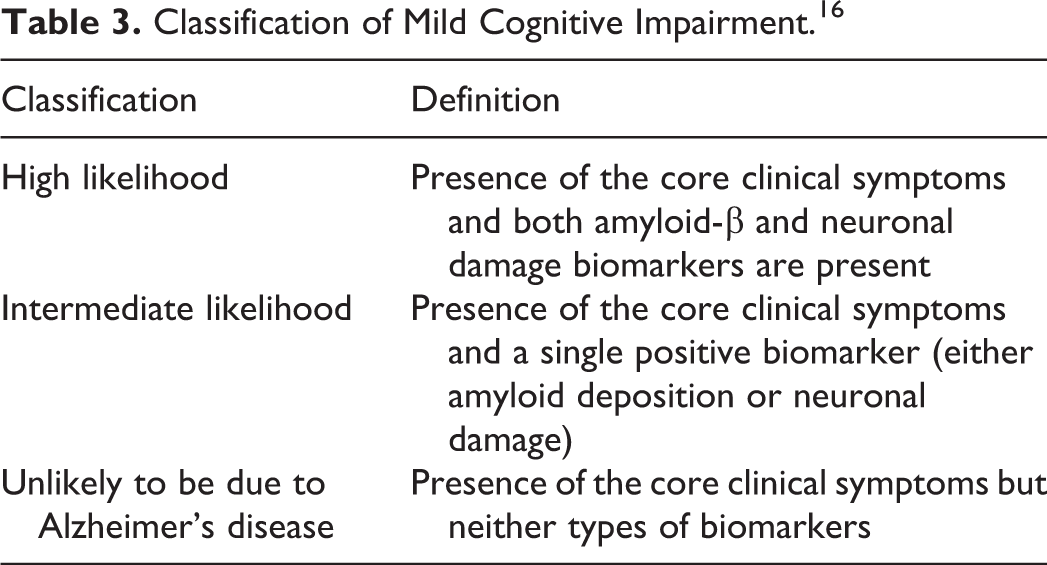
Amnestic MCI is more common than non-amnestic MCI by a ratio of about 2:1. 42 Non-amnestic MCI may result from normal aging and have reversible causes, or it may be the result of a predementia stage of non-Alzheimer’s disease such as frontotemporal lobar degeneration, dementia with Lewy bodies, Parkinson disease with dementia, vascular dementia, or primary progressive aphasia. 42 The National Institute of Aging and the Alzheimer’s Association work group proposed a new diagnostic criterion for Alzheimer’s disease, 16,39,41 which can be applied to preclinical Alzheimer’s disease, MCI due to Alzheimer’s disease, and Alzheimer’s disease dementia. Both the core clinical criteria and the biomarker criteria for MCI due to Alzheimer’s disease are useful for diagnosis. The core clinical criteria are subjective or objective cognitive impairments in one or more domain(s) of cognitive function, which have not disturbed the patient’s social or occupational functions with no other causes of cognitive impairment (neurologic, psychiatric, systemic disorders, metabolic dysfunctions, or medications). There are 3 categories of biomarkers for MCI due to Alzheimer’s disease: amyloid ligands, functional imaging, and structural magnetic resonance imaging, as shown in Table 2. Despite the discovery of these biomarkers for Alzheimer’s disease, new biomarkers are still needed for better prediction of the natural course of dementia. 43 According to the recommendation of the American Academy of Neurology (2018), in case that patients with MCI or families ask about biomarkers, physicians should advice that there are no accepted biomarkers available to date. 9 The likelihood of MCI being due to Alzheimer’s disease can be categorized as high, intermediate, or unlikely (Table 3). 16,41,44,45
Abbreviations: MRI, magnetic resonance imaging; PET, positron emission tomography.
Classification of Mild Cognitive Impairment. 16
There are currently a number of ongoing clinical trials being conducted regarding the pharmacological and nonpharmacological management of MCI (mostly amnestic MCI), but there is still no definite consensus of its management. However, cholinesterase inhibitors (donepezil) have been shown to lead to modest cognitive improvement in patients with amnestic MCI when compared with placebo; however, it did not show clinically meaningful results. 14,46 The practical guideline regarding MCI according to the American Academy of Neurology (2018) stated that there were insufficient data to support the use of cholinesterase inhibitors in general. If physicians offered, they must formerly provide a lack of evidence base. 47 There is currently no data that anti-inflammatory agents (eg, rofecoxib, celecoxib, and naproxen), ginkgo biloba, vitamin E, or vitamin E plus vitamin C have any benefit in this regard. Similarly, flavonoid-containing drink, homocysteine-lowering B vitamins, and V0191 showed insufficient results on cognitive measures. 9 A 6-month of transdermal nicotine (15 mg/d) were likely to gain cognitive test performance but not clinical global impression in patients with MCI who did not smoke. 9,48 In addition, receiving tesamorelin (growth hormone–releasing hormone) injections over 20 weeks might be effective in promoting performance on various cognitive measures, but the sustainability of this effect after 20 weeks was uncertain. 9,49 Cognitive and physical activity have been shown to decrease the risk of dementia and MCI, but more evidence of this is necessary. 14 The benefit of exercise was derived from 2 studies. One was a 6-month randomized controlled trial of twice-weekly resistance training exercise; the results showed improved selective attention/conflict resolution, associative memory, and regional patterns of functional brain plasticity, compared with twice-weekly balance and tone exercises in patients with MCI. It was a 60-minute class under certified fitness instructors. The patients performed 2 sets of 6 to 8 repetitions and loading increased when sets were accomplished with appropriate form. 50 Another study also demonstrated that multicomponent exercise could improve logical memory and preserve usual cognitive function in patients with MCI. The patients performed 90 minutes/d, 40 times for 6 months. 9,10 For cognitive intervention, the evidence is currently insufficient to support or disprove the use of any individual one. It may promote strategy knowledge, internal strategy use, and well-being but not external strategy or memory. 9 A systematic review and meta-analysis reported that cognitive interventions may impact not only on cognitive outcomes alone but also on activities of daily living and metacognition. 51 Therefore, encouraging patients with MCI perform regular physical and cognitive exercise training is recommended. 9
Vascular Cognitive Impairment
Vascular MCI or VCI is a type of MCI that is unlikely to be due to Alzheimer’s disease. It is commonly classified into 2 categories namely, poststroke- and nonstroke-related VCI. 52 Vascular cognitive impairment consists of one or more cognitive impairments including executive/attention, memory, language, and visuospatial functions, ranging from MCI to dementia, caused by clinical features of vascular events or evidence of vascular damage found using neuroimaging. 53 There are multiple domains of cognitive deficits in cases of VCI that have common presentations with dysexecutive syndrome. 18,54,55 Concomitant motor signs with VCI include frontal gait disturbance, lower body parkinsonism, apathy, depression, urinary incontinence, spasticity, hyperreflexia, and frontal release signs. 52 Majority of VCI would be mixed type, with the most common combined with Alzheimer’s disease.
Modification of various vascular risks may potentially reduce the risk of nonamnestic MCI. 56 The pathogenesis of VCI depends on host factors (age, education, genetics, ApoE-4 carrier, vascular risk factors, diabetes), vascular causes (atherosclerosis, microvascular diseases, endothelial disorder), and additional pathologies (amyloid deposition, cerebral amyloid angiopathy, aging). 56,57 There have been criteria proposed for diagnosis of VCI and vascular dementia, and these are presented in Table 4 according to the American Heart Association/American Stroke Association. 53,55,58 Diagnostic criteria are based on the presence of cognitive deficit and vascular disease plus a clinical decision that the cerebrovascular lesions explain the deficit in the affected patient. 52 There are various modifiable risk factors for VCI, the amelioration of which can decrease the degree of vascular injury.
There are abundant studies regarding the benefit of cholinesterase inhibitors and the N-methyl-D-aspartate (NMDA) receptor antagonist in VCI. Overall, they were safe and improved modest cognitive advantages, with inconsistent benefit on overall function. 52,53 Other pharmacological treatment showed insufficient evidence on cognitive measures such as aspirin, calcium channel blockers, statin, and vitamin supplementation. 53,59 Treatment of hypertension and lifestyle modification, such as being active and engaging in regular exercise, getting adequate nutrition, maintaining adequate socioeconomic support from the community, avoiding inappropriate use of medication, avoiding smoking, adherence to a Mediterranean diet, moderation of alcohol consumption, and adopting a positive attitude, may be effective strategies to prevent or delay the progression to vascular dementia 45 ; however, there is limited evidence of these interventions in improving cognitive function in patients with VCI. 59 Benefits of physical activity is probably due to the results of synaptogenesis, neurogenesis, and promoting vascular health similarly to the effect on other causes of dementia. 53 The consequence of lowering blood pressure to prevent cognitive decline beyond stroke prevention is debated; however, treating high blood pressure in persons with hypertension is generally advised due to benefit on other aspects. Focusing on hyperglycemia, there was small evidence regarding the effect on reducing VCI risk in diabetic patients. Given its benefit on various target organs including heart, eye and kidney, appropriate glycemic control in diabetic patients is likely to be worthwhile. 53
Mild Cognitive Impairment in Parkinson Disease
Parkinson disease is the second most common neurodegenerative disorder with progressive loss of dopaminergic neurons of the substantia nigra pars compacta. 57 Mild cognitive impairment in Parkinson disease (PD-MCI) usually takes the form of nonamnestic single-domain MCI and leads to increased health-care costs, increased fall risk, lower quality of life, and disabilities. Risk factors for MCI in Parkinson disease are aging, severe motor symptoms, non-tremor-dominant motor phenotype, and low education. The prevalence of MCI in Parkinson disease is about 20% to 50%; it varies depending on the population, cognitive domain, and utilized definition. 60,61
The pathophysiology of MCI in Parkinson disease may include synucleinopathy and amyloid deposition, neurochemical dysfunction (acetylcholine and dopamine), synaptic loss, and structural change. These processes are no different from Alzheimer’s disease, as they begin with slowly progressive proteinopathy and progress to cellular dysfunction and, eventually, structural change. 48 -50 There has been a criterion proposed for the diagnosis of MCI in Parkinson’s disease, 51 but it still needs validation. Diagnosis of PD-MCI is presented in Table 5. 60 Use of the MoCA as a screening tool for this syndrome has been recommended, as it is validated for both MCI and Parkinson’s disease with dementia. 62
Issues to Consider in the Diagnosis of PD-MCI. 60

Abbreviation: PD-MCI, mild cognitive impairment in Parkinson disease.
Nonpharmacological measures for MCI in Parkinson disease, such as cognitive intervention programs or physical exercise, require further investigation. There is little evidence of cholinesterase inhibitors reducing the risk of falling and improving cognitive outcomes in patients with Parkinson disease using donepezil or memantine. 60,62 Dopaminergic medications such as levodopa or dopamine agonists showed inconstant cognitive effects depending on the nature of task and level of basal dopamine function in corticostriatal circuitry. 63 A 24-week, double-blind, placebo-controlled trial of Rasagiline (a monoamine oxidase type B inhibitor) treatment in PD-MCI failed to improve cognitive function, but it gained motor symptoms and activities of daily living. 64 Atomoxetine, a adrenergic reuptake inhibitor, clozapine, serotonin and dopamine receptors, and second-generation tricyclic antidepressant were found to enhance attention and better control psychosis and depressed mood from placebo-controlled trials. 65 Management of comorbidities, such as depression, anxiety, apathy, and sleep disorder, which can contribute to worsening of cognitive function appears to be helpful. Furthermore, non-PD medications, such as centrally acting medications for pain, bladder function, and sleep, which causes cognitive decline should be avoided. 60
Can MCI Be Prevented?
Mild cognitive impairment may progress to dementia, and approved medications at present could only delay the progression of some types of dementia, particularly Alzheimer’s disease. Additionally, the effects of medications are modest. 12,29 Therefore, prevention of MCI is likely to be the best way against the onset of dementia. The studies regarding prevention of MCI mainly cover dementia syndrome in which MCI due to Alzheimer’s disease is the most common type, followed by vascular dementia. This review then would present the evidence base of effective interventions to prevent MCI in general. The interventions could be classified into 4 groups: pharmacological interventions, over-the-counter (OTC) supplements, physical activity, and cognitive training. 66 -69
Pharmacological Interventions
A systematic review showed that there were no randomized controlled trials regarding protective effect of dementia medication, antihypertensive medication, nonsteroidal anti-inflammatory drugs, aspirin, and testosterone. Similarly, no studies comparing the effect of intensive versus standard antihypertensive medication treatment, intensive versus standard antidiabetic medication treatment, and statin plus fenofibrate versus statin alone were available. Interestingly, estrogen alone and estrogen plus progestin versus placebo increased risk of MCI at hazard ratio of 1.34 and 1.07, respectively. Selective estrogen receptor modulator at 120 mg/d of raloxifene examined the benefit with a relative risk of 0.73 (95% CI: 0.53-1.01). Therefore, currently, no pharmacological interventions consistently show benefit in preventing MCI and estrogen with/without progestin increased risk of MCI. 69
Over-the-counter supplements
A systematic review reported that only few studies examined effects of OTC intervention on clinical MCI due to Alzheimer’s disease. No data comparing placebo and omega-3 fatty acids, soy, folic acid, B vitamin (folic acid plus B12), B vitamin (folic acid plus B6, B12), vitamin D plus calcium, vitamin E, vitamin C, or β-carotene regarding prevention of MCI is available. There was insufficient data to conclude the benefit of Ginkgo biloba, and multivitamin offered no benefit in clinical trial. Generally, existing OTC supplements have limited evidence for cognitive protection. 68
Physical Activity
Multicomponent physical intervention including flexibility, strength, balance, endurance, and aerobic training had insufficient data to conclude the effect. There was no study comparing attention control with aerobic training, resistance training, Tai chi, physical activity plus diet, physical activity plus protein supplement, or physical activity, diet, and cognitive training. Although, single-component physical activity interventions showed unfavorable results, a multidomain intervention appeared to promote cognitive function in older persons with normal cognition. Encouraging to perform in clinical practice is recommended since the benefits also affect to prevent or manage other chronic illnesses. 66
Cognitive Training
There is no clinical trial on the protective effect of cognitive training in adults with normal cognitive function to progress to MCI according to a systematic review. Training with specific domain could improve cognitive performance in the trained domains which were reasoning, executive function/attention/processing speed, and memory. Therefore, there is insufficient evidence of cognitive training regarding prevention or delay of cognitive decline in adults with normal cognitive function. 67
Conclusion
This topic reviewed the common pathophysiology of MCI, particularly in MCI due to Alzheimer’s disease, VCI, and MCI in Parkinson disease. The MoCA is recommended as a cognitive screening test for MCI. More trials are required in order to discover the most effective strategies for the prevention and delay of MCI. However, lifestyle modifications including regular cognitive and physical activity should be promoted through strong health policies in order to promote successful aging.
Footnotes
Acknowledgments
The authors would like to acknowledge Dylan Southard (Research Affairs, Faculty of Medicine, Khon Kaen University, Thailand) for editing this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.


