Abstract
Background:
Conflicting findings prevail about pain in older persons with cognitive impairment. There is evidence of changed pain perception; however, pain is also underrecognized. Pain and cognitive impairment were examined in a national cohort of older persons assessed using the Home Care International Residential Assessment Instrument (interRAI-HC).
Methods:
Participants were 41 459 aged 65+ years receiving a mandated needs assessment to access publicly funded services. InterRAI-HC pain severity and Cognitive Performance Scale analyses covaried for age, gender, and ethnicity.
Results:
Milder pain prevalence increased with age, whereas daily severe-excruciating pain prevalence decreased with age. Daily severe-excruciating pain was reported by 18% of cognitively intact individuals decreasing to 8% in the severe cognitive impairment group. This relationship remained after covarying for age, sex, and ethnicity. Differences among dementia subtypes were found.
Conclusion:
Although severe pain reports decrease with increasing age and cognitive impairment, more nuanced research covarying for dementia severity and subtype is required.
Introduction
“Individuals with neurological disorders such as dementia are susceptible patient groups in which pain is frequently under-recognized, underestimated and under-treated” Hadjistavropoulos. 1 (p1216)
Pain is highly prevalent among older people living in the community, with estimates for current pain of 20% to 46% and for chronic pain ranging from 25% to 76%. 2 There are consistent findings of a higher pain prevalence in women than men 3 -5 ; however, there are inconsistent findings regarding the relationship of pain with age. Abdulla and colleagues’ systematic review found an increase in pain with age in almost half of the 39 studies included; however, in 5 of those, pain reports reduced after 85 years. 2 Ten studies reported a decrease in pain with age and 6 studies reporting no change with age. 2 In New Zealand, the New Zealand Health Survey (NZHS) epidemiology study data for chronic pain support the pattern of increasing pain with age, with around a third of the 75+ years age-group affected; however, the age bands beyond 75 years were not reported separately in that survey. 5 Methodological reasons for these mixed findings include use of differing age categories, differing definitions or type of pain (“any,” chronic, or acute), as well as varying pain measures, samples, and settings studied (epidemiological, clinical outpatients, or long-term care settings).
Pain may be reported in the absence of tissue damage 6 yet may not reported even when pain is generally associated with a specific condition. 3 According to the biopsychosocial model of pain, 7 the impact of pain for an individual is influenced by interactions among somatic stimuli, psychological mood state, beliefs and coping styles, as well as environmental contextual factors including cultural factors. 8 In cognitively intact individuals, subjective pain experience and pain reports may be influenced or amplified by thoughts about the pain—ruminating about why they have pain or pain catastrophizing about the implications of pain for their futures. 9,10 There is therefore considerable variability in individual experiences of pain, including pain sensitivity, psychological, or physiological adaptation to pain, as well as to the overt expressions of pain complaints, or pain or illness behaviors. 8,11 As such, it is difficult to assess objectively and so the gold standard among healthy samples is self-report pain measures. 12,13
Knowledge of pain in those with cognitive impairment or dementia was very limited until the last 20 years as those with dementia were often excluded from research studies. 14 A review of the literature on pain in older persons with cognitive impairment notes inconsistent findings in relation to the frequency of pain reporting in this group. 1 Epidemiological and clinical studies often report reducing pain with increasing levels of cognitive impairment, 15 whereas experimental studies present a contrary perspective. These contradictory results may be due to the complex relationship between pain and cognitive impairment; however, this issue is also compounded by well-recognized methodological difficulties of assessing pain in older persons with severe cognitive impairment. 1,16 The impaired ability to communicate in older persons with severe cognitive impairment contributes to underrecognition and therefore undertreatment of pain in this group. 1 The expression of pain may be different, with withdrawal, agitation, or aggression being possible indications of pain in the severely impaired group. 17,18 Alternative methods to overcome the limitations of self-report in this group include greater use of carer observations and rating forms of pain behaviors (eg, facial expression, vocalization, changes in posture, gait, and other changes from usual behavior) or use of formal behavioral tests. 1
Experimental studies of pain experience in those with cognitive impairment note altered intensity and affective experience of pain in those with cognitive impairment compared to age-matched controls. Defrin and colleagues’ review of experimental studies concluded that pain is more likely to be increased rather than decreased in those with cognitive impairment. 19 Pain experience also appears to vary with the diagnosis related to the cognitive impairment 20 and with the stage of dementia. Experimental methods also affect pain findings in those with cognitive impairment due to variations in the type of pain stimulus used and how pain is measured. 19
New Zealand was the first country in the world to mandate the use of the Home Care International Residential Assessment Instrument (interRAI-HC) to standardize clinical assessments of community-dwelling older persons requiring access to publically funded community services or long-term residential care. The New Zealand interRAI-HC is a comprehensive, standardized, needs assessment tool assessing 20 domains within broad areas of physical and mental health, current treatment, functional independence, social supports, and living environment. Composite scales guide clinical decision-making and care planning. The interRAI-HC is one of the suite of instruments with a core set of assessment items common to all care settings plus additional tailored items. Information from the different versions of the interRAI are transferable and easily interpreted across care settings, in contrast to the previous practice of a variety of nonstandardized tests and measures being used in different settings across New Zealand. Those assessed provide explicit informed consent for use of their de-identified information for planning and research purposes. The interRAI-HC provides an invaluable growing repository of information for health research to improve patient outcomes. It is used widely in international studies (see systematic review). 21 Several New Zealand interRAI studies have recently been published 22 -27 ; however, there is little published regarding the experience of pain in older persons with cognitive impairment in New Zealand. This study offers the first opportunity to better understand this issue using national interRAI-HC data. The aim of this study was to examine the relationship of pain with cognitive impairment (controlling for age, sex, and ethnicity) in community-dwelling older New Zealanders assessed using the interRAI-HC.
Methods
Study Design
This cross-sectional study examines a national, community-based cohort assessed on referral for a needs assessment for access to publicly funded service provision or support. Research related to this data set was approved by the Health and Disability Ethics Committee, New Zealand Ministry of Health (14/STH/140).
Participants
Participants were aged 65 years and older assessed using the interRAI-HC between July 1, 2012, and June 30, 2014, and who had consented to have their data used for research. Those normally community-dwelling individuals who were temporarily hospitalized for any reason at the time of this assessment (n = 3585) were excluded from these analyses, leaving a sample size of 41 459. These participants were excluded as hospitalization frequently represents a deterioration in functioning that is likely to be a trigger for admission to long-term care.
Measures
InterRAI-HC 28 is a comprehensive 236 question clinician-rated measure assessing domains of demographics and living situation, aspects of physical and psychological functioning, diagnostic and symptom variables, treatment, social supports, and an environmental assessment, generating 27 composite scales which quantify severity and inform treatment planning. Trained assessors complete the detailed form using the interRAI-HC manual for item definitions and time frames. 29 Assessments typically take between 1 and 2 hours and scores are based on all sources of information (patient self-report, observation, case notes, family reports) and all items must be completed. Previous work has demonstrated that there were few missing variables, and data reliability and validity were high. 30
For this study, items from the following sections were used: A—Identification information (demographics and living situation); B—Intake and initial history (ethnicity); I—Neurological diagnoses related to cognitive impairment (Alzheimer’s disease, other dementia [not Alzheimer’s], Parkinson disease, stroke/cardiovascular diagnoses); J—Pain control; and the composite scales—pain scale and Cognitive Performance Scale.
The interRAI-HC algorithm–generated pain scale is a 0- to 4-Likert scale with scoring as follows: 0—no pain, 1—less than daily pain, 2—daily pain but not severe, 3—daily severe pain, and 4—daily excruciating pain (see https://www.ccim.on.ca interRAI CHA Key: Outcome Measures and Scales). The pain scale variable was dichotomized for the logistic regression analysis by the presence (or not) of daily severe or daily excruciating pain (levels 3 and 4 on the pain severity scale), as this was considered a clinically significant threshold. The pain control question is rated on a 6-point Likert scale, but for these analyses, we created a dichotomized “inadequate pain control” variable by collapsing items 4: Therapeutic regime followed but pain control not adequate and item 5: No therapeutic regime being followed, pain not adequately controlled.
The composite interRAI-HC Cognitive Performance Scale is a 7-point scale with higher scores indicating increasing levels of cognitive impairment. 31 An algorithm generates composite points based on difficulty in the following domains: decision-making, understanding, short-term memory, procedural memory, and at severe levels, ability to feed self, with the final most severe category 6, being in a coma. Scores are: 0—intact, 1—borderline intact, 2—mild, 3—moderate, 4—moderately severe, 5—severe, and 6—very severe. The Cognitive Performance Scale has been compared to the Mini-Mental State Examination, 32 the Montreal Cognitive Assessment, 33 and with clinician diagnoses. 34 It has excellent specificity (identifying true negatives without cognitive impairment) although sensitivity is sometimes lower than more comprehensive cognitive assessment measures (ie, it may miss true positives—those who do have cognitive impairment). 32 -36
Statistical Analysis
χ2 univariate descriptive statistics were used to characterize the sample in relation to the association of demographic variables (age, sex, and ethnicity) and the presence of daily severe-excruciating pain with scores on the Cognitive Performance Scale. A binary logistic regression (stepwise forward regression) model was used to examine whether increasing severity on the Cognitive Performance Scale predicted the presence of daily severe-excruciating pain, after controlling for age, sex, and ethnicity. 27 Odds ratios are reported quantifying the difference in risk of having daily severe-excruciating pain at different levels of cognitive impairment. All analyses used SPSS version v23. 37 A P value of <.01 was used.
Results
The mean age of the sample was 82.48 (standard deviation: 7.48) years. Percentages within a 10-year age bands were 65 to 74 (16.3%) years, 75 to 84 (40.4%) years, 85 to 94 (39.5%) years, and 95 (3.7%) years and over. Females comprised 61.8% of the sample. Ethnic identification was primarily European (88.9%), with other ethnicities being New Zealand Māori (5.0%), Pacific Islander (3.2%), Asian (2.2%), and “Other” ethnicity (0.7%) categories. All individuals were community living at the point of assessment and most were living independently in their own homes (89.7%). There were significant differences among age, sex, and ethnicity in relation to daily severe-excruciating pain status, with a higher prevalence of daily severe-excruciating pain in younger age bands and females and, in Māori or European, compared to Pacific Islander and Asian ethnic groups.
For the whole sample, a rating of “any” pain was common (58.4%). Percentages in each category for the pain scale were no pain (41.6%), less than daily pain (20.8%), daily pain but not severe (24.6%), daily severe pain (9.1%), and daily excruciating pain (3.4%). The pattern of changes in pain severity ratings across age is illustrated in Figure 1. There is a complex pattern, with pain of mild to moderate intensity increasing with age, but the pattern is reversed for the daily severe and daily excruciating pain categories where prevalence reduces with age.
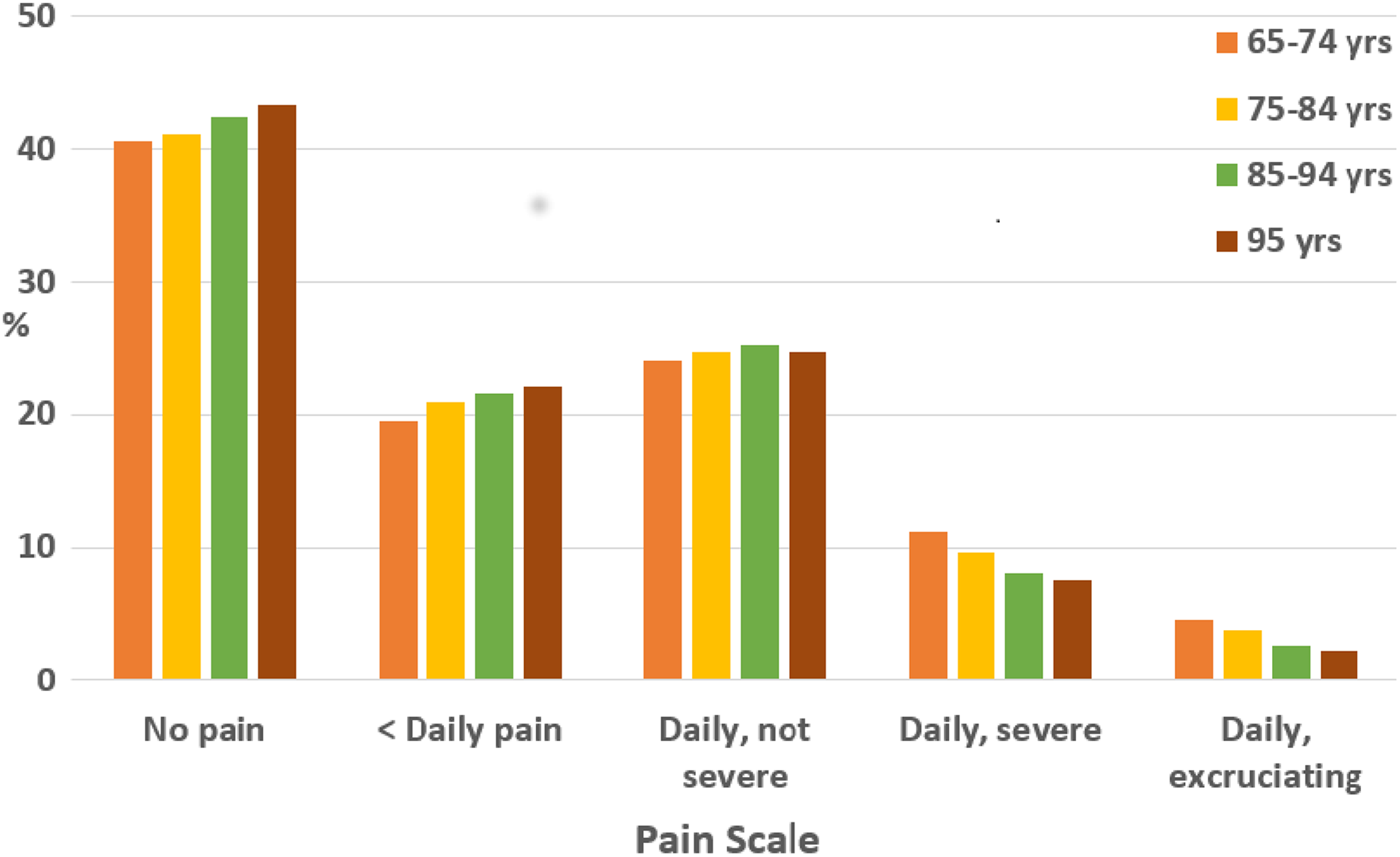
Pattern of pain severity ratings within age groups (unadjusted) in the New Zealand interRAI sample (n = 41 459).
Within the sample, 40.4% had a diagnosis associated with cognitive impairment, with some receiving more than one associated diagnosis. These diagnoses were stroke or cardiovascular (18.2%), Alzheimer’s (8.5%), other dementia (16%), and Parkinson disease (4.1%). Data regarding other causes of cognitive impairment, for example, from head injury or intellectual disability were not available in this data set.
Table 1 presents the uncorrected Cognitive Performance Scale scores in this sample in relation to the presence of daily severe-excruciating pain, along with the adjusted odds ratios of being in the daily severe-excruciating pain group, after covarying for age, sex, and ethnicity.
Rates of Daily Severe-Excruciating Pain With Increasing Levels of Cognitive Impairment in Those Aged 65+ Years in the New Zealand interRAI sample.a
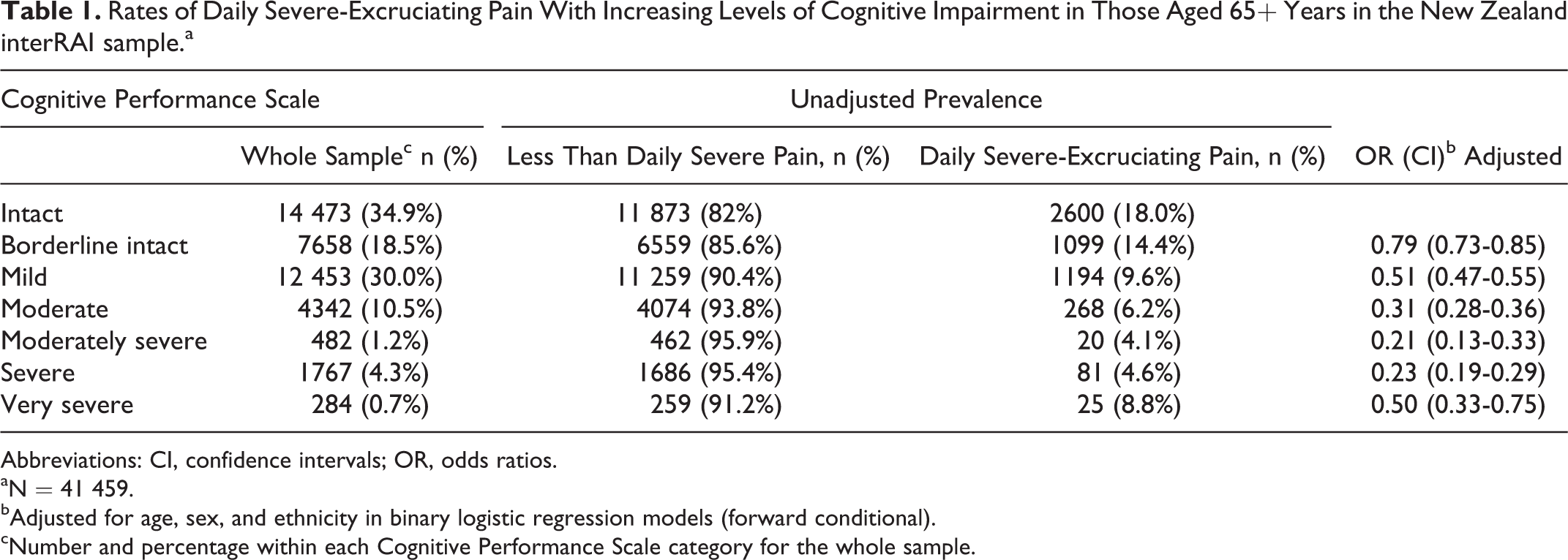
Abbreviations: CI, confidence intervals; OR, odds ratios.
aN = 41 459.
bAdjusted for age, sex, and ethnicity in binary logistic regression models (forward conditional).
cNumber and percentage within each Cognitive Performance Scale category for the whole sample.
In univariate analysis, there was an inverse relationship between levels of cognitive impairment and pain severity. As cognitive impairment became more severe, the chances of being in the daily severe-excruciating pain group decreased until the 2 most severe cognitive impairment categories where there was a slight increase again (hockey stick pattern; see Figure 2). This pattern of results remained in the logistic regression model (stepwise forward regression) after covarying for age, sex, and ethnicity (see Figure 3). This model explained 5.1% of the variance (Nagelkerke R 2) with 87.4% of participants correctly classified. Cases were excluded listwise within the logistic regression model if data on any variable were missing (n = 343, 0.8% of the sample).
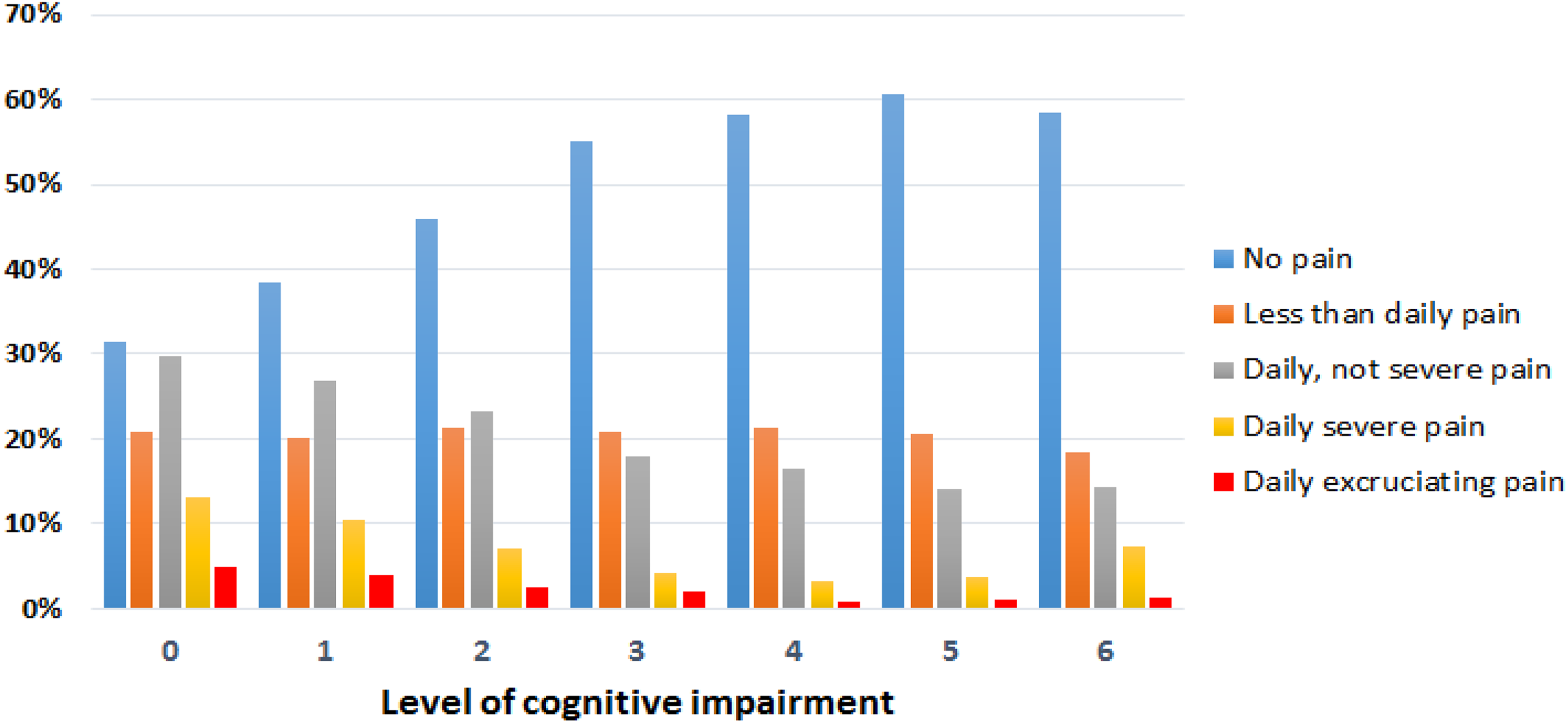
Relationship between severity of pain and cognitive impairment (unadjusted) in the New Zealand interRAI sample (n = 41 459).
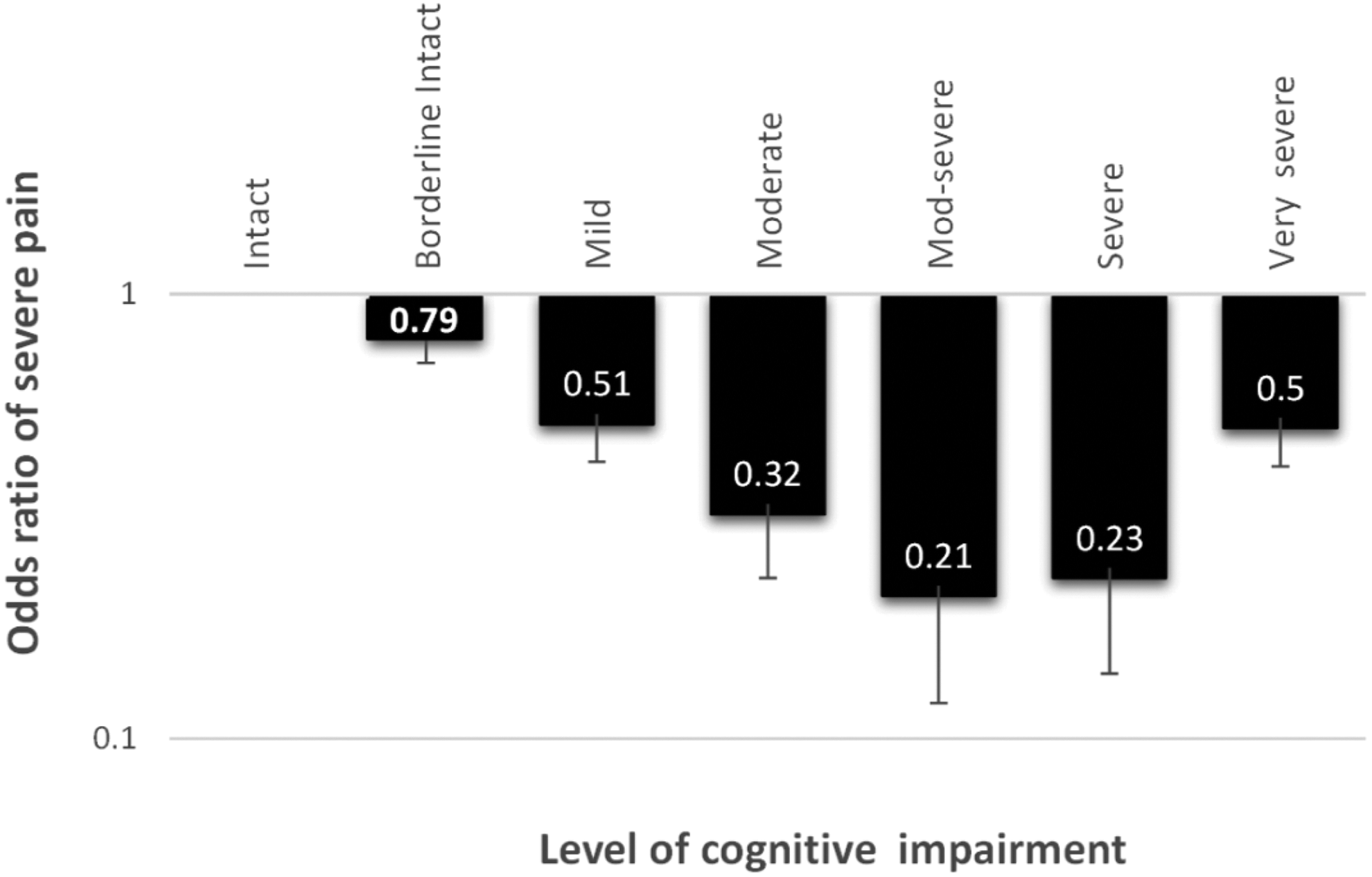
Odds ratios for being in the daily severe-excruciating pain category as level of impairment increases in the Cognitive Performance Scale, after covarying for age, sex, and ethnicity.
The percentage with daily severe-excruciating pain within the whole sample was 12.5% (n = 5661) and within specified diagnoses associated with cognitive impairment were Parkinson disease 10.1% (n = 187) versus no Parkinson diagnosis 12.7% (n = 5474, χ2 = 10.68; P < .01), stroke/cardiovascular 10% (n = 816) versus no stroke/cardiovascular diagnosis 13.1% (n = 4845, χ2 = 60.88; P < .001), Alzheimer’s 4.2% (n = 162) versus no Alzheimer’s diagnosis 13.3% (n = 5499, χ2 = 265.32; P < .001), and other dementia 6.3% (n = 456) versus no other dementia diagnosis (n = 6223, χ2 = 494.30; P < .001). Parkinson disease and stroke/cardiovascular disease diagnoses did not differ from each other however both had significantly higher prevalence of daily severe-excruciating pain than those with Alzheimer’s or other dementia diagnoses which did not differ from each other (χ2 = 279.63; P < .001). Excluding those with no or mild cognitive impairment within the Parkinson and stroke/cardiovascular disease groups to ensure that the cognitive impaired groups were of comparable severity, differences in severe daily pain reports were attenuated: Alzheimer’s 3.9%, stroke/cardiovascular disease 4.4%, other dementia 4.5%, and Parkinson 7.4%.
Within the sample, 9.8% were rated as having daily severe-excruciating pain and an inadequate pain regime or no regime at all. Within this subgroup with daily severe-excruciating pain and an inadequate analgesic regime, there was no statistically significant difference across levels of cognitive impairment in adequacy of the pain control regime (χ2 = 5.8; P = .45).
Discussion
Pain was common in this large sample of community-dwelling older New Zealanders presenting for a comprehensive needs assessment, with higher prevalence of pain in females, relatively younger age bands and Caucasian and Māori ethnicity (compared to Asian and Pacific Island ethnicity). Pain reports increased with age in the mild to moderate pain categories; however, prevalence of daily severe-excruciating pain reduced across age bands. In those with cognitive impairment, after covarying for age, sex, and ethnicity, as the level of cognitive impairment increased, the percentage assessed as having daily severe-excruciating pain decreased substantially.
The prevalence of current (past 3 days) “moderate (daily pain but not severe)” or worse pain severity in the present study (37.4%) is of a similar magnitude to the prevalence of chronic pain (around 32%) for the 65 years and older age band within the self-reported NZHS (n = 14 000 adults). 5 The prevalence in our study is a little lower than the 46% pain prevalence (pain in the past week) reported in a longitudinal study community sample of 833 Mexican American and European Americans 38 and considerably less than the 79% of women and 53% of men reporting pain during the past 14 days in a cohort of community-living Swedish 70-year-olds 39 ; however, the varying time frames may contribute to these differences.
Higher rates of pain have been reported in Caucasian participants compared to other ethnic groups, consistent with our findings, including within the NZHS 5 and also internationally. For example, lower pain was reported in Mexican American participants compared to European Americans. 38
The decreasing prevalence of daily severe-excruciating pain with increasing age in our study differs from the NZHS where rates of self-reported chronic pain increased in prevalence across the life span, from 31% in the 65 to 74 age-group to 34% in the 75 years and over age-group. The variable patterns of pain and age noted in the literature 2 are likely to be due to variations in pain definitions and severity threshold, methods of reporting, and how age groups are categorized across studies. 40 Our current findings suggest that where other studies included those with mild or worse pain, any inverse pattern that may be present in the much smaller daily severe-excruciating pain group is likely to be obscured. Another possible explanation for lower rates of pain in the eldest participants in cross-sectional studies is that people with serious health conditions (with associated pain) might have already died, leaving the relatively healthy in the oldest age categories. Prospective studies following cohorts over time would address this issue.
Our study findings are consistent with some other large studies, including in those in long-term care facilities regarding the inverse relationship of pain severity with increasing cognitive impairment. A Scandinavian study using the Minimum Data Set (MDS) in 6487 older persons in long-term care facilities also found an inverse relationship between pain and cognitive impairment. 41 Those authors specifically commented on the negative association of pain with cognitive impairment as contrary to the positive associations of pain with depression and other conditions. That pattern is consistent with our present findings and that of our previous article where increasing pain severity was positively associated with increased risk of falls, fatigue, and depression. 27
Similar results were reported in a large United States study using the MDS which examined pain and cognitive impairment in nursing home residents aged 65 years and older (n = 71 227). 42 Self-reported pain was more frequent in those with milder dementia, with substantially lower pain reports among the most severely cognitively impaired group. In the half of their sample able to self-report (32.1% of whom had severe cognitive impairment), 67.7% reported moderate to severe pain. The other half of their sample (74.6% of whom had severe cognitive impairment) was unable to provide self-report. Staff rated only 10.7% of this group of patients as having moderate to severe pain using the MDS-Pain Behavior Scale. The group without the ability to articulate pain had half the rate of analgesia use and higher rates of verbal and physical aggression than those able to communicate pain (90% of whom were on pain medication). Another study using the MDS also found the same inverse relationship of pain and cognitive impairment in participants aged 65 years and over in long-term care facilities in the Netherlands (n = 2295), Finland (n = 5761), and Italy (n = 1959). 18 The prevalence of any pain varied across studies, from Italy 32%, the Netherlands 43%, and Finland 57%, but the inverse relationship was present in all 3 countries. In the United States, a retrospective nursing home chart review study in 48 patients with Alzheimer’s disease and advanced cancer pain found an inverse relationship between pain ratings and cognitive impairment. 43 That study used the Cognitive Performance Scale and the Discomfort Behavior Scale (an observational rating scale) from the MDS assessment.
The services and health for elderly in long term care (SHELTER) study (n = 4156), using the interRAI-long term care facilities (LTCF), reported a relatively low prevalence rate (21.3%) for current pain of moderate or greater intensity in their sample of older persons living in long-term care facilities in 7 European Union countries. 44 Although these authors do not report data specifically supporting the inverse relationship of pain with cognitive impairment, their low prevalence of reported pain might be influenced by the fact that two-thirds of their sample was classified as cognitively impaired. 45
Studies inconsistent with our findings include the following community and long-term care facility studies. A Swedish community study assessing 124 male and 117 female 70-year-olds using visual analogue, verbal pain rating scales, and psychometric tests reported no relationship between pain severity and cognitive functioning. 39 Of note, however, those authors commented on the good cognitive functioning in their relatively young sample. Rates of dementia were not reported but might be expected to be lower than in studies with older samples. In the US National Health and Aging Trends epidemiology study (n = 7609), those with “probable” dementia (n = 802) had higher rates of “bothersome” pain (63.5%) and activity limiting pain (43.3%) compared to a matched control group (n = 802) without dementia (bothersome pain: 54.5%; activity limiting pain: 27.2%). 46 That study did not report pain severity in relation to extent of cognitive impairment. In a study of nursing home residents (n = 199) in the Netherlands, higher rates of pain were reported in those with more severe dementia (27%) compared to those with less severe dementia (15%). They noted discrepant findings between a pain rating scale, the mobilization – observation – behaviour – intensity – dementia scale (MOBID-2) which gave higher ratings (but no difference between dementia subtypes), compared to self-report where a vascular dementia group had much higher pain reports (54%) than those with Alzheimer’s (18%) and other dementia types (14%). Although the rates of pain reported in that study were much higher than in our study, we also found lower severe daily pain reports in those with Alzheimer’s disease than those with a cardiovascular/stroke diagnosis; however, when only those with moderate or greater cognitive impairment were included, there was little difference between these groups.
Integration of the mixed findings above is challenging due to methodological differences across studies, particularly the inconsistent use of pain thresholds and differentiation of levels of cognitive impairment. Those studies with similar methods, that is, using the MDS, the forerunner of the interRAI tend to be consistent with our findings. 18,41 -43
Evidence from experimental and clinical studies that the mechanism of pain experience may be different in those with significant cognitive impairment is perhaps not unexpected given the overlap in brain areas affected by both dementia and pain processing. Pain and suffering are experienced following the transmission of noxious stimuli via the spinal cord, thalamus, and the cerebral cortex, in particular the sensory cortex and the frontal lobe. Kunz and colleagues speculated that dementia-related neurodegeneration in prefrontal areas might contribute to impaired executive functioning and loss of pain inhibitory potency, increasing vulnerability to pain in those with cognitive impairment. 47
The complexities however are illustrated in the following studies. Pain reports in those with cognitive impairment in one study did not differ from a healthy control group after controlling for analgesic use; however, when assessed after a movement task likely to elicit pain, the group with cognitive impairment reported less pain than the cognitively intact group. 48 Reduced physiological reactivity during venipuncture has also been reported in persons with dementia compared to those without dementia. 49 Similarly, Monroe et al reported that people with Alzheimer’s did not demonstrate the same level of pain behavior as expected, making behavioral assessment problematic. 43 In summary, the evidence for the relationship of pain in those with dementia suggests that for some subtypes at least, there is altered pain experience, but the impaired capacity to communicate pain also contributes to lower reports of pain in this group compared to those with no cognitive impairment.
Difficulties in assessing pain in this vulnerable group, limitations in current assessment methods, and other factors contribute to an underrecognition and undertreatment of likely pain especially in long-term care facilities. 42,44 The Australian and New Zealand Society for Geriatric Medicine considers pain to be “the 5th vital sign,” and called for better assessment and management of pain to be a quality indicator in care facilities. 50 In community samples though, pain may be undertreated due to a substantial proportion (30.3%) of older persons with “bothersome pain” choosing not to use pain medication. 46 In a previous review, we suggested that this might be a cohort effect related to stoicism related to experiencing earlier adversity. 51
The strengths of this study include that this is the first report examining the very large national interRAI-HC data set to examine pain and cognitive impairment in older persons in New Zealand. The nationally mandated use of this well-validated instrument, high consent rate for research use, and inclusion of all sources of information in the assessment increases confidence in these findings. Use of such big data sets makes possible more fine-grained analyses addressing methodological issues such as the complex relationship of pain and age identified in this study.
There are limitations for this study. Although the interRAI-HC is an extremely useful comprehensive screening tool, breadth in coverage comes at the expense of depth. Issues with the sensitivity of the Cognitive Performance Scale have been raised, 32 -36 meaning clinicians need to be aware that it may miss some cases of cognitive impairment. The interRAI-HC manual appropriately advises caution in rating pain in those with severe cognitive impairment, given the acknowledged difficulties in assessing pain in this group, again with the risk of missing those with pain. 16 Other limitations include that these participants were assessed at a point of change in health so this group may not be representative of all older persons living in the community. Statistical significance in large samples may not translate to clinically relevance; however, we believe that these findings, although of small magnitude, are clinically meaningful and have service delivery implications.
Conclusions
Mild to moderate pain complaints increased with age in this large cross-sectional interRAI data set of older New Zealanders; however, a different pattern was found for those with severe pain, where reports decreased with both age and with increasing cognitive impairment. Future prospective research needs to be more nuanced in taking into account severity of dementia and dementia subtypes. The interRAI cohorts offer the potential to examine the relationship of cognitive functioning and pain over time in individuals, with a focus on monitoring change in pain in dementia subtypes.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was received by Dr Jamieson from the Royal Australasian College of Physicians (New Zealand Fellows Research Establishment Fellowship 2016) and the Canterbury Healthcare of the Elderly Research Trust for the wider interRAI research project. Funders had no input into the writing of this manuscript.
