Abstract
Elderly patients with Alzheimer’s disease (AD) and other dementias are at high risk of polypharmacy and excessive polypharmacy for common coexisting medical conditions. Polypharmacy increases the risk of drug–drug and drug–disease interactions in these patients who may not be able to communicate early symptoms of adverse drug events. Three acetylcholinesterase inhibitors (ACHEIs) have been approved for AD: donepezil (Aricept), rivastigmine (Exelon), and galantamine (Razadyne). They are also used off-label for other causes of dementia such as Lewy body and vascular dementia. We here report evidence from the literature that ACHEI treatment, prescribed for cognitive impairment, can reduce the load of medications in patients with AD by also addressing cardiovascular, gastrointestinal, and other comorbidities. Using one drug to address multiple symptoms can reduce costs and improve medication compliance.
Introduction
Elderly patients with Alzheimer’s disease (AD) and other dementias are at high risk of polypharmacy (5-9 medications) and excessive polypharmacy (10+ medications) for common coexisting medical conditions. 1 –4 Polypharmacy in this population is particularly worrisome because of the high risk of drug–drug and drug–disease interactions and the decreased cognitive ability to communicate early symptoms of adverse drug events (eg, gastrointestinal [GI] bleeding, dizziness, etc). Traditional methods to reduce polypharmacy include adjusting treatment goals to the patient’s value and expected life expectancy, pharmacist monitoring, use of a computerized drug database to catch duplication, comprehensive geriatric assessment, and the recent public health campaign via Choosing Wisely to educate both prescribers and patient stakeholders about low-value medical care. 5 –9 Another method to reduce polypharmacy and medication burdens in the elderly patients with chronic disease such as AD is the use of one medication to address the multiple comorbidities and symptoms for which many medications are prescribed. 10,11 In this review, we discuss preliminary evidence supporting the efficacy of the US Food and Drug Administration (FDA)-approved anti-Alzheimer’s medications (acetylcholinesterase inhibitors [ACHEIs]) for the treatment of cardiovascular, GI, and other comorbid symptoms in elderly patients living with AD and other dementias. 6
Three medications have been approved for AD under the category of ACHEIs. 12 Examples of this medication include donepezil (Aricept), rivastigmine (Exelon), and galantamine (Razadyne). Donepezil, the first and most widely used of these 3,13 has also been used in those with Parkinson’s disease and Lewy body dementia. 14,15 Memantine, approved by the FDA in 2003 for use in patients with AD, operates along different pathways from ACHEIs. 16 Acetylcholinesterase inhibitors have modest and temporary effects on improving cognitive function in patients with AD; however, their actual and potential benefits may extend beyond the cognitive. The overall effect of ACHEIs is to reduce the breakdown of acetylcholine (ACh), increasing the activity of the cholinergic load centrally and peripherally. The peripheral effects occur via an increase in the parasympathetic/vagal drive to visceral organs and systems: heart, GI, genitourinary, and other systems. The ACHEI-induced increase in parasympathetic drive outside of the central nervous system largely explains the wide range of physiological effects in patients using ACHEIs. Their physiological pro-parasympathetic effects on the cardiovascular system, GI, eyes, and bladder have the potential to help in reducing polypharmacy in older patients living with AD. Patients with dementia, of which AD is the most common, have a high prevalence of such comorbidities as Parkinson’s disease, stroke, diabetes, atherosclerosis, fluid and electrolyte disorders, insomnia, incontinence, 17 pneumonia, 17 glaucoma, 18 and xerostomia. 19 These conditions require additional medications for their management, placing such patients at greater risk of polypharmacy. 20
We begin with the assumption that very many patients are already being prescribed ACHEIs. If they have any of these other conditions, the double benefit of ACHEIs should be considered before adding another medication to the regimen of such patients. The current review will provide evidence from the literature (summarized with level of evidence 21 in Table 1) that ACHEI treatment, prescribed for cognitive impairment, can potentially reduce the load of medications in patients with AD by also addressing comorbidities. By using one drug to address multiple symptoms, costs can be reduced and medication compliance improved.
Secondary Effect of Acetylcholinesterase Inhibitors (ACHEIs) in Patients With Alzheimer’s Disease (AD) or Other Dementias.a
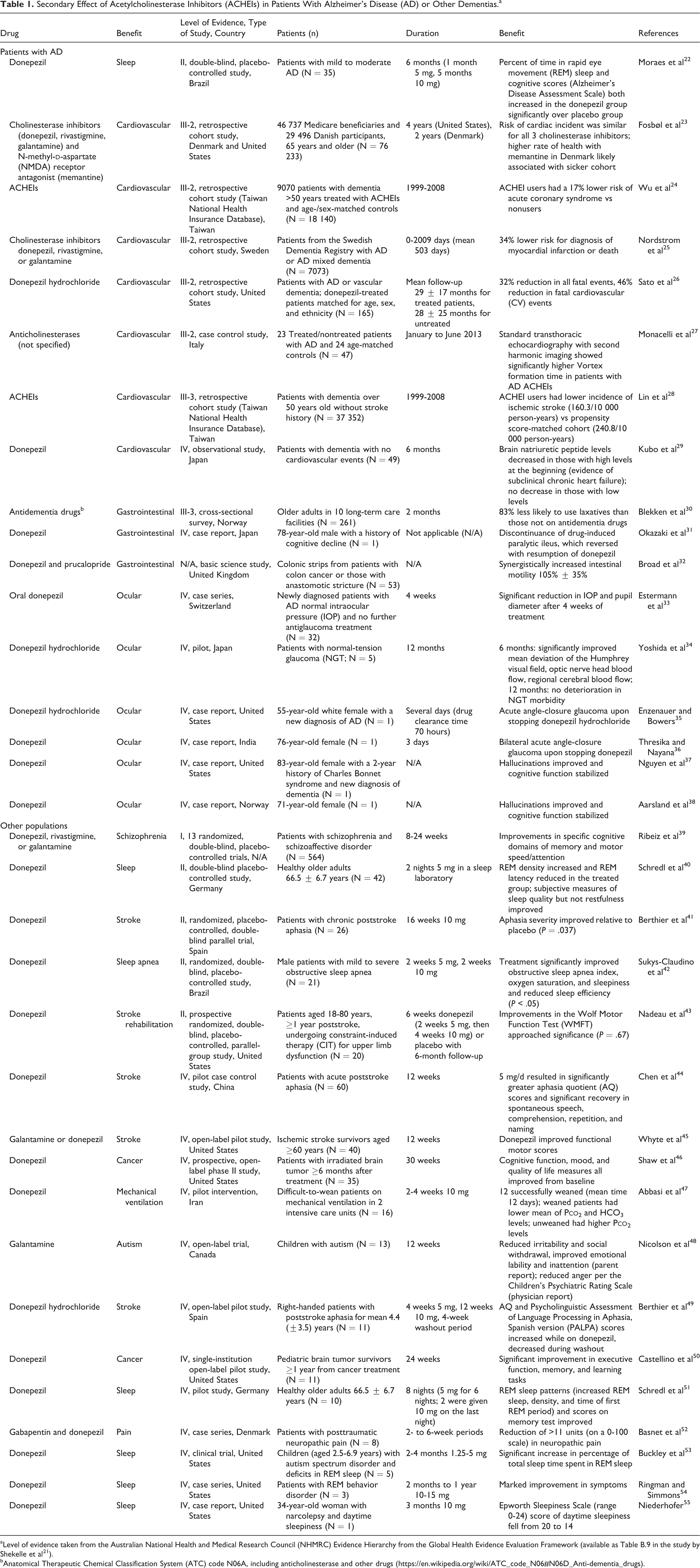
aLevel of evidence taken from the Australian National Health and Medical Research Council (NHMRC) Evidence Hierarchy from the Global Health Evidence Evaluation Framework (available as Table B.9 in the study by Shekelle et al 21 ).
bAnatomical Therapeutic Chemical Classification System (ATC) code N06A, including anticholinesterase and other drugs (https://en.wikipedia.org/wiki/ATC_code_N06#N06D_Anti-dementia_drugs).
Beneficial Effects of ACHEI
Tachycardia
Numerous studies link heart disease and AD. 56,57,58 Tachycardia (from atrial fibrillation [AF]), atrial flutter, and other causes) represents a very common, potentially devastating problem in the elderly population. As people age, the left ventricle becomes stiff and then increased contractility of the left atrium is required during diastole to push the blood into the left ventricle. Tachycardia is thus not well tolerated in the elderly patients because it leads to the loss of the atrial push, triggering flash pulmonary edema. The most common causes of tachycardia in the elderly patients include AF, supraventricular tachycardia, infections, and the side effects of certain medications. 59 Atrial fibrillation is a common comorbidity in patients with dementia. Many of these patients are on several medications, such as digoxin, β-blockers, and calcium channel blockers, to control the AF-related tachycardia. These medications have myriad cognitive and noncognitive side effects. The major control of heart rate is the parasympathetic cholinergic (vagus) drive to the heart, responsible for slowing down the heart (negative chronotropic effect), unlike the sympathetic component which subserves positive chronotropy. 60 Evidence from past studies show that Aricept and other ACHEIs can slow down the heart rate in patients with AF and other tachycardia. 61 In fact, slowed heart rate (bradycardia) was the most common adverse drug reaction to donepezil in a French study of 6 years of data from the French Pharmacovigilance Database. 62 One review characterized the risk from bradycardia and syncope as “small but significant.” 61
Myocardial Infarction, Heart Failure, Cardiovascular Events
Numerous studies indicate a possible cardioprotective effect of cholinesterase inhibitors (ChEIs). 63 The effect, as indicated in 1 study of more than 76 000 older adults in the United States and Denmark (evidence level-III-2), appeared similar for all ChEIs. 23 A prospective noninterventional study by Kubo et al presented evidence supporting the possible benefits of donepezil in heart failure. The Donepezil Cardiac Test Registry study enrolled 49 patients with dementia (43 with AD and 6 with diffuse Lewy body) to assess the cardiovascular effects of donepezil treatment. 29 The patients ranged in age from 65 to 95 years and were treated with 3, 5, or 10 mg donepezil daily over a 6-month period. Patients were evaluated at baseline and at the 1-month, 3-month, and 6-month mark. Cardiovascular evaluation consisted in part of a 12-lead electrocardiogram, plasma brain natriuretic peptide (BNP) levels, and heart rate. None of the patients in the study experienced cardiovascular events. Brain natriuretic peptide levels were unchanged in patients with low-level BNP but were substantially decreased in patients with high-level BNP. 29 This result suggested the safety of donepezil in patients without cardiovascular symptoms and possible benefits in those with subclinical heart failure.
Nordstrom et al found ACHEI use in patients with AD correlated with decreased myocardial infarction (MI) and death risk in a cohort study of 7073 patients from the Swedish Dementia Registry (evidence level-III-2). 25 The patients had been diagnosed with AD or Alzheimer’s mixed dementia. After accounting for confounders, the researchers found that patients who used ACHEI had a 34% lower risk of either MI or death compared to those who did not. The authors reported the lowest risk in patients on the highest recommend dose of ACHEI (ie, for donepezil 10 mg, for rivastigmine >6 mg, for galantamine 24 mg). 25
A retrospective cohort study by Sato et al (evidence level-III-2) reported reduced cardiovascular mortality risk in older adults with AD or vascular dementia treated with donepezil. 26 In this cohort, 76 patients on donepezil were compared to 80 age- and sex-matched controls. After a mean follow-up of more than 2 years (29 ± 17 months in the donepezil group and 28 ± 25 months in the controls), the donepezil-treated patients had a 32% reduction in fatal events and a 46% reduction in fatal cardiovascular events, compared to controls, as assessed using Cox proportional hazard regression analysis. Survival advantage continued through 4 years of follow-up but decreased over time, possibly because of the small sample size. The authors cautioned against overinterpreting the results and argued for further study. Others 64 have called for studies to explore enhancing cholinergic signaling to reduce cardiac morbidity and mortality.
One Taiwan population-based retrospective cohort study (evidence level-III-2) 24 looked at patients with dementia from the Taiwan National Health Insurance Database from 1999 to 2008. This study found that ACHEI use was associated with decreased risk of acute coronary syndrome compared to age- and sex-matched nonusers (hazard ratio: 0.836; 95% confidence interval [CI]: 0.750-0.933; P < .001]). Another Taiwan observational retrospective cohort study (evidence level-III-3) 28 investigated the risk of ischemic stroke in ACHEI use in patients with moderate to severe dementia without stroke history. This study included 37 352 patients with dementia over 50 years old without stroke history from the same database over the same period. The ACHEI users had a 0.508 adjusted hazard ratio for ischemic stroke (95% CI: 0.434-0.594; P < .001). There was no significant difference in all-cause mortality between ACHEI users and nonusers.
A related review article 64 discussed recent studies that implicate ACh as a major regulator of cardiac remodeling and provide support for the notion that enhancing cholinergic signaling in human patients with cardiac disease can reduce morbidity and mortality. These recent results support the idea of developing large clinical trials of strategies to increase cholinergic tone, either by stimulating the vagus nerve or by increasing availability of ACh in heart failure. These articles state that increasing ACh levels, through either vagal stimulation or ChEIs, can act through a variety of pathways to reduce cardiac remodeling in several different models of cardiac disease. As such, the cholinergic system may serve as an important pharmacological target in patients with heart disease. 64 Another study (evidence level-III-2) 27 found protective effects in patients with Alzheimer’s dementia treated with anticholinesterases, by improving diastolic filling through increasing Vortex formation time. Data from an animal study 65 support the protective effects of increasing the cholinergic drive as a mechanism to reverse sympathetic hyperactivity-induced cardiac dysfunction. In patients with dementia on medications (Senna, Miralax, or lactulose) for constipation or medications (β-blockers, calcium channel blocker, or digoxin) for slowing down the heart rate, especially in the setting of AF/flutter, the use of ACHEI and its cholinomimetic effects on the cardiovascular and GI systems can allow clinicians to discontinue 2 or more of the medications.
Gastrointestinal Motility
Slow transit in the GI system is common in the elderly population, especially those with AD. 66 This is worsened by decreased physical activity in many such patients. The consequences of slowing of GI motility include decreased gastric emptying of food, eating, constipation, obstipation, fecal obstruction, and diverticulosis. Chronic constipation can potentially lead to diverticulosis, intestinal obstruction, increased risk of urinary tract infection, and other comorbidities. These comorbidities are common reasons for hospital admission in the elderly patients and are a major driver of health-care costs. 67 Many studies have shown that ChEIs improve GI emptying and colonic motility. 68 -70 This improvement reflects increased activity of the cholinergic drive to the GI system. One study (evidence level-III-3) of constipation among nursing home patients in Norway found that those taking antidementia drugs (including ACHEIs) were 83% less likely to use laxatives than those not on antidementia drugs. 30 A randomized study of 30 patients with diabetes mellitus and constipation showed that oral pyridostigmine (a ChEI) improved overall colonic transport time. 71 Clearly, ACHEI has the potential to reduce the use of laxatives in older adults with dementia. Many patients with AD take 4 to 5 laxatives, and laxative use is very common in nursing homes. 72,73
An in vitro experiment demonstrated preliminary evidence suggesting the treatment of GI dysmotility by combining donepezil with prucalopride (a serotonin type 4 [5-HT4] receptor agonist). Broad et al obtained macroscopically normal full-thickness ascending and descending colonic tissue samples from 53 patients undergoing surgery, mostly for bowel cancer. 32 Muscle contraction and relaxation in the samples were elicited using electrical field stimulation (EFS). Donepezil (30 nM) combined with prucalopride (3, 10 μM) markedly increased EFS-evoked contractions compared with either alone (P = .04). Thus, following further clinical studies, the combination of these 2 drugs could be used to treat GI dysmotility, with possibly less cardio-bronchial side effects than that experienced with neostigmine. 74 -76
Okazaki et al reported paralytic ileus in a 78-year-old patient with dementia following abrupt discontinuation of donepezil. 31 This patient had been on 5 mg/d donepezil treatment for dementia for 3 years prior. The patient stopped the medication due to loss of appetite during hospitalization for severe dehydration. Three days following the discontinuation, the patient experienced abdominal pain, distention, and constipation. Plain film X-ray also indicated intestinal distention with significant gas retention. Despite treatment, the gas retention persisted for 1 week. Upon resuming donepezil, the gas retention and the patient’s intestinal symptoms improved “immediately and dramatically.” This again not only indicates the need for caution when stopping ACHEI in patients with AD but also supports the usefulness of donepezil to treat GI dysmotility such as pseudo-obstruction.
Vision Problems
Glaucoma
Glaucoma is a common cause of low vision and blindness in the elderly population, particularly in those with AD.18 Usually painless and therefore undetected until late in the disease progression, glaucoma usually requires 2 or more expensive eye drops to reduce intraocular pressure (IOP). Patients with AD can find adherence to this medication very complicated and may have difficulty in following the complex medication regimen. One mechanism for increased IOP is decreased flow of aqueous humor through the anterior uveal tract, a process that is facilitated by the ACHEI-mediated increase in parasympathetic drive to the eye. One way to reduce IOP is to improve the flow of aqueous humor and the cholinergic drive to the eye via the parasympathetic innervation of the eye. The main neurotransmitter for the parasympathetic drive is ACh. Acetylcholine is broken down by acetylcholinesterase (AChE). One way to improve ACh is to block AChE using ACHEI, such as Aricept. 77 If a patient with AD is on 2 or 3 medications, the number of eye drops prescribed can potentially be reduced. Evidence for this mechanism has been published in several studies. 33 -35,78,79
Several reports indicate a strong relationship between AD and glaucoma. 80 -83 In a study by Yoshida et al, patients with normal-tension glaucoma (NTG) exhibiting AD-like decreased cerebral blood flow (CBF) benefited from donepezil treatment. 34 All 5 patients in the study were treated with 5 mg/d of oral donepezil hydrochloride for 12 months. Although no significant change in IOP was noted, Yoshida et al report improved regional CBF, mean deviation of the Humphrey visual field, and optic nerve head blood flow. This result suggests the possibility of improving optic neuropathy in patients with NTG via oral donepezil hydrochloride. The effect of donepezil on IOP was further demonstrated by another study with a slightly larger sample size. Estermann et al report treating 32 newly diagnosed patients with AD with normal IOP using oral donepezil hydrochloride over 4 weeks, in the absence of other glaucoma treatment. Comparison of pretreatment and posttreatment measurements of all 63 eyes indicated a mean 8.8% reduction in IOP and a mean 7.4% reduction in pupil diameter. 33 This evidence further supports the possibility of using donepezil to treat glaucoma.
Conversely, angle-closure glaucoma has been reported after abrupt discontinuation of oral donepezil. 35,36 Per Enzenauer and Bowers, a 55-year-old newly diagnosed patient with AD was on 10 mg daily donepezil hydrochloride and no other pharmacotherapy for 4 to 6 months. 3 5 Within 3 to 5 days of running out of the donepezil, the patient presented to the emergency department with 12 hours’ severe right-sided headache, photophobia, reduced vision, nausea, and vomiting. Eye examination and IOP measurement indicated acute closed-angle glaucoma of the right eye and occludable angles in the left eye. In addition to topical antiglaucoma treatment, the patient underwent laser iridotomy of the right eye, and the next day, prophylactic iridotomy of the left eye. Although this may suggest the importance of screening eye examinations prior to commencing donepezil chloride in patients with AD, this adverse effect is suggestive of the possible cholinergic effects of donepezil chloride in glaucoma management.
Charles Bonnet Syndrome
Donepezil has also been shown to reduce symptoms in those with Charles Bonnet syndrome, which evidences visual hallucinations. 84 Those with Lewy body dementia or AD may be unable to identify the hallucinations as unreal. For both populations, donepezil has been shown to reduce hallucinations and increase the ability to identify the visions as illusory. 37,38,85
Putative Benefits of ACHEIs and Similar Medications (Neostigmine)
Dry Mouth Syndrome (Xerostomia)
Dry mouth is a very common and potentially debilitating condition in the elderly patients, especially in patients with AD. 19 Chronic dry mouth in the elderly can lead to malnutrition, poor dental hygiene, loss of teeth, and recurrent mouth infections. 86,87 Dry mouth or xerostomia in elderly patients can be a result of several conditions such as Sjögren syndrome and other autoimmune diseases, as well as being a consequence of radiation therapy. 88 It can also be a side effect of polypharmacy, 89 -93 including anticholinergic medications. 86 Patients with AD with dry mouth are particularly prone to poor appetite, aspiration pneumonia, and weight loss from malnutrition. Many of the medications for dry mouth, such as artificial saliva, are complex to use in patients with dementia with poor compliance and cost. The main mechanism for saliva production is increased activity of the cholinergic system. Acetylcholinesterase inhibitors have been shown in many studies to improve production of saliva. 94 Neostigmine, which also blocks acetylcholinesterase, likewise increases saliva production. 95
Atonic Bladder
Atonic bladder conditions are common and potentially devastating in the elderly patients, particularly in patients with AD. They may result from injury, 96 disease, 97 stroke, 98 surgery, 99,100 or complications of diabetes. 101 Atonic bladder, when untreated, leads to recurring bladder infections, kidney stones, and kidney failure. 102 While appropriate medications to improve function in atonic bladder do not exist, the major cholinergic pathway for stimulating bladder contraction is via parasympathetic cholinergic drive to the bladder. The activity of this pathway is enhanced by Aricept and other ACHEIs, as well as similarly acting neostigmine. 103 Evidence from published studies showed that ACHEIs can increase bladder contractility, 104,105 especially in men with large prostates. 106
Sleep Disorders
Sleep apnea is a risk factor for AD and a common comorbidity in such patients. 20 Since cholinergic mechanisms help regulate rapid eye movement (REM) sleep, 107 ACHEIs should increase this type of sleep in those with a deficit. Several reports bear this out. In 2000, Ringman and Simmons reported marked improvement in 3 patients with REM sleep disorder with administration of donepezil. 54 In 2 studies of healthy older adults, Schredl et al 40,51 studied the effect of donepezil on sleep in healthy older adults. In the first, conducted in 2001, 10 healthy older adults (aged 66.5 ± 6.7 years) were administered 5 mg donepezil for each of 6 nights and their sleep patterns observed and recorded in a sleep laboratory. Their scores on a memory test taken from the Alzheimer’s Disease Assessment Scale (ADAS) 108 were also recorded. The percentage of time spent in REM sleep and REM density (a measure of eye movement during sleep) increased; REM latency (time until first REM sleep) decreased. Of the 8 patients who took the memory test, the mean score over 8 trials increased significantly from the baseline night to the last night of medication. The greatest increase in parameters occurred after the first dose of donepezil, but the increase persisted throughout the study period. 51 As a follow up, in 2006, Schredl et al studied 42 health older adults (age range 60-77 years) for 2 nights in a sleep laboratory (level II evidence). Twenty-two received 5 mg donepezil and 20 received a placebo. Sleep architecture was recorded and all patients were given a sleep questionnaire the next morning for them to report their level of being refreshed and subjective experience of waking during the night and other aspects of their sleep. Both REM latency (69.5 ± 31.1 minutes vs 88.2 ± 50.6 minutes) and REM density (32.0% ± 9.2% vs 24.5% ± 8.8% of the total night) were significantly improved in the donepezil group over the placebo group (P > .05). Patients in the donepezil group did not report being more refreshed but did report more time awake (53.3 ± 57.6 minutes vs 28.1 ± 67.3 minutes) and more times waking (3.5 ± 2.9 vs 2.1 ± 1.1) than the placebo group. 40
In a double-blind placebo-controlled study conducted over 6 months in Brazil (level II evidence), Moraes et al studied the effect of donepezil on sleep in patients with AD. 22 Thirty-five patients with mild to moderate AD were assessed after 6 months of donepezil (5 mg in month 1, 10 mg thereafter) or placebo for cognition, polysomnographic parameters, and brain wave patterns (overall, frontal, and temporal). At 6 months, treated patients had significantly more time spent in REM sleep (17.0% ± 9.9% vs 8.4% ± 5.4%) and better ADAS cognitive subscale scores (28.3 ± 12.3 vs 42.8 ± 18.7, lower scores represent better cognition) than the placebo group (P < .01). 108
Donepezil has also been used to treat sleep-related disorders in those with obstructive sleep apnea (evidence level II), 42 autism, 53 and narcolepsy. 55 Care must be taken, however, because ACHEIs can cause sleep disorders. One study reported REM sleep behavior disorder in an 88-year-old man after his nightly dose of rivastigmine was increased; adding 0.5 mg clonazepam at bedtime resolved the nocturnal behaviors. 109
Other Effects
While the mechanism is unclear, donepezil has been reported to increase libido in some older adults 110 ; one hypersexual patient with AD was successfully treated by switching to rivastigmine. 111 Other potential benefits of ACHEIs include a reduction in neuropathic pain, 52 assist with weaning of patients from mechanical ventilation, 47 improved cognition after cancer 46,50,112 or cancer treatment, 113 improved cognitive function after stroke, 45 treatment of obstructive sleep apnea, 42 increased REM sleep in children with autism, 53 decreased sleepiness in a patient with narcolepsy, 55 reduced aggressive behaviors associated with autism, 48 and in those with schizophrenia and schizoaffective disorder. 39 Both donepezil and memantine have been used to treat poststroke aphasia 41,43,44,49,114,115 as summarized by Berthier et al. 116
Discussion
Although the focus of this review is on the salutary effects of ACHEI, several clinically relevant side effects (nausea, vomiting, and syncope) warrant mentioning. 117 -119 These side effects are amplification of the physiological cholinomimetic effects of the ChEIs, especially in the setting of higher starting dose or rapid upward dose titration in frail elders with dementia and multimorbidities. Indeed, the side effects of nausea (2%-8%) and vomiting (1%-5%) were the most commonly reported reasons for ACHEI discontinuation across all ChEI clinical trials. 117 In a meta-analysis of randomized placebo-controlled trials, Kim et al found a significant and independent relationship between the use of ChEIs and increased odds of syncope (odds ratio = 1.53, 95% CI: 1.02-2.30). 118 This relationship was independent of type and severity of dementia, site of care, or length of follow-up. However, use of ChEIs was not associated with risk of falls, fracture, or accidental injury in these studies. 118 A key approach to reducing the risk of these cholinomimetic side effects is by starting at low dose (eg, at 25% of recommended starting dose) and by slow titration over months instead of weeks as suggested by the manufacturer’s recommended prescribing information. Another approach is switching to a different ChEI, as the minor pharmacokinetic and dynamic differences between ACHEIs may alter an individual patient’s risk of side effects. 119
We considered the preponderance and variety of these studies to be worth of note. Bringing them all together in one place provides the opportunity to consider some aspects of ACHEI prescription that may indicate the need for larger, more rigorous studies of these potential benefits. In particular, we need to understand how genetic differences in drug metabolism among patients affect the likelihood of experiencing some of the beneficial effects of ACHEI (as well as side effects) described in this article. Such studies have potential to yield new indications of ACHEIs for diseases that currently have no effective therapies. Example of this abound in science, as in the repurposing of bupropion (an antidepressant) for the new indications of smoking cessation and obesity. 120
One limitation of this review is that these studies lack the “gold standard” rigor of randomized controlled trials. We consider the number and variety of positive effects as indicating a trend, which we attempted here to document. Randomized controlled trials or database analysis of a large number of patients may be the next step before any recommendations should be made in terms of prescribing changes.
Conclusion
Acetylcholinesterase inhibitors have several positive pleiotropic activities that can help in reducing polypharmacy in elderly patients living with dementia and other serious comorbidities. In fact, treatment with ACHEI may be reducing the incidence of those conditions (ie, vision and hearing problems, diseases of the musculoskeletal system, lipoprotein disorders, and hypertension), which are less likely to be diagnosed in older adults with dementia than in those without dementia. 17
The evidence reviewed in this article underscores the usefulness of Aricept and other ACHEIs in the treatment of the comorbid symptoms of glaucoma, dry mouth, gastroparesis, slow transit GI, fecal obstruction, and atonic bladder. The effectiveness of ACHEI in patients with AD with deficient cholinergic function in systems other than the neurological—cardiovascular, GI, ocular, urinary—may reflect its enhancement of overall acetylcholinergic neurotransmission. Inasmuch as the effects of ACHEIs (both positive and negative) are multisystemic, they warrant more rigorous studies on how we can harness the positive effects while minimizing the risk of negative effects. The usefulness of ACHEIs in reducing polypharmacy and costs in patients with AD merits further study in a large randomized, controlled, clinically comparative trial.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
