Abstract
Objective:
To test the effect of angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin-receptor blockers (ARBs) on reducing the risk of dementia in patients with heart failure (HF).
Methods:
This retrospective, longitudinal study used a cohort of HF patients identified from a local Medicare advantage prescription drug plan. Multivariable time-dependent Cox model and marginal structural model using inverse-probability-oftreatment weighting were used to estimate the risk of developing dementia. Adjusted dementia rate ratios were estimated among current and former ACEI/ARB users, as compared with nonusers.
Results:
Using the time-dependent Cox model, the adjusted dementia rate ratios (95% confidence-interval) among current and former users were 0.90(0.70-1.16) and 0.89 (0.71-1.10), respectively. Use of marginal structural model resulted in similar effect estimates for current and former users as compared with the nonusers.
Conclusion:
This study found no difference in risk of dementia among the current and former users of ACEI/ARB as compared with the nonusers in an already at-risk HF population.
Heart failure (HF) and dementia are common medical conditions in older adults. 1 More than 5.5 million adults have HF in the United States, and the incidence approaches 10 per 1000 population after 65 years of age. 2 Despite a decline in cardiovascular mortality in developed countries over the last 3 decades, the burden of HF has risen. 3 Dementia is an independent predictor of increased mortality in patients with HF. 4 –6 One in 6 individuals older than 70 has dementia with prevalence increasing exponentially with advanced age. It is estimated that the number of cases with dementia in 2050 will increase 3-fold compared with the year 2000. 7 Alzheimer’s disease (AD) is the most common followed by vascular dementia (VaD) and other forms of dementia. There is considerable overlap emerging in the risk factors and pathogenesis of vascular dementia and AD. 8
Heart failure is associated with more than an 80% increase in the risk of dementia and AD. 1,9 The probable biological pathways linking HF to dementia, and AD in particular, include considerably impaired cerebral circulation from HF, clinical and silent brain infarcts, leukoaraiosis, and cerebral thrombosis for which HF is a risk factor. Numerous studies have found that hypertension is also a risk factor for dementia. 10 –12 Therefore, its control in patients with HF should be beneficial in reducing the incidence of dementia. 13 Our previous work showed that angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) were better than other antihypertensive agents in reducing the risk of dementia in elderly diabetic patients. 10
Studies have shown ACEIs/ARBs to restore normal cerebral autoregulation, reverse inflammatory reactions, decrease neuronal cell death, and have antiplatelet aggregating and atrial antifibrillatory effects. 14,15 However, there are studies that have found no association with use of ACEIs/ARBs and risk of dementia. 16,17 To our knowledge, there is no study in patients with HF that assessed the effect of ACEIs/ARBs on time to dementia diagnosis. Moreover, literature showed that ACEIs/ARBs have different effect on cognitive impairment in patients with and without HF, 18 and dementia significantly increased the risk of hospitalization and mortality in patients with HF. 4 –6 Given the significant clinical impact of dementia associated with HF and the current urgency for effective treatments to reduce the incidence of dementia, the present study examined the effectiveness of ACEI/ARB that is widely prescribed for the treatment of HF.
The key challenge in comparison of true treatment (causal) effects in an observational study is the presence of confounding, especially confounding by indication for treatment. Furthermore, in longitudinal studies involving chronic illnesses such as HF, some confounders change with time during the follow-up. A model that allows incorporation of time-varying confounders is the extended Cox model. However, it has been argued that this model is not sufficient to give causal effect estimates in the presence of time-dependent confounders that are affected by previous treatment. 19,20 To estimate the causal effect of treatment on outcome in the presence of such confounding, marginal structural models (MSM) have been proposed. 21 In the present study, we have considered occurrence of hospitalization to be a time-dependent confounder affected by previous treatment. Physicians may adjust ACEI/ARB therapy after occurrence of a hospitalization that may be indicative of decompensation in overall health that could herald increased risk of dementia. However, (prior) ACEI/ARB therapy should benefit patients and reduce their risk of hospitalization. Therefore, hospitalization may be a time-dependent confounder in this study because it is predictor of subsequent treatment, predicted by previous treatment, and may be associated with risk of dementia. This relationship is illustrated in Figure 1.
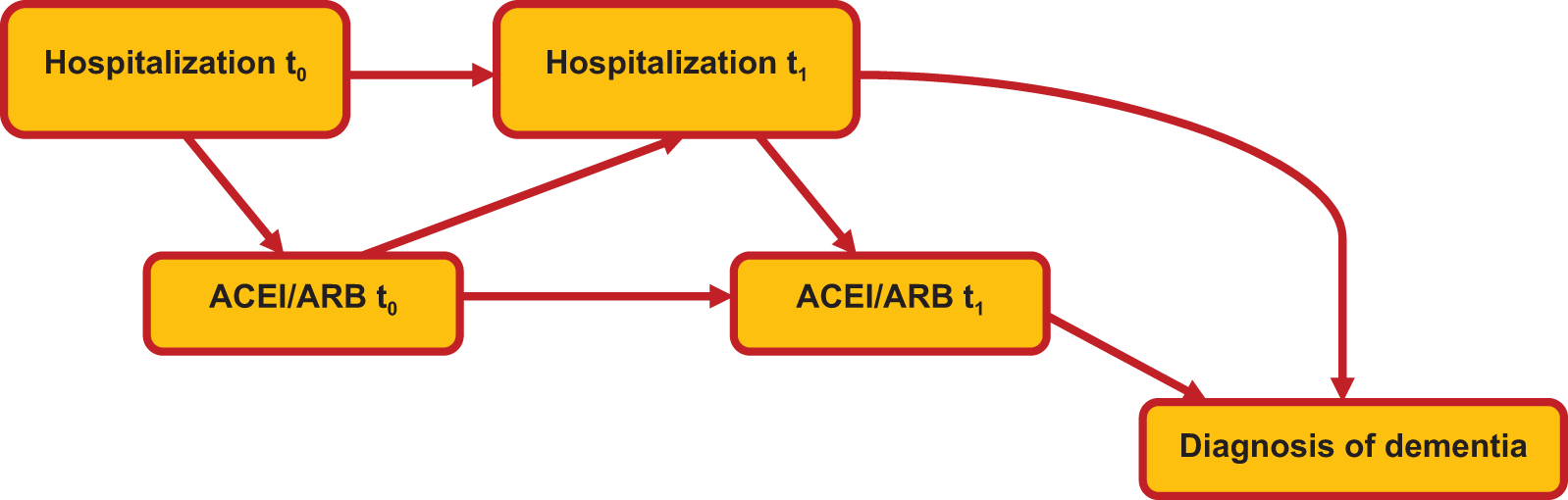
Illustration of time-dependent confounding of ACEI/ARB use and risk of dementia by hospitalization. ACEI indicates angiotensin-converting enzyme inhibitors; ARB, angiotensin receptor blocker.
The goal of this study is to test the causal effect of ACEIs/ARBs on reducing dementia in patients with HF using extended Cox model and Marginal structural model to account for time-dependent confounding by hospitalization.
Methods
Data Source
The study design was a retrospective longitudinal cohort of all patients with HF in a Medicare advantage prescription drug (MAPD) plan in Texas from January 1, 2008, to December 31, 2011. Several computerized data files including membership file, member summary file, institutional claims file, professional claims file, quest lab, CCMS, and pharmacy file were used. Membership and member summary files include demographic, severity scores, and cost data for beneficiaries for each year. Institutional claims include information on all inpatient claims. The files contain diagnostic information in the form of International Classification of Diseases, Ninth Revision (ICD-9) codes, and procedure information in form of Current Procedural Terminology (CPT) codes. Professional claims contain information on all outpatient encounters. The files contain diagnostic information in the form of International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes and procedure information in form of CPT codes. Quest lab files contain 153 applicable laboratory tests that include low-density lipoprotein cholesterol (LDL-C), hemoglobin A1c (HbA1c), and creatinine. The CCMS file includes the hospitalization records, date of admission, date of discharge, and length of stay. Pharmacy files contain Part D pharmacy data provided by pharmacy benefits manager. The pharmacy records include patient and drug identifying information, fill dates, days of supply, quantity dispensed, and dosing information for each prescription filled.
Selection of Patients
Patients were included in the study sample if they had at least 1 claim after the index date and diagnosed with HF (ICD-9-CM 402.01, 402.11, 402.91, 415.0X, 416.9X, 425.4X-425.9X, 428.XX, 429.4X, 785.51) between January 1, 2008, and December 31, 2011. Patients were excluded if they were diagnosed with dementia or died before the index date. Index date was defined as (1) January 1, 2009, for patients who had HF diagnosis on or before January 1, 2009, and their coverage prior to January 1, 2008 and (2) date of HF diagnosis for patients who had 1 or more years of records prior to HF diagnosis. For patients who did not have records 1 year prior to HF diagnosis, we assigned the index date as the date at which these patients with HF had at least 1 year prior records. Finally, records with missing values except for the laboratory measures were deleted from the final sample, as suggested by Little and Rubin. 22
Outcome Measure
Time to dementia diagnosis
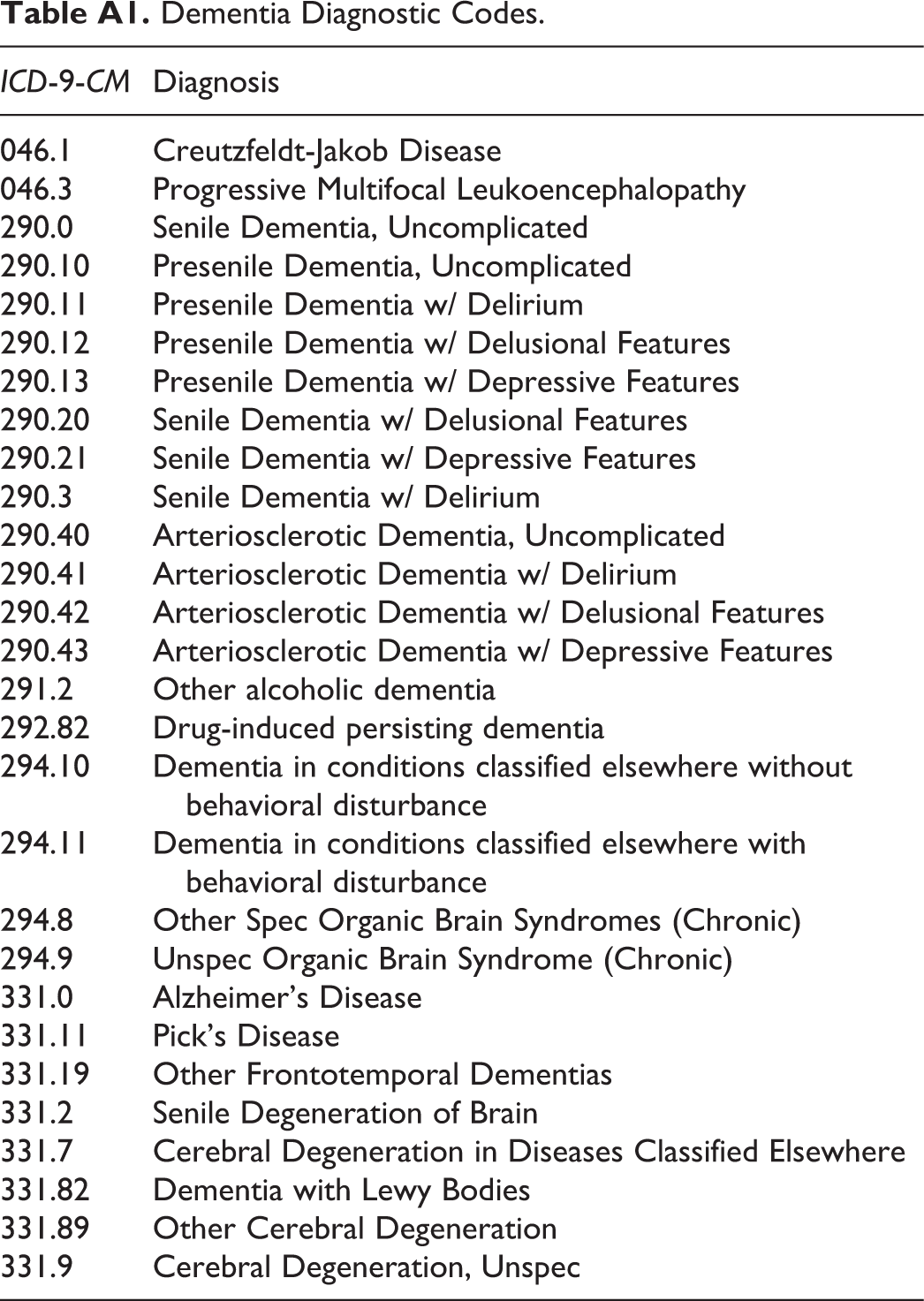
Dementia was defined based on the occurrence of any relevant ICD-9-CM codes in the inpatient or outpatient records from the index date. The diagnosis of dementia was examined through December 31, 2011. The ICD-9 codes and their description to identify dementia diagnosis are given in Appendix A. 10,11
Exposure
A patient’s exposure to ACEI/ARB was assessed for the study period (January 01, 2009-December 31, 2011). Exposure was classified on a monthly basis by assessment of the days’ supply of the filled prescription. Gaps were measured between the end of one prescription and the pick-up of the next. Every person-month during study follow-up was classified according to the use of ACEIs/ARBs. Current use in a month was defined as use when the days’ supply for ACEIs/ARBs covered 15 or more days in that month. Former use in a month was defined as use after the current use or when days’ supply for ACEIs/ARBs covered less than 15 days in that month. Nonuse in a month was defined as no prescribed use of ACEIs/ARBs or days’ supply covered less than 15 days in that month or any preceding months. In other words, nonusers had no prescription for ACEI/ARB for 15 days or more in any prior month, whereas former users did. Former users and nonusers could become current users of ACEIs/ARBs during follow-up. Exposure to ACEI/ARBs was therefore time varying or time dependent and could change over the course of follow-up (Figure 2).
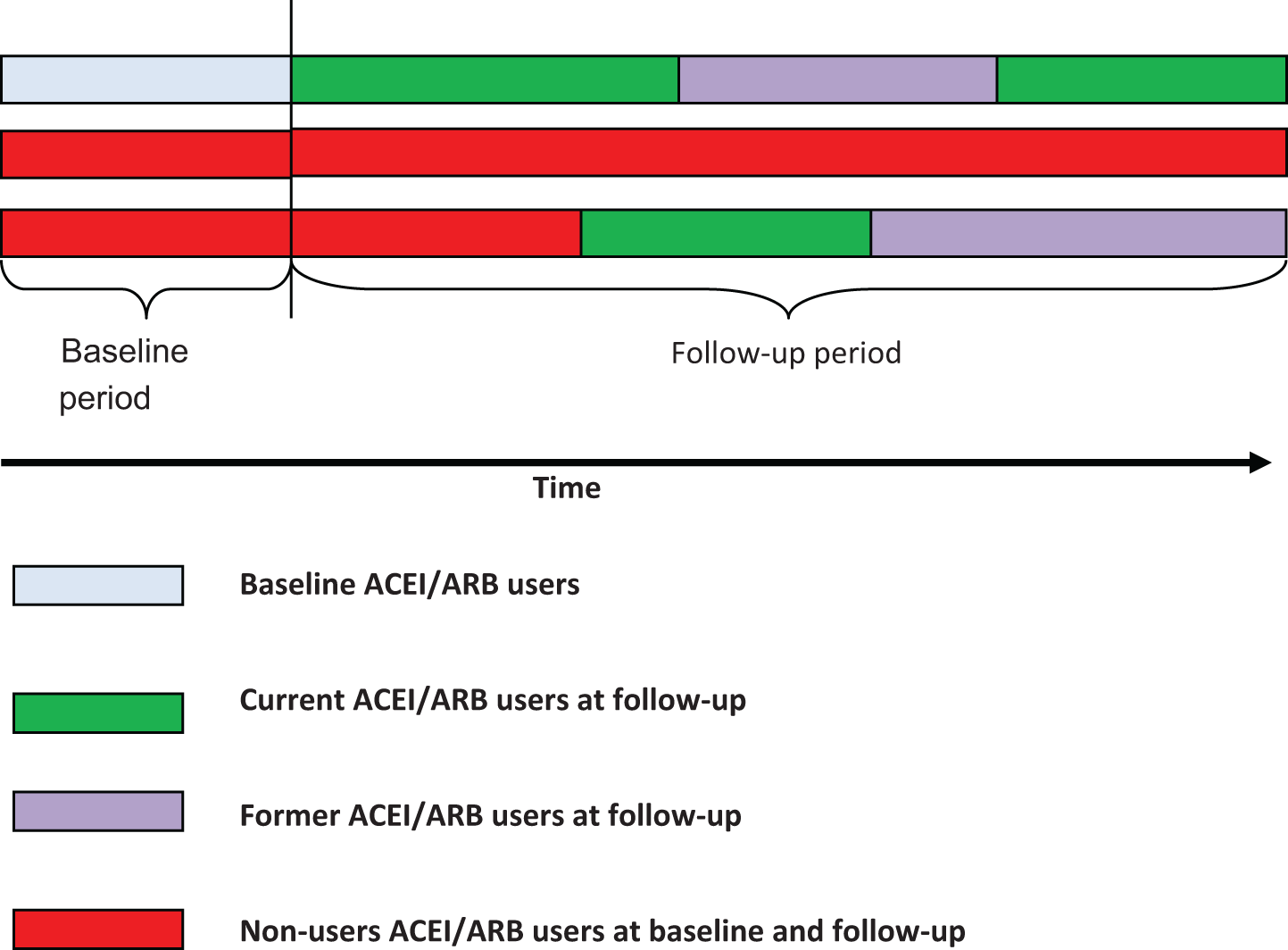
Illustration of exposure to ACEI/ARB on monthly basis classified as current users, former users, and nonusers. ACEI indicates angiotensin-converting enzyme inhibitors; ARB, angiotensin receptor blocker.
Average daily dose and achievement of targeted dose were measured at the 6-month baseline period. Average daily dose was calculated at the baseline as the total quantity of ACE/ARB divided by the available days of ACEI/ARB therapy during the 6-month preindex period. Dose was standardized to 20 mg of enalapril equivalent. After calculating average daily dose for each patient, a categorical variable for attainment of targeted dose was created as below targeted dose and at or above targeted dose. 23
Covariates
The variation in survival rates may be associated with the time-independent sociodemographic factors, treatment factors, and clinical factors, therefore the study included 43 such covariates. Sociodemographic factors included age and gender. Treatment factors included various comedications taken 1 year prior to the index date. We included diuretics (thiazide diuretics, potassium sparing diuretics, aldosterone antagonist, and loop diuretics), digitalis glycosides, statins, calcium channel blockers (CCBs), beta blockers (BBs), vasodilators, antiarrythmics, alpha blockers, insulin, oral hypoglycemics, and nonstatin lipotropics. Clinical factors included 22 comorbid conditions 24 –26 and appropriate laboratory measures such as serum creatinine, LDL-C, and HbA1c. Severity of illness could not be assessed by ejection fraction (EF) or New York Heart Association (NYHA) HF classification due to lack of information in the database. Instead, we used number of years in the cohort and prior hospitalization as a substitute for HF severity. 25,27 We also included number of all drugs taken, number of comorbid conditions, and Centers for Medicare and Medicaid Services (CMS) risk score to account for medication burden and disease severity. The CMS risk score is calculated based on data taken from a large pool of beneficiaries to estimate the average predicted costs for each of the component factors (eg, age sex, low-income status, and individual disease groups). It consists of 189 disease classifications for use in risk adjusting of clinical outcomes in Medicare populations. 11
In addition to the time-independent covariates, the time-dependent covariates considered for the study were concurrent hospitalization, cumulative duration of ACEI/ARB use, and adherence defined on a monthly basis. A patient was considered hospitalized in a particular month if she was admitted to the hospital in that month. If the length of stay in hospital was more than 30 days, then the patient was considered hospitalized for 2 consecutive months and so on. Duration of use was calculated as cumulative duration of use by summing the duration of all prescriptions and updated monthly. Adherence-as medication possession ratio (MPR) was calculated as follows:
MPR = ([Total months supply]/time available) × 100.
A dichotomous variable was created as less than 80% MPR and ≥80% MPR 26 on monthly basis.
Statistical Analysis
Analyses were performed using SAS software version 9.3 (SAS Institute, Cary, North Carolina). Bivariate associations between the predictor variables and the outcomes were tested using the chi-square test for categorical variables and the t test for continuous variables. Two different approaches were used to test the effect of ACEI/ARB use on the risk of the dementia diagnosis. First, a time-dependent Cox model was developed, and second a marginal structural Cox model using inverse probability weights was constructed.
Time-Dependent Cox Model
Crude dementia rates for baseline ACEIs/ARBs use versus nonuse were compared. Kaplan-Meier method was used to estimate unadjusted dementia diagnosis by baseline use of ACEIs/ARBs, and the log-rank test was used to compare the groups. Next, a multivariable time-dependent Cox model of time to dementia diagnosis was constructed. The independent variables used in the Cox regression were 43 time-independent factors such as sociodemographic factors, treatment factors, and clinical factors and time-dependent factors updated monthly such as exposure groups, duration of use, medication possession ratio, and hospitalization. Patients were censored if they did not reach the outcome till December 31, 2011 (end of the study) or if the patient died before dementia diagnosis. Hazards ratios (HR)/rate ratios were obtained from the model after adjusting for the covariates mentioned above. Hospitalization may be a time-dependent confounder in the present study. It is an intermediate variable affected by previous treatment and predicting future treatment and also an independent risk factor for dementia. Thus, simply adding this variable in the time-dependent Cox model may introduce bias and cannot provide causal effect of ACEI/ARB on dementia 19,20
Marginal Structural Cox Model
To estimate the causal effect of ACEI/ARB use (current use and former use) versus nonuse on dementia diagnosis in the presence of time-varying confounding factors, a marginal structural model employing inverse probability treatment weighting (IPTW) was used. In longitudinal studies with time-varying exposure, the marginal structural models (MSMs) use the IPTW that is updated at various time points to achieve the balance between the groups not only at baseline but also at different time points. Therefore, MSM allows controlling for effects of time-dependent confounders that predict the subsequent treatment and that are predicted by previous treatment. 28
The MSM using IPTW is related to propensity scoring. 29,30 The IPTW is the inverse of the propensity scores that are calculated using multinomial logistic regression in case of multiple treatment settings (treatments A, B, and control). 25,27 If the propensity score distribution has large variability (possibly because some covariates are highly correlated with treatment) then the treatment patterns will have extremely large weights. 28 Robins et al 21 and Hernán et al 31 recommend replacing the IPTW with stabilized weights to reduce this variability and ensure the estimated treatment effect remains unbiased. 28
The estimates obtained from MSM were compared with the HRs obtained from time-dependent Cox model to determine the bias due to presence of time-varying confounding factors (hospitalization).
Subgroup Analysis
Two separate subgroup analysis were carried out that restricted the age-group to ≥65 years and that used a new user design for drug exposure. 32 The study was approved by the Institutional Review Board of University of Houston.
Results
A total of 8062 patients with HF were included in the final analysis that met inclusion and exclusion criteria. Of these, 1135 (14.1%) patients were diagnosed with dementia over a follow-up to 36-month period between January 01, 2009, and December 31, 2011. The median length of follow-up for the cohort was 22 months for a total follow-up of 180 381 patient months. Of these, 76,948 patient months were of current use of ACEI/ARB, 51 021 patient months were of former use of ACEI/ARB, and 52 412 patient months were of nonuse of ACEI/ARB.
Patient Characteristics
Table 1 compares the baseline characteristics of patients with and without dementia. The mean age (± standard deviation) of the HF cohort was 74.5 (±9.21) years. Patients with dementia were older (78.2 ± 8.5 years) than those without dementia (73.9 ± 9.2 years, P < .0001). The incidence of dementia increased with age from 7.0% in the <65-year age-group to 9.2% in the 65 to 74, to 17.5% in the 75 to 84, and to 26.3% in the >85-year age-groups (Figure 3). Sex was not associated with a diagnosis of dementia. (P = .199).
Characteristics of Patient With CHF—8062 Patients.
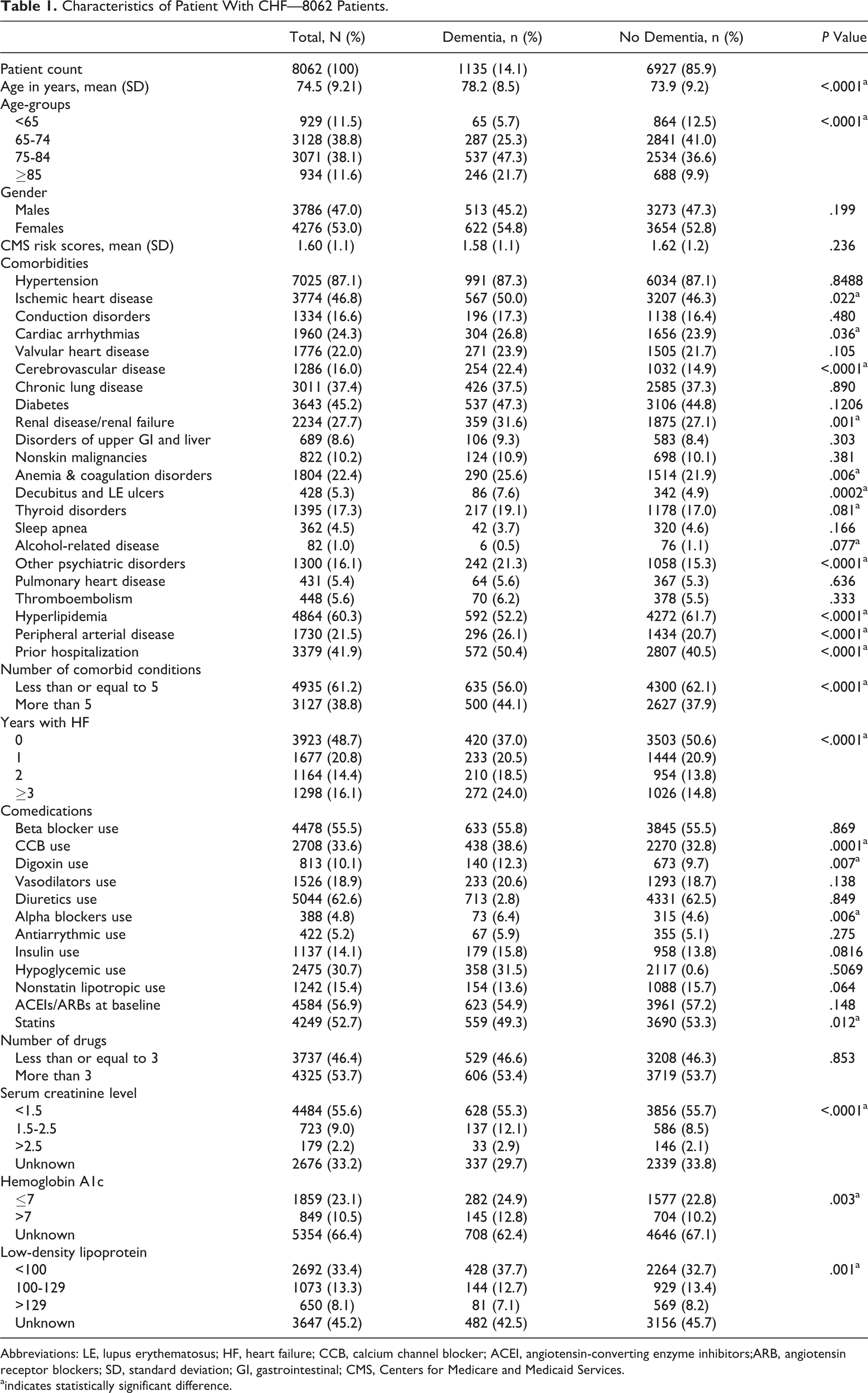
Abbreviations: LE, lupus erythematosus; HF, heart failure; CCB, calcium channel blocker; ACEI, angiotensin-converting enzyme inhibitors;ARB, angiotensin receptor blockers; SD, standard deviation; GI, gastrointestinal; CMS, Centers for Medicare and Medicaid Services.
aindicates statistically significant difference.
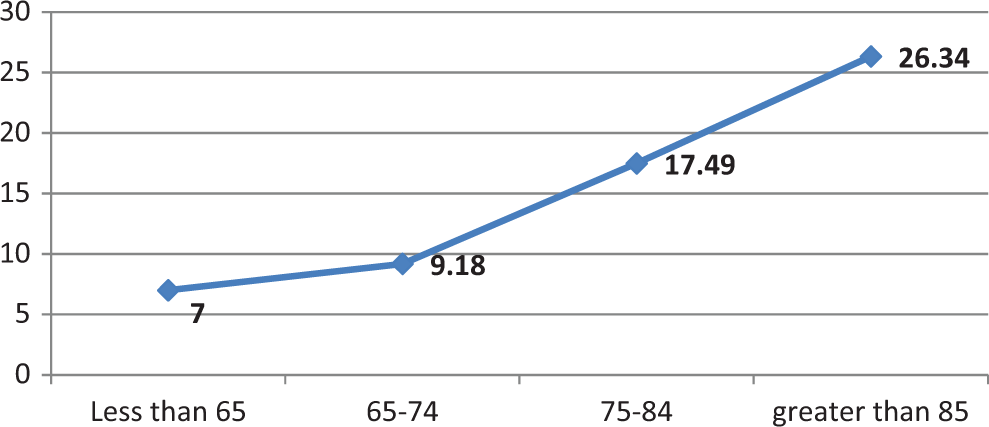
Incidence of dementia among different age-groups.
About 39% patients had more than 5 comorbid conditions. Of the 22 comorbid conditions, top 5 comorbid conditions were hypertension (87.1%), hyperlipidemia (60.3%), ischemic heart disease (46.8%), diabetes (45.2%), and chronic lung disease (37.4%). Approximately 42% of the patients had prior hospitalization, which was significantly associated with dementia (P < .0001). Increasing years with HF and higher comorbid conditions were each associated with a higher incidence of dementia (P < .0001).
In unadjusted analysis, patients taking statins (P = .012) had a lower incidence of dementia, whereas those taking CCBs (P = .0001), digoxin (P = .007), and alpha blockers (P = .006) had a higher incidence of dementia. The average daily dose for baseline use of ACEI/ARB (enalapril equivalent) was 21.4 (±12.6) mg, and 65.0% (n = 2978) of patients received the target dose among the baseline users of ACEI/ARB (n = 4584).
Time-Dependent Cox Model
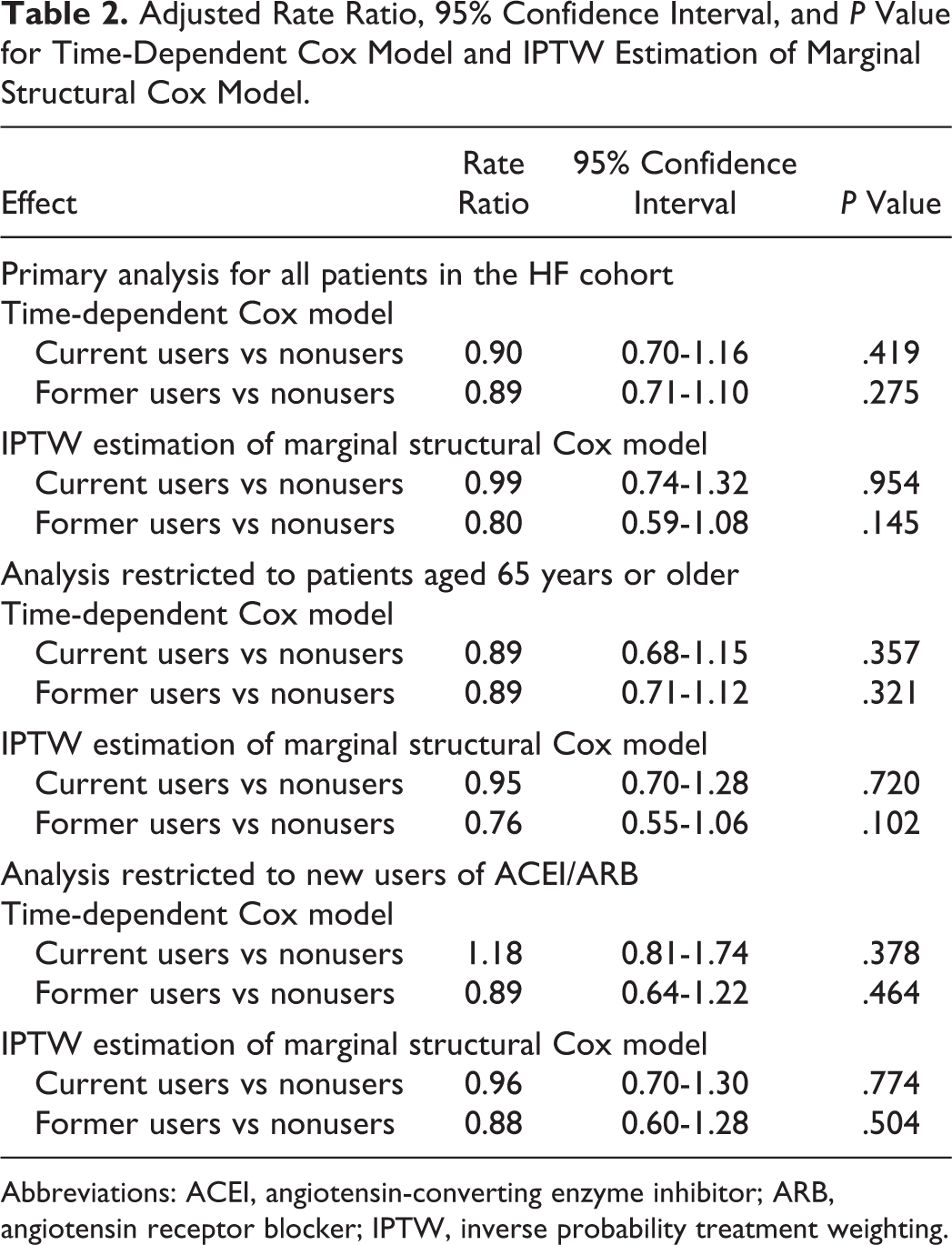
There were no significant differences in the crude dementia rates for baseline users of ACEI/ARB (13.59%) versus nonusers (14.72%; P = 0.148). However, the Kaplan-Meier curve showed that unadjusted survival was significantly different across baseline ACEI/ARB users versus nonusers (P = .028 for log rank test). Multivariable time-dependent Cox model found that as compared with the nonusers, the adjusted HR did not differ significantly among the current users of ACEIs/ARBs (HR, 0.90; 95% confidence interval [CI], 0.70-1.16) or among former users (HR, 0.89; 95% CI, 0.71-1.10; Table 2).
Adjusted Rate Ratio, 95% Confidence Interval, and P Value for Time-Dependent Cox Model and IPTW Estimation of Marginal Structural Cox Model.
Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; IPTW, inverse probability treatment weighting.
Marginal Structural Cox Model
The distribution of stabilized weights was symmetric and centered around 1 at all times (mean = 1.007, SD = 0.228). After adjusting for the time-dependent confounder (hospitalization) in addition to the other confounders (listed in Table 1) and applying stabilized weights to a pooled logistic regression, the estimated causal dementia rate ratio was 0.99 (95% conservative CI: 0.74-1.32) for current users and 0.80 (95% conservative CI: 0.59-1.08) for former users. Thus, under our assumptions of positivity, consistency, no model misspecification, and no unmeasured confounding, ACEI/ARB current use or former use does not appear to decrease the risk of dementia. (Table 2) These estimates were similar to the HRs obtained from the standard time-dependent Cox model.
Subgroup Analysis
We performed 2 subgroup analyses to test the robustness of study findings (Table 2). To assess for the possible bias from inclusion of patients <65 years of age (patients with end stage renal disease [ESRD] or disability), we restricted our analysis to all persons aged 65 years of age or older. Findings were essentially identical to those of the primary analysis for both time-dependent Cox model (current users, HR, 0.89; 95% CI, 0.68-1.15; former users, HR, 0.89; 95% CI, 0.71-1.12) and marginal structural Cox model (current users, rate ratio, 0.95; 95% CI, 0.70-1.28; former users, rate ratio, 0.76; 95% CI, 0.55-1.06). In the analyses that included only the new users (defined as no use of ACEI/ARB during the 6 months prior to the index date), we found no association between ACEI/ARB use (current and former) and risk of dementia.
Discussion
This study tested the hypothesis that ACEI/ARB would reduce the risk of dementia in patients with HF. These results demonstrate that with the current or former use of ACEI/ARB, there was no significant difference in the risk of dementia as compared with the nonuse. Unbiased estimation of the effect of treatment in an observational study requires adjustment for time-dependent confounding. 33 Standard time-dependent Cox model that incorporates time-varying variables give biased estimates of the true effect of treatment in presence of these time-varying confounders. Therefore, to estimate the causal effect of ACEI/ARB on risk of dementia, we used both a time-dependent Cox model and IPTW estimation of a marginal structural Cox model. The rate ratios obtained from the standard Cox model were similar to that of the MSM, indicating that the mediation by the time-varying confounder (hospitalization) considered in the study was minimal.
We performed subgroup analyses to test the robustness of our findings. We restricted our analysis to all persons aged 65 years or older to assess for the possible bias from inclusion of patients <65 years of age with ESRD or disability. We also performed an analysis restricted to new users to address bias that would be introduced from the inclusion of prevalent users in the cohort. 32 The findings from the subgroup analyses were essentially identical to those of the primary analysis.
To the best of our knowledge, this is the first study to examine the effect of ACEI/ARB use on risk of dementia in patients with HF. One observational study by Zuccala et al 18 assessed the effect of treatment with ACEI on cognitive improvement in patients with HF. The study found that starting the treatment with ACEIs was significantly associated with improvement in cognitive performance (OR, 1.57; 95% CI 1.18-2.08) among patients with HF. Our conclusion differs from theirs in reporting no difference in effectiveness of ACEIs/ARBs use versus nonuse. One reason for this could be the outcome considered: Zuccala et al examined cognitive performance, whereas our study assessed the risk of dementia. However, studies have shown that mild cognitive impairment is a strong predictor of dementia and AD. 34 Another reason could be the median follow-up time: Zuccala et al had a very short median follow-up time of 13 days, whereas our study had a considerably longer median follow-up time of 22 months. Third reason could be the study population and the exposure group considered: Zuccala et al failed to mention whether the patients included were incident or prevalent cases with HF and considered the effect of ACEI therapy only, whereas our study represented real-world practice settings that included a mixture of incident and prevalent cases with CHF. Also, the exposure group considered was use of either ACEI or ARB.
There are several theoretical reasons through which ACEI/ARB could offer neuroprotection. The ACEI/ARBs in addition to controlling comorbid hypertension can increase the impaired cerebral blood flow that is common in AD. This effect can be attributed to the improvement in left ventricular systolic function with use of these drugs. 18,35,36 Heart failure is associated with increased angiotensin II activity in the brain, and angiotensin II mediates inhibition of acetylcholine release and produces endothelial dysfunction in the cerebral circulation. 18,37 Thus, inhibition of the signaling by angiotensin II might provide beneficial effects in AD. The ACEI/ARB may inhibit or block the angiotensin II-mediated activities as well as interrupt the upregulation of inflammatory nitric oxide synthase isoenzymes that are associated with AD and dementia. 14,35 Despite these probable pharmacological mechanisms, our results suggest no statistical difference in the effectiveness among current and former users as compared with nonusers in reducing dementia in HF.
The Literature on the Effect of ACEI/ARB Use on Risk of Dementia in Population Without HF is Mixed
Some prior studies have shown a statistically significant decrease in risk of AD and other dementia among persons receiving ACEI/ARB therapy. In a 1-year, prospective, randomized, parallel-group cohort trial, Ohrui et al 38 found that brain-penetrating ACEI decreased the progression of AD significantly as compared with a nonpenetrating ACEI or CCB. The Heart Outcomes Prevention Evaluation (HOPE) 39 Study and the Perindopril Protection Against Recurrent Stroke Study (PROGRESS) 40 demonstrated a 41% reduction in cognitive decline associated with stroke and a 34% reduction in dementia among patients with recurrent stroke with the use of an ACEI. The post hoc analysis of Study on Cognition and Prognosis in the Elderly (SCOPE) found that the cognitive decline in the ARB group (candesartan) was significantly lower (RR, 0.49), suggesting that candesartan is neuroprotective. 41 Our prior work did find a significant association between ACEIs, ARBs and risk of dementia in patients with diabetes. When compared to the nonusers, there was 24% and 18% decrease in the risk of dementia in patients on ARB and ACEI, respectively. 10
In contrast, other studies have failed to show a lower risk of AD and dementia with ACEI/ARB use. A study by Khachaturian et al 16 examined the relationship of antihypertensive drugs use and the incidence of AD in an elderly population (age ≥65 years) of Cache County, Utah. This observational study involved more than 3000 participants and revealed increased protection from AD with the use of antihypertensive medication as a whole. However, the study failed to show any association with the use of ACEIs and risk of dementia. Hanon et al 42 conducted a cross-sectional study of 1241 patients with hypertension from a cohort of elderly patients who reported memory loss and were attending a geriatric memory clinic. 12 No significant associations were found with the use of ACEI or ARBs and incidence of AD. None of the aforementioned studies was done to assess the effects of ACEI/ARB use on an already at-risk HF population.
A possible explanation for our findings is that follow-up period was relatively short with median length of follow-up of 22 months to observe incident dementia. However, this study found significant increases in dementia rates as age increased, thus giving face validity to the study results (Figure 3). 10 Another reason could be confounding by severity of illness, as we could not account for information on EF and NYHA HF classification. However, we attempted to control for this by adjusting for the number of years with HF, number of all medicine, and prior hospitalization of the patients, as in other large studies of patients with HF. 24,25,27 We also adjusted for 21 comorbidities, CMS risk scores, and accounted for time-dependent confounding by indication by adding monthly hospitalization records as a severity of illness factor in MSM. Finally, our findings are a cross-sectional view of real clinical practice in a cohort of patients with HF, with a follow-up of 3 years for a long-term condition. These results may not be generalizable to patients without HF, as previous literature shows an interactive effect between use of ACEI and HF on cognitive impairment. 18
Our study possesses several unique strengths. Our cohort, with a high prevalence of comorbidities and abundantly prescribed comedications, adequately represents the patient population in real-world practice settings. Our study uses some important clinical factors such as the serum creatinine, HbA1c, and LDL-C values that are not usually recorded in retrospective databases. Use of dose information of the ACEIs/ARBs strengthens the results, as it plays an important role in predicting the clinical outcomes in patients with HF. 43 We addressed the issue of nonrandomized treatment allocation in an observational study by creating a pseudo-randomized sample by applying inverse probability treatment weights to the sample. 20 As time-dependent Cox model cannot address the issue of time-dependent confounding by indication, we also carried out the analysis using MSM. Our results were robust independent of the analytical strategy and the subgroup analyses performed.
The interpretation of results of this study should be made in light of several limitations. First, for MSM to have valid estimates, we make an assumption of no unmeasured confounding, which remains untestable. However, it is likely that any potential unmeasured confounders would be somewhat correlated with the numerous sociodemographic, clinical, and severity of illness measures that were measured, thus reducing residual confounding. Future research is needed that may include prescriber-related factors such as physician specialty, size of institution, and location, as these may influence the receipt of ACEIs/ARBs. In addition to lacking information on disease severity (EF and NYHA HF classification), the ICD-9-CM codes used to identify HF cannot differentiate between systolic and diastolic left ventricular dysfunction. Hogg et al 44 estimated patients with HF to be equally distributed between systolic and diastolic dysfunction, suggesting it is reasonable to consider them same and thus studying effects of treatment on all patients with HF. 23 Dementia was identified using a clinician-reported ICD-9 diagnosis code that likely is a much cruder end point than a valid instrument measuring cognitive function administered in a standard fashion and frequency. Distinguishing vascular from Alzheimer-type dementia was probably not reliable in our data. Also, we could not control for potential risk factors for dementia, such as race, education, diet, smoking, and alcohol use. Finally, dispensed prescriptions were considered as actually consumed. However, in general, pharmacy claims are demonstrated to be an accurate measure of prescription drug consumption. 45 To the extent possible, measures of adherence or persistence of use were created in a cumulative and time-varying manner during follow-up to strengthen analyses.
In conclusion, in the absence of randomized clinical trials, appropriate adjustment for time-varying confounding by indication may provide the best evidence to estimate the treatment effect in observational studies. This exploratory study using time-dependent Cox model and MSM found no association between the current and the former use of ACEI/ARB as compared with the nonuse in a HF cohort. However, we note that MSM provided similar effect estimates to the time-dependent Cox model indicating that the mediation by time-dependent confounder considered in the study was minimal. Further research needs to be done in HF population in which other measures such as changing blood pressure or EFs could be handled as time-varying confounders. Also, it would be interesting to compare the effects of ARBs versus ACEIs and furthermore compare brain-pentrating ACEIs versus non brain-penetrating ACEIs.
Footnotes
Appendix A
Dementia Diagnostic Codes.
| ICD-9-CM | Diagnosis |
|---|---|
| 046.1 | Creutzfeldt-Jakob Disease |
| 046.3 | Progressive Multifocal Leukoencephalopathy |
| 290.0 | Senile Dementia, Uncomplicated |
| 290.10 | Presenile Dementia, Uncomplicated |
| 290.11 | Presenile Dementia w/ Delirium |
| 290.12 | Presenile Dementia w/ Delusional Features |
| 290.13 | Presenile Dementia w/ Depressive Features |
| 290.20 | Senile Dementia w/ Delusional Features |
| 290.21 | Senile Dementia w/ Depressive Features |
| 290.3 | Senile Dementia w/ Delirium |
| 290.40 | Arteriosclerotic Dementia, Uncomplicated |
| 290.41 | Arteriosclerotic Dementia w/ Delirium |
| 290.42 | Arteriosclerotic Dementia w/ Delusional Features |
| 290.43 | Arteriosclerotic Dementia w/ Depressive Features |
| 291.2 | Other alcoholic dementia |
| 292.82 | Drug-induced persisting dementia |
| 294.10 | Dementia in conditions classified elsewhere without behavioral disturbance |
| 294.11 | Dementia in conditions classified elsewhere with behavioral disturbance |
| 294.8 | Other Spec Organic Brain Syndromes (Chronic) |
| 294.9 | Unspec Organic Brain Syndrome (Chronic) |
| 331.0 | Alzheimer’s Disease |
| 331.11 | Pick’s Disease |
| 331.19 | Other Frontotemporal Dementias |
| 331.2 | Senile Degeneration of Brain |
| 331.7 | Cerebral Degeneration in Diseases Classified Elsewhere |
| 331.82 | Dementia with Lewy Bodies |
| 331.89 | Other Cerebral Degeneration |
| 331.9 | Cerebral Degeneration, Unspec |
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported with the use of facilities and resources of the VA HSR&D Center for Innovations in Quality, Effectiveness and Safety (CIN13-413). The views expressed reflect those of the authors and not necessarily those of the Department of Veterans Affairs, the US government or Baylor College of Medicine.
