Abstract
Background/Rationale:
This report describes a quality improvement continuing medical education activity designed to enhance the recognition and treatment of residents with Alzheimer’s disease (AD) or other dementias in skilled-nursing facilities (SNFs).
Methods:
Charts were compared in 6 areas prior to and following (stages A and C) a live, faculty-led workshop (stage B). Four SNFs completed stages A (n = 67 residents) and B, and 3 SNFs completed stage C (n = 52 residents). All charts came from residents with AD or a diagnosis of dementia or dementia-like symptoms.
Results/Conclusion:
The SNFs had >95% baseline performance in both the frequency of cognitive assessments and documented medication reviews. The percentage of residents who received a quality-of-life assessment and those who had a mental health care plan in place represent areas for improvement. As part of this activity, a toolkit was developed to help guide facilities and clinicians in instituting care improvements for residents with AD/dementia.
Background/ Rationale
Alzheimer’s disease (AD) is estimated to affect 5.4 million individuals in the United States, and individuals live anywhere from 4 to 20 years after their symptoms become noticeable to others, depending on age and other coexisting conditions. 1 Of all individuals older than 65 years of age, an estimated 6% to 8% have AD or another form of dementia, and this rate exceeds 30% at ages 85 and older. 2 In June 2011, 47% of all nursing home residents had a diagnosis of dementia in their nursing home record. 3
People with AD can display a number of symptoms that impact behavior, memory, and thinking. These symptoms can be referred to as the ABCs of AD: activities of daily living (ADL), behavior, and cognition. However, a hallmark feature to watch for is a decline in any of the aforementioned domains and from previous levels of functioning. As the disease advances, so can symptom severity, such that patients may experience increasing disorientation, mood, and behavior changes; deepening confusion about events, time, and place; unfounded suspicions about family, friends, and professional caregivers; more serious memory loss and behavior changes; and difficulty speaking, swallowing, and walking. The other symptoms can actually lessen in severity. Caring for residents with AD or other forms of chronic dementia in nursing homes is difficult due to residents with AD requiring more supervision when performing ADLs; a lack of resources; failing resident health complicated by the presence of medical comorbidities; and the rapidly growing, affected population. More research is needed on interventions that may be used in the nursing home environment to reduce falls and any other negative outcomes in residents with a cognitive impairment (CI). When staff are properly trained, caring can become easier.
An international working group convened by the National Institute on Aging and the Alzheimer’s Association (NIA-AA) published diagnostic criteria for both the predementia phase and for AD dementia. 4 Prior to the publication of the 2011 guidelines, the diagnostic criteria used by practitioners primarily came from criteria published in 1984. The 2011 guidance put forth a model for the trajectory of AD and indicates that AD is fundamentally a continuum of brain changes. The continuum is composed of a preclinical phase, during which pathophysiological changes start, to mild cognitive impairment (MCI), during which CI is detectable, and finally to AD dementia. 5 Alzheimer’s disease is the most common type of dementia, but vascular disease, Lewy body disease, and frontotemporal degenerative syndromes are other common causes of dementia. 6
Due to the growing burden of dementia and the exponentially increasing number of older patients with complex health conditions, we examined charts from residents with either the more general diagnosis of CI and dementia or AD dementia in skilled-nursing facilities (SNFs). To this end, a quality improvement (QI) continuing medical education (CME) activity was designed. This activity was designed to be responsive to changing health care needs of SNFs and to enhance the recognition and treatment of residents with AD or other dementias through the joint sponsorship of Indiana University School of Medicine, Division of Continuing Medical Education and the Academy for Continued Healthcare Learning (ACHL). The activity allowed participating facilities to retrospectively assess their current clinical practice; select areas for improvement; implement interventions based on treatment guidelines and health care standards; and then reevaluate their practices and procedures. This article describes the ways in which participating SNFs sought to improve their performance, and the action plans they initiated to meet their goals in an effort to contribute to both the knowledge and the practice of caring for residents with AD and other dementia types in these settings.
Methods
The QI model used for this activity was based on well-established models of performance improvement and QI, including the Institute for Healthcare Improvement Model for Improvement. 7 -10 In this QI activity, data from SNFs participating in the baseline assessment (stage A) were aggregated and compared against data from SNFs participating in reassessment phase (stage C; Table 1).
QI CME Model.a
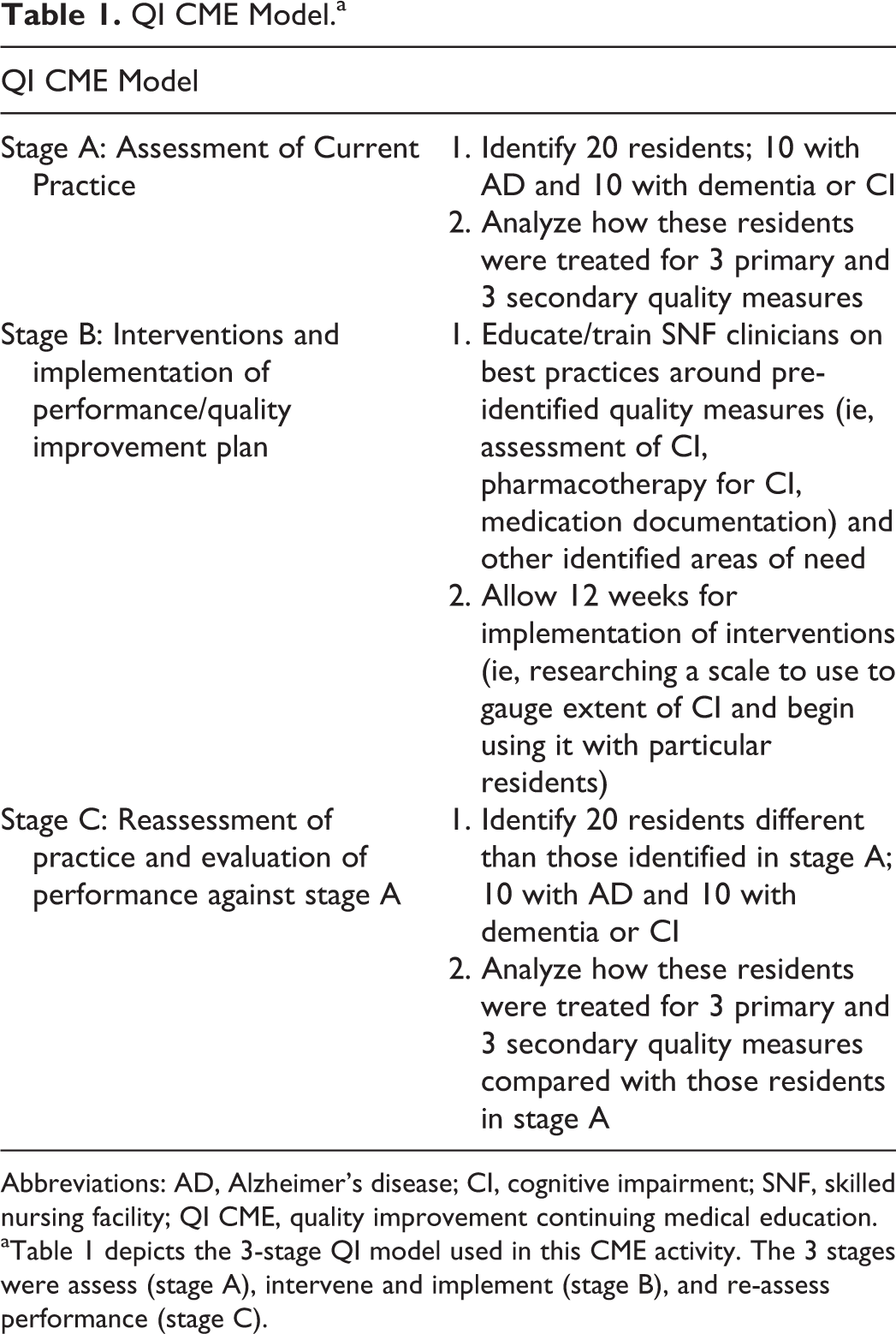
Abbreviations: AD, Alzheimer’s disease; CI, cognitive impairment; SNF, skilled nursing facility; QI CME, quality improvement continuing medical education.
a Table 1 depicts the 3-stage QI model used in this CME activity. The 3 stages were assess (stage A), intervene and implement (stage B), and re-assess performance (stage C).
The planning phase for the activity consisted of inviting faculty regarded as experts in AD and dementia care. An activity chair was identified and led the efforts on recruiting additional faculty as well as identifying the quality measures and interventions for the activity. Next, outreach was conducted to recruit volunteer SNFs with an interest in the QI activity. Upon confirmation, a champion, or an employee of the SNF, was identified at each facility to help coordinate the activity. Their responsibilities included the overall management of participating clinicians at their respective SNF; reviewing charts and entering data onto the data collection forms (during baseline [stage A] and reassessment [stage C]); and coordination of a live workshop conducted at their SNF (part of the stage B intervention). Designated champions were then provided instructions on completing the activity, and an ACHL program manager was available to answer questions.
Each faculty member had an opportunity before each live workshop meeting to customize a core slide set according to the SNF’s interests and needs. After a literature review, the following QI primary measures were selected for this activity (Table 2).
Primary and Secondary Quality Measures.a
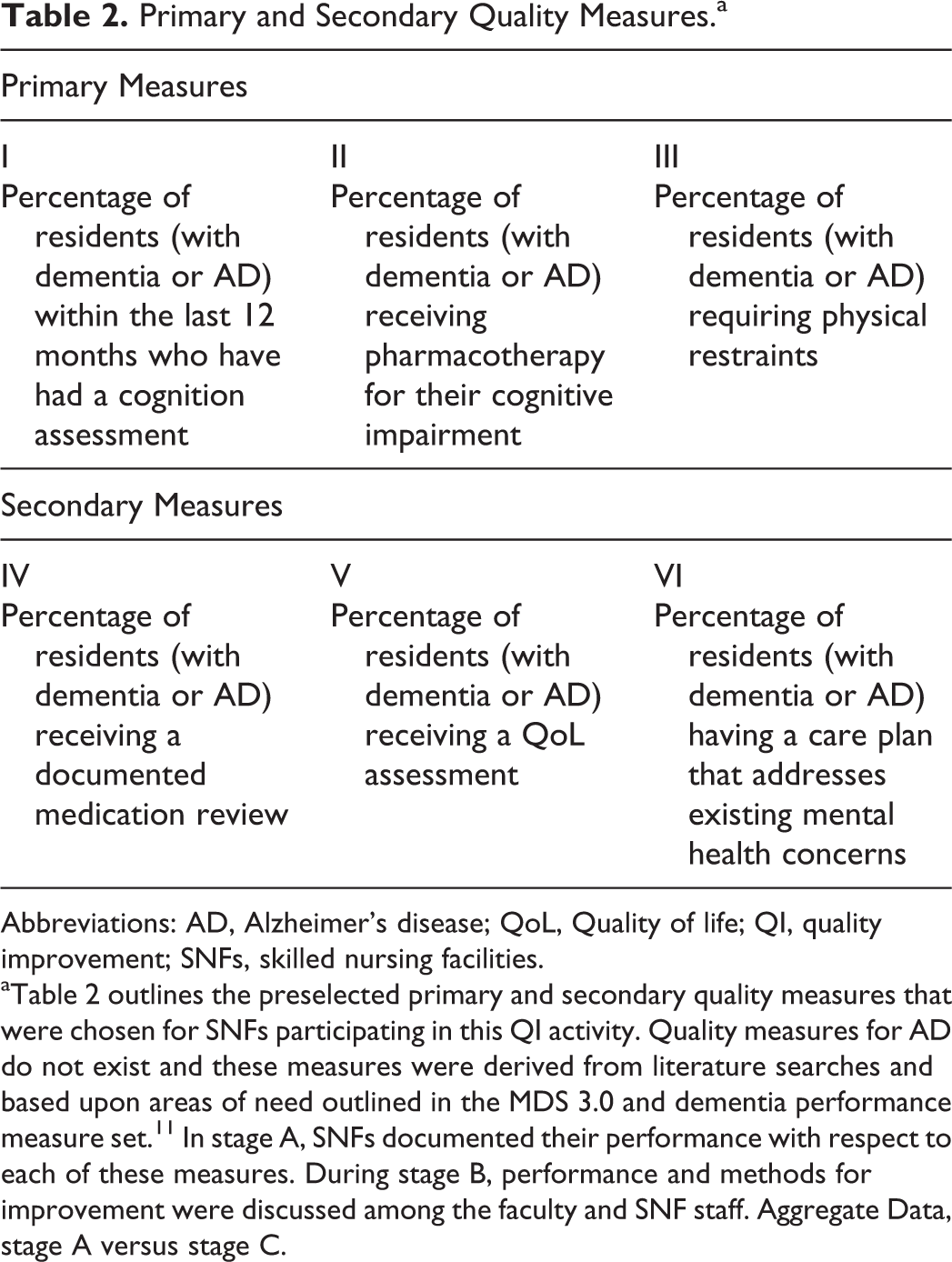
Abbreviations: AD, Alzheimer’s disease; QoL, Quality of life; QI, quality improvement; SNFs, skilled nursing facilities.
a Table 2 outlines the preselected primary and secondary quality measures that were chosen for SNFs participating in this QI activity. Quality measures for AD do not exist and these measures were derived from literature searches and based upon areas of need outlined in the MDS 3.0 and dementia performance measure set. 11 In stage A, SNFs documented their performance with respect to each of these measures. During stage B, performance and methods for improvement were discussed among the faculty and SNF staff. Aggregate Data, stage A versus stage C.
Furthermore, “documented” refers to written evidence indicating whether a procedure/discussion was indicated/conducted or not indicated/not conducted. The denominator for these calculations was the number of residents with AD (diagnosed and reported on their charts) combined with the number of residents with any dementia (a maximum of n = 20 per SNF). An n = 20 was chosen as it was feasible to get this number of residents with AD from an SNF regardless of whether it had an AD special care unit.
It is important to stress that these measures were chosen in the absence of formal, society (or institute)-endorsed quality measures for nursing home residents with AD but were based on existing tools and measures for residents with dementia, including the Minimum Data Set 3.0 (MDS, nursing home assessment tool) and Centers for Medicare & Medicaid Services (CMS) nursing home quality measures. 11 -13
During stage A, each champion was instructed to randomly select 20 resident charts and enter data in a predefined data collection form. Patient identifiers were removed prior to data analyses. No institutional review board was involved, as we did not perform interventions at the resident level but rather reviewed chart data and facility performance in aggregate. In stage C, champions randomly selected 20 different resident charts for review at least 12 weeks after implementation of the stage B interventions (ie, live workshop and action plans). Different charts were chosen between stages A and C to allow SNFs to circumvent age-related and disease complications (ie, deaths and transitioning to hospice care). Analyzing performance between stages A and C allowed us to gauge the effectiveness of the interventions at a systems level. A total of 5 SNFs applied to participate in this QI activity, of the 1500 SNFs that were recruited and of the 50 that expressed initial interest. One SNF dropped out from the activity prior to stage A. Thus, the data analyses included 4 SNFs that provided stage A baseline data and participated in stage B. Because an additional SNF dropped out after stage B, only data from 3 of the SNFs were included in stage C.
The stage B intervention consisted of live workshops customized for each SNF. The workshop offered CME and continuing education (CE) credit for physicians (MDs), nurses (RNs), and nurse practitioners (NPs) who comprised the target audience. A total of 47 clinicians attended the stage B workshops (2 MDs, 19 RNs, 1 NP, 2 occupational and physical therapists (OT/PTs), and 23 other health care practitioners). Prior to each workshop, the chair-appointed faculty member reviewed a particular SNF’s stage A data (ie, SNF performance on the 3 primary and the 3 secondary measures) and determined any performance areas in need of improvement. Each dialectic lecture began with a review of how the SNF performed on each measure and was followed by best practices that could be used to achieve still higher performance levels as well as both pharmacological and nonpharmacological strategies that could be used to meet these ends. Additional content and discussion centered on implementation challenges and also focused on other areas of need within the SNF as identified by the staff attending stage B and as they related to residents with dementia. In addition, as part of the stage B intervention, workshop attendees received a syllabus, which included learning objectives; a resources listing that included performance measures as they related to dementia; clinical practice guidelines; NIA-AA recommendations; and relevant online CME activities for the management of AD. Stage B components can be found in the toolkit at http://www.achlqicme.org/alz/Toolkit.aspx. All toolkit components were also faculty approved. Furthermore, space was allotted within the syllabus for the development of an action plan. At the end of the workshop, faculty led a discussion with attendees to develop a customized action plan that focused on 2 to 3 goals that could be implemented at their facility using strategies discussed at the workshop to further improve the care and health outcomes of residents with dementia. The SNFs were free to also choose other areas for improvement unrelated to the activity’s primary and secondary measures if staff felt that there were more pressing measures in need of improvement.
Statistical Analysis
This study was designed to identify the impact of the stage B interventions on 3 primary and 3 secondary measures serving as indicators of change in the management of residents with AD and/or other dementia. The data analyses were based on independent samples obtained via random selection of patient charts at 4 SNFs before the workshop (stage A) and at 3 of those facilities following the workshop (stage C). The data comprised 2 sets of records: the first set collected during stage A (n = 67) and the second collected during stage C (n = 52). Binary (yes/no) responses were compared between stages A and C by a chi-square general association test.
Results
When this QI program was designed, it was not done so according to the Reach Effectiveness Adoption Implementation Maintenance (RE-AIM) criteria 14 of validating whether the intervention was applicable and/or sustainable. The RE-AIM methodology also allows investigators to translate research findings into practice. An attempt is made to frame the outcomes according to this methodology so as to provide a more useful organization of the findings. The RE-AIM framework allows for the evaluation of the “robustness, translatability, and public health impact” of health behavior interventions by assessing a project on the following dimensions: reach into the target population; efficacy; adoption of procedures by staff; implementation (consistency and cost); and maintenance of the intervention over time.
Reach (a Description of the Target Population)
A total of 5 SNFs were recruited to participate in this QI activity. All 5 facilities were state licensed and signed agreements to participate in this QI activity. Again, 4 of the 5 recruited SNFs provided stage A baseline data and participated in stage B (live workshop/action plan); however, only 3 SNFs completed stage C. The 4 participating SNFs were located in Illinois, Missouri, New Jersey, and North Carolina.
Three of the SNFs accepted individuals who were self-pay or had either Medicare or Medicaid, and only 1 was strictly self-pay. Of the 3 care facilities that completed the activity (through stage C), 2 had specific units that housed residents with AD exclusively (ie, special care units). Among the residents sampled, sex and age were not significantly different for stages A and C. Approximately 75% of residents were female, and the mean ages were 82.9 years (stage A) and 84.9 years (stage C).
The chart data collected from each SNF was based on 10 residents with a documented diagnosis of AD, in combination with 10 residents without a diagnosis of AD (these residents could have other dementias [type not specified] or dementia-like symptoms), and this yielded a maximum of 20 resident charts per SNF.
Efficacy or Aggregate Data Analyses
There were no statistically significant differences noted in any of the primary or secondary measures between stages A (n = 67) and C (n = 52; P > .05; nonsignificant [NS]; Figure 1). In some cases, high initial, baseline performance was present (ie, a ceiling effect). For instance, the percentage of residents who received a cognitive assessment on at least an annual basis was 97% in stage A (90.4% in stage C, P = NS). Similarly, documented medication reviews occurred in 97% of patients in stage A (94.2% in stage C, P = NS). In addition, a floor effect was present for the percentage of residents who experienced restraint use in stage A (2.3%) versus stage C (2.2%; P = NS).
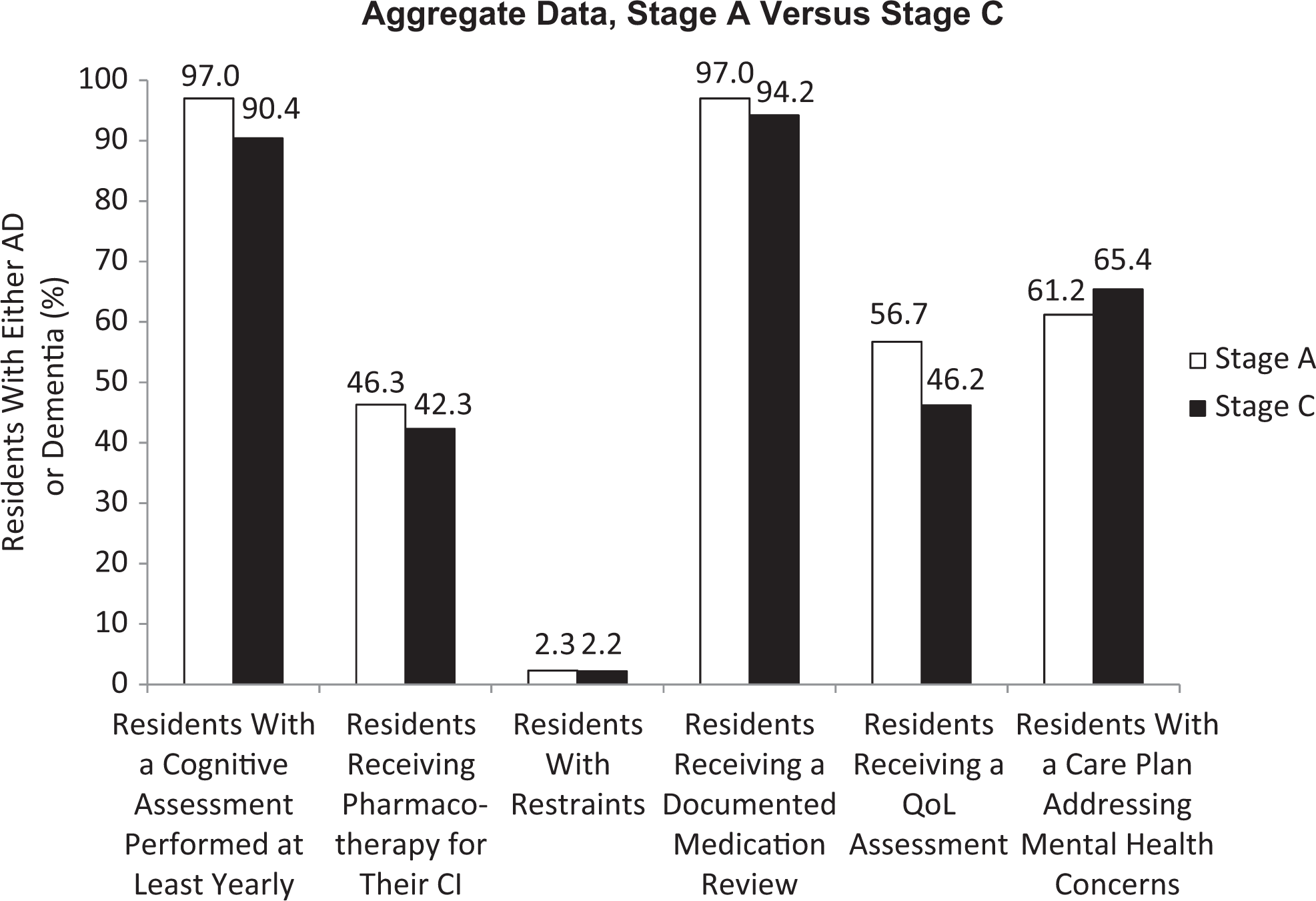
There were no statistically significant differences noted in any of the primary or secondary measures between stages A and C (P > .05). Note that stage A performance was high for residents receiving a cognitive assessment on at least a yearly basis as well as those residents receiving a documented medication review (>95%). Areas for improvement among the SNFs sampled include the percentage of residents receiving pharmacotherapy or a QoL assessment as well as the percentage of residents with a care plan that addresses mental health concerns. CI indicates cognitive impairment; QoL, quality of life; SNFs, skilled nursing facilities.
Areas for potential improvement among SNFs include the percentage of residents who receive pharmacotherapy for their CI (stage A, 46.3%; stage C, 42.3%; P = NS); the percentage of residents who receive a quality-of-life (QoL) assessment (stage A, 56.7%; stage C, 46.2%; P = NS); and the percentage of residents with a care plan that addresses mental health concerns (stage A, 61.2%; stage C, 65.4%; P = NS). Additionally, a 2 × 2 analysis revealed that there was a positive interaction between QoL and interventions to modify behavior (which included both nonpharmacological and pharmacological strategies) from stages A to C (P = .002).
A trend toward increased severity of dementia was noted among residents from stages A to C (P = .058). The percentage of residents with mild–moderate dementia tended to decrease from stages A to C (70.4% vs 33.3%), whereas the percentage of residents with moderate–severe dementia tended to increase (29.6% vs 47.6%). Similarly, the percentage of residents whose basic activities of daily living were affected from stages A to C approached statistical significance (78.9% vs 96.2%, P = .054). Thus, it seems that the group of residents analyzed in stage C was more disabled, and likely needed more assistance, than the group of residents analyzed during stage A.
Implementation and Maintenance
In addition to reporting on the primary and secondary measures, facilities often implemented additional interventions to further improve the care of residents with AD/CI within their facilities. One facility had an in-service for all staff on fall preventions and installed cameras and monitors in places where falls occurred the most. The same facility also brought on an activity assistant to help with more daytime activities for the residents. A separate facility started to draft protocols to gauge pain levels in residents with dementia and will eventually implement the protocols as part of the patient logs. A third facility hired a new champion for facility quality improvement. Results from these interventions were not immediately available, and activity champions noted that it could take up to a year before protocols were implemented and results were documented. Thus, while significant changes in the primary and secondary performance were not detected in this activity, additional procedures to improve the care of residents with AD or other dementias were both implemented and maintained over the long term.
Conclusion
Although this activity did not produce statistically significant changes in the identified measures, facilities that agreed to participate in the activity are likely at the higher end of performance in the SNF industry. Two of the 3 facilities completing the activity had special care units for residents with AD. Thus, the high baseline levels for these facilities are not characteristic of all facilities and in fact may represent practices that occur at facilities with more staffing and resources, especially given that all facilities accepted self-pay residents. The utility of this activity rests in the types of interventions that were implemented as part of the QI CME program such as those used to improve QoL, measure and document pain, and reduce falls among residents with AD/CI. Activities such as this are important to help propel the long-term care industry forward, as there is a severe dearth of performance and quality measures that SNFs can utilize to improve care for residents with AD and other dementias.
Our outcomes demonstrate that the 4 SNFs within our sample are screening at least 90% of their residents with AD and/or dementia for changes in their CI on at least an annual basis. The high baseline performance for this measure is most likely due to SNFs using the MDS which has staff evaluate resident cognitive status via resident questioning with either the Brief Interview for Mental Status or staff assessment (section C). 12 The MDS is required by CMS to be completed by all nursing homes and swing bed providers in order to have a standardized method of performance assessment. All 4 SNFs in our sample were also performing a documented medication review, mostly on a monthly basis and sometimes on a daily basis, especially for specific residents. As with cognitive screens, SNFs periodically assess for changes in medications/regimens via the MDS (section N). Thus, the reason for the high baseline performance is that many SNFs already screen for these measures at resident intake or if the residents are insured by Medicaid. Data that would have been useful to include would be the percentage or types of medication errors that occurred; however, due to data collection form length, we were wary of asking the champions more questions and asking them to spend more time collecting data. While selection bias may exist as these 4 SNFs were highly motivated and saw the QI activity through at least stage B, the intent of the activity is for any SNF to be able to improve the care of their residents with either AD or other dementias using the learnings from, and the tools created during, this activity. Performance measures specific to AD do not exist and are urgently needed to help standardize the diagnosis, management, and treatment of persons with AD. However, even when formalized performance measures are lacking, facilities can continue to improve resident care by critically and systematically assessing current practices and identifying recurrent, problematic issues for either or both residents and staff.
The percentage of residents who had care plans that addressed mental health concerns (ie, behavioral/psychological strategies to provide care and mitigate the disruption that may stem from either AD or other dementias) tended to increase from stages A to C. Moreover, because of a significant interaction between QoL and behavioral modification (which included both nonpharmacological and pharmacological approaches), educating staff on methods, including both pharmacologic and nonpharmacologic strategies to address behavioral issues, may contribute to a better QoL for both residents and caregivers and potentially make caregiving easier. 13,15 This activity surpasses the MDS as it attempts to examine QoL directly; in contrast, the MDS uses the effects of pain on resident functioning as well as section F (preferences for customary routine, activities, community setting) as an indirect QoL gauge. In the literature, no “gold standard” for assessing QoL exists, and general practice uses a combination of methods and sources to infer a resident’s QoL. 15 Again, the SNFs that participated go above and beyond the MDS to report on, more frequently monitor, and continually improve the QoL of residents with AD/dementia.
One SNF that completed stage C noted that the use of pharmacologic agents tended to decline over the course of the activity, as many of the residents with either dementia or AD who were sampled in stage C were in the end stages of their disease. Furthermore, the staff at this SNF felt that the medication’s effects were no longer beneficial in residents transitioning to hospice. The trend toward a decrease in dementia medication use from stages A to C may have been influenced by a higher proportion of residents with moderate–severe dementia in stage C compared with stage A (P = .058). Thus, although overall prescription medication use did not significantly change from stages A to C, the workshop may have resulted in more appropriate medication use (ie, residents with vascular dementia stopping/discontinuing medications specific for AD dementia). Although not significant, the result represents an opportunity for further clinical education. An interesting finding from our results revealed that there was a significant association in stage C between the use of behavior modification and being prescribed a prescription medication for AD such that 38.5% of the residents who were prescribed medication for their AD also received behavior modification (P = .025). One interpretation may be that clinicians for residents with AD/dementia who were receiving medication were also more likely to employ a broader range of other therapeutic modalities in the care of their patients.
In the current activity, the type of dementia that a resident had was not specified in the inclusion criteria for half of the patients (ie, vascular, mixed, Parkinsonian, Lewy body, frontotemporal, etc). There is literature to support dementia medication discontinuation among patients with end-stage dementia. If dementia medication is discontinued, it is often at the discretion of both medical providers and caregivers; medication discontinuation is also recommended when there is no improvement or there is a further decline in cognition and/or function following a duration of use of at least 6 months; adverse events are experienced by the patient and are attributable to the medication; or when other symptom management therapies are prescribed and started. 16 In addition, among many primary care providers as well as the public, there is a perception that AD medications are not useful because they are not curative. However, it has been demonstrated that the time spent providing care for patients with AD (ie, ADL assistance) could be reduced by an hour a day for patients receiving donepezil for over a year as compared with patients receiving placebo. 17 Additional medications that are available to treat mild–moderate AD include galantamine and rivastigmine. Memantine is usually reserved for the treatment of moderate–severe AD. In addition to dementia type, residents’ comorbidities were also not included as data points within the activity. Individuals with AD tend to have more comorbidities than patients without dementia, 18 and the prescribing practices for AD/dementia may also be influenced by a patient’s comorbidities. 19 Thus, these factors (ie, dementia type; severity and stage of dementia; and comorbidities) may have an impact on prescribing practices in SNFs. Not asking these targeted questions on our data collection form was a major study limitation, and it is likely that these data would have helped ascertain more information on the types of care provided by SNFs for residents with AD and other dementia types.
Additional nonpharmacologic strategies that the SNFs in our activity instituted as part of their action plan following the stage B intervention included the consistent assessment of QoL; standardization of cognitive scales (increased/consistent use of the Brief Interview for Mental Status [BIMS]); and music therapy/musical opportunities for residents.
The CMS released statistics in September 2012 regarding aggregate nursing home performance on a number of quality measures. Approximately 40.4% of long-stay residents had a cognitive impairment; 17.7% of long-stay residents had a formal diagnosis of AD while 45.8% of long-stay residents had a diagnosis of dementia. 20 Among the facilities that responded to the survey, 2.4% used restraints (ie, trunk or limb) with their residents. These numbers mirror our own findings: 2.2% to 2.3% of residents in our sample were restrained. The CMS report also notes that rates of restraint use have declined in the past decade (2002-2012). The MDS contains section P for staff to document the frequency with which restraints are used on residents. The floor effect seen with restraint use may be due to an increased awareness and practice among SNFs, similar to that seen with periodic screening for CI and documented medication reviews.
Also contained within the CMS report was the number of residents who have required increasing aid with ADLs (ie, bed mobility, transfers, eating, and toileting). This statistic was estimated to be 16.7% with a standard deviation of 9.3%. We noted that the residents in our sample had more moderate–severe dementia in stage C (47.6%) compared with stage A (29.6%). Additionally residents with mild–moderate dementia decreased from stage A (70.4%) to stage C (33.3%). Trends in data such as ours and reports like those from CMS demonstrate the need for more effective interventions in SNFs and for residents with AD/dementia.
The authors of the CMS report also state that residents’ functional decline may be lessened by specialized nursing care/other interventions. Authors comment that one reason for including this measure on MDS 3.0 was to prompt SNFs with poor performance to develop approaches to either improve or maintain residents’ involvement and control of ADLs.
It is also possible that discussions during stage B of our activity spurred participating SNFs to initiate other, positive changes in the care of their residents with AD/dementia. In fact, one SNF in our activity listed as one of their action plan goals the reduction in falls by 10% and, to this end, provided a fall-prevention training program for their staff and installed cameras in areas where many of the falls were occurring. Thus, although changes in our measures did not meet statistical significance, education provided to clinicians and caregivers within SNFs was utilized to initiate changes and improve the care of their residents with AD. This activity may also serve to initiate discussions regarding quality measures in long-term care facilities, which are less regulated and have significantly fewer reporting requirements than SNFs.
Based on the QI activity and the action plans provided by the participating SNFs, we developed an algorithm that may serve as a framework to implement a tailored QI activity in facilities that wish to further improve care for their residents with AD. In addition, a toolkit with resources (webcast, slides, and web links to references/helpful information) from the activity is available to the public at http://www.achlqicme.org/alz/Toolkit.aspx. We believe these resources contribute to the literature and bring important attention to a “path forward” for care quality of residents with AD.
Footnotes
This article was accepted under the editorship of the former Editor-in-Chief, Carol F. Lippa.
Acknowledgements
The authors would like to thank the following SNFs for their participation in this QI activity: Champaign County Nursing Home (Urbana, IL); Delmar Gardens Chesterfield (Chesterfield, MO); and Francis Parker Memorial Home, Inc (Piscataway, NJ). The authors would also like to thank Mark Van Buskirk (Data Reduction LLC; Chester, NJ) for statistical analysis. Finally, the authors would like to thank the accredited provider, Indiana University School of Medicine, and planning committee members Mikey Vogel, RN, Tarra Barot, PhD, Mindi Daiga, MBA, Lisa Keckich, MS, and Vanessa Senatore for planning and managing the QI activity.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: George T. Grossberg, MD: Dr Grossberg has served as a consultant for Accera, Inc; Baxter; Eli Lilly and Company; Forest Pharmaceuticals, Inc; Lundbeck; and Novartis Pharmaceuticals Corporation. He has also received research funds through St. Louis University from Baxter; Janssen Pharmaceuticals, Inc; and Pfizer Inc. He has also served on a safety monitoring committee for Merck & Co, Inc.
Martin Farlow, MD: Dr Farlow has served as a consultant for Accera, Inc; Alltech; Astellas Pharma US, Inc; Avanir Pharmaceuticals, Inc; Bayer AG; Bristol-Myers Squibb Co; Eisai Med Res, Inc; Eli Lilly and Company; GE Co; Helicon Therapeutics, Inc; Hoffman-La Roche Inc; MedAvante, Inc; Medivation, Inc; Merck and Co. Inc; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Prana Biotechnology Ltd; QR Pharma; Sanofi-Aventis US; Shire Pharmaceuticals; Toyama Pharmaceuticals; and UCB Pharma. He has received grant and research support from Eisai, Inc; Eli Lilly and Company; Genentech Inc; Hoffman-La Roche Inc; Novartis Pharmaceuticals Corporation; and Sanofi-Aventis US. Dr Farlow also has served on speakers bureaus for Eisai, Inc; Pfizer Inc.; Forest Pharmaceuticals, Inc; Novartis Pharmaceuticals Inc, and received funds for a patent sold to Elan Corporation, PLC.
The following authors have reported no conflicts of interest: Soo Borson, MD, Stephen Connor, PhD, and Mary Mittelman, DrPH.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This activity was supported by an educational grant from Pfizer Inc. (unrestricted educational grant number: 039880). Full control of methodology, analysis, content, and manuscript creation resided with the authors in full compliance with ACCME® Accreditation Policies and the ACCME Standards for Commercial SupportSM.
