Abstract
Objective:
Analysis sought to determine whether Wechsler Memory Scale-Logical Memory (LM)-correct responses and errors were related to magnetic resonance imaging (MRI) brain volume measurements.
Methods:
The LM immediate (LM-I) and LM delay (LM-D) free recall correct responses and related and unrelated errors were scored. Principal components analysis yielded a 3-factor solution: LM-I and LM-D correct responses, LM-I and LM-D-unrelated errors, and LM-I/-D-related errors. The MRI total cerebral brain volume, frontal brain volume, temporal horn volume (THV), and white matter hyperintensities volume (WMHIV) were obtained.
Results:
Increasing THV (suggesting greater regional atrophy) was associated with lower scores on the LM-correct responses factor. Extensive WMHIV was associated with higher scores on the LM-related errors factor.
Conclusion:
These results suggest that LM-correct responses could relate to emerging brain alterations. Longitudinal research might enhance the sensitivity of this test to identify preclinical impairment and persons at risk of mild cognitive impairment and dementia.
Keywords
Introduction
Impairment on memory tests is often an early indicator of an incipient or emerging mild cognitive impairment (MCI) or dementia. The 2 types of memory tests most often used to assess for amnesia and to characterize amnestic states in MCI and dementia are serial list-learning tests 1 –3 and paragraph recall tests such as the Wechsler Memory Scale (WMS)—Logical Memory (LM 4 ) subtest.
Research conducted with verbal serial list-learning tests over the past 30 years has demonstrated unique and specific patterns of impairment in Alzheimer’s disease (AD) and other dementia syndromes including Parkinson’s disease, Huntington’s disease, and vascular dementia (VaD) associated with subcortical white disease with each dementia syndrome presenting with distinctive patterns of performance regarding both correct responses and unique constellations of seen on free recall, cued recall, and recognition test conditions. 5 –9 Similar serial list-learning patterns of performance have recently been documented in patients with MCI. 10 In the aggregate, this research suggests that the analysis of errors and process in conjunction with traditional quantitative test scores are useful in characterizing dementia and MCI phenotypes. 11,12
In addition to serial list-learning tests, recall of prose passages such as the WMS LM 4,12 subtest is another frequently administered verbal memory test used to assess for amnestic-related disorders. Indeed, LM test performance is part of the National Alzheimer’s Coordinating Center Unified Data Set. 13 However, unlike research using verbal serial list-learning tests, an analysis of process and errors obtained from LM immediate (LM-I) and LM delayed (LM-D) free recall has not been conducted. In the current research, an exploratory analysis of LM-I and LM-D free recall correct responses, and immediate and delayed recall errors that are related and unrelated to the stimulus material were scored from a large cohort of community–dwelling participants without stroke or dementia.
The purpose of the current research is 2-fold. First, using exploratory principal component analysis (PCA), we sought to determine what, if any, factor structure might be obtained from the LM subtest when LM-correct responses, LM errors related to stimulus content (ie, LM-related errors), and LM errors unrelated to stimulus content (ie, LM-unrelated errors) were included in the model. Second, we examined the association between different anatomic structures as defined with magnetic resonance imaging (MRI) scans as related to identified LM factors in a sample free of stroke, MCI, or dementia.
Methods
Research Participants
Established in 1948, the Framingham Heart Study (FHS) initiated research designed to identify common characteristics contributing to cardiovascular disease via longitudinal observation of an original cohort of 5209 men and women. The FHS Offspring Cohort (n = 5124) was recruited in 1971 for similar purposes. 14 Biological offspring of the Original FHS Cohort and their spouses were eligible for enrollment. Participants were invited to regular health examinations (eg, detailed medical history, physical examinations, and laboratory tests) approximately every 4 years. 15 From 2005 to 2008, 2037 offspring participants took part in a longitudinal study on brain aging that included administration of a neuropsychological test protocol. We excluded 61 participants with prevalent clinical stroke and 22 participants due to incomplete LM test data. Thus, a total of 1954 participants comprised the study sample for the determination of the LM factor structure. Of the 1954 participants, 1564 had available MRI data. For the analysis of the association of the LM factors and MRI brain volume measurements, we additionally excluded 8 participants with prevalent or unknown dementia status, resulting in a sample size of 1556. The institutional review board at Boston University Medical Center approved the study protocol. Informed consent was obtained from all participants.
Neuropsychological Test Protocol and Administration Procedure
A neuropsychological test protocol was administered to study participants by trained examiners that included the immediate and delayed recall conditions of story A from the Wechsler Logical Memory Test (LM; 4). Test examiners were trained to adhere to the standardized administration of all measures and convened weekly with a licensed clinical supervisor to discuss general scoring criteria and specific case examples. Test sessions were digitally recorded, and participant responses were transcribed verbatim to ensure accurate data collection. A clinical neuropsychologist and postdoctoral psychology fellow supervised examiners and performed monthly quality control (QC) checks by listening to the digital voice recording of the test session to ensure accurate administration and transcription of verbal information. Additionally, QC procedures were applied to check scoring accuracy of correct and error responses. Test batteries were randomly selected for QC checks and were evenly distributed across all examiners.
Classification of LM Errors
The analysis and quantification of errors produced on the FHS neuropsychological protocol were implemented in 2005 to complement traditional quantitative measures for the purpose of increased sensitivity and potential-enhanced detection of subtle impairments that are not captured by standard quantitative scores. Table 1 describes the 8 qualitative error types and subtypes that were coded.
Examples of Logical Memory Errors.

A confabulation is a component of the response provided by the participant that was not an accurate detail from the story as administered. An intrusion is a response comprised of information the participant has taken from a prior conversation or prior test that was administered as part of the overall neuropsychological protocol but was provided as if it were part of the current test. Confabulation and intrusion errors were further subcategorized into errors that were related to the context of the administered story and errors that were unrelated to stimulus context. For LM-D free recall, related and unrelated confabulation and intrusion errors were additionally subdivided to reflect errors repeated from the immediate free recall condition and new errors that were only produced on the delayed free recall trial.
Magnetic Resonance Imaging and Neuroanatomic Regions of Interest
The MRI techniques used in the FHS Offspring Study have been described previously. 16 Briefly, participants were evaluated with a 1.5-T Siemens Magnetom. T2-Weighted double spin-echo coronal sequences were acquired in 4-mm contiguous slices. All images were read centrally blind to any demographic or clinical information including age and parental dementia status. We computed total cerebral brain volume (TCBV), frontal brain volume, and the volume around the temporal horns (temporal horn volume [THV]), which served as a surrogate measure of the hippocampus. All volumetric measures were calculated as a percentage of total cranial volume to correct for differences in head size. 17 The presence of extensive white matter hyperintensity volume (WMHIV) was defined as having WMHIV >1 standard deviation above age-group specific means. 18
Definition of Covariates
Educational achievement was studied as a 3-class variable (<high-school degree, high-school degree, and ≥college degree). Cardiovascular risk was measured using the Framingham Stroke Risk Profile 19 and included hypertension, current smoking (yes/no), diabetes mellitus, history of cardiovascular disease, atrial fibrillation, and left ventricular hypertrophy. Participants were categorized according to the presence or absence of at least 1 apolipoprotein E ∊4 (APOE ∊4) allele (∊2∊4/ ∊3∊4/ ∊4∊4 vs ∊2∊2/∊2∊3/∊3∊3).
Statistical Analysis
An exploratory principal components factor analysis for the LM-I and LM-D tests was performed, using the PROC FACTOR procedure (SAS statistical software, version 9.2). 20 The following 8 variables were entered into the factor analysis: LM-I quantitative score, LM-I number of related confabulations, LM-I number of unrelated confabulations, LM-D quantitative score, LM-D number of new related confabulations, LM-D number of new unrelated confabulations, LM-D number of repeated related confabulations, and LM-D number of repeated unrelated confabulations. Intrusion errors were not included because of the low frequency of occurrence (<1.0%). A total of 3 factors were retained based on the inspection of scree plots. The factors were rotated using the VARIMAX procedure, and factor scores for each of the 3 factors were calculated for each participant.
Brain measurements for THV and WMHIV were log transformed to correct for skewness. All brain volume measurements were standardized to a mean of zero and a standard deviation of 1 to facilitate comparisons between the results. Linear regression models were constructed to examine the association between a standard deviation increment of each brain volume measurement and each of the 3 PCA-derived factors. The β coefficient from this model represents the change in outcome per standard deviation increment of brain volume measurement. Three multivariable models were constructed. Model 1 was adjusted for age at MRI, sex, education group, and time between neuropsychology and MRI. Model 2 was adjusted for the model 1 covariates plus current smoking status, diabetes, hypertension, and history of cardiovascular disease, atrial fibrillation, and left ventricular hypertrophy. Model 3 was adjusted for all of the covariates in models 1 and 2 plus APOE ∊4. A P value of <.05 was considered statistically significant.
Results
Study sample characteristics of the 1954 participants used to derive the LM factors and of the subset of 1556 participants used in the analysis of the LM factors and MRI measures are presented in Table 2. Principal components analysis yielded a 3-factor solution, accounting for 53.7% of the variance (Table 3), using the 8 LM variables. As seen in Table 3, the traditional quantitative scores loaded separately from the variables measuring LM errors. Factor 1 was comprised of correct quantitative LM-I and LM-D free recall raw responses (LM-correct responses factor; 21.1% of variance). Factor 2 was comprised of LM-I and LM-D unrelated errors (LM-unrelated errors factor; 16.5% of variance) and consisted of LM-I free recall unrelated confabulations and LM-D free recall repeated unrelated confabulations. Finally, factor 3 consisted of the LM-related errors (LM-related errors factor; 16.1% of variance) and was comprised of LM-I free recall-related confabulations and LM-D repeated related confabulations. Age-adjusted Spearman’s correlation coefficients were calculated for each of the derived factors and each brain MRI measure and no statistically significant correlations were observed (data shown in online supplemental Table 1).
Study Sample Characteristics for Framingham Heart Study Offspring Participants Who Completed the Logical Memories Test During Neuropsychological Examination.

Abbreviations: APOE, apolipoprotein E; CVD, cardiovascular disease; MRI, magnetic resonance imaging; NP, neuropsychology; SD, standard deviation.
Rotated Factor Pattern for Logical Memories Immediate (LMI) and Delayed (LMD) Test Measures.a
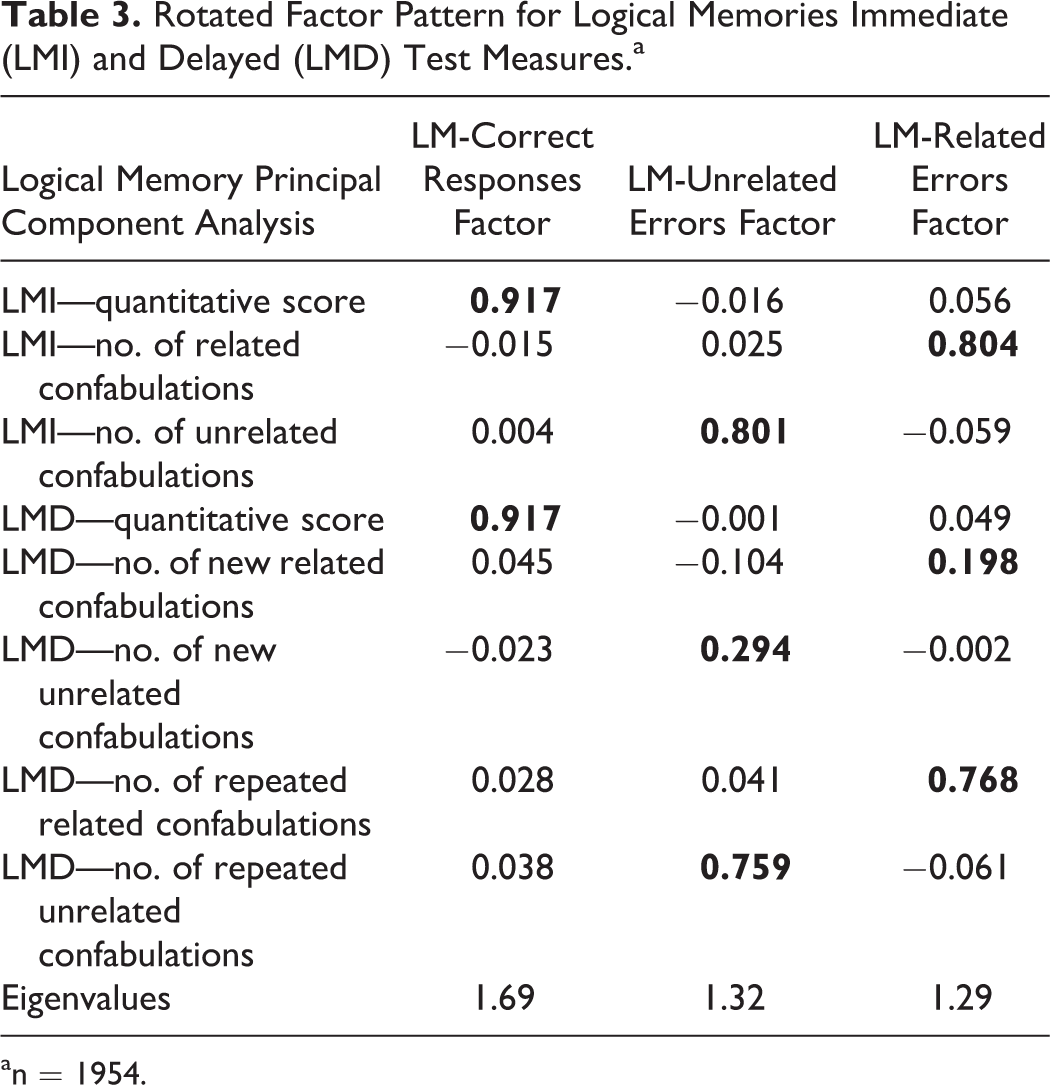
an = 1954.
Table 4 presents the linear regression results examining associations between each MRI brain measure and the 3 LM factors derived from the principal components analysis. No association was found for TCBV or frontal lobe volume with any of the LM factors. However, there was a statistically significant association between increasing THV and the LM-correct responses factor (β = −.063, P value = .009) in model 1. The β coefficient was attenuated after further adjustment for vascular risk factors in model 2 (β = −.056, P value = .2) and after adjustment for vascular risk factors and APOE ∊4 in model 3 (β = −.053, P value = .03) but remained statistically significant. The THV was not associated with either the unrelated errors or the related errors factors.
Linear Regression Results for the Association Between Standard Deviation Incrementa of MRI Brain Measures and Logical Memory Test Derived Factors.b
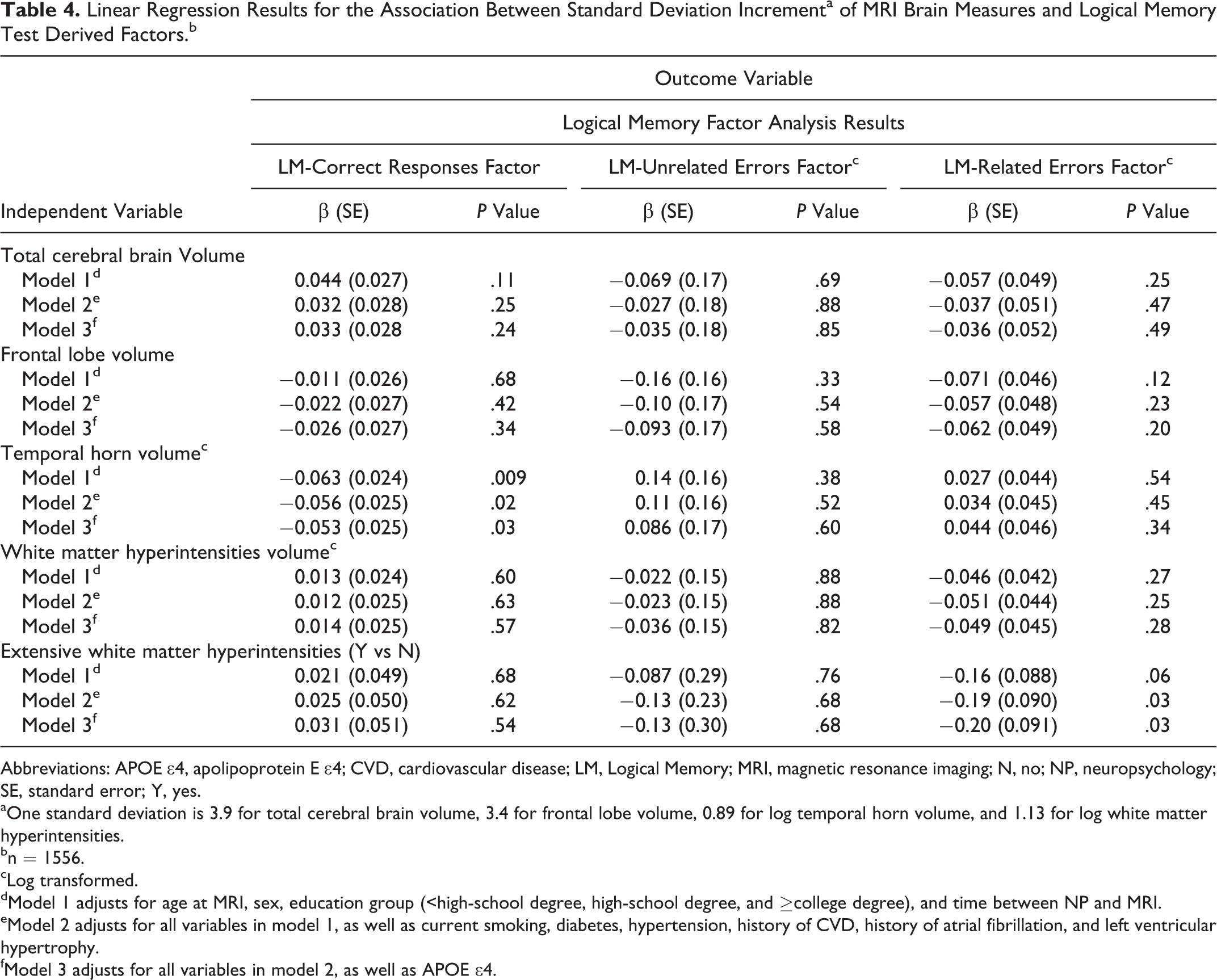
Abbreviations: APOE ∊4, apolipoprotein E ∊4; CVD, cardiovascular disease; LM, Logical Memory; MRI, magnetic resonance imaging; N, no; NP, neuropsychology; SE, standard error; Y, yes.
aOne standard deviation is 3.9 for total cerebral brain volume, 3.4 for frontal lobe volume, 0.89 for log temporal horn volume, and 1.13 for log white matter hyperintensities.
bn = 1556.
cLog transformed.
dModel 1 adjusts for age at MRI, sex, education group (<high-school degree, high-school degree, and ≥college degree), and time between NP and MRI.
eModel 2 adjusts for all variables in model 1, as well as current smoking, diabetes, hypertension, history of CVD, history of atrial fibrillation, and left ventricular hypertrophy.
fModel 3 adjusts for all variables in model 2, as well as APOE ∊4.
The presence of extensive WMHIV was associated with a lower score on the related errors factor for models 2 and 3. Extensive WMHIV was not associated with either the correct responses factor or the unrelated errors factor for any of the models.
We additionally examined the association between the MRI brain measures and the individual LM-I and LM-D quantitative scores (online supplemental Table 2). Statistically significant associations were observed only for THV. The THV was associated with a lower LM-I quantitative score in model 1 (β = −.21, P value = .04) but the association was no longer statistically significant in models 2 and 3. The THV was inversely associated with the LM-D quantitative score for model 1 (β = −.31, P value = .003), model 2 (β = −.27, P value = .01), and model 3 (β = −.25, P value = .02).
Discussion
The WMS LM subtest is an economical and popular neuropsychological test used to assess for alterations in memory associated with MCI and dementia. In our exploratory PCA, we observed that immediate and delayed free recall, LM-unrelated errors, and LM-related errors do, indeed, load on separate factors. The variables loading on the LM-correct responses factor were the traditional quantitative raw scores for immediate and delayed free recall. Thus, the LM-correct response factor appears to reflect the capacity for encoding and retaining novel information. The LM-related errors factor was comprised of related immediate free recall errors and the reoccurrence of these same and new errors after a delay. Similar behavior was noted for the LM-unrelated errors factor for immediate free recall-related errors and the reoccurrence of these same and additional errors after a delay. To the best of our knowledge, this is the first study to assess error performance from the LM subtest as related to MRI-defined regions of interest in a community-based sample.
It is possible that the cognitive constructs underlying the LM-unrelated errors and the LM-related errors factors might best be viewed as a continuum or interaction between degraded lexical-semantic content on the one hand; as well as impaired access to this same lexical-semantic knowledge on the other hand. Some support for this notion has been provided in prior research 5 where patients with AD and VaD associated with MRI-defined white matter hyperintensities were compared using a 9-word serial list-learning test. Patients with AD produced more initial intrusion errors and tended to perseverate and continue to produce these same errors on subsequent test trials. 5 Such behavior might suggest dysexecutive impairment. This research 5 also found that these same serial list-learning errors tended to be rather prototypic errors (eg, “apple”) as related to its respective semantic category; and, that the volume of these errors produced by patients with AD was negatively correlated with the semantic integrity of patient’s “animal” fluency output. In the aggregate, the initial emergence of prototypic free recall errors coupled with the fact that these errors are subjected to perseveration suggests the presence of a complex interaction involving derailed and inefficient access to lexical/semantic information.
An objective of the current research was to test the hypothesis that unique MRI regions of interest would be differentially associated with each of the factors obtained from the LM exploratory principal components analysis. In the current research, we confirmed previous findings that AD-related brain structure was associated with the LM-correct responses factor, that is, the amount of information encoded and retained. The new findings obtained in the current research show that the presence of extensive WMHIV was associated with higher scores on the LM-related errors factor, that is, increased error production.
Given the exploratory nature of the current research, it is unclear whether the association between distinct anatomic regions of the brain and LM-correct responses versus LM-related responses represents a single or combined underlying neurocognitive construct. This determination must await further research, perhaps with a wider array of neuropsychological measures and other imaging technology, as well as longitudinal follow-up for patterns of incident change. Nonetheless, recent research regarding MCI and Alzheimer’s/VaD has found that the errors made on memory, language, and executive tests are highly distinctive and related to different MCI and dementia phenotypes. 10,11,21 Moreover, there is research suggesting that neuropsychological impairment can provide early indications of emerging neurocognitive illness. 22 A longitudinal analysis of MRI regions of interest and the pattern of performance on the LM subtest might provide more insight regarding these questions.
The association between LM-correct responses, LM errors, and specific brain regions as measured with MRI has implications for the characterization and diagnosis of preclinical impairment thought to be associated with AD 23 and MCI also as related to AD. 24 The MCI is believed to be a prodrome leading to the eventual emergence of a dementia, while criteria for preclinical impairment 23 are uncertain and recommended only for research purposes. It has been suggested that as dementia such as AD emerges, MRI biomarkers precede cognitive alterations. 25 However, in 1 study, 26 impaired performance on delay free recall obtained from the Rey Auditory Verbal Learning Test preceded both MRI and CSF biomarkers in assessing measures of insidious progression to dementia. In another recent study 22 involving patients with MCI and dementia evaluated from a memory clinic, neuropsychological assessment was found to be more sensitive than MRI measures of brain atrophy for detecting disease progression. Also, errors made on tests of naturalistic action have also recently been associated with specific MRI-defined regions of interest. 27,28 These previous findings 21,22,27,28 along with data from the current research suggest that detection of preclinical MCI/dementia may be enhanced by extending assessment of cognition to include error responses in combination with selected MRI biomarkers and cardiovascular risk factors.
One of the strengths of the current research is that to our knowledge this is the first community-based study that has analyzed both traditional and error measures from a paragraph recall test in association with MRI parameters. Possible limitations in the current research include the fact that participants were Caucasian, highly educated, and relatively healthy compared to the general population, suggesting some limits regarding external validity. Additionally, because these analyses were exploratory, we did not adjust for multiple comparisons in the analysis so we cannot exclude the possibility that our findings may be due to the presence of false positives. Further studies and longitudinal follow-up are needed to confirm these initial findings. Another possible weakness is that the MRI measures used here are quite minimal in comparison to what has been used in studies of functional and structural neuroimaging in relation to human memory function. A more thorough and informative analysis may be possible through the application of comprehensive neuroimaging methods such as voxel-based morphometry or multivoxel pattern analysis. Finally, we also acknowledge the small number or low occurrence of unrelated and related LM errors. However, in response to this critique, the low base rate for unrelated and related errors may have significant practical clinical utility. For example, it is possible that excessive numbers of LM errors, even when the traditional quantitative immediate and delayed recall scores of correct responses are intact, might indicate an emerging preclinical dementia syndrome, perhaps with vascular features. Future research will be needed to see whether baseline LM errors are clinically meaningful as an initial marker of cognitive decline and future risk of MCI or clinical dementia.
In sum, the analysis of process and errors obtained on the LM tests in combination with traditional scoring methods provide additional information over the correct total scores alone. Further research is warranted to determine the usefulness of error measures in relation to the occurrence of MCI and dementia.
Footnotes
Acknowledgments
The authors thank the extraordinary participants and families of the Framingham Heart Study who made this work possible. We also acknowledge the great work of all the research assistants and study staff.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Framingham Heart Study’s National Heart, Lung, and Blood Institute contract (N01-HC-25195), by grants (R01-AG16495, R01-AG08122, R01-AG033040) from the National Institute on Aging, and by grant (R01-NS17950) from the National Institute of Neurological Disorders and Stroke.
