Abstract
Background/Rationale:
Adherence to cholinesterase inhibitor (ChEI) is associated with treatment effectiveness in the treatment of Alzheimer’s disease (AD). We investigated the clinical adherence to donepezil in Taiwan.
Methods:
This was a retrospective study. Patients treated with donepezil were recruited from Kaohsiung Medical University Hospital and Kaohsiung Municipal Ta-Tung Hospital from February 2004 to April 2013. We analyzed their treatment duration in months.
Results:
A total of 273 patients were included in our analysis. Sixty-seven patients withdrew from donepezil treatment with mean treatment duration of 28.0 ± 25.9 months. Better initial scores on the Mini-Mental Status Examination (P = .007), Cognitive Abilities Screening Instrument (P = .003), and the Clinical Dementia Rating Scale (CDR) Sum of Boxes (P = .011) were positively associated with clinical adherence. The clinical adherent rate was higher in the CDR-0.5 group than in the CDR-2.0 group with significant difference.
Conclusion:
Although there are some limitations in our study, these findings indicate that early intervention with ChEI in patients with AD should be emphasized and may lead to a better clinical adherence.
Introduction
Alzheimer’s disease (AD), a progressive neurodegenerative disease, is the most common form of dementia for which a cure remains to be found. The clinical situation worsens as the disease progresses and eventually leading to death. The AD prevalence in Taiwan is 5.7% and estimated to double by 2030. 1 Donepezil is a cholinesterase inhibitor (ChEI) agent that is used most widely to delay the progression of cognitive dysfunction and improve other symptoms in patients with AD2-5; in addition, donepezil is safe for use during long-term AD therapy. 6 Sustained use of ChEI for 3 or more months may delay the progressive cognitive, functional, and behavioral degeneration caused by AD. 7,8 Clinical adherence is critical to benefiting from ChEI. In a double-blind study, the cognition of donepezil-treated patients decreased to a level similar to that of placebo-treated patients within 6 weeks after they discontinued medication therapy. 9
Information pertaining to the treatment duration may provide insight into the therapeutic effectiveness, safety, and long-term tolerability of donepezil when used in general care settings. However, withdrawal from the medication program is frequently a barrier to effective therapy. 10,11 Determining the reasons for withdrawal may provide clinicians with a clearer understanding of optimal donepezil therapy. Poor drug tolerability is likely to decrease medication adherence. 12 In a previous study, the adverse effects of the drug were consistent with its cholinergic actions, and withdrawal from the medication treatment program occurred primarily within the first 12 weeks of treatment. 13 Several studies have reported that the most common adverse effects include nausea, vomiting, diarrhea, muscle cramping, dizziness, fatigue, and anorexia; these adverse affects are dose dependent. 13 Moreover, prescribers can influence individual treatment 14 because the published medical guidelines or government policies followed by prescribers vary according to country; and their prescribing practices influence medication adherence. 14
Reimbursement for donepezil prescription is based on the policy established by the National Health Insurance (NHI) of Taiwan. Donepezil is not approved for patients with AD exhibiting clinical decline after treatment. Clinical decline is defined as progression of the clinical dementia rating (CDR) to the next stage or when the Mini-Mental Status Examination (MMSE) score is lower than the initial score by more than 2 points. Because of the progressive course of AD, patients excluded from the NHI may not be reimbursed, thus affecting the prescription of donepezil. Changing medication can be approved if side effects appear within 3 months after donepezil prescription and can cause nonadherence to donepezil.
The rate of clinical decline seems to vary during the course of AD and may influence the therapeutic effectiveness at various AD stages. Seltzer et al 15 observed little or no decline in the Alzheimer's Disease Assessment Scale—Cognitive Subscale or MMSE scores of the patients with early-stage AD in a placebo group, suggesting that the therapeutic response of patients with early-stage AD may differ from that of patients at advanced stages of AD. Therefore, varied treatment responses during various stages of AD should be investigated to determine the most effective benefit from donepezil and promote clinical adherence. This retrospective study investigated the clinical adherence to donepezil in Taiwan and the causes for nonadherence in patients with various stages of AD.
Methods
Patients
All patients with AD receiving donepezil were recruited from the neurologic department at Kaohsiung Medical University Hospital (KMUH) and Kaohsiung Municipal Ta-Tung Hospital (KMTTH), 2 medical centers in Southern Taiwan, from February 2004 to April 2013. The patients had to fulfill the physical requirements to receive donepezil and adhere to the NHI prescription guidelines. Alzheimer’s disease was diagnosed according to the criteria specified by the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA) 16 and with reference to the comprehensive neuropsychological assessments: MMSE or CDR scale. Patients with mild or moderate AD were selected, and patients with other conditions that possibly contributed to dementia diagnosis, such as cerebrovascular disease and Parkinson’s disease, were excluded.
Evaluation
Evaluation procedures were approved by the institutional review board of the KMUH, and patients or their legal representatives provided written informed consent. Patient information, such as age, sex, and educational attainment, as well as the neuropsychological assessments including the patient’s initial MMSE, 17 Cognitive Abilities Screening Instrument (CASI), 18 CDR scale, 19 and Clinical Dementia Rating Scale Sum of Boxes (CDR-SB) 20 scores were collected. Higher scores on the MMSE (which ranged from 0 to 30) and CASI (0-100) indicated enhanced cognitive function. Clinical Dementia Rating is a categorical scale with only 4 categories: 0, 0.5, 1, 2, and 3, and the CDR-SB is a numerical scale ranging from 0 to 18; on both scales, larger numbers represent worse function. Every 6 months, a series of neuropsychological assessments was conducted by psychologists to evaluate the AD progression and the relative therapeutic effectiveness. During every assessment, the patient caregivers were present.
Donepezil was prescribed according to the NHI guidelines in Taiwan, including the following principles. (1) The clinical diagnosis of AD had to fulfill the criteria established by the NINCDS-ADRDA, the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, or the International Classification of Diseases. (2) The MMSE scores of the patients had to be within the range of 10 to 26 or the patients had to have global CDR scores of 1 or 2. (3) If patients had a decline in MMSE score that exceeded 2 points, or if the CDR score progressed to the next stage compared with the score from the first year, donepezil was not prescribed to the patients. (4) All the tests were administered annually to ensure that patients fulfilled the reimbursement criteria of the NHI.
During July 2013, we conducted a large-scale retrospective survey of patients who discontinued treatment, investigating the reasons for withdrawal during the therapeutic period. Age was defined as the age at first treatment, and data from patients who continually received the treatment during the follow-up period were censored on the date of their final treatment. Therapeutic duration was defined as the number of months from the initial therapy to the final approved donepezil treatment by the NHI. Withdrawal from the treatment program was established when patients with AD ceased to follow-up at the neurology outpatient department or changed to treatment with other drugs. The reasons for withdrawal were ascertained face-to-face or through a telephone interview with a spouse or key family member.
Statistical Analysis
Data analysis was performed using SPSS statistical software (V17.0; SPSS Inc, Chicago, Illinois). All statistical tests were 2-tailed, and an α of .05 was adopted to indicate significance.
Kaplan-Meier survival curves were employed to analyze the clinical adherence to and withdrawal from treatment of donepezil among patients at various stages of AD. The log-rank test was used to test the differences, and Cox proportional hazard models were used to adjust for demographic variables, such as age, sex, and educational attainment, and initial neuropsychometric test results including the MMSE, CASI, and CDR-SB. The CDR was used to analyze the adherence at various stages of AD until the end of treatment.
Results
A total of 273 patients with mild to moderate AD were recruited for the statistical analysis (Table 1). Among them, 67 (24.5%) patients withdrew from donepezil treatment within the therapeutic period. The mean therapeutic duration was 28.0 ± 25.9 months, with a maximum of 128 months and a minimum of 0.5 months. The 12-month and 24-month adherence rates were 90.1% and 84.8%, respectively (Figure 1).
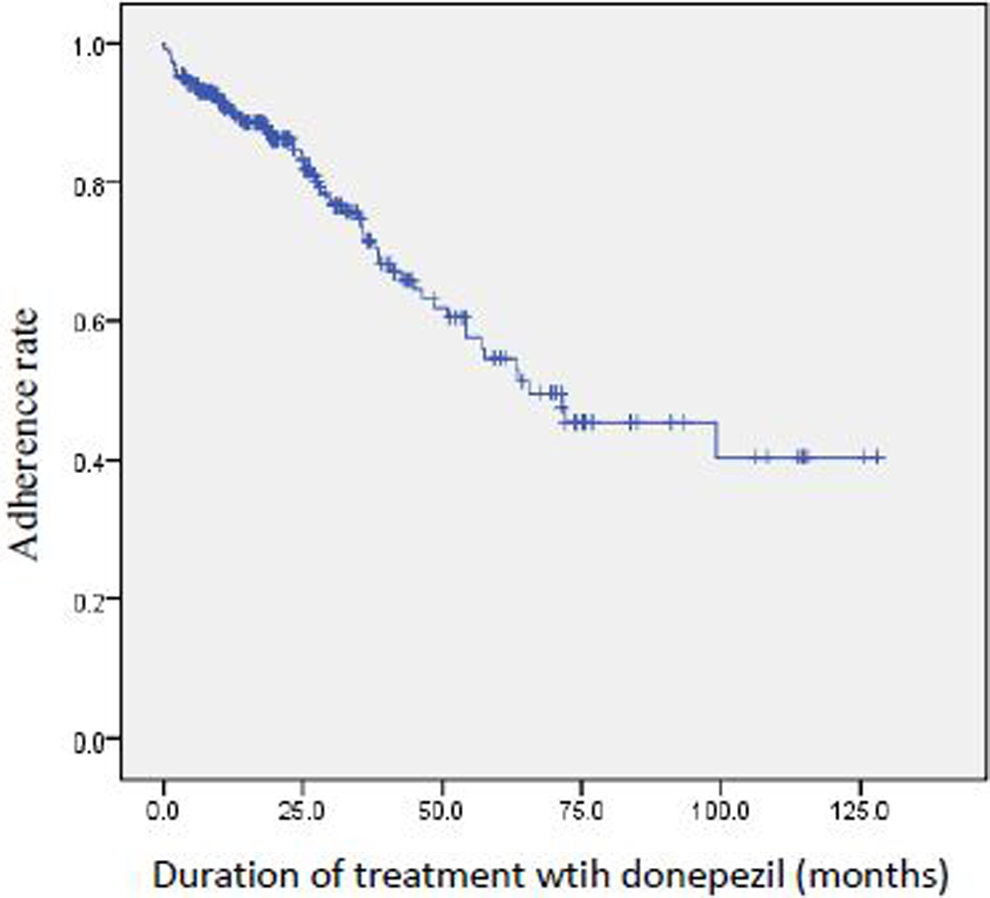
Kaplan-Meier survival estimates for the clinical adherence of taking donepezil in patients with mild to moderate Alzheimer’s disease (AD). The mean therapeutic duration was 28.0 ± 25.9 months with a maximum of 128 months and a minimum of 0.5 months. The 12- and 24-month adherence rates were 90.1% and 84.8%, respectively.
Demographic Characteristics of the Participants With AD.
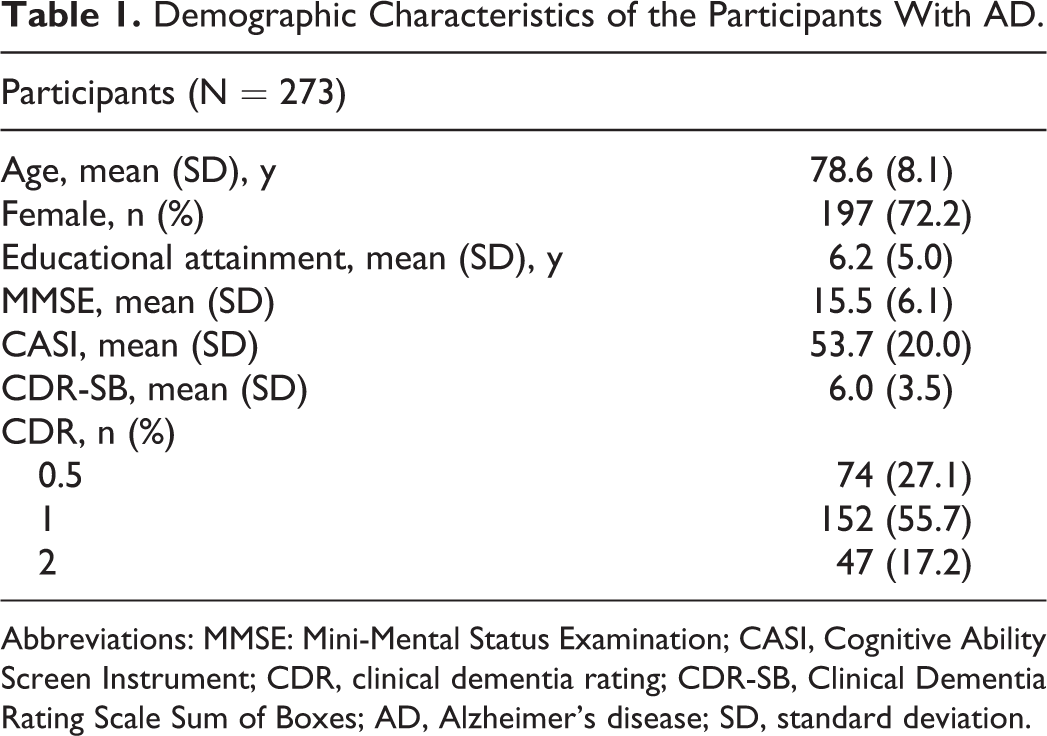
Abbreviations: MMSE: Mini-Mental Status Examination; CASI, Cognitive Ability Screen Instrument; CDR, clinical dementia rating; CDR-SB, Clinical Dementia Rating Scale Sum of Boxes; AD, Alzheimer’s disease; SD, standard deviation.
Among factors influencing clinical adherence, significant associations were observed among the initial scores on the MMSE (P = .007), CASI (P = .003), and CDR-SB (P = .011), but not among age, sex, or education (Table 2). For every 1-unit increase in the initial MMSE score, the risk of withdrawing from donepezil treatment decreased by 6% (hazard ratio [HR] = 0.940, 95% confidence interval [CI]: 0.899-0.983). For every 1-point increase in the initial CASI score, the risk of withdrawing from treatment decreased by 2% (HR = 0.98, 95% CI: 0.968-0.993). Regarding the CDR-SB, every 1-point increase in the initial test score increased the risk of withdrawal by 10.5% (HR = 1.105, 95% CI: 1.023-1.194). Among the 67 patients with AD who withdrew from donepezil treatment, 12 had a global CDR of 0.5, 38 had a CDR of 1.0, and 17 had a CDR of 2.0; the clinical adherence rate was higher in the CDR-0.5 group that had a therapeutic duration of 91.1 ± 9.0 months (Figure 2). Comparing the CDR-0.5 and CDR-2.0 groups suggested that the clinical adherence significantly increased in the group of CDR-0.5 and CDR-2 (P = .017) but did not increase when comparing the CDR-0.5 and CDR-1.0 groups or the CDR-1.0 and CDR-2.0 groups.
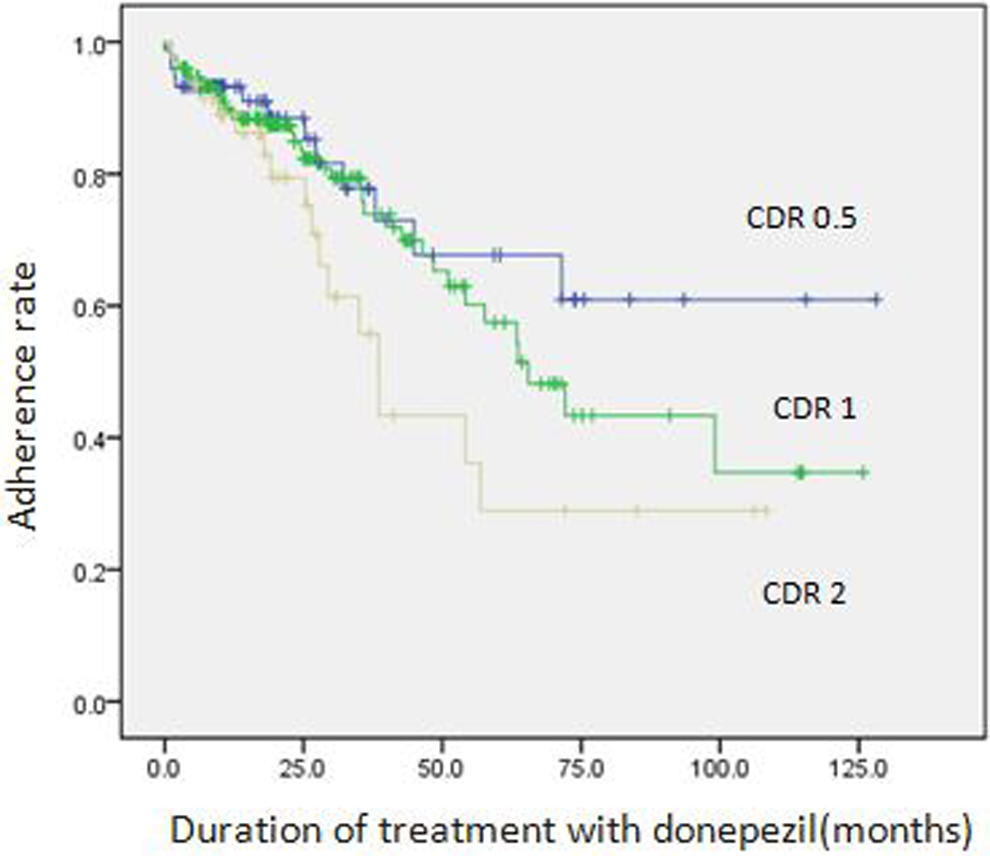
Kaplan-Meier survival estimates of adherence rate in different CDR stages were significantly different in the CDR-0.5 and CDR-2.0 groups (P = .017 with the use of the log-rank test). CDR indicates Clinical Dementia Rating.
Adjusted Effects of Factors Influencing Adherent Rate.
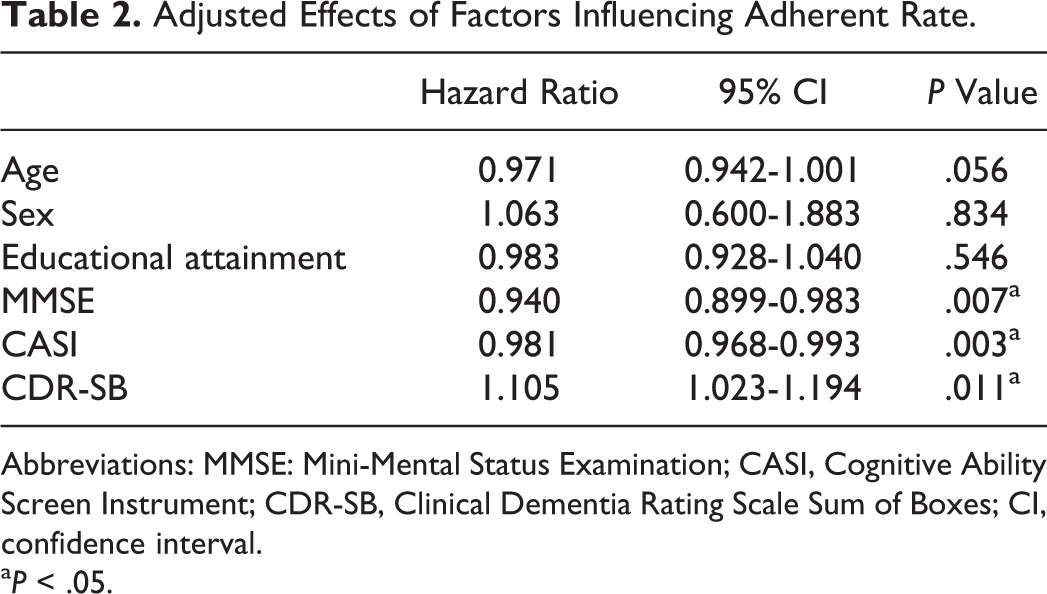
Abbreviations: MMSE: Mini-Mental Status Examination; CASI, Cognitive Ability Screen Instrument; CDR-SB, Clinical Dementia Rating Scale Sum of Boxes; CI, confidence interval.
a P < .05.
Among the reasons leading to withdrawal (Table 3), the NHI policy in Taiwan (43.3%) substantially influenced the patients who discontinued treatment. Clinical decline was the most frequent reason. Nine patients with AD discontinued donepezil because of adverse side effects. Among them, 7 (77.8%) patients had gastrointestinal problems: 5 experienced nausea and vomiting and 2 had diarrhea. Of the remaining 2 patients, 1 patient experienced drowsiness and 1 patient had insomnia.
Reasons of Withdrawal From Donepezil Treatment Stratified With CDR Stages.
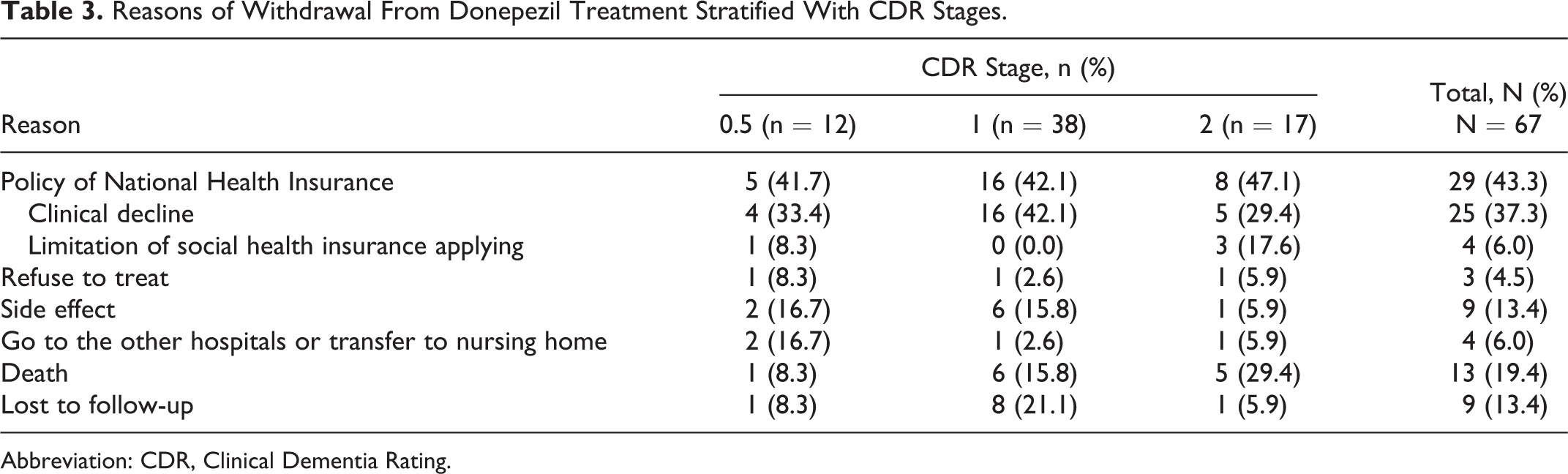
Abbreviation: CDR, Clinical Dementia Rating.
Discussion
The clinical adherence rate of donepezil observed in this study was high (mean therapeutic duration: 28 months) compared to that in another study conducted in Taiwan in which the mean therapeutic duration was 14 months. 21 Only 9.6% of the patients investigated in that study continued ChEI use after 3 years. 21 The discrepancies may have resulted from various study designs. In the previous study, patients and data were obtained from the NHI system. Data from the NHI system are heterogeneous, including all types of hospitals in Taiwan, such as local and area hospitals, and medical centers. Data from both neurology and psychiatric departments were included into the analysis, thus ensuring that conditions were more heterogeneous.
Several studies conducted in other countries have reported that the mean therapeutic duration of donepezil was relatively short, ranging from 17 to 80 weeks, and the risk of withdrawal during the therapeutic period was 10.7% to 67.3%. 22 -26 These heterogeneous results may be attributable to the various study designs and reimbursement regulations pertaining to the treatment.
In the current study, we determined that age (P = .056) and sex (P = .834) were not associated with clinical adherence; however, the results varied widely among countries. Previously, a retrospective study performed in the United States 27 indicated that men and old age (≥86 years) were associated with an improved adherence to oral AD medication. However, Massoud and Gauthier 7 indicated that the risk of discontinuation was significantly higher in patients ≥80 years old, compared with that in patients aged <70 years, an observation that was similar to the results of a study conducted in Taiwan 21 that determined the withdrawal rate in older patients with AD (age ≥ 76 years) was higher. The interaction between age and sex may variably influence the clinical adherence to donepezil among patients in various countries. These topics must be clarified in further studies investigating genetic factors, donepezil tolerance, and clinical adherence among patients belonging to various ethnic groups.
We determined that clinical adherence significantly increased in the CDR-0.5 group than in the CDR-2.0 group; no difference was observed in age, sex, or education between these 2 groups. This observation may indicate that an early donepezil intervention among patients with AD yielded a more favorable therapeutic response. The progression of the AD is not distinct at the early stage. Therefore, taking the medicine at the early stage may slow the rate of deterioration, and the mean therapeutic duration may be longer. Moreover, our results suggested that patients who demonstrated enhanced initial function (higher initial MMSE and CASI and lower initial CDR-SB) tended to exhibit improved clinical adherence. Few studies have investigated determinants of long-term donepezil persistence other than age, sex, and ChEI type. However, the research by Amuah et al 2 indicated that lower MMSE scores may lead to an increased risk of discontinuing ChEI, a result that was similar to ours. Although donepezil is the medication most widely used to delay the progression of cognitive dysfunction in patients with AD, it cannot cure the disease. Based on our finding, patients with AD at the CDR-2 stage had higher mortality rate (29.4%), compared with the patients at the CDR-0.5 and CDR-1 stage. A previous study 28 also indicated that a higher mortality rate was noted in patients with advanced dementia because they experienced more and frequent complications, such as pneumonia, febrile episodes, and eating problems. Thus, donepezil should be taken at the early stage of AD to slow the rate of clinical decline and obtain a favorable therapeutic response.
Brady and Weinman 13 described factors associated with adherence to ChEI, such as drug side effects, comorbid illness, and physical and cognitive limitations. Our analysis indicated that the NHI policy was the most common reason leading to withdrawal from donepezil treatment in Taiwan. Donepezil is not approved for patients with AD exhibiting clinical decline. The second most frequent reason for discontinuing ChEI was adverse effects, primarily gastrointestinal symptoms, a result that was consistent with results obtained by other studies. 24,29 The donepezil tolerance of patients may affect the clinical adherence; decreased adherence is likely to result from poor drug tolerability.
Our study had some limitations. First, the sample size was small: only 273 patients with AD treated with donepezil were recruited in this retrospective study and 67 patients with AD withdrew from the medication treatment. Moreover, all patients with AD were recruited from KMUH and KMTTH. The results could not be extended to other setting in Taiwan, such as regional hospitals, local hospitals, or clinics. An additional large-scaled survey should be conducted in order to resolve this concern. Second, we performed a retrospective cohort study, and basic data were obtained from hospital medical records. The lack of integrity in the medical records may decrease validity, and the therapeutic duration could not be accurately recorded. Furthermore, whether the patients with AD actually took the medication was difficult to confirm based on the hospital medical records. Third, we did not address the comorbidity and behavioral and psychological symptoms of dementia in the study patients. Finally, the clinical compliance of patients with AD, particularly those at the moderate stage, might be related to their caregivers. Basic information about the caregivers was collected in our previous study. 30 Further analysis of the correlation between the characteristics of caregivers and the compliance should be included in further research.
Conclusion
In this study, we determined that the NHI policy regarding ChEI payment, not the drug side effects, was the most common factor leading to withdrawal from donepezil treatment. The results indicated that the clinical adherence rate was significantly higher in the CDR-0.5 group than in the CDR-2.0 group. In addition, enhanced cognitive function before treatment positively influenced clinical adherence. Thus, early intervention with ChEI in patients with AD should be stressed.
Footnotes
Acknowledgments
We acknowledge and appreciate the contribution from the staff of the Department of Neurology, KMUH, Kaohsiung Medical University, that facilitated completing this study.
Authors’ Note
Yun-Ping Chang and Chiu-Hsien Yang contributed equally to this work. This article is based on research that was not funded by outside sources.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
