Abstract
Toxoplasma gondii is an intracellular protozoan infecting 30% to 50% of global human population. Recently, it was suggested that chronic latent neuroinflammation caused by the parasite may be responsible for the development of several neurodegenerative diseases manifesting with the loss of smell. Studies in animals inoculated with the parasite revealed cysts in various regions of the brain, including olfactory bulb. Development of behavioral changes was paralleled by the preferential persistence of cysts in defined anatomic structures of the brain, depending on the host, strain of the parasite, its virulence, and route of inoculation. Olfactory dysfunction reported in Alzheimer’s disease, multiple sclerosis, and schizophrenia was frequently associated with the significantly increased serum anti–T gondii immunoglobulin G antibody levels. Damage of the olfactory system may be also at least in part responsible for the development of depression because T gondii infection worsened mood in such patients, and the olfactory bulbectomized rat serves as a model of depression.
Keywords
The olfactory route for various infectious and/or toxic agents may initiate or exacerbate classical neurodegenerative and autoimmune diseases, especially in persons with genetic predisposition. 1 –9 Several authors showed that many neurologic and neurodegenerative abnormalities are first demonstrable in the olfactory system with loss of smell (anosmia) up to 10 years before the onset of cognitive or motor dysfunction. 7 Neuroinflammation is a common feature of these diseases mostly emerging in the elderly individuals 9 and marked by activated glial cells that secrete numerous pro- and anti-inflammatory cytokines and other neurobiomediators. 10 For example, an exacerbation of Alzheimer’s symptoms lasting for few months following a systemic infection was also capable of elevating serum interleukin (IL) 1β. 11 Recently, it was suggested that chronic T gondii infection may be the key infectious agent responsible for triggering and development of several neurodegenerative diseases associated with an increased generation of several pro- and anti-inflammatory cytokines, including IL-1β. 12 –14
Toxoplasmosis is one of the most frequent infections affecting both healthy and immunocompromised humans with approximately 6 billion people infected. 15,16 During its life cycle, the pathogen interacts with approximately 3000 host genes or proteins, and many of them represent an extensive Toxoplasma gondii host–pathogen interactome enrichment in several psychiatric and neurological diseases. 17 At present, in immunocompetent individuals T gondii infection is believed to be asymptomatic, 18,19 but an increasing body of literature strongly suggests that the parasite is slowly emerging as a global health threat, 16,19 –22 especially in neurodegenerative diseases. Seroprevalence of the parasite measured by specific serum anti–toxo immunoglobulin G (IgG) antibodies varies widely in different countries depending on diagnostic tests used, environmental and socioeconomic conditions, including eating habits, health-related practices, and host susceptibility (Table 1). All these factors considerably hinder attempts to establish clear-cut connections between the highly prevalent infection of T gondii and the development of neurological diseases that are heralded by anosmia. In 1994, the National Health Interview Survey data obtained from 42 000 US households showed a 1.4% prevalence of self-reported olfactory dysfunction exponentially increasing with age. 24 Pregnancy is one of potential risk factors for olfactory disorders, 25 and the relationship between development of these abnormalities and chronic latent T gondii infection may be supported by the fact that at that time hormonal storm markedly affecting cellular and humoral immunity of pregnant woman may also exacerbate latent toxoplasmosis and increase the risk of congenital infection in the fetus. At present, one cannot exclude that isolated/syndromic congenital anosmia 26 is due to perinatal infection with the parasite, especially that structural differences in the brains of individuals with congenital anosmia are extending well beyond olfactory bulb and tract, including the piriform and orbitofrontal cortices. 27,28 Thus, from the first days/weeks of life, these neuroinflammatory processes may play an important role in the progress of pathophysiological abnormalities developing in the brain that finally lead to the olfactory system dysfunction also in neurodegenerative diseases.
Seroprevalence of Toxoplasma Gondii–Specific IgG Antibodies in Pregnant Women in a Selected Number of Countries.a
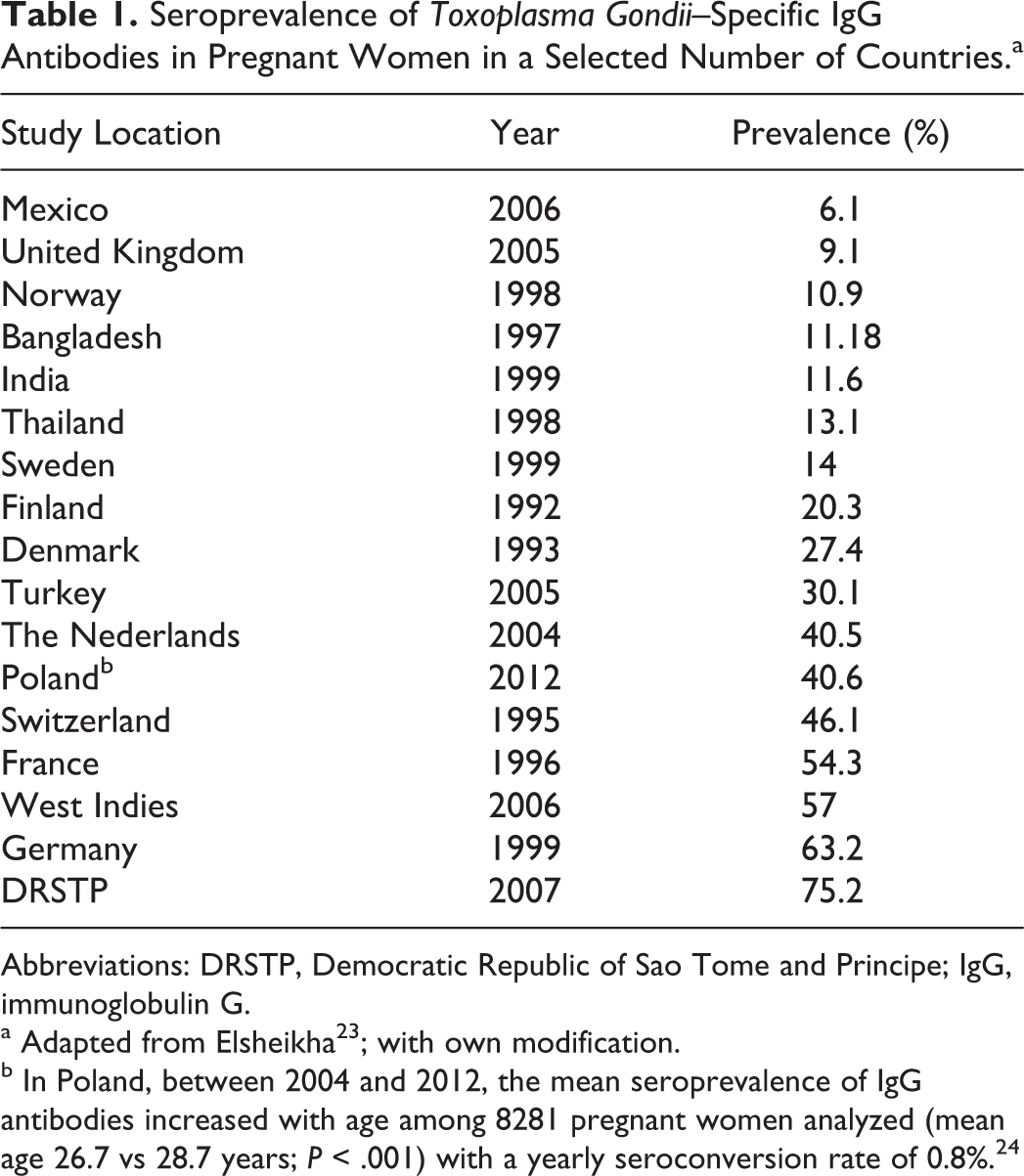
Abbreviations: DRSTP, Democratic Republic of Sao Tome and Principe; IgG, immunoglobulin G.
a Adapted from Elsheikha 23 ; with own modification.
b In Poland, between 2004 and 2012, the mean seroprevalence of IgG antibodies increased with age among 8281 pregnant women analyzed (mean age 26.7 vs 28.7 years; P < .001) with a yearly seroconversion rate of 0.8%. 24
Toxoplasma gondii tachyzoites may invade different types of brain cells including neurons, astrocytes, microglial cells, and Purkinje cells in the cerebellum. Intracellular tachyzoites manipulate signaling pathways and several signs for transduction mechanisms involved in apoptosis, immune cell maturation, and antimicrobial effectors functions. 29 It was demonstrated that in neurons infected by T gondii not only parasitic cysts but also the host cell cytoplasm and some axons were stained positive for the parasite antigens, thus supporting the notion that it may interfere with neuronal function. 20,30 It must be noted that in mice (at day 60 postinoculation with the parasite type II ME49 strain), a calculation of total cyst number per brain volume of various regions of the brain revealed that although cyst number decreased in cortex, thalamus, hippocampus, and striatum, their number slightly increased in olfactory bulb, hypothalamus, cerebellum, and brain stem. 30,31 The development of behavioral changes was paralleled by the preferential persistence of cysts in defined anatomic structures of the brain, 30 –32 depending on the host, strain of the parasite, its virulence, and the route of inoculation. 32 –34 Localization of T gondii cysts in different brain regions and cell types in both embryonal and adult animal brain tissues are presented in Tables 2 to 7. Immunochemistry study revealed that all major parts of neurons including the soma, dendrites, and axons harbored cysts, whereas intraneuronal T gondii antigen was present in the cytoplasm of cyst harboring neurons, and the parasite antigen–positive axons could be followed over long distances. 31 Astrocyte interactions with neuronal cysts were frequently observed. 33 Exposure of lipopolysaccharide (LPS) to neurons in the central nervous system (CNS) induced strong neurodegeneration in vivo and in vitro in substantia nigra and midbrain dopaminergic neurons 49 –51 as well as in hippocampal and cortical neurons. 52,53 Similar neuronal cell death was also reported in the enteric nervous system (ENS). 54,55 It was found that the increased production of nitric oxide (NO) by inducible nitric oxide synthase was a major cause of cell death in LPS-treated cell cultures. 49,52 Toxoplasma gondii infection of different host brain cells was associated with an enhanced generation of various cytokines, including interferon (IFN) γ, tumor necrosis factor (TNF) α, IL-1β, NO, and reactive oxygen/nitrogen species 56,57 as well as with an increased production of many neurotic biomolecules (Table 8). These molecular disturbances could affect the sense of smell also in children with autism, 13,64 Asperger’s syndrome, 65 and migraine patients 66,67 and result in olfactory impairment along with age 68 –70 (Table 9). This reasoning may be supported by the progressive decline in the levels of serum heat shock protein (HSP) 60 and HSP70 with age, whereas HSP70 antibody levels tend to increase (Table 10). On the other hand, it is known that host-derived HSPs play an important role in the development of innate immune defense against T gondii infection. 72 It must be noted that different strains of T gondii induced several constellations of cytokine responses 73 important for the development of various clinical signs and symptoms in the infected host. Virulence of the parasite has been linked with strain-dependent distinct dendritic cell responses and reduced number of activated CD8+ T cells. 74 In animals, oral/peritoneal inoculation with T gondii genotypes I to III resulted in atrophy or hypoplasia of some segments of the gastrointestinal tract and death/hypertrophy of part of myenteric neurons. 75 –77 Similar morphometric abnormalities of the ENS may be responsible for the development of gastrointestinal tract dysfunction reported in patients with autism, inflammatory bowel and/or autoimmune diseases, and in many other gastrointestinal tract disturbances. 55 Glial cells in the ENS appear to be very similar in origin, gross morphology, and ultrastructure to astrocytes of the CNS and bear similar relationships with neuronal cell bodies and processes to peripheral Schwann cells. 78 All these abnormalities in the brain and other organs associated with chronic T gondii infection strongly suggest that similar neuroinflammatory processes are also taking place in the olfactory system, leading to its progressing damage.
Preferential Localization of Toxoplasma Gondii ME49 Strain Cysts in Different Regions of Murine Brain at 2 and 6 Months Postinoculation.a
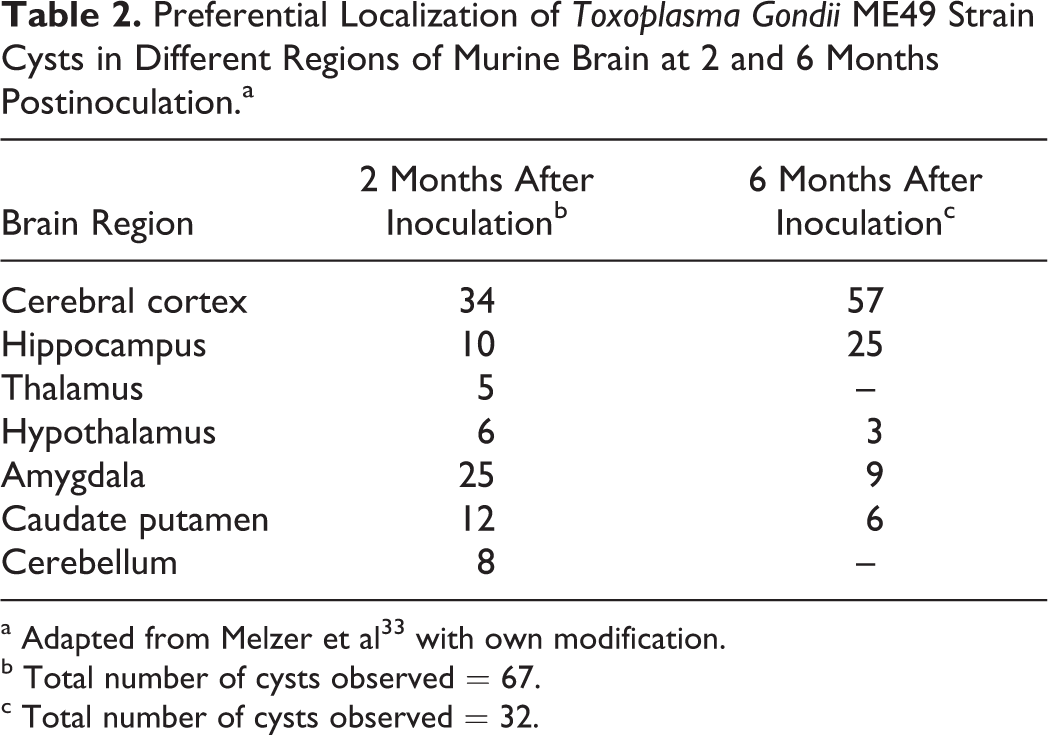
a Adapted from to Melzer et al 33 with own modification.
b Total number of cysts observed = 67.
c Total number of cysts observed = 32.
Distribution of Reactivation Foci in the Central Nervous System After Acute Toxoplasma Gondii Infection in Mice.a
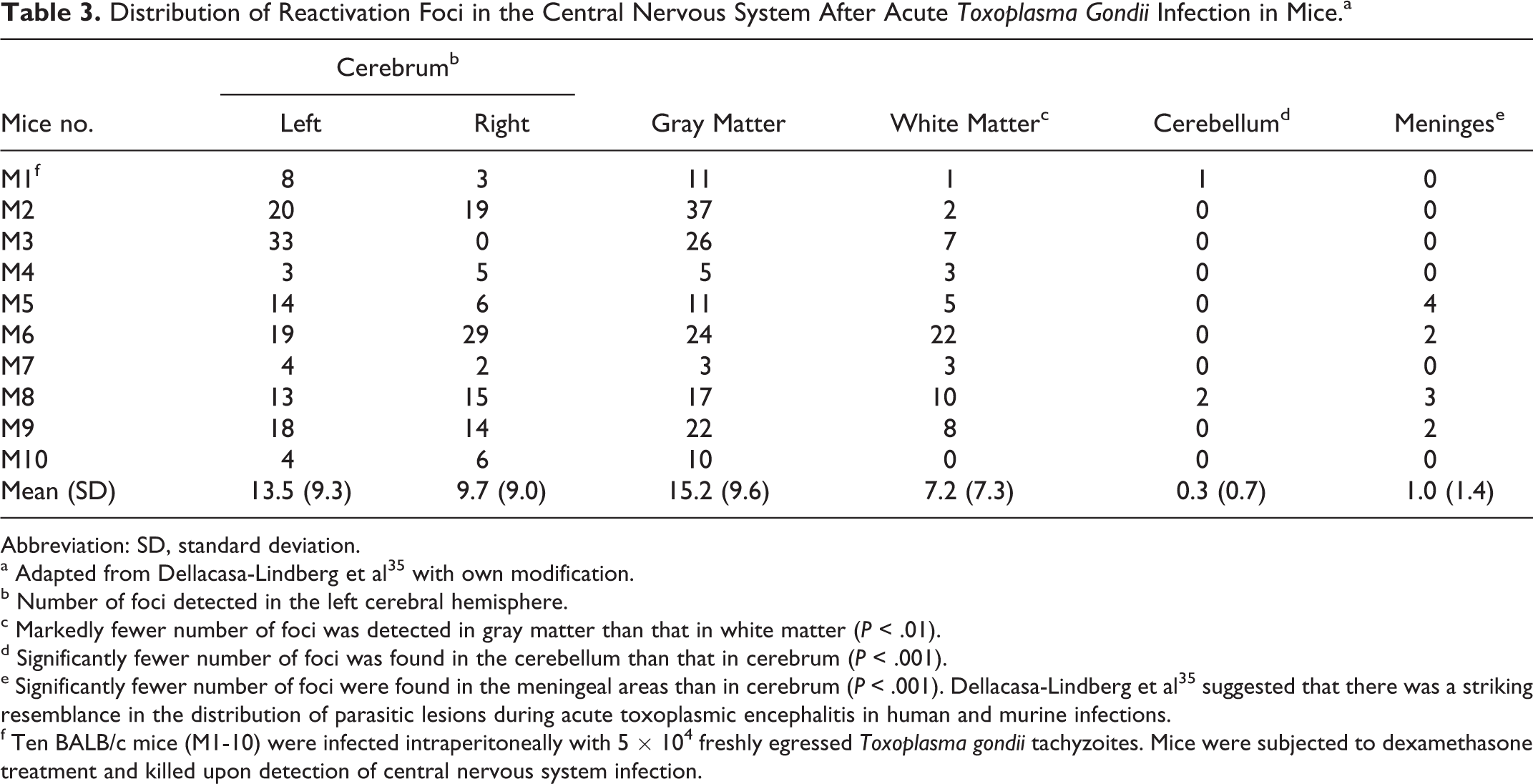
Abbreviation: SD, standard deviation.
a Adapted from Dellacasa-Lindberg et al 35 with own modification.
b Number of foci detected in the left cerebral hemisphere.
c Markedly fewer number of foci was detected in gray matter than that in white matter (P < .01).
d Significantly fewer number of foci was found in the cerebellum than that in cerebrum (P < .001).
e Significantly fewer number of foci were found in the meningeal areas than in cerebrum (P < .001). Dellacasa-Lindberg et al 35 suggested that there was a striking resemblance in the distribution of parasitic lesions during acute toxoplasmic encephalitis in human and murine infections.
f Ten BALB/c mice (M1-10) were infected intraperitoneally with 5 × 104 freshly egressed Toxoplasma gondii tachyzoites. Mice were subjected to dexamethasone treatment and killed upon detection of central nervous system infection.
The Number of Cysts Load in Brain Tissue and Congenital Transmission Rate From the Offspring.a
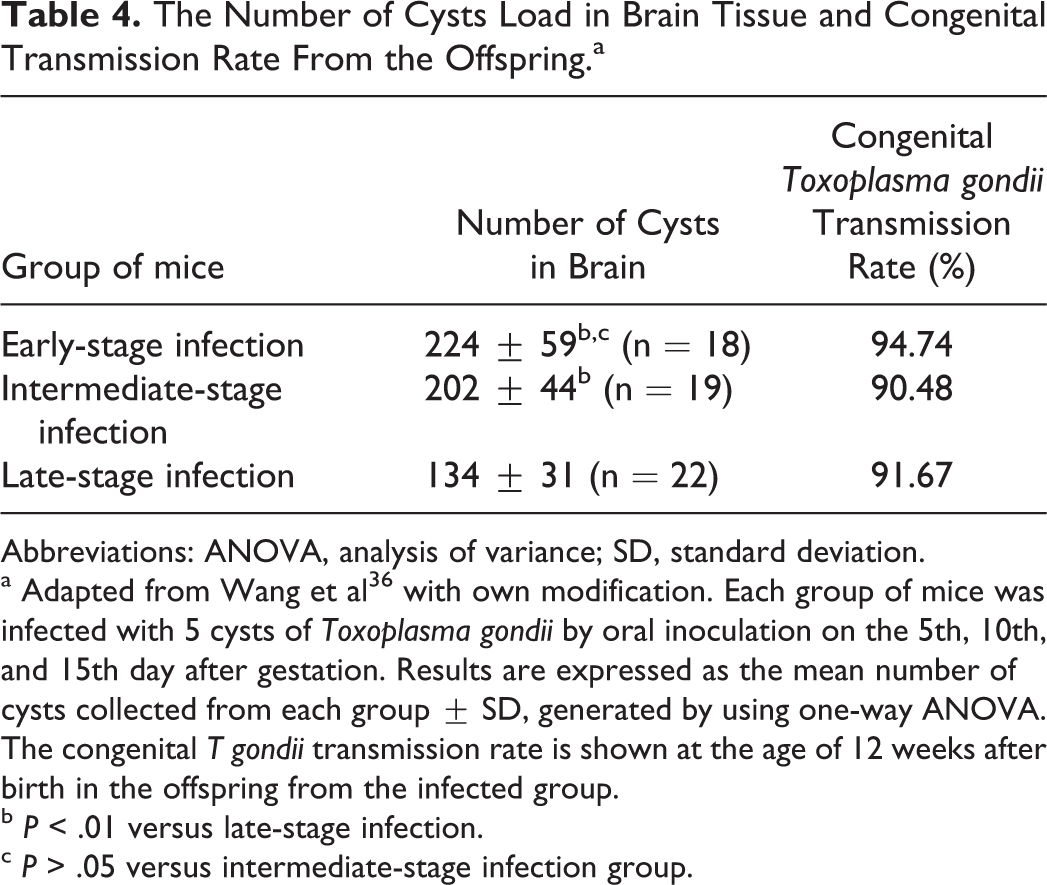
Abbreviations: ANOVA, analysis of variance; SD, standard deviation.
a Adapted from Wang et al 36 with own modification. Each group of mice was infected with 5 cysts of Toxoplasma gondii by oral inoculation on the 5th, 10th, and 15th day after gestation. Results are expressed as the mean number of cysts collected from each group ± SD, generated by using one-way ANOVA. The congenital T gondii transmission rate is shown at the age of 12 weeks after birth in the offspring from the infected group.
b P < .01 versus late-stage infection.
c P > .05 versus intermediate-stage infection group.
Infection Rates of Different Cell Types From Embryonal Rat Cortices (E15) After In Vitro Infection With Toxoplasma Gondii Tachyzoites.a
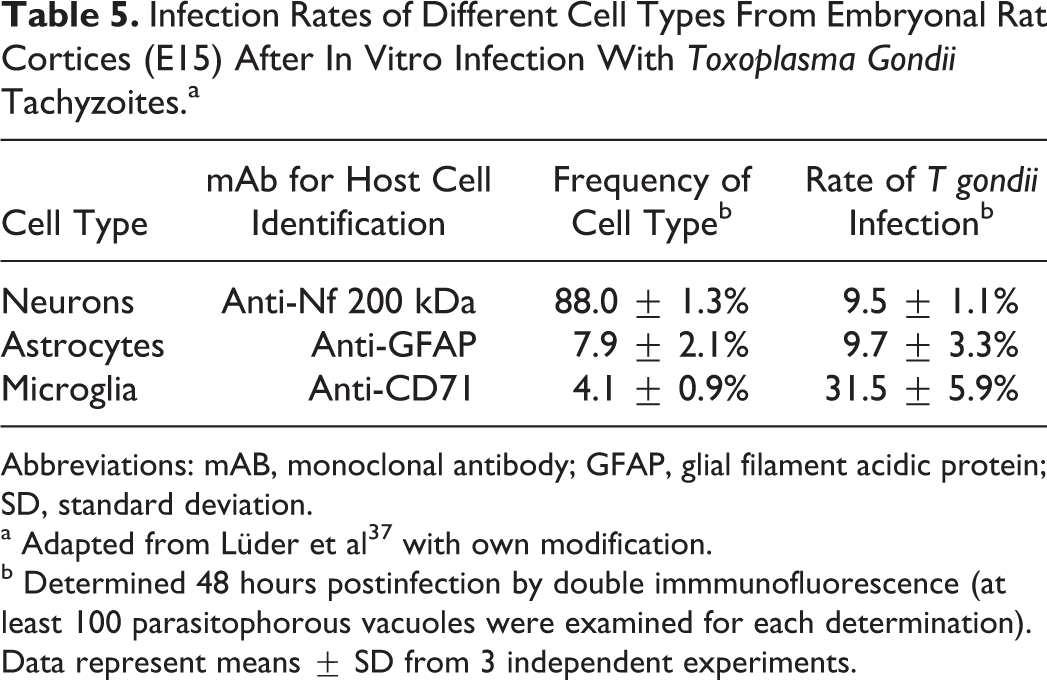
Abbreviations: mAB, monoclonal antibody; GFAP, glial filament acidic protein; SD, standard deviation.
a Adapted from Lüder et al 37 with own modification.
b Determined 48 hours postinfection by double immmunofluorescence (at least 100 parasitophorous vacuoles were examined for each determination). Data represent means ± SD from 3 independent experiments.
Replication and Morphology of Toxoplasma Gondii in Different Cell Types From Embryonal Rat Cortices (E15).a
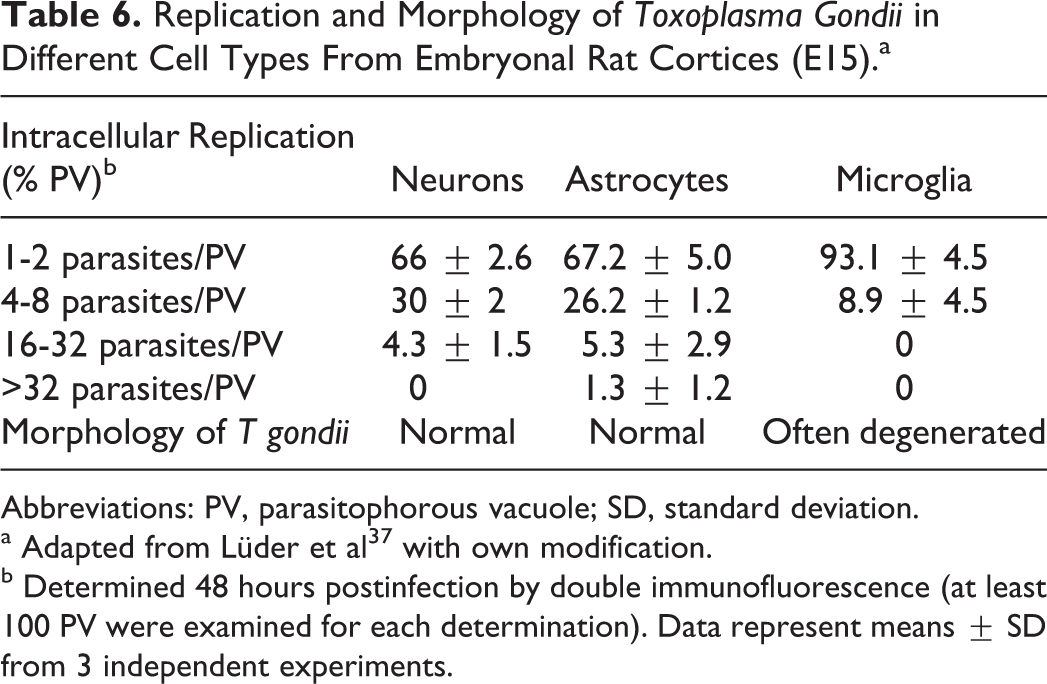
Abbreviations: PV, parasitophorous vacuole; SD, standard deviation.
a Adapted from Lüder et al 37 with own modification.
b Determined 48 hours postinfection by double immunofluorescence (at least 100 PV were examined for each determination). Data represent means ± SD from 3 independent experiments.
Toxoplasma Gondii Infection and Cyst Formation in Primary Cultures of Cells of the Central Nervous System.a
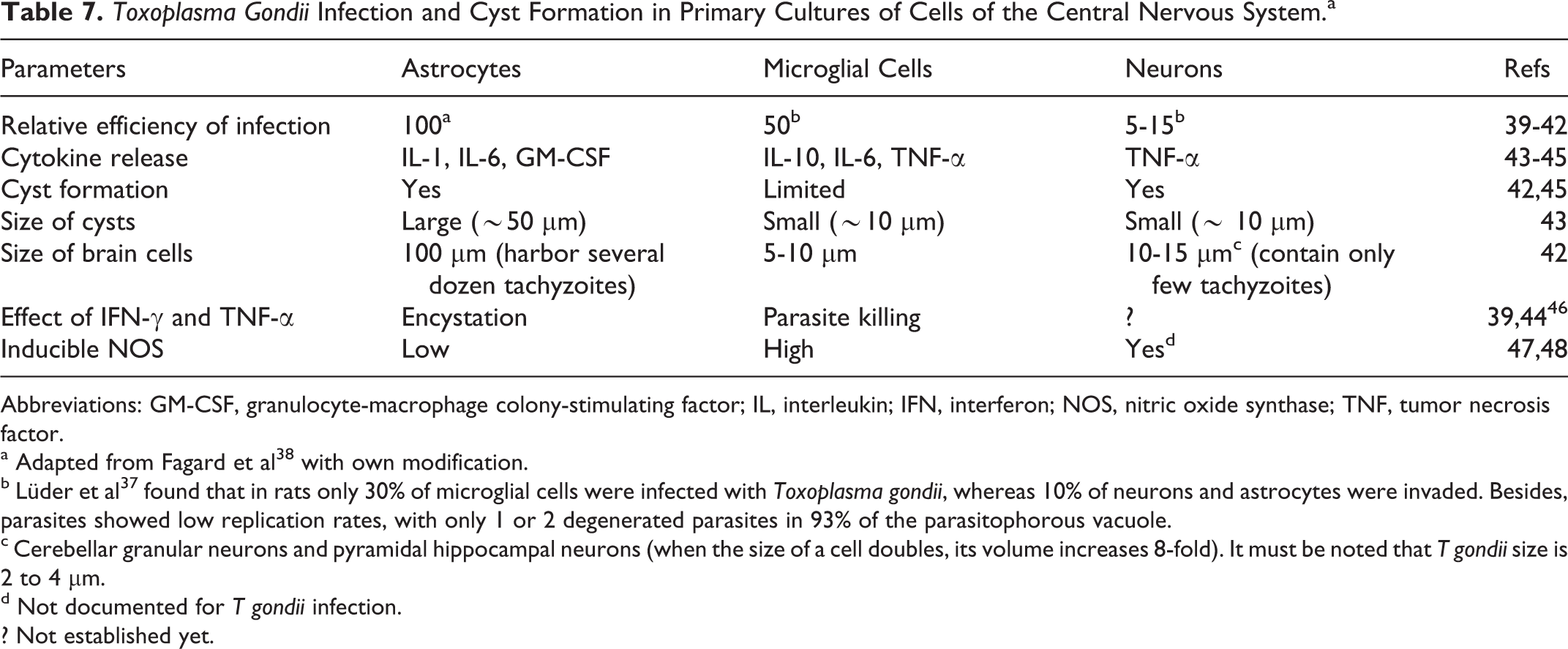
Abbreviations: GM-CSF, granulocyte-macrophage colony-stimulating factor; IL, interleukin; IFN, interferon; NOS, nitric oxide synthase; TNF, tumor necrosis factor.
a Adapted from Fagard et al 38 with own modification.
b Lüder et al 37 found that in rats only 30% of microglial cells were infected with Toxoplasma gondii, whereas 10% of neurons and astrocytes were invaded. Besides, parasites showed low replication rates, with only 1 or 2 degenerated parasites in 93% of the parasitophorous vacuole.
c Cerebellar granular neurons and pyramidal hippocampal neurons (when the size of a cell doubles, its volume increases 8-fold). It must be noted that T gondii size is 2 to 4 μm.
d Not documented for T gondii infection. ? Not established yet.
Possible Consequences on Neurons of Cytokines and Biomolecules Secreted Upon Toxoplasma Gondii Infection.a

Abbreviations: RNI, reactive oxygen intermediates; PAF, platelet-activating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; IL, interleukin; IFN, interferon; NO, nitric oxide; TNF, tumor necrosis factor.
a Adapted from Fagard et al 38 with own modification. It must be noted that Toxoplasma gondii infection caused a significant increase in dopamine metabolism in neural cells, which may lead to psychobehavioral changes in humans infected with toxoplasmosis. 58 Dopamine concentrations were 14% higher in the brain of mice with chronic infections than in controls. 59 In addition, induction of indoleamine 2,3-dioxygnase expression and decreased levels of tryptophan and increased formation of kynurenine were found in the brain, lungs, and serum of mice infected with the parasite. 60 Moreover, dopamine stimulated tachyzoite proliferation in human fibroblast and primary neonatal rat astrocyte cell cultures, 61 thus further enhancing the harmful effects of the parasite on the brain function. In addition, chronic latent T gondii infection is associated with overproduction of various cytokines, and it was postulated that cytokines may induce changes in mood and behavior, leading to depressive illness in man. 62,63
Percentage of Toxoplasma Gondii–Positive Individuals Among 214 Nonpsychiatrically Affected Controls Depending on Age Analyzed During a Large Epidemiologic Study of 869 Psychiatric Patients.a
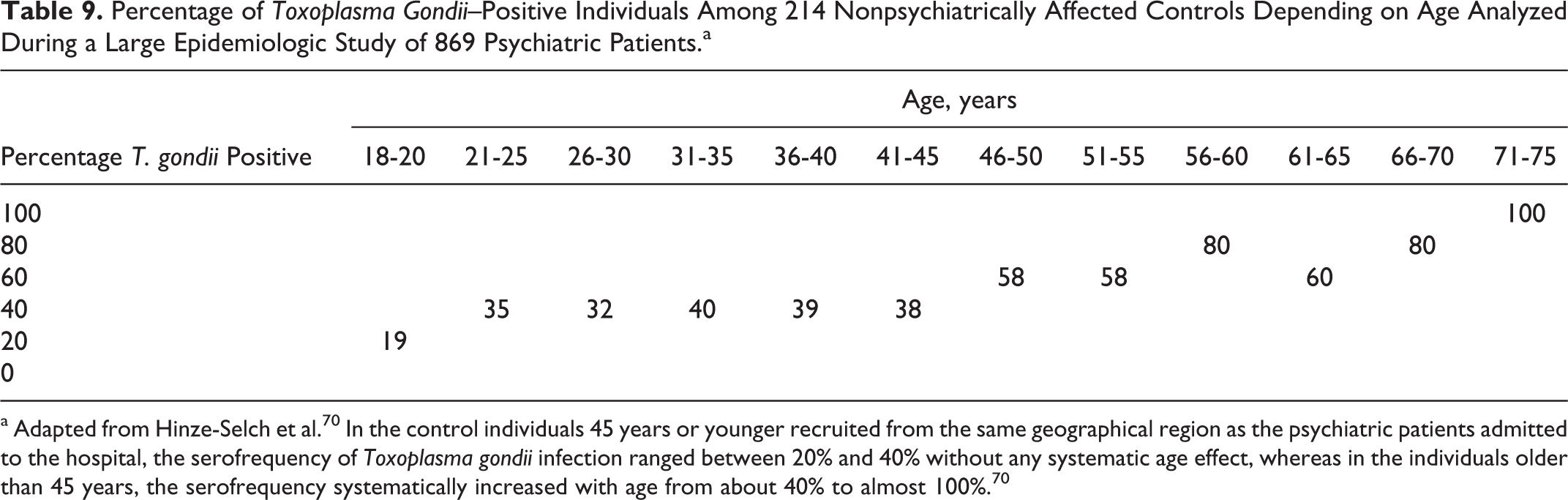
a Adapted from Hinze-Selch et al. 70 In the control individuals 45 years or younger recruited from the same geographical region as the psychiatric patients admitted to the hospital, the serofrequency of Toxoplasma gondii infection ranged between 20% and 40% without any systematic age effect, whereas in the individuals older than 45 years, the serofrequency systematically increased with age from about 40% to almost 100%. 70
Changes in Serum Heat Shock Protein (HSP) and anti-HSP Antibody Levels in Aging.a
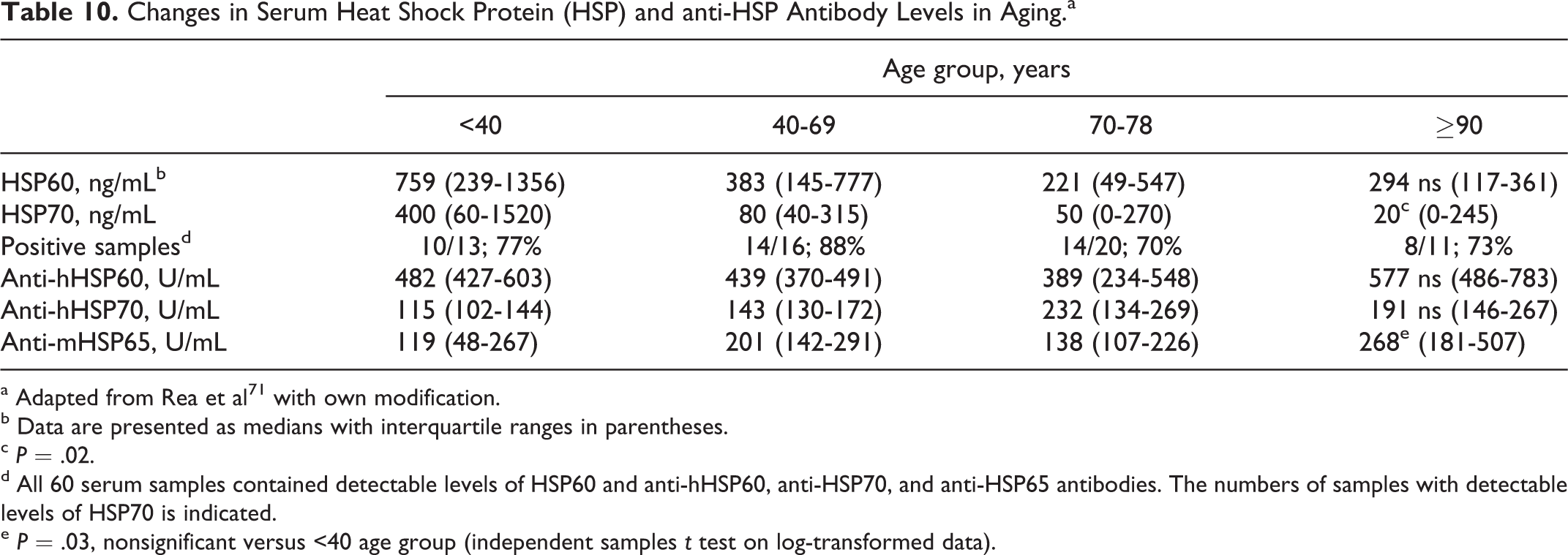
a Adapted from Rea et al 71 with own modification.
b Data are presented as medians with interquartile ranges in parentheses.
c P = .02.
d All 60 serum samples contained detectable levels of HSP60 and anti-hHSP60, anti-HSP70, and anti-HSP65 antibodies. The numbers of samples with detectable levels of HSP70 is indicated.
e P = .03, nonsignificant versus <40 age group (independent samples t test on log-transformed data).
Xiao et al 79 showed that in male mice, infection with the parasite led mainly to modulation of genes associated with olfactory function, such as downregulation of the number of olfactory receptors and dopamine receptor D4. However, general olfactory tests and sensorimotor gating were normal in both male and female infection. 79,80 The discrepancy between the findings in rodents and impaired sense of smell reported in the patients with Alzheimer’s disease, as well as in the individuals with various autoimmune diseases having chronic T gondii infection, may be at least in part explained by the markedly greater morphometric parameters of rhinencephalon in animals (lobus olfactorius) than in humans (bulbus olfactorius), 81 which must be clearly associated with a considerably smaller extent of the olfactory tissue subjected to neuroinflammatory destruction. 82 It must be emphasized that low olfactory bulb volumes have been found in patients with schizophrenia (left and right bulb) and their first-degree relatives (right bulb) as compared with healthy individuals (Table 11). 83 In 1 study, the significant atrophy was also reported in 43.9% of 150 patients with systemic lupus erythematous (SLE), with progression of reduction in right and left hippocampal volumes related to disease duration (P < .001). 84 Moreover, patients with neuropsychiatric SLE had amygdala damage. 85 In patients with Parkinson’s disease, olfactory loss was considered as a marked early symptom that correlated with the progression of the disease, 86 and parkinsonian symptoms have been observed as an initial manifestation in a Japanese patient with acquired immunodeficiency syndrome and T gondii infection. 87 Olfactory dysfunction has also been reported in HIV-infected and AIDS individuals, 88,89 in patients with Alzheimer’s disease, 90 in patients with Down’s syndrome, 91 in patients with multiple sclerosis, 92 in patients with SLE, 93 in patients with schizophrenia 94 and their relatives, 95 and during several pregnancies, 96 that is, the clinical entities with significantly increased serum anti–T gondii IgG antibody levels compared with healthy controls. 23,97 –104 Furthermore, the above-mentioned disturbed brain regions were consistently more infected than other sites in animals with toxoplasmosis. 31 –33
Olfactory Bulb Volumes in Patients With Schizophrenia, First-Degree Relatives, and Healthy Controls.a
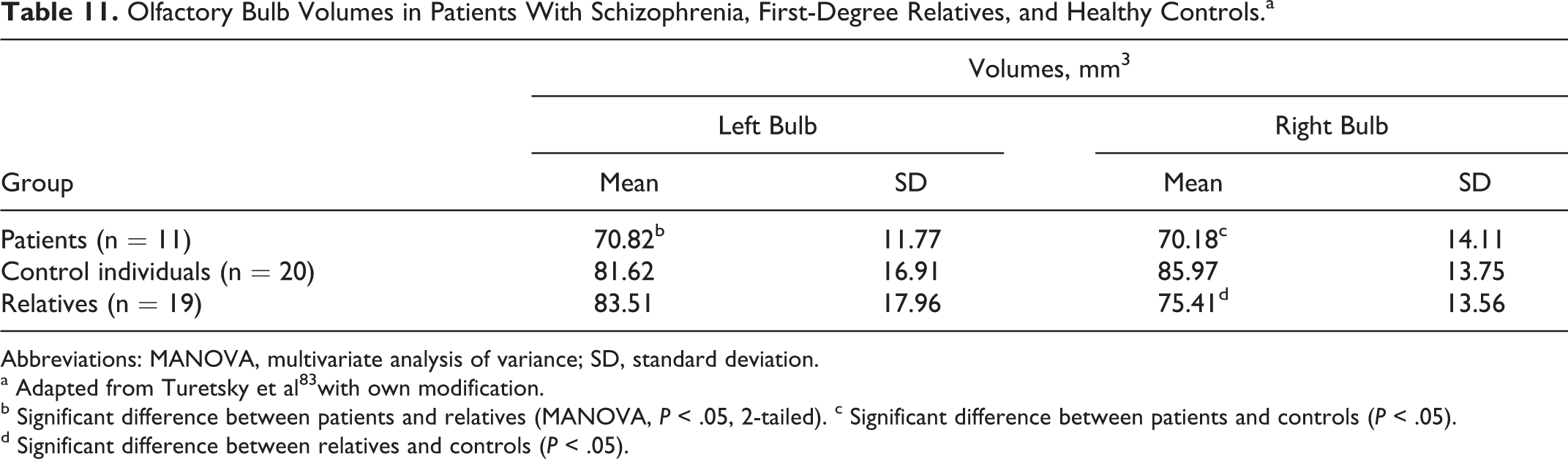
Abbreviations: MANOVA, multivariate analysis of variance; SD, standard deviation.
a Adapted from Turetsky et al 83 with own modification.
b Significant difference between patients and relatives (MANOVA, P < .05, 2-tailed). c Significant difference between patients and controls (P < .05).
d Significant difference between relatives and controls (P < .05).
Depression is highly prevalent in various medical conditions, including infectious, autoimmune, and neurodegenerative diseases. It seems that damage of the olfactory system is at least in part responsible also for development of depression because it was found that T gondii infection worsened mood in pregnant women, 105 female veterans, 106 older persons, 107 and patients with multiple sclerosis. 108,109 Higher incidence of depression also preceded the onset of Parkinson’s disease, 110 and the olfactory bulbectomized rat is usually serving as a model of depression. 111 Moreover, depression was reported in a sample of patients with obsessive–compulsive disorder, 112 and an important role of the parasite was suggested in the pathogenesis of this clinical entity. 113 Also, patients with recurrent mood disorders with history of suicide attempt had higher T gondii antibody titers than nonsuicide attempters (P = .004). 114 It must be added that antidepressants act on the host immune system causing neuroendocrine alterations associated with an increased generation of several bioneurotic molecules (Table 12). 115 Thus, the relationship between chronic latent T gondii infection and brain damage resulting in the development of depression should be seriously taken into consideration. In such patients, treatment of the infestation together with estimation of clinical course of depression would be helpful in more beneficial modification of actual therapeutic regimens. This suggestion is in line with the finding that the antipsychotic haloperidol and the mood stabilizer valproic acid most effectively inhibited the parasite growth in vitro with synergistic activity. 29
Antidepressant Effects on the Host Immune System.a
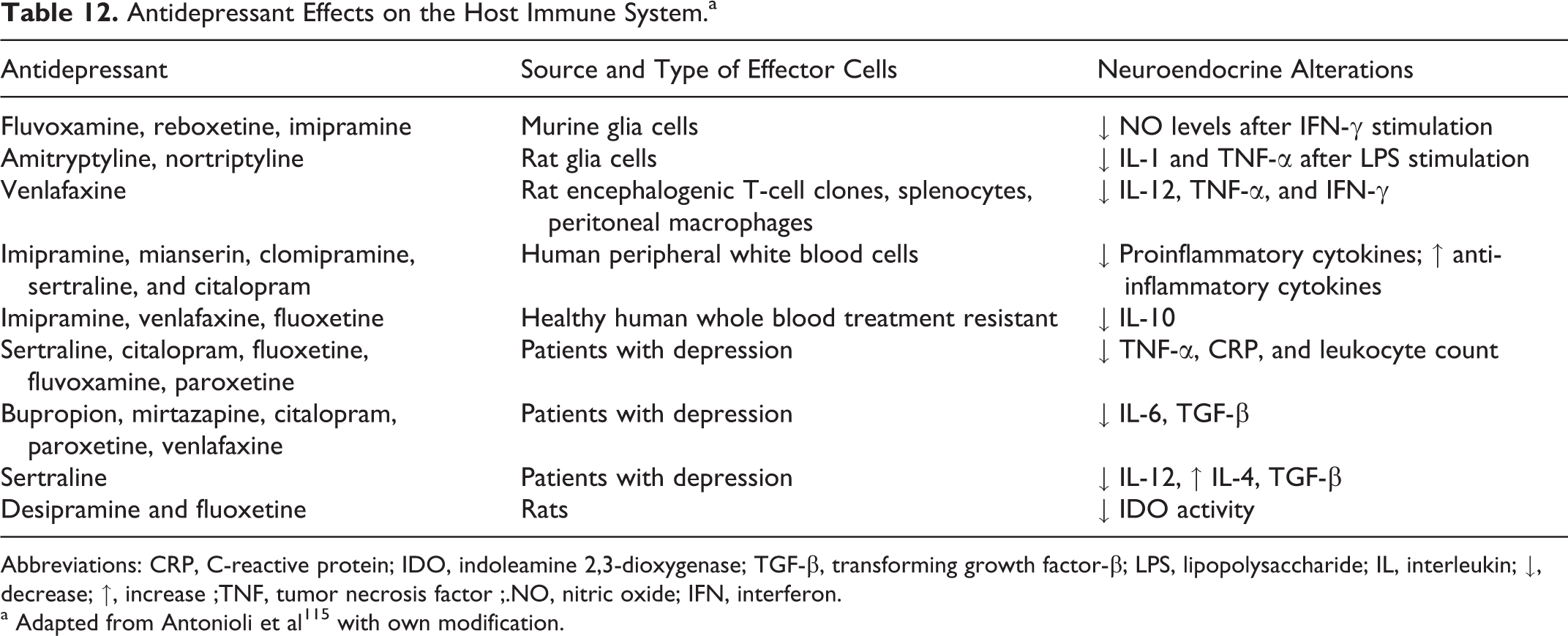
Abbreviations: CRP, C-reactive protein; IDO, indoleamine 2,3-dioxygenase; TGF-β, transforming growth factor-β; LPS, lipopolysaccharide; IL, interleukin; ↓, decrease; ↑, increase ;TNF, tumor necrosis factor ;.NO, nitric oxide; IFN, interferon.
a Adapted from Antonioli et al 115 with own modification.
Interestingly, T gondii infection can convert the rodents’ natural aversion to cat odors into attraction, 82,116,117 probably because of altered neuronal activity in limbic brain regions that is necessary for innate defensive behavior associated with the activation of adjacent sexual arousal pathways 79,80 and generation of various neurotransmitters. 115 It should be noted that a surface SAG1 antigen of T gondii combined with nontoxic mutants of cholera toxin and enterotoxin (powerful mucosal adjuvants) administered intranasally in mice provided a beneficial high-level protection after virulent challenge infection with the parasite cysts. 118 In addition, treatment with monoclonal antibody against IL-6 resulted in a remarkable decrease in inflammation and numbers of cysts in the brain of animals with toxoplasmic encephalitis. 119 This beneficial effect may be partly explained by the fact that IL-6 enhances intracellular replication of the parasite acting through interactions with IFN-γ and TNF-α molecular activities. 120 Unfortunately, despite development of many serological and molecular methods in recent years, diagnosis of toxoplasmosis still faces difficulties because most of the commercially available tests are not fully specific and sensitive, representing wide variations in accuracy due to the fact that the parasite exhibits several protein and LPS antigens depending on its virulence, strain type, infection stage (tachyzoites, bradyzoites, oocysts), innate and/or acquired host immunity, and so on. 121 –123
In summary, damage of the olfactory system caused by chronic latent T gondii infection may affect olfactory bulb volume and various olfactory functions, therefore being responsible for the smell impairment in patients with several neuropsychiatric and/or autoimmune diseases. In addition, it seems that damage of the olfactory system may also be at least in part responsible for development of depression, which is frequently observed in those individuals.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
