Abstract
Background/Objective:
Apolipoprotein E (apoE) plays an important role in the pathogenesis of Alzheimer’s disease (AD). Altered cerebrospinal fluid (CSF) and plasma levels have been previously reported in patients with AD. We hypothesized that CSF apoE levels of patients with newly diagnosed AD might be associated with their cognitive performance.
Methods:
Patients with AD (N = 71) enrolled into an observational study underwent neuropsychological testing (Consortium to Establish a Registry for AD [CERAD] plus) at time of diagnosis. The CSF was obtained, and apoE concentrations were determined. Generalized linear models were constructed to assess the associations of apoE and neuropsychological measures while adjusting for important potential confounders.
Results:
No association of CSF apoE levels and cognitive function could be demonstrated. Still, the use of neuroleptic drugs, female gender, preprogression time, and lower education were linked to worse cognitive function in some domains.
Conclusion:
The CSF apoE appears not to be suitable as a biochemical surrogate of cognitive function in AD under the given circumstances. By means of longitudinal analyses, potential associations with the velocity of decline will be investigated in the near future.
Introduction
Alzheimer’s disease (AD) is a major public health problem today with steadily increasing socioeconomic impact on aging societies in the near- and mid-term future. 1 Thus, advancing the research of AD pathogenesis and biomarkers for early diagnosis and disease monitoring is crucial. Furthermore, biomarkers as biochemical surrogates reflecting the presence of the disease itself, the disease stage, or the patients’ cognitive status are essential for monitoring purposes in potential therapeutic trials.
Apolipoprotein E (apoE) plays a central role in AD pathogenesis. First clues originated from epidemiological studies associating the APOE polymorphism with the disease risk in sporadic, late-onset AD (LOAD). 2 The risk modifying role of the APOE ∊4 allele has subsequently been validated in genome-wide association studies (GWAS). 3 The protein itself is expressed in a variety of organs, predominantly in hepatic and brain tissue. The ApoE, which exists in 3 isoforms (E2, E3, and E4) has a physiological role as a ligand in the endocytosis of lipoproteins and is therefore associated with lipid transport. 4 In the cerebrum, this is essential regarding the redistribution of lipids in neuronal degeneration and regeneration including membrane repair or axon remyelination. 5 Moreover, apoE is able to modulate glutamate receptor function as well as synaptic plasticity in an isoform-dependent fashion. 6 In AD, apoE has been associated with amyloid β (Aβ) fibrillization, deposition as well as plaque formation and therefore appears to be directly involved in AD pathogenesis. 7 On the histological level, immunoreactivity to apoE in amyloid plaques has been demonstrated already more than 20 years ago, thus putting emphasis on the link between apoE and Alzheimer’s disease. 8 Additionally, this has also been shown for neurofibrillary tangles (NFT) as the second important neuropathological hallmark of AD. 9 Underlining apoE’s role in plaque formation, Holtzman and colleagues were able to show that Aβ deposition is greatly reduced in apoE-deficient transgenic mice. 10
Considering the available pathomechanistic evidence linking apoE to AD, apoE has been studied as a potential biomarker of AD alongside the established biomarkers such as Aβ, τ, and its phosphorylated isoforms. Its concentrations in cerebrospinal fluid (CSF) and plasma have been investigated in several studies. The results are contradictory. Increased levels of apoE were reported in the CSF of patients with LOAD. 11,12 In a small study, Hahne and colleagues did not find any difference between AD and healthy controls. 13 However, Blennow et al discovered decreased levels in AD. 14 Reasons for these discrepancies could be diverse, that is, laboratory methods, medication, timing of CSF acquisition considering potential circadian rhythms and stage effects, and so on. Regarding the latter, conflicting results regarding longitudinal changes in CSF apoE in AD have also been published previously. 15,16
In a recent study conducted by Song et al, it was attempted to relate plasma concentrations of apoE and other relevant apolipoproteins to cognitive status and decline in healthy individuals as well as in patients with mild cognitive impairment (MCI). 17 Regarding apoE, higher levels were found in plasma of patients with MCI. Again, it must be mentioned that the available evidence regarding plasma apoE is also inconsistent. However, often decreased levels have been found in AD. 18,19
Since apoE appears not to cross the blood–brain barrier, 20 the reasons for altered apoE concentrations in the CSF might be different from those in plasma. Thus, CSF apoE may reflect pathophysiologic processes in the cerebrum better than plasma apoE. Considering the available evidence and Song’s results 17 in mind, we hypothesized CSF apoE concentrations to be associated with the cognitive status of patients newly diagnosed with AD. Thus, we analyzed CSF levels of apoE in a cohort of patients with AD from an ongoing, observational study. In univariate preanalyses, CSF apoE was associated with the performance in the Boston Naming Test (BNT, Pearson correlation coefficient −0.255, P =.048). By fitting generalized linear models (GZLMs), we attempted to unveil potential associations of CSF apoE with Consortium to Establish a Registry for AD (CERAD) test battery subscale measures adjusting for important potential influencing factors.
Methods
Patients
Patients included into this analysis were enrolled in the “rapidly progressive Alzheimer’s disease (rpAD)” study. This study comprises the entire continuum of AD phenotypes—from slowly to rapidly declining forms. It is of observational and prospective design. Dubois’ revised research criteria were used to establish the diagnoses. 21 These criteria are derived from the classic and mainly clinical National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer's Disease and Related Disorders Association criteria, 22 but CSF biomarkers’ analyses and neuroimaging are additionally used. All patients underwent extensive neuropsychological testing (CERAD-NP and Trailmaking test-A [TMT-A] 23 ) at the time of diagnosis. Furthermore, neurological examinations were performed by study physicians, and neuroimaging was obtained. Lumbar puncture was performed, and blood specimens were taken as part of the routine diagnostic process. All patients or legal caregivers provided informed consents. The study has been performed in accordance with the Declaration of Helsinki. The local ethics committee has approved the study (Georg August University, Göttingen, Germany, votum No 9/6/08).
Biomarker Measurements
Determination of CSF Aβ1-42, Aβ1-40 τ, and hyperphosphorylated τ (pτ) was done by means of standard methods using commercially available kits (Innogenetics, Gent, Belgium). The CSF apoE was quantified using enzyme-linked immunosorbent assay kits/monoclonal antibodies for recognition of human apoE (E276 mouse Ig [immunoglobulin] G1 [capture antibody], E887 mouse IgG1 [detection antibody]) by Mabtech AB (Nacka, Sweden).
Statistical Analyses
Consortium to Establish a Registry for AD subscale raw values and a CERAD “composite” value were used as the outcome measures. The CERAD subscales included Mini-Mental State Examination (MMSE), phonemic fluency, semantic fluency, BNT, word learning, word recall, drawing (copying figures), drawing recall (figure recall), and TMT-A. A CERAD “composite” value was obtained by extracting one principal component from the highly intercorrelated CERAD subscale scores. Thereby, the 9-dimensional data array (CERAD subscales) was reduced to 1 single variable reflecting most of the observed variance of the original data. The following variables were used as independent variables: CSF apoE levels, age, preprogression time (PPT; estimated time between the disease’s first clinical manifestation and diagnosis 24 ), sex, smoking, diabetes mellitus, medication (intake of benzodiazepines, antidepressants, neuroleptic drugs, and antidementia drugs such as acetylcholinesterase inhibitors or memantine), APOE genotype (∊4- vs non-∊4 carriers), and education (≤ 10 years vs >10 years of formal education).
For every dependent variable, 1 GZLM was fitted. First, all independent variables were included into the models. Successively, the GZLMs were fitted by means of stepwise backward selection, eventually utilizing only some selected independent variables that represented an optimal subset in terms of model fit. Akaike’s corrected information criterion (AICC) was used as a marker of model fit (the lower, the better). The P values below.05 indicated statistical significance. The P values were adjusted for multiple testing using Bonferroni’s procedure. SPSS version 20 (IBM corporation, New York) was used to compute the statistics.
Results
The patient sample examined consisted of 71 (N = 71) patients. Table 1 provides full overview regarding sociodemographic, medical, and genetic data as well as cognitive status. The results of CSF biomarkers are shown in Table 2.
Sociodemographic, Medical, Genetic, and Neuropsychological Data at Baseline (Time of Diagnosis).a
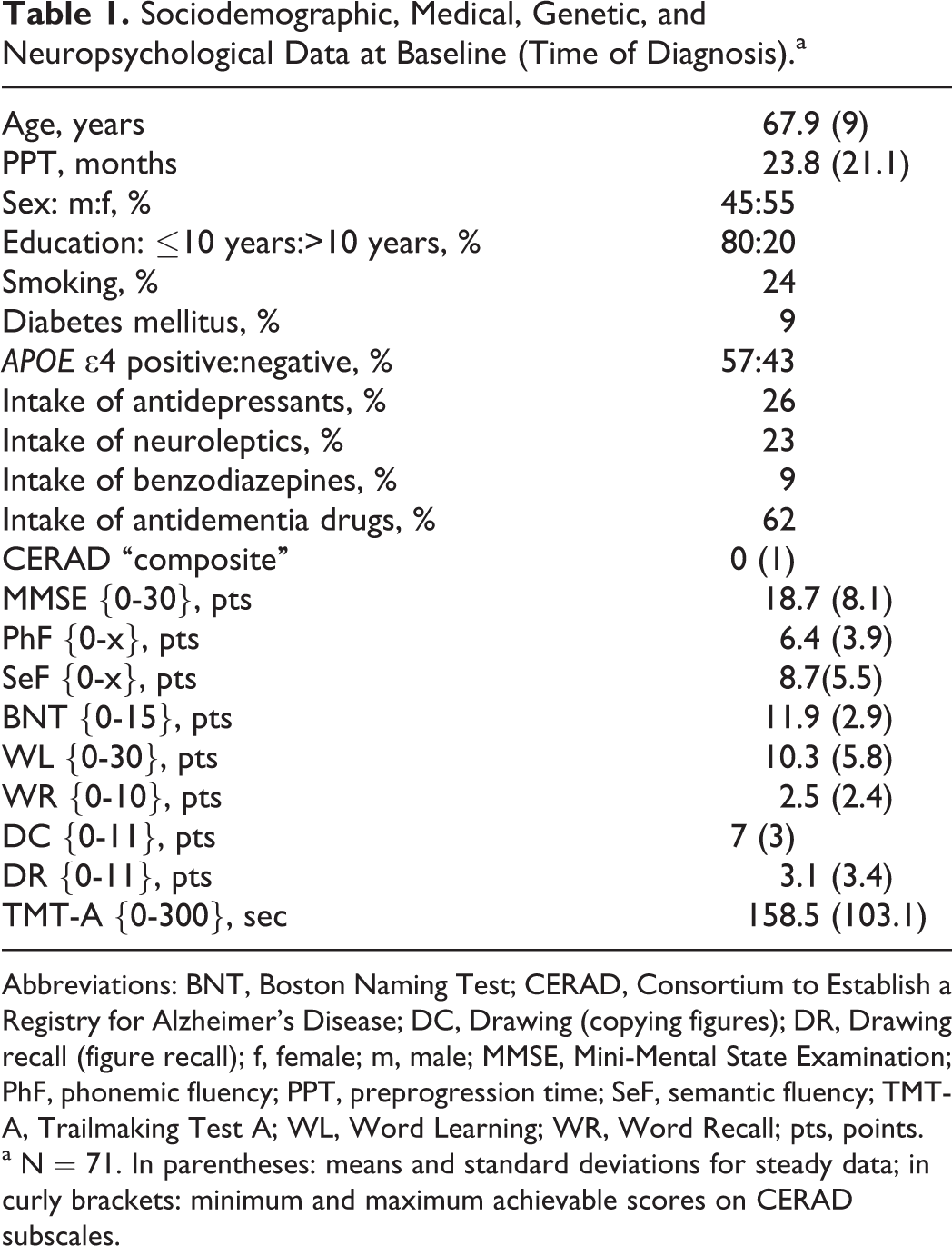
Abbreviations: BNT, Boston Naming Test; CERAD, Consortium to Establish a Registry for Alzheimer’s Disease; DC, Drawing (copying figures); DR, Drawing recall (figure recall); f, female; m, male; MMSE, Mini-Mental State Examination; PhF, phonemic fluency; PPT, preprogression time; SeF, semantic fluency; TMT-A, Trailmaking Test A; WL, Word Learning; WR, Word Recall; pts, points.
a N = 71. In parentheses: means and standard deviations for steady data; in curly brackets: minimum and maximum achievable scores on CERAD subscales.
Results of CSF Biomarker Measurements (Mean, Standard deviation).a

Abbreviations: Aβ, amyloid β; AD, Alzheimer’s Disease; CSF, cerebrospinal fluid.
a The “Cut-Off” is the diagnostic cutoff value for AD diagnostics standardized for our laboratory.
PreAnalyses
In preanalyses, univariate significant correlations of CSF apoE with the Aβ ratio (Pearson correlation coefficient = −0.38, P =.001) and the BNT score (Pearson correlation coefficient = −0.255, P =.048) could be uncovered. There was no significant difference in CSF apoE levels across the APOE polymorphism groups (CSF apoE in ∊4-positive patients = 2 ± 1 µg/mL; in ∊4 negative patients = 2.3 ± 0.8 µg/mL; P =.155). To seek confirmation, GZLMs were applied with the CSF apoE level as the dependent variable and age, gender, PPT, presence of diabetes mellitus, nicotine use, Aβ ratio, τ, pτ, and APOE genotypes as the independent covariates or cofactors. Significant associations were found for age (B = 0.22, P = 0.037), Aβ ratio (B = −0.998, P =.001), and diabetes (estimated marginal means; no diabetes: 2.1 [0.1] µg/mL; diabetes present: 3.2 [10.35] µg/mL; P =.003 [Bonferroni adjusted]).
Main Analyses
With 10 cognitive scales as the outcome variables, 10 distinct GZLMs were fitted in a multistage process. First, all variables were included into the model (see Methods section). Subsequently, independent variables not associated with the outcome were removed from the model if this led to a superior model fit as indicated by a reduction in the AICC. These optimized models are shown in Table 3.
Each column represents an individual GZLM.
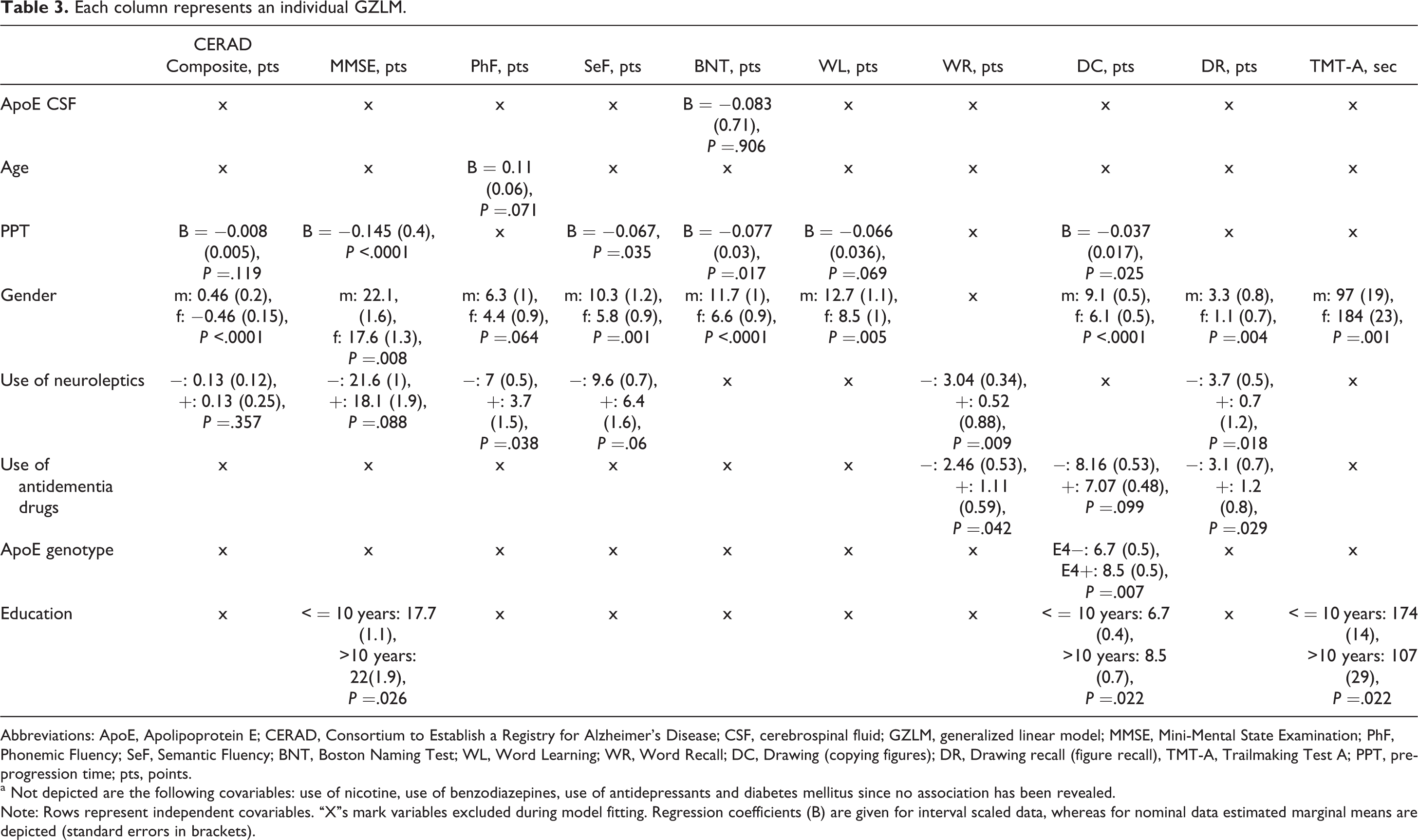
Abbreviations: ApoE, Apolipoprotein E; CERAD, Consortium to Establish a Registry for Alzheimer’s Disease; CSF, cerebrospinal fluid; GZLM, generalized linear model; MMSE, Mini-Mental State Examination; PhF, Phonemic Fluency; SeF, Semantic Fluency; BNT, Boston Naming Test; WL, Word Learning; WR, Word Recall; DC, Drawing (copying figures); DR, Drawing recall (figure recall), TMT-A, Trailmaking Test A; PPT, preprogression time; pts, points.
a Not depicted are the following covariables: use of nicotine, use of benzodiazepines, use of antidepressants and diabetes mellitus since no association has been revealed. Note: Rows represent independent covariables. “X”s mark variables excluded during model fitting. Regression coefficients (B) are given for interval scaled data, whereas for nominal data estimated marginal means are depicted (standard errors in brackets).
As the main finding, CSF apoE was not associated with cognitive status. The univariate association of apoE and BNT scores could not be confirmed in the GZLMs. Furthermore, age, smoking, diabetes mellitus, and use of antidepressants or benzodiazepines were not predictive of performance on the CERAD subscales. Patients with a longer duration of illness performed worse on some scales. So did female patients and those taking neuroleptic or antidementia drugs. APOE status was only associated with visuoconstructive abilities (∊4-positive patients performed better), and lower education was related to lower scores on the MMSE scale as well as tests of visuoconstruction and executive function (Table 3). The P values (Bonferroni adjusted) are indicated. For steady variables, coefficients (B) and standard errors (in parentheses) indicating associations adjusted for possible effects of all other independent variables are given. For categorical data, estimated marginal means and standard errors (in parentheses) are depicted.
Discussion and Conclusion
The exact functional role of apoE in AD pathogenesis is not clearly defined. The available evidence from neuropathological and functional studies, as well as the well-established risk-modifying properties of the APOE polymorphism in epidemiological studies and GWAS, strongly support the notion that apoE is a significant factor in AD pathogenesis. The finding of apoE immunoreactivity in Aβ plaques and NFT is intriguing, 8,25 and an active role of apoE in Aβ plaque formation has been demonstrated. 7,10,25 The revelation of ApoE functions other than sole lipid transport, such as modulation of glutamatergic receptor signaling and synaptic plasticity 6 as well as possible exertion of immediate neurotoxic effects 26 may help to establish a more direct link to cognitive function. Additionally, cerebral cholinergic levels and thereby cognition may be influenced by apoE which interacts with Aβ and the butyrylcholinesterase. 27
It has been shown that apoE does not cross the blood–brain barrier. 20,28 Hence, it appears conceivable that CSF levels of apoE reflect those in the brain, although—to our knowledge—no convincing studies relating apoE CSF concentrations to neuropathology have been published to date. Brought into a clinical context, CSF apoE levels have not been consistently examined as a diagnostic biomarker for AD. Some contradictory evidence exists. 11 –14 Studies associating cognitive function to apoE levels are scarce. Higher CSF levels are related to worse cognitive function in APOE ∊4-positive HIV-infected patients. 29 Song and colleagues assessed plasma lipoproteins in context with cognitive status. Among others, plasma apoE levels were higher in patients with MCI compared with healthy individuals and were negatively correlated with cerebral gray matter volume. 17
Herein, we endeavored to uncover potential associations of cognitive status of patients newly diagnosed with AD and their CSF apoE levels. In preanalyses, significant associations with age, Aβ ratio, and the presence of diabetes could be revealed. Higher apoE levels with higher age are consistent with previously published findings. 30 The inverse association of CSF apoE with Aβ ratio might be hinting at an interaction between both the molecules. 31,32 The reason why CSF apoE concentrations might be higher in patients with AD with diabetes has not convincingly been investigated yet. In contrast to the literature, no significant difference according to APOE genotype groups was found in our study. 27
As this study’s main result, we were not able to reveal any association of CSF apoE and cognitive status of patients with AD at the time of diagnosis. Especially the preanalytic, univariate finding of higher CSF ApoE being related to lower BNT results was reduced to a nonsignificant “trend” in the GZLMs. Irrespective of the negative outcomes, other significant associations were found. The influence of neuroleptic drug use, gender, duration of illness, and education could be consistently demonstrated. Antipsychotic drugs have already been linked to lower cognitive status. 33,34 This can be interpreted in 2 ways, either worse cognitive function is associated with more psychotic symptoms in AD thus leading to an increased antipsychotic drug use or the use of neuroleptic drugs leads to impairment of cognition. The same applies to “gender” and is therefore consistent with previous reports. 35,36 Also compatible with previous studies is the finding that higher education is independently correlated with better performance on some CERAD subscales. The paradigm of cognitive reserve must be mentioned in this context. 37 Moreover, the preprogression time 24 is predictive of cognitive function; it is the nature of neurodegenerative diseases to progress over time. Often during this preprogression time primary care physicians prescribe antidementia drugs before formal diagnoses are established. This observation corresponds well to experiences in the clinical routine. This might explain worse performance of patients taking antidementia drugs in some parts of the CERAD tests.
A few more variables (use of nicotine, use of benzodiazepines, use of antidepressants, and diabetes mellitus) possibly associated with cognitive status have been considered in the models as described earlier (Table 3). But these did not exert any relevant effect on the outcome measures. Inclusion of these covariables into the GZLM enabled control of potential confounding effects. Nevertheless, residual confounding by unknown factors cannot entirely be excluded. Analysis of all potential confounders in this respect is important. Several studies omit the inclusion of important covariables such as the intake of neuroleptic drugs or preprogression times.
Our study has strengths and limitations. Regarding the latter, this analysis is cross-sectional. However, the underlying cohort is being followed up prospectively. Therefore, it will be possible to uncover longitudinal trends with future analyses. Another limitation is the lack of a global dementia severity scale. Yet, by extracting a CERAD composite score, one variable comprising all cognitive domains has been created. Furthermore, possible floor and ceiling effects also have to be acknowledged as study limitations. The most important limitation is the sample size (N = 71). Our population is rather small compared to the studies performed on an epidemiologic scale. However, it is noteworthy that CSF biomarker analyses were available. Although by now CSF analyses should have been implemented into the routine diagnostic workup of dementia, this is often not the case. Thus, CSF specimens for scientific investigations are rare goods. This is one reason for a small sample size. Conversely, the use of CSF biomarkers combined with updated research criteria increasing the certainty of the diagnoses is a strength of this study.
It has to be emphasized that the usability of CSF apoE as a diagnostic biomarker has not been examined here. This would necessitate inclusion of demented non-AD patients and healthy controls.
In summary, we were not able to confirm the suggested associations between CSF apoE concentrations of patients having AD and their cognitive status. Thus—under the reservation of study limitations—CSF apoE appears not be a biochemical surrogate for cognitive function. However, disease stage, gender, education, and use of neuroleptic drugs were demonstrated to be associated with worse cognitive function. Since the majority of patients examined here had mild to moderate AD, it still remains possible that CSF apoE levels may be a marker of AD severity at earlier or later stages of the disease.
Further studies should apply additional neuropsychological tests differentially sensitive to cognitive changes in very early or very late stages (floor/ceiling effects). Also, the evaluation of CSF apoE as a prognostic marker for longitudinal cognitive change (decline) should be investigated in prospective studies.
Footnotes
Acknowledgement
We thank Mrs Barbara Ciesielcyk and the laboratory team for skillfully performing the CSF analyses and ELISAs.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Bundesministerium für Bildung und Forschung grant within the German Network for Degenerative Dementia (KNDD-2, 2012-2015, determinants for disease progression in AD, grant No. 01GI1010C).
