Abstract
Objective:
The aim of this study was to describe the performance in Clock Drawing Test (CDT) of the elderly individuals assessed in a geriatric clinic, with at least 1 year of schooling, comparing with other groups with higher education and with Clinical Dementia Rating (CDR) levels. The study also aims to correlate the results of CDT and other used diagnostic tests for dementia by CDR levels, providing additional validity evidence to the CDT.
Methods:
Cross-sectional study with 426 elderly individuals, >60 years old and at least 1 year of education. All participants searched for medical assistance at Geriatric and Gerontology Ambulatory of Jundiaí city, in Brazil. The community-dwelling outpatients previously undergone a detailed clinical examination and neuropsychological evaluation: Cambrigde Cognitive Examination (CAMCOG), Mini-Mental State Examination (MMSE), andCDT. To differentiate data from diagnostic groups based on CDR, it Kruskal-Wallis test was used. Pearson statistics were calculated to compare data from CDT and CDR. The statistical analyses were 2-tailed and were considered significant when P < .05.
Results:
Regarding CDT, groups with more years of schooling showed similar means in CDR = 0 and CDR = 0.5 and in CDR = 1 and CDR = 2. Shulman and Sunderland scale were high score in groups with more years of education and above of cutoff points in all CDT score. On the contrary, in Mendez scale we did not observed similar means. Otherwise, in the group with less years of schooling greater means differences in the CDT were observed.
Conclusion:
The CDT did not show a strong correlation with MMSE and CAMCOG, both important instruments in Brazilian population to investigate dementia. For elderly individuals with high education levels, the CDT did not seem to be a good test to detect cognitive impairment.
Keywords
Introduction
The Clock Drawing Test (CDT) is one of the most important diagnostic instruments for to test dementia in elderly patients with high sensitivity and specificity. 1,2 In 1953, the CDT was used to analyze functional performance in parietal lobes. 3 Nowadays, it is used to evaluate memory, executive function, and verbal comprehension. 1,2,4,5
Shulman et al 5 and Bourke et al 6 report that execution errors can be observed in the position of the hands to indicate the correct time or size (pointers that exceed the circle) in elderly individuals with frontal dysfunction. Another common mistake is evidenced when the patient put the hands, indicating 11 and 50 minutes rather than 11 and 10 minutes (11:10). This task suggests abstraction performance. Literature indicated many scales for the CDT 7 –11 with a variety of sensibility (42%-97%) and specificity indices (48%-94%). 12
Damasceno 13 and Teixeira et al 14 suggest Alzheimer’s disease (AD) in Brazil represent 50% to 70% of whole dementia cases. With the evolution of the disease, especially in patients with advanced dementia, the impairment of the cortical region leads to disorientation, confusion, urinary incontinence, and inability to care for oneself. 13 To verify the stages of dementia some scales were developed to determine its severity. 15 Clinical Dementia Rating (CDR) is the most widely used because it measures individual changes regardless of the age and education, with high consistency in the diagnostic value. 15 –17 Higher scores on CDR indicate greater severity of dementia. 17
Thus, considering the CDT as one of the most important diagnostic tests for dementia, a widely applied instrument in the world, to have great relevance to the international literature since it cited in many articles published, and to be able to assess several cognitive impaired, the aim of this study was to analyze CDT the performance in of the elderly assessed in a geriatric clinic, with at least one year of schooling compared with other groups with higher education and with high CDR levels. Our study also aims to correlate the results of CDT and other used diagnostic tests for dementia by CDR levels, providing additional validity evidence to the CDT.
Methods and Procedures
Cross-sectional study included 426 participants aged >60 years with at least 1 year of education. The educational group was divided as 1 to 4 years (group 1), 5 to 8 years (group 2), 9 to 11 years (group 3), and >11 years (group 4). All participants searched for medical assistance at the Geriatric and Gerontology Ambulatory of Jundiaí city, in Brazil. The community-dwelling outpatients previously underwent a detailed clinical examination and neuropsychological evaluation. The neuropsychological examinations were Cambrigde Cognitive Examination (CAMCOG) 18 ; Mini-Mental State Examination (MMSE), 19 CDT, 7 –9 Geriatric Depression Scale (GDS) with 15 items, 20 and Pfeffer Functional Activities Questionnaire (PFAQ). 21 Criteria for AD were based on Mckhann et al 22 and Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition [DSM-IV]). 23
Patients with severe dementia (CDR ≥3), history of stroke, major depression (GDS ≥ 7), parkinsonism, visual or auditory difficulties and participants who did not accept to participate for neuropsychological examination were excluded from the study. The CDR score of 0 is considered as the inclusion criteria for normal control group (NC).
Participants were separated into 3 groups according to the CDR scores: CDR = 0, CDR = 0.5, and CDR = 1 and CDR = 2. The CDR levels were then correlated with CDT to verify whether dementia stage influenced the CDT results by the education level.
Statistical Analysis
All information were analyzed by SPSS 15.0 (2007). Sample distribution was tested by Kolmogorov-Smirnov test, and it was found that cognitive testing (CDT) corresponded to a nonparametric distribution. To differentiate data from diagnostic groups based on CDR, Kruskal-Wallis test was used. Pearson statistics were calculated to correlate data from CDT and CDR. The statistical analyses were 2-tailed and were considered significant when P < .05.
Results
In present study, 312 (73.20%) were female, 230 (54%) had 1 to 4 years of schooling, 74 (17.40%) had 5 to 8 years, 59 (13.80%) had 9 to 11 years, and 63 (14.80%) had more than 11 years of education. Average age was 60 years. A total of 144 (33.80%) participants had a CDR score of 0, 60 (14.10%) had a score of 0.5, and 131 (30.80%) and 91 (21.40%) patients had a CDR score of 1 and 2, respectively. Age, gender, and schooling showed statistically significant differences between groups ( Table 1).
Sample Characteristics Regarding Age, Gender, and Education Level
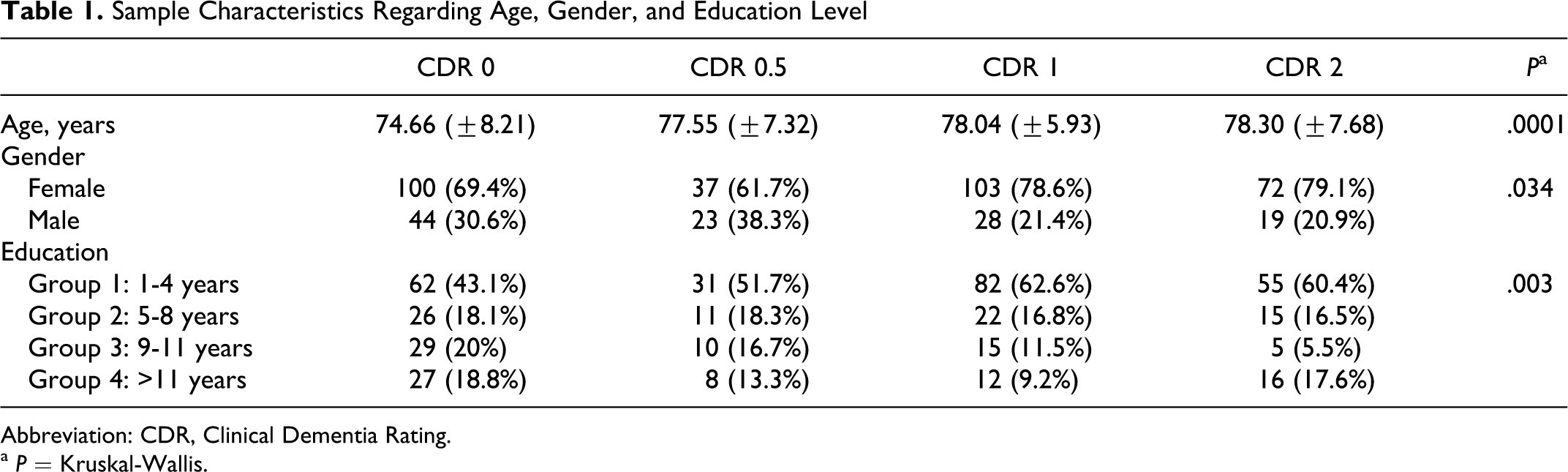
Abbreviation: CDR, Clinical Dementia Rating.
a P = Kruskal-Wallis.
Considering CDT, groups with more years of schooling showed similar means in CDR = 0 and CDR = 0.5 (Table 2) and in CDR = 1 and CDR = 2 (Table 3). In groups with more years of education the Shulman and Sunderland scale score were high and all the CDT scores were more than the cutoff points. But in Mendez scale different means were observed. In the group with less years of schooling, greater CDT mean differences were observed. Mann-Whitney analyses showed significant statistical differences between CDT and low educational level in those with a CDR score of 0 0.5 (Table 2) and between the groups 1 to 4 years and 9 to 11 years with a CDR score of 1 and 2 (Table 3). The CDT was not capable to differ patients with high education levels among the CDR (0 to 2) and the group with 9 to 11 years of schooling showed no statistical significance in the differences between CDR 0 and 0.5, as can be ascertain in Table 2.
The CDT scale scores were poorly correlated with MMSE and PFAQ results in those with a CDR score of 0. Moderate and significant correlations were observed in those with a CDR score of 0.5, 1, and 2 between these measures, mainly between CDT scales and MMSE results (Table 4). Correlation results between CDT scales and PFAQ were most important (significant and moderately associated) in those with a CDR score of−0.5 and 1. We also observed moderate and significant correlations between CDT scales with CAMCOG results in all levels of CDR groups.
Mendez, Shulman, and Sunderland CDT Means and SD of the Participants With CDR = 0 and CDR = 0.5 Separated by Schooling Groups
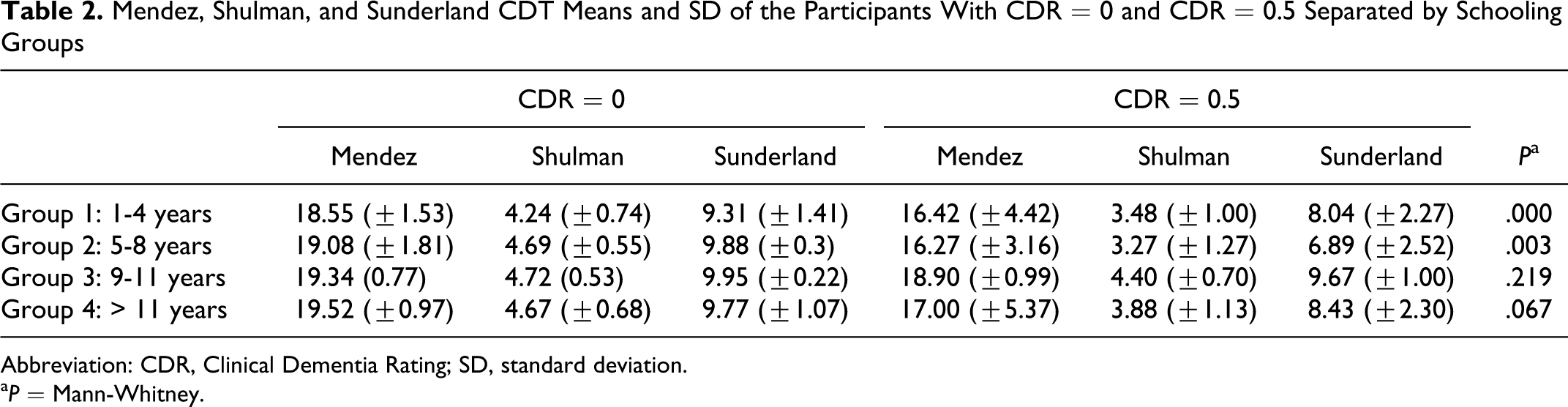
Abbreviation: CDR, Clinical Dementia Rating; SD, standard deviation.
a P = Mann-Whitney.
Mendez, Shulman, and Sunderland CDT Means and SD of the Participants With CDR = 1 and CDR = 2 Separated by Groups of Schooling
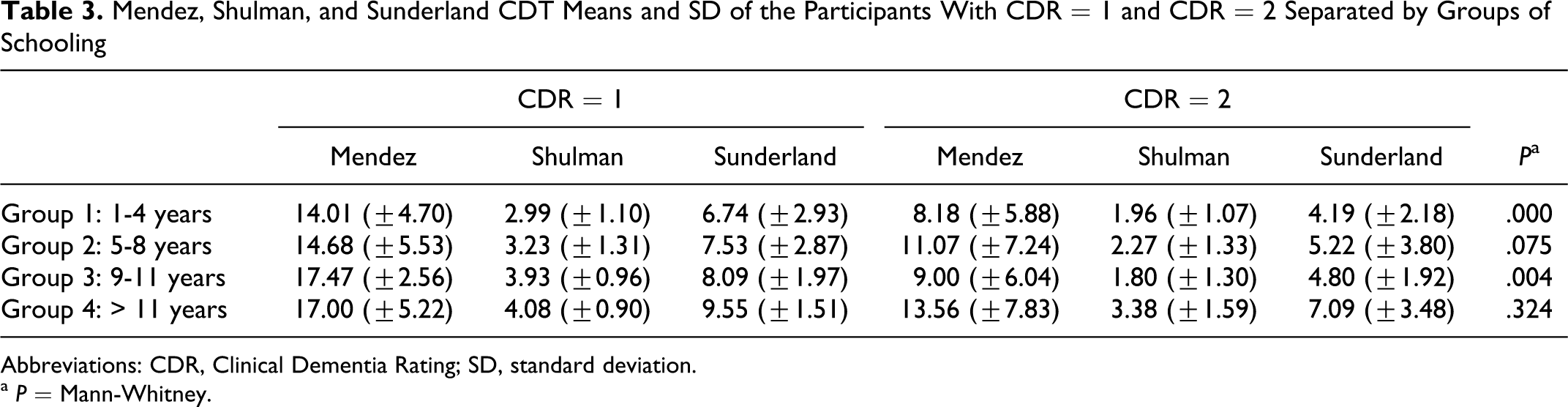
Abbreviations: CDR, Clinical Dementia Rating; SD, standard deviation.
a P = Mann-Whitney.
Pearson’s Correlation Analysis Coefficients and Significance Values Between CDT Scales, MMSE, CAMCOG, and PFAQ by CDR Levels
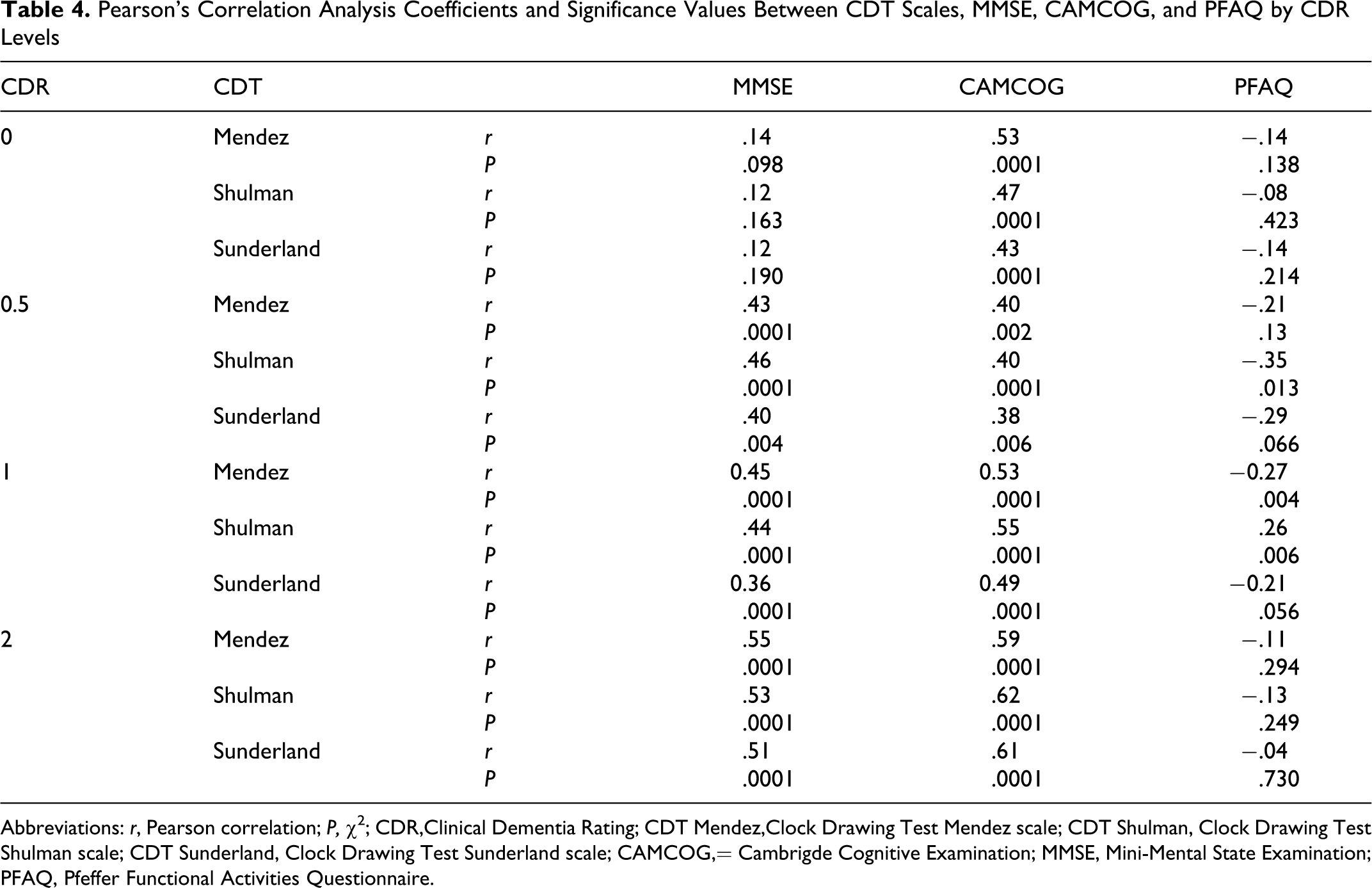
Abbreviations: r, Pearson correlation; P, χ5; CDR,Clinical Dementia Rating; CDT Mendez,Clock Drawing Test Mendez scale; CDT Shulman, Clock Drawing Test Shulman scale; CDT Sunderland, Clock Drawing Test Sunderland scale; CAMCOG,= Cambrigde Cognitive Examination; MMSE, Mini-Mental State Examination; PFAQ, Pfeffer Functional Activities Questionnaire.
Discussion
The aim of this study was to compare the Mendez and Shulman and Sunderland CDT scales in different CDR scores, with respect to the years of education. The CDT has a high sensitivity and specificity in the diagnosis of dementia. 1,5 It is widely used because it is simple to correct and easy to be administered (does not require a qualified professional to administer the test). 1,5 It was shown that in those with more than 8 years of schooling, the CDT had similar scores at different levels of dementia.
Fuzikawa et al 24 found a moderated correlation between MMSE and CDT in elderly individuals with low education levels and had no significant differentiation with regard to age, gender, and schooling levels. In Fuzikawa et al 24 study, the education mean was 2 years. Unlike the study by Fuzikawa et al, in the present study 122 patients had more than 8 years of education. Similar mean CDT scores were observed in Shulman and Sunderland scale, suggesting that CDT is not a good test to discriminate elderly patients with dementia with high education.
The correlation between CDT Mendez scale and MMSE was moderate in more severe dementia (CDR = 2). Although CAMCOG showed a moderated correlation with CDT scales. We cannot expect low MMSE and CAMCOG scores in elderly patients with high education levels will also result in low CDT scores. One can suppose that the elderly individuals with higher schooling years seem to have better cognitive function and patients with severe of dementia can draw a satisfactory clock. Corroborated with this study, Heinik et al 4 ,25,26 consider preservation of cognition in early dementia suggesting a complementary test together with CDT to increase diagnostic accuracy. Ainslie et al 27 and Manos 28 enfatizes that neuropsychiatric assessment should considerate educational level, age and depressed mood, since these factors may influence patient outcomes. Heterogeneous population studies suggest that CDT can be used concurrently with other tests to evaluate dementia. 25,29
The CDT Mendez scale seems to be a good instrument when the mean is compared with the different CDR scores. Storey et al 30 found a good sensitivity (98%) in Mendez scale but the CDT was not a useful predictor of the dementia diagnosis. Seigerschmidt et al 31 also did not find a statistically significant difference in CDT in identifying the earlier stages of dementia. The authors considered the CDT lacks information in prodromol cases, besides collected data of age, gender, and education levels. The same data were observed in Powlishta et al 32 study of patients with mild dementia, proving that CDT is not a useful screening instrument.
A limitation of the present study is the lack of sensitivity and specificity data from CDT scales in those with high education levels. In future studies, the ROC curve seems to give an idea of the cutoff points in patients with many years of schooling.
In conclusion, the CDT does not have a strong correlation with MMSE and CAMCOG, both considered important instruments in diagnosing dementia in Brazilian population. For elderly individuals with high education levels, the CDT does not seem to be a good instrument to indicate cognitive impairment.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
