Abstract
Alzheimer’s disease (AD) is characterized by the progressive impairment of mental and emotional functions, including the processing of emotional facial expression (EFE). Deficits in decoding EFE are relevant in social contexts in which information from 2 or more sources may be processed simultaneously. To assess the role of contextual stimuli on EFE processing in AD, we analyzed the ability of patients with AD and healthy elderly adults to identify EFE when simultaneously performing another task. Each of the 6 basic EFEs was presented to 15 patients with AD and 35 controls in a dual task paradigm that is in parallel with a visuospatial or a semantic task. Results show that the decoding of EFEs was impaired in patients with AD when they were simultaneously processing additional visuospatial information, yet not when they were performed in conjunction with a semantic task. These findings suggest that the capacity to interpret emotional states is impaired in AD.
Keywords
Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder that produces severe deficits in cognitive function, affecting memory, planning, working memory, attention, and communication. The deterioration of communication skills is directly related to deficits in the nonverbal communication of emotions, including the ability to recognize the emotional states of others, 1 and more specifically the ability to interpret emotional facial expression (EFE).
The greatest known risk factor for AD is increasing age and indeed, the majority of patients with AD are over 65 years of age. The impact of aging on the ability to recognize EFE is well documented. Aging has been linked with impairment of the ability to identify facial expressions of anger, sadness, and fear but not happiness, surprise, or disgust. 2 -4 The accepted hypothesis is that in normal aging, these specific deficits are induced by deterioration in brain areas that mediate emotional processing; sadness is processed via the anterior and subcallosal cingulate, 5 while fear and disgust are processed in the amygdala 6,7 and basal ganglia, 8,9 respectively.
Structural and morphological brain damage in patients with AD is more pervasive than that observed in healthy aged adults, and such damage may underlie the impairment of EFE decoding in these patients, affecting certain basic emotions more than others. 10,11 Several studies have described specific deficits in patients with AD in identifying discrete emotional faces, that is negative emotions, 12 -14 with the exception of recognizing disgust, which is preserved. 15 Importantly, results can vary depending on the type of the EFE identification task employed. Thus, patients with AD exhibit deficits in EFE decoding in tasks that demand a high level of cognitive effort, such as discrimination 16 and matching, 13 and these observations are supported by neuroimaging studies 17,18 demonstrating damage to the emotional processing areas of AD brains. However, the origin and nature of this impairment remains unclear. When reviewing the facial emotion recognition in AD, 19 several methodological inconsistencies in the literature were highlighted, arguing that the identification of deficits is dependent on the cognitive resources associated with each type of task (discriminating, matching, or identifying EFEs). Indeed, it was concluded that patients with AD have no difficulties in identifying particular EFEs, suggesting that studies in this area should employ tasks that more closely approximate to basic social interactions.
In social contexts, nonverbally communicated emotions are processed constantly, and they must be interpreted correctly during communication. 20 This processing provides additional information to facilitate the interpretation of other people’s words and actions in everyday life. As interpersonal communication takes place in parallel with other activities, information from various sources must be processed simultaneously (eg, decoding EFEs while linking them to verbal information). This continuous flow of information requires a network of processes that simultaneously manipulate and evaluate information from multiple sources. As EFE processing depends partly on the ability to split attention resources between at least 2 concurrent tasks, emotional dual task (DT) paradigms offer the most accurate means of assessing the processing of emotional stimuli that occurs in everyday life.
The majority of studies investigating deficits in divided attention employ DT paradigms in which 2 tasks are performed individually or in combination. Studies of DT 21,22 indicate that the division of attention is associated with a decrement in task performance, particularly when both tasks are of the same nature (eg, visual or semantic). 23 Our prediction that patients with AD perform poorly in attention-demanding tasks is supported by an abundance of behavioural and neurological data, 24 -30 demonstrating a disproportionate impairment in the simultaneous performance of 2 tasks.
Given that successful communication and appropriate interactions are dependent on the ability to detect and react to the affective state of others while processing information from additional sources, it is important to determine whether these fundamental processes are impaired in AD. As patients with AD exhibit deficits in the simultaneous performance of 2 tasks, 31,32 functional deficits in EFE identification associated with this disease may influence the impaired interpersonal communication observed in these patients in social contexts.
The main objective of this study was to investigate the role of context stimuli in the interpretation of facial expressions by patients with AD. Specifically, we assessed the capacity of patients with AD to identify EFEs while simultaneously performing a visuospatial or semantic task. We predicted greater deficits in EFE identification in patients with AD when required to simultaneously process additional information. Moreover, we predicted greater DT impairment when EFE identification was performed in parallel with a visuospatial rather than a semantic task (ie, when both tasks depend on the same cognitive resources). This hypothesis was based on our previous studies of emotional processing, 33 -36 which revealed that neurological patients performed worse than healthy elderly adults in a DT paradigm, demonstrating that deficits in EFE identification are due to pathological effects rather than those of aging.
Methods
Participants
The AD patient group was comprised of 15 individuals (9 female and 6 male), ranging in age from 65 to 85 years (mean, 76.87; standard deviation [SD], 6.64), who were recruited from the outpatient practices of the Hospital Universitario La Paz in Madrid. All patients had a diagnosis of probable AD according to National Institute of Neurologic, Communicative Disorders and Stroke-Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA) criteria. 37 Individuals with a history of cerebrovascular accident, head injury, neurological disorders (other than AD), cardiovascular disease, chronic alcoholism, and/or drug abuse were excluded from the study. No participants were using neuroleptic medications, and all performed normally in general medical and neurological screening tests. Inclusion criteria also included normal corrected eyesight and a mild stage of the disease process, as determined by the Mini-Mental State Examination 38 (MMSE) and the Blessed Dementia Scale 39 (BDS). The lower cutoff for mild impairment was 18 out of 30 and the mean MMSE score for participants with AD was 23.33 (SD, 3.37; range, 18-29). The control group was composed of 35 healthy adult volunteers (22 female and 13 male), from 66 to 85 years of age (mean, 73.86; SD, 3.94). The same criteria described above were applied to the control participants, with an MMSE score of 24 or higher as the cutoff for inclusion as a control participant to screen out individuals with undiagnosed clinical impairments. 40,41 The mean MMSE score for the control group was 29.31 (SD, 0.71; range, 27-30). Participants in the control group were recruited from a variety of community-based social organizations for the elderly individuals. An additional exclusion criteria for dementia was a BDS score >4.
A final exclusion criterion for both groups was major depression, as indicated by scores >7 on the 15-item version of the Geriatric Depression Scale (GDS), 42,43 and a diagnosis of any psychiatric disorder. Written informed consent was obtained from all participants or from caregivers when appropriate. Background data for both groups are presented in Table 1.
Demographic Characteristics of Alzheimer’s Disease (AD) and Control Groups
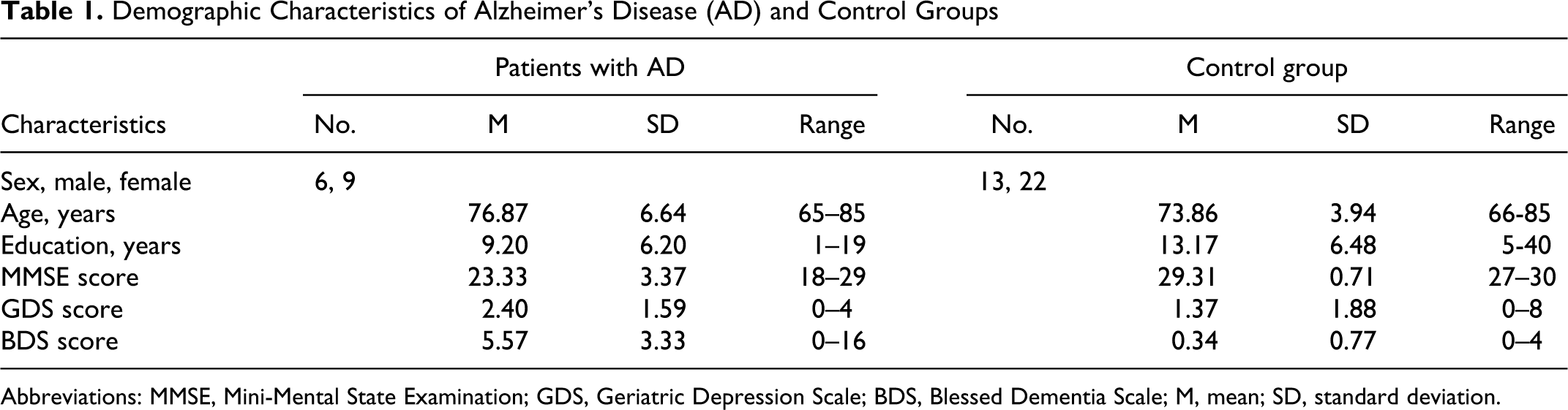
Abbreviations: MMSE, Mini-Mental State Examination; GDS, Geriatric Depression Scale; BDS, Blessed Dementia Scale; M, mean; SD, standard deviation.
The control and AD groups did not differ significantly in terms of age, t(48) = 1.99, P > .050; gender ratio, AD: χ 2 (1) = .60, p > .05; Control group : χ 2 (1) = 2.31, P > .050; or years of education, t(48) = −2.00, P > .050; or GDS t(48) = 1.85, P = .050. As expected, significant differences in MMSE, t(48) = −10.09, P > .000, and BDS scores, t(48) = 8.39, P > .000, were observed between groups, demonstrating the impaired cognitive capacity of the AD group with respect to controls.
Experimental Tasks
Emotional facial expression identification task
The EFE identification was performed as described previously, thereby facilitating comparison with the present findings. 33 -36 In a forced-choice task, participants were presented 24 EFEs one at a time (4 representing each basic emotion: happiness, sadness, anger, fear, surprise, and disgust), and they were asked to identify the emotion that best described the target stimulus. The participants were provided with a response list with the names of the 6 emotions and they were given as much time as necessary to make their decision. The stimuli were generated using a 3-dimensional (3D) imaging technique (Poser 6, Curious Labs, Santa Cruz, California), which creates virtual human faces whose expressions are determined by the single action units described in the handbook of the Facial Action Coding System (FACS). 44,45 The emotions were portrayed by 4 virtual actors (2 male and 2 female avatars) and the expressions were enacted by an experienced FACS coder (Johannes Heiner Ellgring [JHE]), activating their virtual muscles with respect to single action units in the computer animation. These stimuli elicit facial electromyographical reactions 46 and amygdalar activation, 47 and thus they represent a valid method for producing standardized expressions of emotions for the assessment of emotional processing. The main dependent variable was accuracy (ie, the number of correct EFEs identified). The total score ranged from 0 to 4, with a maximum of 24 correct responses. “Correctness” was defined as an answer corresponding to the label given according to the Facial Action Units. The chance level was considered as 1 correct answer for a single emotion and 4 for the entire set of 24 items.
Visuospatial task (Corsi Blocks)
The Corsi Blocks (CB) task was used to create visuospatial interference. This task is commonly used to assess short-term visuospatial memory in adults, children, and patients with neuropsychological deficits. The CB task involves a representation of 9 circles in a 3 × 3 horizontal and vertical array. In each trial, a specific arrangement of the 9 circles (7 white and 2 red) was displayed, with the 2 red circles (targets) appearing in a predetermined sequence at a rate of 1 per second. Participants were asked to mark the correct sequence of the red circles on an answer sheet.
Semantic task (backward digit recall)
For semantic interference, we used a backward version of the digit recall task (BDR), a commonly used measure of working memory. A total of 24 sequences of 2 digits (selected from 1 to 9 without repetition) were presented aurally at a rate of 1 per second from a recording of a male human voice. Participants were required to recall the digits in the reverse order of presentation.
To calculate any loss in DT performance, all participants also performed the single tasks described above.
Dual Tasks
To experimentally simulate EFE processing in a social context, participants performed an EFE identification task while simultaneously processing a visuospatial or semantic task.
The EFE identification concurrent with a visuospatial task (EFE/CB)
Participants performed the EFE identification task concurrent with a CB trial. A total of 24 trials were conducted, each beginning with an acoustic signal, the tone “F” at a frequency of 500 Hz that lasted 150 ms. A visual signal was presented simultaneously, consisting of a white plus sign on a black screen that remained visible for 1200 ms. Subsequently, a black screen was displayed for 1200 ms beginning 600 ms prior to the EFE/CB trial. Each test was followed by another black screen displayed for 600 ms before the subsequent trial. Participants were required to indicate the sequence in the CB test and identify the EFE, always in this order. In each trial, the order of the EFE and the position (left or right) of the EFE and CB were randomized.
The EFE identification concurrent with a semantic task (EFE/BDR)
As in the EFE/CB DT, each EFE was displayed simultaneously with a BDR trial for 1200 ms. After a black screen was displayed for 600 ms, the participants were required to first indicate the sequence of the BDR and to then identify the EFE, always in this order.
Procedure
Before beginning the experiment, the aims of the study were explained to each participant and written informed consent was obtained. Participants were informed that they were free to terminate their participation in the study at any point, although this did not occur. The protocols for research involving human participants were approved by the Ethical Committee of the University (UNED).
The trials were split into 2 different sessions. In the first session, participants performed the single tasks and they were assessed using neuropsychological scales. The DT trials were performed in the second session and the order in which the tasks were presented was counterbalanced to avoid learning effects. All participants were tested individually in a quiet room and the experiment was conducted using a 17-inch color display. Stimulus presentation and data recording was controlled by E-prime software developed for this experiment, and instructions were given verbally and presented onscreen before each test condition. All responses were recorded by the experimenter.
Experimental Design
The experiment was of a mixed factorial design with 2 × 2 factors: group, AD patients and controls; condition, EFE identification concurrent with a visuospatial task and EFE identification concurrent with a semantic task. The within-subjects variable was “condition” and the between-subjects variable was “group.” The dependent variable was the number of correct responses provided for all EFEs.
Results
The EFE Identification Tasks
A primary analysis compared the accuracy of patients with AD and control participants in EFE identification in the 3e conditions tested. The number of EFEs correctly identified revealed that patients with AD performed worse than healthy controls in the simple EFE identification task (12.27 vs 15.29, respectively), as well as when EFE identification was performed concurrent with a visuospatial task (9.47 vs 12.83, respectively) or a semantic task (10.07 vs 13.43, respectively; Figure 1).
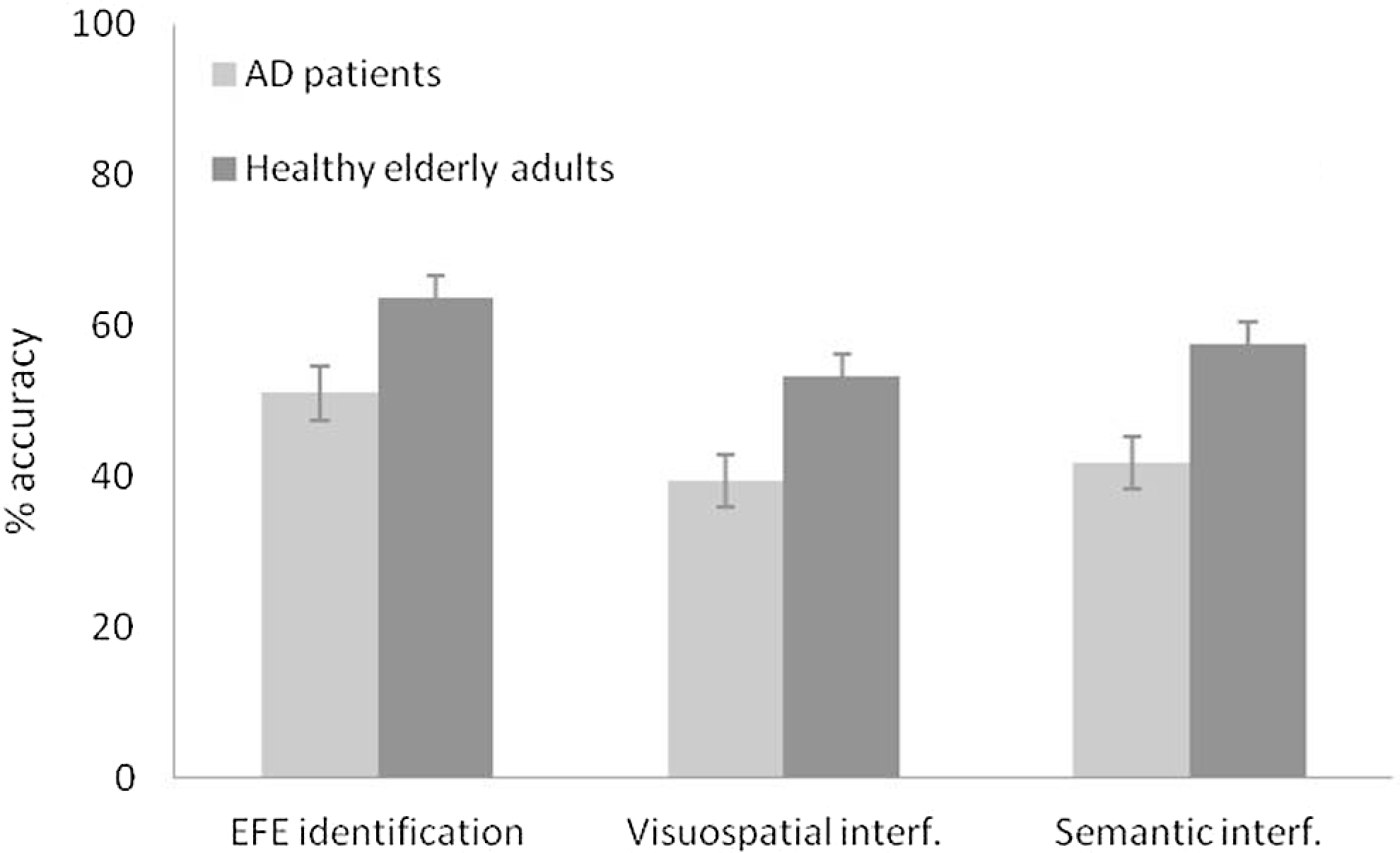
Accuracy of EFE identification (%) by patients with AD and control participants in the 3 experimental conditions. EFE indicates emotional facial expression; AD, Alzheimer’s disease.
Correct responses in each of the 3 conditions were analyzed by multivariate analysis of variance (MANOVA), which revealed significant differences in the single EFE identification task, F (1, 48) = 6.48, MSE = 95.70, P < .050, ηp 2 = .11, in EFE identification concurrent with a visuospatial task, F (1, 48) = 13.98, MSE = 118.67, P = .001, ηp 2 = .22, and in EFE identification concurrent with a semantic task, F (1, 48) = 8.28, MSE = 118.67, P < .050, ηp 2 = .15. Thus, the performance of patients with AD was significantly poorer than that of the control group in all conditions.
Visuospatial Task
The number of correct responses in the CB task was lower for patients with AD than their control counterparts in both the simple CB task (18.73 vs 23.51, respectively) and the CB DT (7.27 vs 14.51, respectively). The performance of patients with AD was significantly poorer than that of the control group in both conditions, and MANOVA revealed significant differences for the single CB task, F (1, 49) = 27.32, MSE = 240.00, P = .000, ηp 2 = .36, and CB DT, F (1, 49) = 26.64, MSE = 551.54, P = .000, ηp 2 = .35.
Semantic Task
Fewer correct responses were obtained from the patients with AD than the control group in the simple BDR task (23.97 vs 24, respectively), yet in the BDR DT (21.93 vs 23.69, respectively), the performance of the AD group was significantly poorer than that of the control group in the DT condition. Indeed, MANOVA revealed no significant differences in the single BDR task, F (1, 49) = .42, MSE = .009, P > .050, ηp 2 = .00, but a significant worsening in the performance of patients with AD in the BDR DT condition, F (1, 49) = 8.97, MSE = 32.24, P < .005, ηp 2 = .15.
Dual Task Performance
To determine whether the differences in DT performance can be attributed to effects already present at baseline or whether they are due to combining the tasks, and to account for the various strategies adopted when simultaneously performing 2 tasks, a formula has been proposed to calculate an overall decrement score: μ = (1 − [(Pm + Pt)/2])*100, where μ is the combined DT score, Pm is the proportional decrease in correct EFE responses between single (Xsingle) and DT (Xdual) conditions [(Xsingle − Xdual)/Xsingle], and Pt is the equivalent proportional decrease in the scores of the visuospatial task (CB) or the semantic task (BDR) task. 48 Thus, a score of 100% indicates no change in the DT score, while lower scores reflect a decrease in this value. We calculated 2 new variables, for the DT combining EFE identification and a visuospatial task (EFE/CB), and for EFE identification combined with a semantic task (EFE/BDR). Analysis of these variables by MANOVA revealed larger decrement scores in the EFE/CB, F (1, 49) = 7.77, MSE = 5709.72, P < .005, ηp 2 = .14, than the EFE/BDR combination, F (1, 49) = 4.00, MSE = 850.51, P > .050, ηp 2 = .08, with significant differences between the 2 groups only in the former. When the EFE task was performed in conjunction with the CB task, the observed decrease in accuracy was greater in the AD group (47.44%) than in the control group (70.76%). However, when EFE identification was performed in parallel with the BDR task, similar decreases in accuracy were observed in AD and control groups (86.58% and 95.58%, respectively; Figure 2).
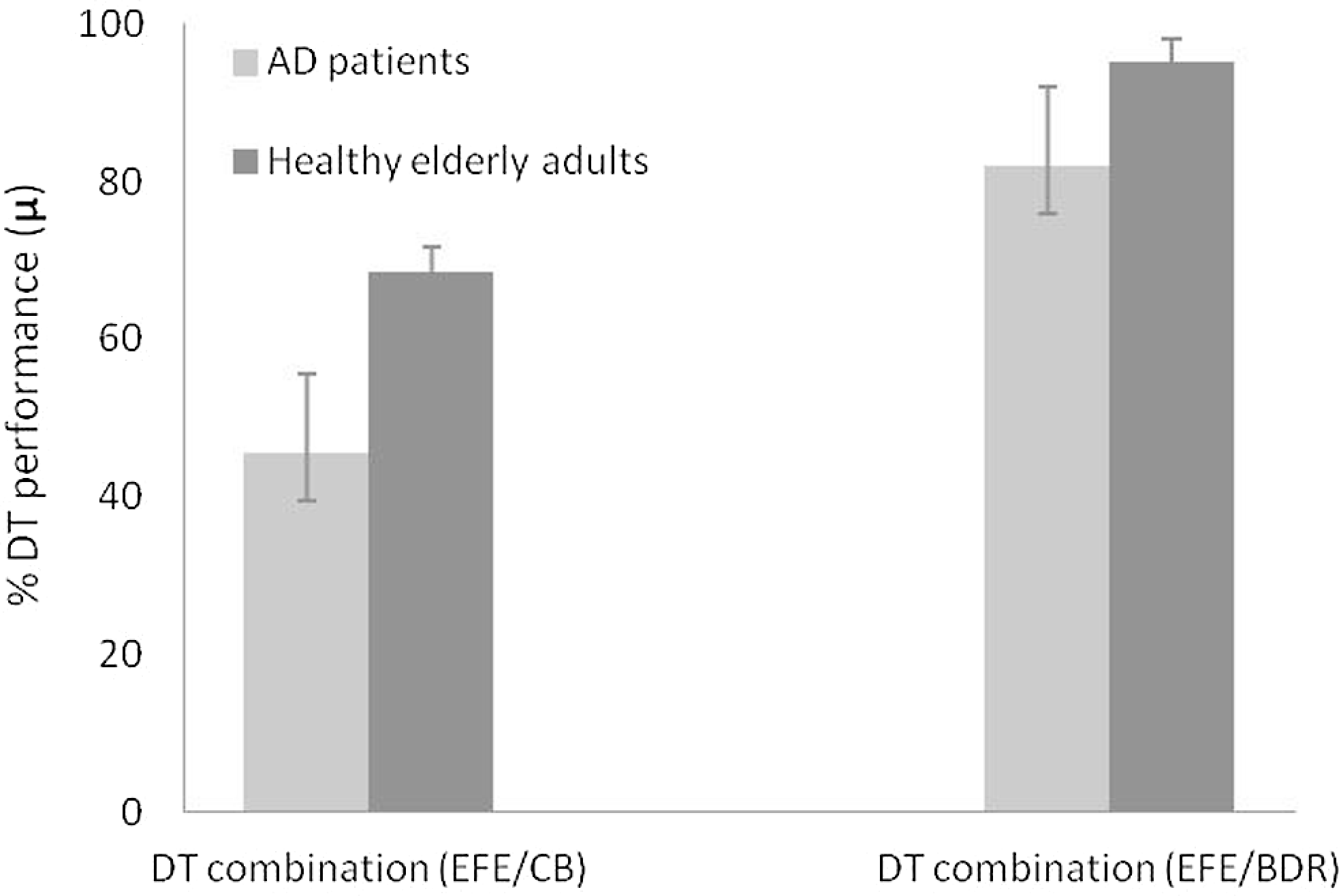
Performance (%) for each DT combination of the AD and control groups. DT indicates dual task; AD, Alzheimer’s disease.
In summary, our results show that EFE identification is impaired in patients with AD as compared with controls, and that the performance of these patients decreases significantly and disproportionately when tested while concurrently performing a visuospatial but not a semantic task. These results suggest that patients with AD have more difficulty integrating information relating to affective states with other environmental stimuli, leading to difficulties in interpersonal communication.
Discussion
The aim of the present study was to assess the role of context stimuli in the interpretation of EFEs by patients with AD. We assessed the capacity of both groups to identify EFEs while simultaneously processing other stimuli that occur during interpersonal communication. The performance of the AD group was significantly poorer than that of the control group in all experimental conditions, EFE identification both alone and in the DT conditions. These findings demonstrate that patients with AD have difficulties identifying EFEs even when not processing additional information. In agreement with previous reports, 5,49 these findings suggest that the specific neurodegeneration associated with AD may provoke such deficits, that is pathological alterations in the neural substrates underlying emotion processing impair EFE recognition.
Analyzing EFE identification in DT conditions revealed that as predicted, patients with AD exhibited functional deficits in identifying emotional faces while simultaneously processing other stimuli. Moreover, when facial emotional processing was performed in parallel with another visuospatial task, competition for the same resources resulted in greater DT decrements than when the concurrent task was of semantic nature. This effect was especially evident in the AD patient group, suggesting that emotional processing is impaired in patients with AD when performed in parallel with a task of the same nature. This is probably an important factor in the social communication deficits described in patients with AD.
The present findings corroborate those of our previous studies, 33 -36 demonstrating that neurodegenerative diseases are associated with deficits in emotional processing in complex stimuli contexts, particularly when processing 2 tasks of the same nature. These results challenge the proposed automaticity of emotional processing, 50 and show that cognitive resources are required to interpret the emotional content of stimuli, suggesting that emotional identification is a nonautomatic process dependent on cognitive state. The basic assumption of the DT paradigm is that automatic tasks are unaffected by simultaneous execution of another task. In AD patients, EFE identification and visuospatial processing were significantly impaired when performed concurrently, indicating that both are controlled processes that are dependent on normal cognitive resources. Moreover, in line with previous studies, 24 -26 we show that attentional control of executive function declines in the early stages of AD and that the performance of 2 simultaneous tasks is markedly impaired, suggesting specific DT-processing deficits.
A key aspect of the present study is the use of experimental tasks with significant ecological validity. The facial emotional processing system is constantly activated and ready, 20 and one of the key functions of this system is to accept or reject other messages, which involves the simultaneous processing of information from more than one source. This study identifies some of the specific challenges experienced by patients with AD when attempting to integrate affective information with normal cognitive activity.
The present findings represent an important contribution to our understanding of deficits in facial emotional recognition in with AD. Communication in everyday life depends on correct perception and interpretation of different emotional and nonemotional cues in a social context. Accurate interpretation of these cues plays an important role in maintaining successful relationships and healthy psychological function. The difficulties of AD patients in recognizing facial expressions may to contribute to their impaired social behavior, resulting in poor communications and interpersonal relations, or inappropriate social behavior. Furthermore, EFE recognition may represent an important indicator of well-being and quality of life in these patients.
Footnotes
Acknowledgments
This research was funded by a grant from the Ministerio de Ciencia e Innovación, Spain (Ref: PSI2009-13598-C02-01, Programa PSIC) awarded to Beatriz García-Rodríguez.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
