Abstract
Dementia with Lewy bodies (DLB) is commonly associated with excessive daytime somnolence (EDS). Modafinil is a wakefulness-promoting agent that is considered to have limited interaction with the dopaminergic system. As individuals with DLB are predisposed to psychotic symptoms that might be exacerbated by dopaminergic stimulation, modafinil is considered to be an attractive option for the treatment of EDS in DLB. We describe 2 cases in which administration of modafinil exacerbated agitation and hallucinations in DLB, and we also review data that may explain the mechanisms underlying this effect. In both cases, psychotic symptoms emerged concomitantly with modafinil administration and remitted following its discontinuation. Although definitive data regarding the benefits and adverse effects of modafinil for the treatment of EDS in DLB await controlled prospective randomized studies, our observations warrant caution regarding its use in this context.
Introduction
Dementia with Lewy bodies, or DLB, is the second most common cause of neurodegenerative dementia. 1 In addition to parkinsonism and visual hallucinations, marked fluctuations in cognition and arousal characterize DLB. Rapid eye moment behavior disorder (RBD) and excessive daytime somnolence (EDS) are among the alterations in arousal that occur in DLB. Excessive daytime somnolence is also common in Parkinson’s disease (PD), and various medications have been tried for its treatment. Traditional psychostimulants can be effective, but they can also precipitate hallucinations and other psychotic symptoms, likely due to their dopaminergic effects. 2
Modafinil (2-[(diphenylmethyl)sulfinyl] acetamide) is a wakefulness-promoting agent that is currently approved by the US Food and Drug Administration to treat EDS in association with narcolepsy, shift work sleep disorder, and obstructive sleep apnea/hypopnea syndrome. 3 Modafinil may also be efficacious for a variety of off-label indications, including treatment-resistant depression, attention-deficit/hyperactivity disorder, and schizophrenia. Another emerging target for modafinil is cognitive dysfunction, as evidence has indicated that it can improve working memory, episodic memory, and processes requiring cognitive control. 4 Modafinil has also been used for EDS in PD because it is thought to have limited interaction with the dopaminergic system and, therefore, reduced risk of exacerbation of behavioral symptoms. Because individuals with DLB are also predisposed to psychotic symptoms, modafinil is an attractive option for EDS in DLB as well. 5 In an open-label study, 5 of 8 patients with various forms of dementia showed improvement in EDS symptoms at doses of 100 to 200 mg/d of modafinil. 6 The effect of modafinil for the treatment of sleep disturbance in DLB has not, to our knowledge, been documented. In this report, we describe 2 cases that suggest the administration of modafinil may exacerbate agitation and hallucinations in DLB, and we review data that may explain the mechanisms underlying this effect.
Case Reports
Case 1
The first participant was an 80-year-old Caucasian male with 12 years of education and a history of benign prostatic hyperplasia and noninsulin-dependent diabetes mellitus who had a history of progressive cognitive decline since the age of 76. His symptoms began with the insidious onset and progression of memory deficits, hypersomnolence, and visual hallucinations. He was initially diagnosed with Alzheimer’s disease (AD) by an outside physician at the age of 78. He was treated with donepezil at 5 mg but when the dose was increased to 10 mg he was thought to have worsened confusion. The dose was, therefore, decreased and maintained at 5 mg/d. Olanzapine was added for hallucinations and titrated upward to 7.5 mg/d with improvement in his hallucinations without substantial worsening in his motor function. Memantine was added and titrated upward to 10 mg twice per day. At the age of 79, he was placed on modafinil 100 mg/d for hypersomnolence. Two days later, the patient’s hallucinations worsened, and the dose of modafinil was decreased to 50 mg; there was a gradual reduction in the psychotic symptoms during the following week. When initially seen by our group at age 80, the patient was described as having persisting hypersomnolence, intermittent confusion, and episodes of physical hyperactivity and talking in his sleep consistent with RBD. On examination, he had a Mini-Mental Status Examination (MMSE) score of 20/30 and was found to have hypomimia and bradykinesia, and DLB was diagnosed. Two months later, modafinil was discontinued because of ongoing hallucinations; improvement was observed 1 week after discontinuation. Three months later, a single dose of 200 mg modafinil was administered by the caregiver so that the patient would be awake for a specific social event. He became agitated, displayed an increased pathological obsession with specific events of the past, and experienced more intense visual hallucinations. These symptoms gradually improved over the following week.
Case 2
A 70-year-old Caucasian male Holocaust survivor with 12 years of education began developing memory problems, increased irritability, and agitation simultaneously. He underwent a routine evaluation at an outside institution, including an magnetic resonance imaging that was interpreted as showing discrete global atrophy. A diagnosis of dementia was made and treated with donepezil instituted at 10 mg/d. Two years later, treatment with memantine was initiated, the dose titrated to 10 mg twice a day, and the patient displayed improved memory and behavior. He was initially evaluated at our center at age 74, by which time he had begun having visual hallucinations of people and delusions, including the idea that his wife was being persecuted by “hundreds of people.” The patient’s wife described him as having significant behavioral fluctuations. A long-standing history of limb thrashing during sleep in a manner consistent with RBD was initially documented at this time, and the patient’s physical examination revealed the presence of mild cogwheel rigidity of the upper extremities bilaterally. His MMSE score was 13/30, and a diagnosis of probable DLB was made. The patient also demonstrated EDS, and, consequently, an outside physician initiated treatment with modafinil. The exact dose and duration of the patient’s treatment is unknown, but the patient’s wife indicated that he soon began having symptoms of agitated mania and sleeplessness, was abusive and loud, and destroyed things in his house. His condition improved markedly with discontinuation of modafinil.
Discussion
We report 2 patients with clinically diagnosed DLB who had marked behavioral worsening on initiation of therapy with modafinil for EDS. Excessive daytime somnolence is a common and significant problem in DLB, and its pharmacologic treatment is challenging due to the potential of most psychostimulant medications to worsen behavioral symptoms.
Dementia with Lewy bodies results from the accumulation of intracellular aggregates of α-synuclein, secondary cellular injury, and apoptotic neurodegeneration. 7 Because DLB and dementia associated with idiopathic PD (PD dementia or PDD) have many overlapping features, the degree to which they represent distinct diseases is a matter of controversy. 8 These diseases are distinguished clinically by the time course of symptom onset: DLB begins with dementia which is complicated by visual hallucinations and parkinsonism, while PDD begins with the motor symptoms of PD and progresses over at least 1 year to also feature cognitive impairment. 9 Although some differences in the nature and distribution of pathology in PDD and DLB exist, there is also substantial overlap between the 2 diseases. Considering the clinical and neuropathological overlap between PD and DLB, we can extend the broader understanding of PD psychosis to explain the mechanism of psychosis in DLB. 10
Psychotic symptoms in PD and DLB result from a complex interplay of extrinsic and disease-related (intrinsic) factors. 11 Intrinsic factors include visual processing deficits, neurochemical and structural abnormalities involving site-specific Lewy body deposition, genetic influences, and sleep dysregulation. 12 Extrinsic factors include the use of dopaminergic-stimulating medications and cholinergic inhibition. 13 Visual hallucinations in PD and DLB may be related to the degeneration of the dopaminergic pathways and high-density aggregation of α-synuclein in the temporal lobe. 10 , 14 Cholinergic deficits and Lewy body pathology in acetylcholine-producing neurons in the brain are also associated with the occurrence of psychosis in both disorders. 15 Many studies demonstrate that nigrostriatal dopamine denervation, caused either by the neurodegenerative process or by selective lesioning, results in a significant compensatory upregulation of striatal dopamine receptors (denervation supersensitivity). The resulting dopaminergic overactivity due to stimulation of supersensitive receptors by replacement therapy may provide a favorable environment for the development of psychosis. 13 The dopaminergic model of psychosis has been expanded to include a cascade of activations and inhibitions, a serotoninergic–dopaminergic imbalance, and a monoaminergic–cholinergic imbalance. 16 Nevertheless, alterations in glutaminergic systems have also been observed in DLB, thus making it difficult to determine the underlying mechanism for any given cognitive or behavioral symptom. 17
To minimize the likelihood of exacerbating agitation or psychosis in DLB, a nondopaminergic drug for the treatment of EDS would be optimal. Modafinil is thought to have a different mechanism of action than amphetamine, and its use has become widespread because of a decreased risk for abuse and a lower risk of cardiovascular effects. Mignot and colleagues 18 and Simon and colleagues 19 published studies demonstrating a lack of action on dopamine by modafinil. Subsequent research has suggested that modafinil’s effects in the brain involve hypocretin, histamine, epinephrine, gamma-aminobutyric acid, and glutamate. 20,21 Modafinil has been shown to directly bind to the dopamine transporter (DAT) and to the norepinephrine transporter. 22 -24 Modafinil administration also leads to significantly elevated extracellular dopamine, norepinephrine, serotonin, glutamate, and histamine levels, and to decreased gamma-aminobutyric acid levels. 25 These effects are particularly prominent in the neocortex and are generally less potent or minimal in subcortical areas. The effects of modafinil on dopamine and norepinephrine appear to be primary; the effects on serotonin, gamma-aminobutyric acid, glutamate, orexin, and histamine may be secondary to catecholamine effects. 3,26 Consistent with this hypothesis, mice lacking DATs or D1/D2 receptors demonstrated no response to the arousal effects of modafinil. 27,28 Finally, Volkow and colleagues 29 showed that at clinically relevant doses, modafinil significantly increases dopamine in the human brain by blocking DATs. These authors propose a reconsideration of the hypothesis of the nondopamine mechanism of action of modafinil. Thus, there is a lack of consensus about the precise mechanism of action of modafinil although dopaminergic effects appear to be present. We argue that the recently demonstrated dopaminergic action of modafinil is associated with psychosis and agitation exacerbation in DLB. Probably, the dopamine supersensitivity observed in DLB promotes a brain environment with a high liability for psychosis and agitation, and a propensity for dopaminergic overactivity with modafinil use.
Even though the safety profile and tolerance of modafinil are generally good, precipitation or exacerbation of psychosis has been reported. The manufacturer’s prescribing information 2 stated that one healthy male had psychosis and sleep deprivation in association with multiple daily doses of modafinil at 600 mg. Another patient with schizophrenia was reported 30 to have an exacerbation of psychosis after taking 100 mg of modafinil for 4 days. Four more case reports are shown in Table 1. 31 - 34 As shown, the exacerbations of psychosis in the first 3 cases were directly influenced by modafinil because the onset of psychosis was temporally related to medication changes. The psychosis in case 4 might have resulted from the combination of modafinil and caffeine rather than either alone. Muscle twitches, hypertension, tachycardia, agitation, and psychosis induced by caffeine usually result from consumption of greater than 1 g. 35 This patient also took 500 mg of modafinil, which is a higher dose than is typical. Modafinil and caffeine may have synergistic effects, leading to psychosis in this patient.
Case Reports of Psychosis Induced by Modafinil
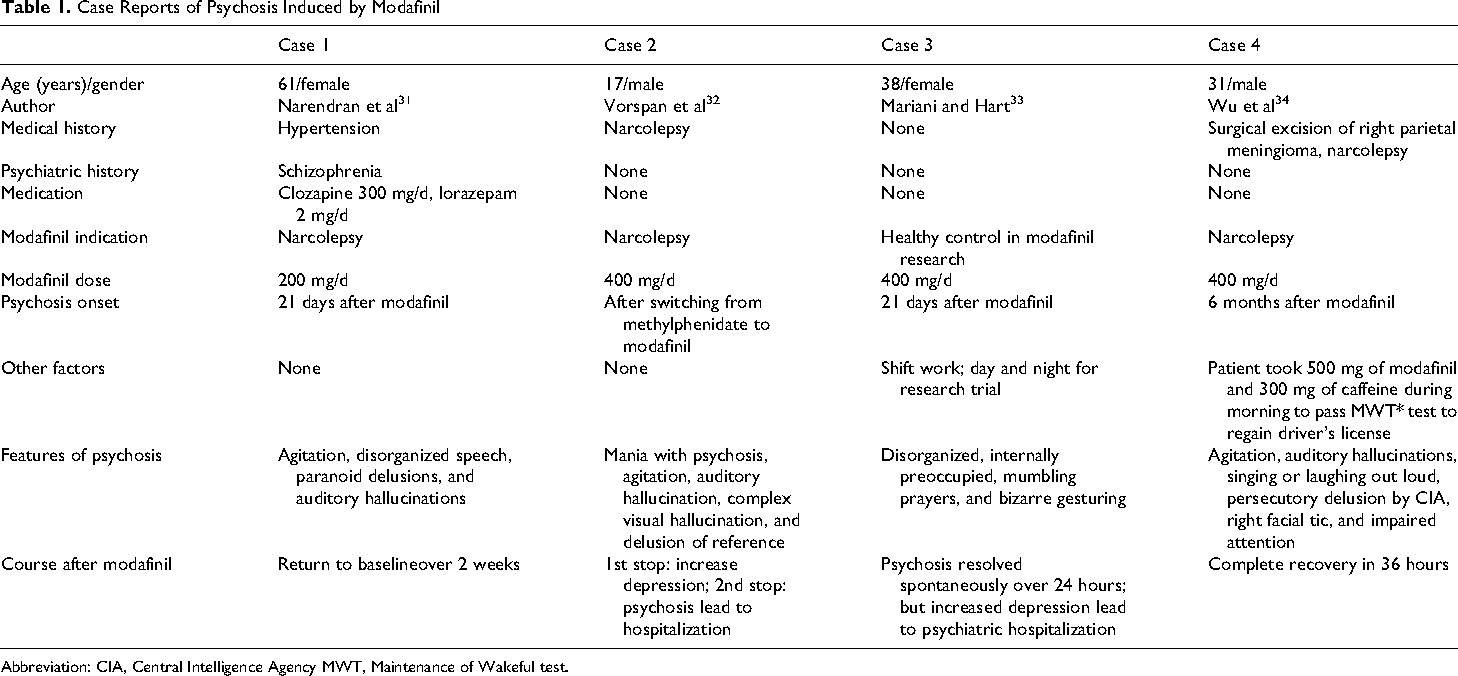
Abbreviation: CIA, Central Intelligence Agency MWT, Maintenance of Wakeful test.
Overall, modafinil is well tolerated, with mild-to-moderate side effects, and there is growing evidence for its utility in EDS in other neurological diseases. It has been used for pramipexole-associated somnolence in PD followed by EDS in PD itself. 36 , 37 Subsequently, several small studies showed a significant reduction in EDS at doses of 100 to 200 mg/d of modafinil, with few side effects. 38–42 On the other hand, a larger, randomized, double-blind, placebo-controlled study (N = 37) failed to show the superiority of modafinil to placebo. 43 Evidence regarding the efficacy of modafinil for EDS in PD is therefore equivocal. Although a small open-label study found some evidence regarding the efficacy of modafinil for EDS in dementia, evidence of its efficacy in DLB is lacking.
Conclusion
We report 2 patients with DLB who demonstrated disabling agitation and hallucinations in association with the initiation of treatment with modafinil for EDS. Considering the complexity of neurotransmitter alterations in DLB and the varied neurochemical effects of modafinil, the exact mechanism underlying the behavioral changes we observed cannot be established with certainty, but we cannot discount the possibility that stimulation of the dopaminergic system is involved. Although definitive data regarding the potential benefits and risks of modafinil and related medications for the treatment of EDS in DLB await randomized controlled trials, our observations warrant caution in the use of modafinil in DLB.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Sidell Kagan Foundation, the Shirley and Jack Goldberg Trust, the Alzheimer’s Disease Research Center Grant P50 AG-16570 from the National Institute on Aging, a California Alzheimer’s Disease Center grant, and the Easton Consortium for Alzheimer's Disease Drug Discovery and Biomarkers. Further support for this study came from the John Douglas French Foundation and the Institute for the Study of Aging.
