Abstract
Keywords
Introduction
Alzheimer’s disease (AD) is the most common neurodegenerative disease presenting with dementia. In most of the cases, the initial symptom is a mild cognitive impairment, caused by an increasing number of neurofibrillary lesions in the cortical transentorhinal region, the hippocampus, and in the limbic areas. As the disease advances, these lesions spread through the cerebral cortex. 1 –3 Neuropathologically, AD is characterized by the presence of β-amyloid (Aβ) extracellular deposits in neuritic plaques, and neurofibrillary alterations that include intracellular deposits of hyperphosphorilated tau protein in neurofibrillary tangles, dystrophic neurites, and neuropile threads. 4
Frontotemporal lobar degeneration (FTLD), formerly known as Pick’s disease, constitutes 5% of the cases of dementia in autopsy series. 5 Frontotemporal lobar degeneration include a group of heterogeneous neurodegenerative diseases, both regarding clinical presentation and histopathologic and genetic characteristics. 6,7 Clinical manifestations appear insidiously, and with the evolution of the disease, diverse characteristics of the personality deteriorate, depending on which prefrontal areas are affected. 8,9 Neuropathologically, the brain shows atrophy of frontal and temporal lobes, and abnormal protein aggregates in the neurons or the glia. 10
Management of AD involves preventing vascular risk factors and symptomatic treatment with acetylcholinesterase inhibitors (AChEIs) and memantine, an uncompetitive N-methyl-
Despite the evidence-based clinical practices guidelines, clinical experience suggests that a high percentage of patients with FTLD are treated with AChEI and/or memantine. The main aim of this study was to describe the use of central nervous system (CNS) drugs in patients with AD and FTLD and to detect the main variables associated with the use of AChEI and memantine in patients with FTLD. Our study used the data collected from 2007 to 2009 by an epidemiological surveillance system focused on dementia. We combined this information with the information obtained from a drug prescription database.
Methods
Design, Geographical Area of Reference, and Study Population
This study used a cross-sectional design and was based on data from the cases registered by Registry of Dementias of Girona (ReDeGi) from 2007 to 2009, which was approved by the Review Board of the Institut d’Assistència Sanitària. The ReDeGi registers demographic and clinical data of all the new dementia cases diagnosed in the hospitals of the public health care system of the Health Region of Girona (HRG). The HRG is located in the North-East of Catalunya, which is, in turn, in the North-East part of Spain. It has an area of 5517 km2, a population of 737 344 inhabitants (according to the Citizens Municipal Registry 2010), and a population density of 133.6 inhabitants/km2.
Registry Procedure and Registered Variables
The ReDeGi registers all the incident cases of dementia diagnosed in the 7 hospitals belonging to the HRG, using standardized criteria for case definition, and following the guidelines proposed by the Center for Disease Control and Prevention for a surveillance system. 14 The methodological principles and the functional structure of the ReDeGi have been previously described. 15 –17 All diagnoses are based on the medical chart, an interview with the patient and the caregiver, a general medical examination, hematology and blood chemistry tests, and neuroimaging diagnosis if required. A specialist technician of the ReDeGi periodically reviews the medical chart of the cases of dementia notified in each of the 7 hospitals of the HRG and registers the information in a clinical research form containing (1) center identification, date of admission, and clinical history identification number; (2) sociodemographical characteristics; (3) characteristics of the diagnosis based on the Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition), 18 date of onset of the symptoms, and date of diagnosis; (4) clinical characteristics (date of administration and score of the Mini-Mental State Examination, 19 the Blessed Dementia Rating Scale, 20 and the Clinical Dementia Rating (CDR), 21 family history of dementia, diagnosis of hypertension, diabetes mellitus, dyslipidemia, cerebrovascular adverse events, thyroid disease, depression history, or personal history of psychosis. The collected information of the ReDeGi meets the confidentiality requirements stated by the Spanish legislation.
Drug Consumption Information
The ReDeGi database was linked with the HRG Pharmacy Unit database from the Public Catalan Healthcare Service (PCHS). This database contains all the drugs prescribed by the PCHS professionals that have been dispensed in pharmacies. Patient records were linked using the personal identification code included in their medical chart. All drugs were provided by the PCHS database at 5th level Anatomical Therapeutic Chemical classification system, and benzodiazepines were grouped according to their half-life (short-acting, intermediate-acting, and long-acting).
Data Analysis
A descriptive analysis of the variables was performed using central tendency measures and dispersion for quantitative variables, whereas absolute and relative frequencies were calculated for qualitative variables. The clinical and demographic characteristics between patients with AD and FTLD, and their drug consumption patterns were compared using χ 2 tests for categorical variables and the Mann-Whitney U test for continuous variables. Normality was checked using Shapiro-Wilk test. Binary logistic regression models were performed to detect the main variables related to AChEI and memantine use in patients with FTLD and AD. Results are expressed as absolute numbers and percentages, means, standard deviations, odd ratios, and 95% confidence intervals (CIs). Statistical tests were considered to be significant with a 2-tailed P value <.05. Processing and analysis of the data were performed using the statistical package SPSS v15.0 for Windows (SPSS, Inc, Chicago, Illinois).
Results
During the period 2007 to 2009, 1894 dementia cases were registered by the ReDeGi in the HRG, of which 57.7% (n = 1092) had AD and 3.4% (n = 64) had FTLD. Of the latter, 79.7% (n = 51) corresponded to the behavioral variant of FTLD, whereas the rest (20.3%, n = 13) were primary progressive aphasia.
The mean age of the patients was 79.4 (standard deviation = 6.9) years, and 62.6% (95% CI: 59.8-65.5) were women. Only 6.8% (95% CI: 5.0-8.0) lived in long-term care residences, and presenile forms accounted for 6.1% (95% CI = 4.6-7.5) of the cases. Regarding severity, 66.9% (95% CI: 64.1-69.6) of the cases were mild (CDR = 1), 24.3% (95% CI: 21.8-26.9) were moderate (CDR = 2), and 8.8% (95% CI: 7.1-10.5) were severe (CDR = 3).
Sociodemographic and clinical characteristics of patients with AD and FTLD are shown in Table 1.
Clinical and Sociodemographic Characteristics of the Patients With Alzheimer’s Disease and Frontotemporal Lobar Degeneration Registered in the Registry of Dementias of Girona
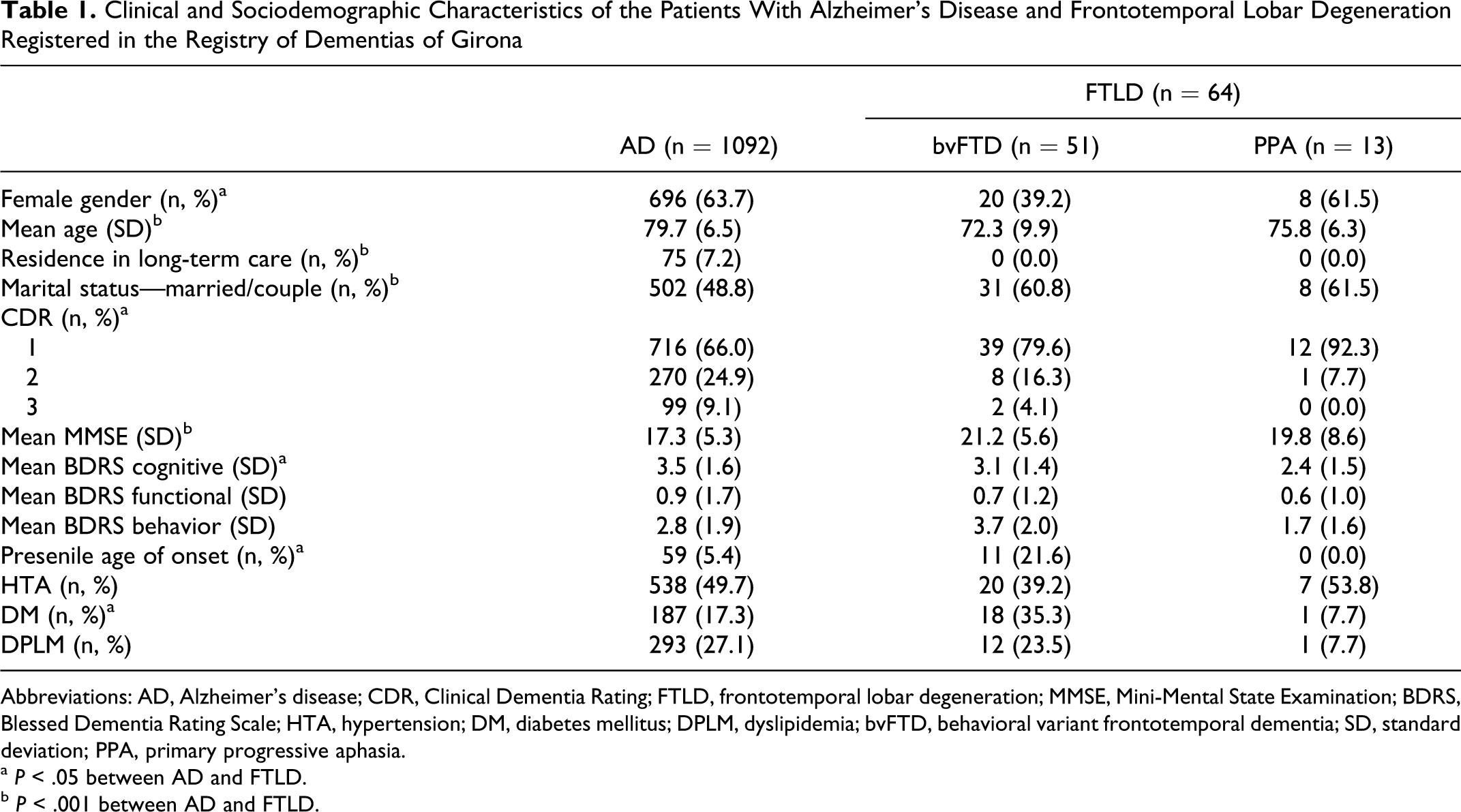
Abbreviations: AD, Alzheimer’s disease; CDR, Clinical Dementia Rating; FTLD, frontotemporal lobar degeneration; MMSE, Mini-Mental State Examination; BDRS, Blessed Dementia Rating Scale; HTA, hypertension; DM, diabetes mellitus; DPLM, dyslipidemia; bvFTD, behavioral variant frontotemporal dementia; SD, standard deviation; PPA, primary progressive aphasia.
a P < .05 between AD and FTLD.
b P < .001 between AD and FTLD.
Antidementia drugs were prescribed to 62.9% of the cases with AD and to 43.8% of the cases with FTLD. Specifically, 57.6% and 17.2% of the patients with AD were treated with AChEI and memantine, respectively. In turn, 42.2% and 10.9% of the patients with FTLD used AChEI and memantine, respectively (Table 2).
Central Nervous System Drugs Consumption by the Patients With Alzheimer’s Disease and Frontotemporal Lobar Degeneration Registered in the Registry of Dementias of Girona (ReDeGi)
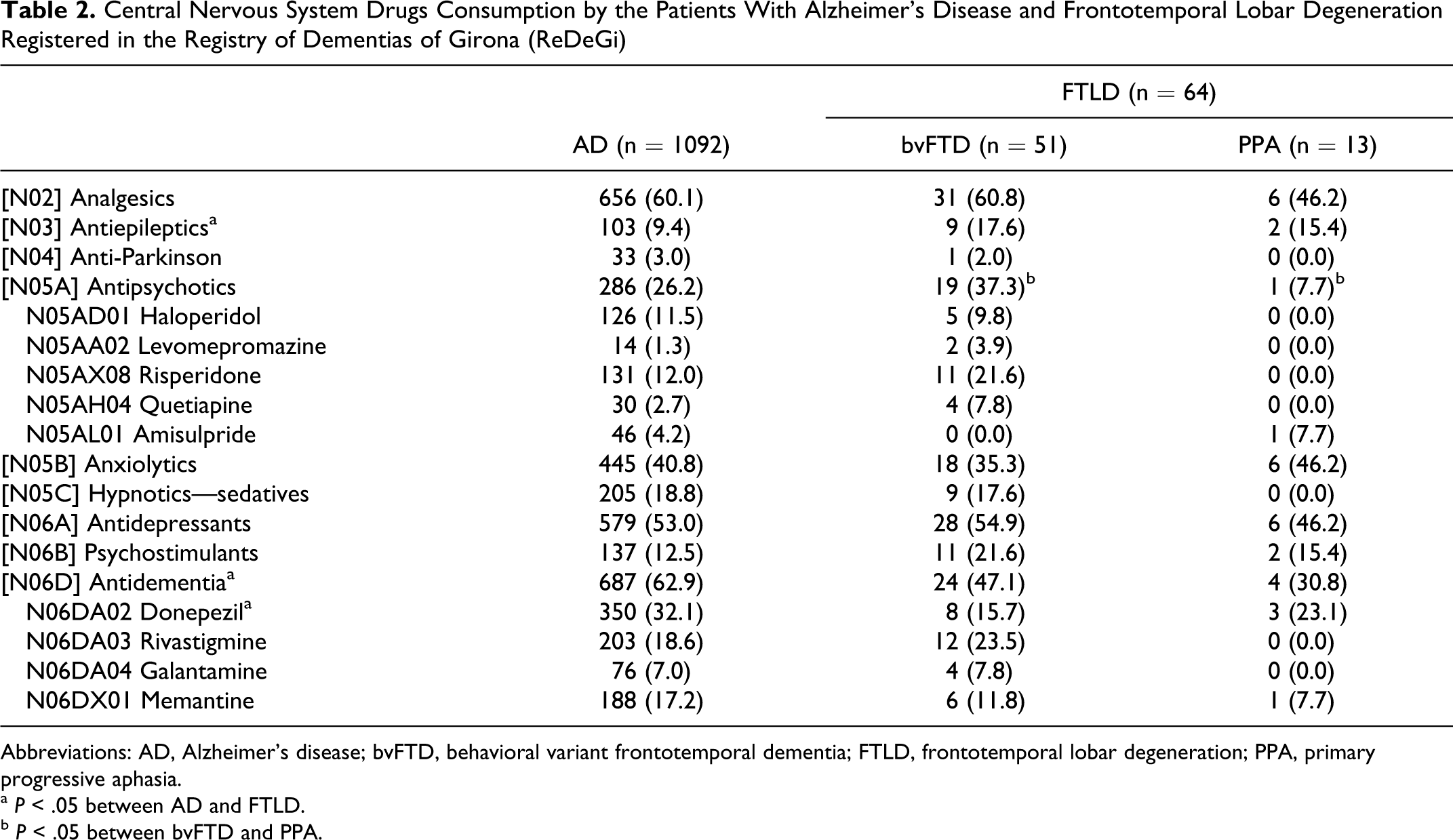
Abbreviations: AD, Alzheimer’s disease; bvFTD, behavioral variant frontotemporal dementia; FTLD, frontotemporal lobar degeneration; PPA, primary progressive aphasia.
a P < .05 between AD and FTLD.
b P < .05 between bvFTD and PPA.
Antipsychotics were used in 26.2% of the patients with AD and 31.3% of the patients with FTLD. The use of haloperidol, risperidone, and other antipsychotics with a frequency of use of 2% or more was analyzed and is shown in Table 2.
Anti-Parkinson drugs frequency of use was 3.0% in AD cases and 1.6% in FTLD. Psychostimulants were used by 12.5% of the patients with AD and 20.3% of patients with FTLD. In particular, psychostimulants included methylphenidate, dextroamphetamine, amphetamine, lisdexamfetamine, atomoxetine, modafinil, and armodafinil (Table 2). Anxiolytics were prescribed to 40.8% and 37.5% of the cases with AD and FTLD, respectively. Hypnotics and sedatives were used by 18.8% and 14.1% of the patients with AD and FTLD. Of all patients with AD, 37.4% used intermediate-acting benzodiazepines (alprazolam, bromazepam, ketazolam, lorazepam, lormetazepam, and flunitrazepam), 12.3% used long-acting compounds (diazepam, clobazam, clorazepate, and flurazepam), and only 0.6% consumed short-acting benzodiazepines (bentazepam, midazolam, and triazolam). The patients with FTLD used intermediate-acting benzodiazepines in 26.6% of the cases (alprazolam, bromazepam, ketazolam, lorazepam, lormetazepam, and flunitrazepam), long-acting (diazepam, clobazam, clorazepate, and flurazepam) in 23.4% of the cases, and no patient with FTLD consumed short-acting benzodiazepines (Table 3).
Consumption of Benzodiazepines by the Patients With Alzheimer’s Disease and Frontotemporal Lobar Degeneration Registered in the Registry of Dementias of Girona (ReDeGi)
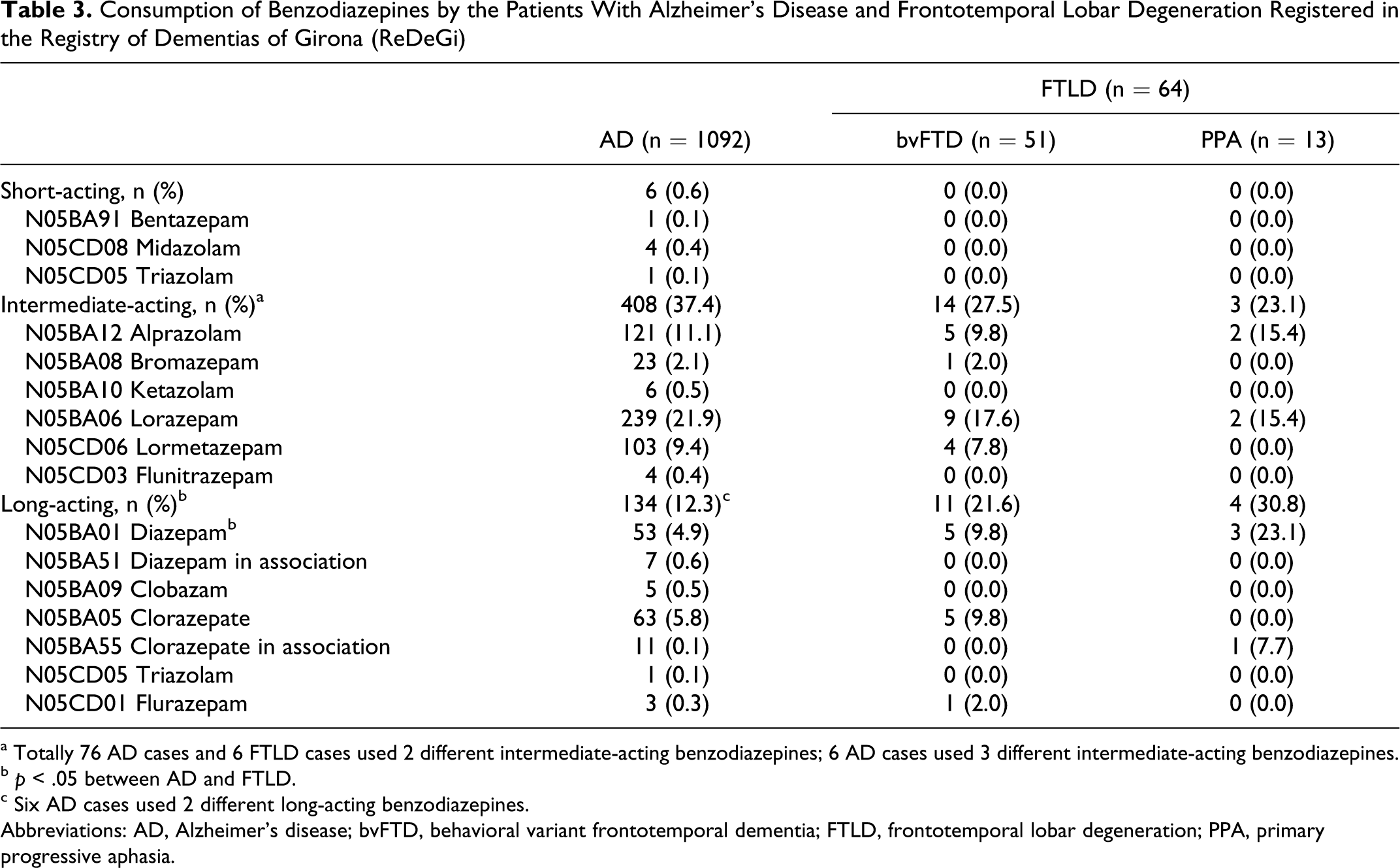
a Totally 76 AD cases and 6 FTLD cases used 2 different intermediate-acting benzodiazepines; 6 AD cases used 3 different intermediate-acting benzodiazepines.
b p < .05 between AD and FTLD.
c Six AD cases used 2 different long-acting benzodiazepines.
Abbreviations: AD, Alzheimer’s disease; bvFTD, behavioral variant frontotemporal dementia; FTLD, frontotemporal lobar degeneration; PPA, primary progressive aphasia.
Antidepressants were prescribed to 53.0% and 53.1% of the patients with AD and FTLD, respectively. Specifically, 36.6% AD cases, and 31.3% FTLD cases used selective serotonin reuptake inhibitors, and 20.0% and 23.4% used tetracyclic antidepressants, respectively (Table 4).
Consumption of Antidepressants by the Patients With Alzheimer’s Disease and Frontotemporal Lobar Degeneration Registered in the Registry of Dementias of Girona (ReDeGi)
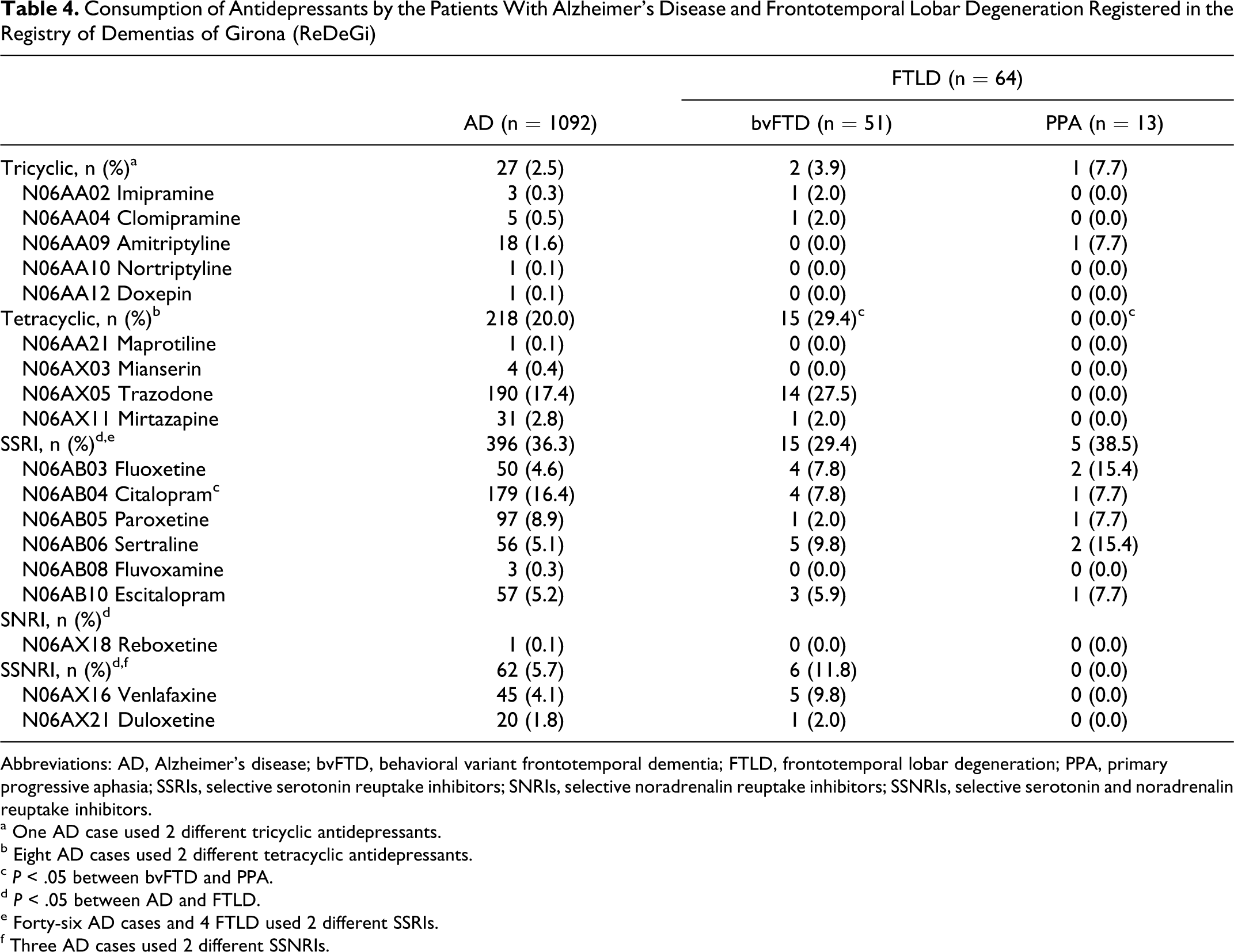
Abbreviations: AD, Alzheimer’s disease; bvFTD, behavioral variant frontotemporal dementia; FTLD, frontotemporal lobar degeneration; PPA, primary progressive aphasia; SSRIs, selective serotonin reuptake inhibitors; SNRIs, selective noradrenalin reuptake inhibitors; SSNRIs, selective serotonin and noradrenalin reuptake inhibitors.
a One AD case used 2 different tricyclic antidepressants.
b Eight AD cases used 2 different tetracyclic antidepressants.
c P < .05 between bvFTD and PPA.
d P < .05 between AD and FTLD.
e Forty-six AD cases and 4 FTLD used 2 different SSRIs.
f Three AD cases used 2 different SSNRIs.
Multiple regression models using all available sociodemographic and clinical variables were performed in order to detect any association with AChEI and memantine consumption, both in FTLD and AD. There were no variables related to antidementia drug consumption.
Discussion
Despite the lack of efficacy of antidementia drugs, and even though they are not indicated in the treatment of FTLD, according to our results, approximately half of the patients were prescribed this medication. Our study is in agreement with a population study performed in California, 22 which showed the same percentage of patients with AD and FTLD taking any antidementia drug (donepezil, galantamine, rivastigmine, or memantine). Nevertheless, in our population, patients with AD most frequently used donepezil, whereas FTLD cases used either donepezil or rivastigmine.
Moreover, unlike in the American study, our patients were more frequently treated with CNS drugs, specially anxiolytics (37.5% vs 6.8%), antidepressants (53.1% vs 43.2%), and antipsychotics (31.3% vs 4.5%).
In most of the cases, the usefulness of CNS drugs to help control behavioral and psychological symptoms in FTLD is controversial. While in some studies, fluvoxamine and paroxetine significantly improved behavior, results by other authors indicated poor benefit of fluoxetine, sertraline, and lithium carbonate. In several studies, the use of trazodone improved behavioral but not cognitive symptoms. Neuroleptics in general, and risperidone as the most common antipsychotic used, do not improve behavior or cognition. 23,24 Despite this, as much as 31.3% of the patients with DLFT were prescribed an antipsychotic, and of them, 55% used risperidone. This extensive use of neuroleptics should be seriously reconsidered, especially if taking into account their serious adverse effects such as cognitive impairment, Parkinsonism, and increased risk of stroke. 25 –27
The use of psychostimulants was low, both in our study and in the American population study by Bei Hu et al, 22 and both in AD and FTLD cases. However, in our population, 20.2% of the cases with FTLD were prescribed a psychostimulant. The most frequently used psychostimulant was methylphenidate, even when it does not improve cognitive symptoms, and the extent of its efficacy is uncertain. 23,28
No differences were detected between treated and not treated FTLD patients with antidementia drugs (AChEI and/or memantine). Age, sex, residence, education, risk factors, clinical history and clinical symptomatology, and other CNS drugs consumption were variables used in the multivariate analysis that yielded no associations. This leads us to think, in agreement with Bei Hu et al, 22 that maybe, these drugs are used only because of medical uncertainties in making a precise diagnosis in the first stages of the disease, and/or due to the need to offer a pharmacological treatment when the patient or the caregiver asks for it. Finally, it might also be due to a general tendency to medicate all patients with dementia.
Current knowledge clearly shows that patients with FTLD have deficiencies in dopaminergic and serotoninergic neurotransmission systems but have no alteration in acetylcholine system. 29 The low prevalence and high clinical complexity of FTLD hinder clinical trials, and the scarce studies performed with drugs authorized to treat AD, revealed no benefits when used in FTLD. 12,13,30 –33
Some limitations must be considered when interpreting the results obtained in this study. Information on the dosages prescribed would be required, as well as the duration of the treatment, to calculate defined daily doses for each drug. In addition, the lack of information on adherence, or treatment withdrawal, might influence the interpretation of the results. Finally, we do not have information on eventual comorbidities in the registered cases, and we cannot definitely assert that all CNS medications were used to treat dementia-related behaviors.
In summary, the existing discrepancy regarding clinical practice and the recommendations based upon clinical evidence warrant further multicentric studies aiming at finding the best means to treat patients with FTLD.
Footnotes
Acknowledgments
The ReDeGi is founded by the Health Region of Girona from the Department of Health of the Generalitat de Catalunya (Spain).
Co-investigators
Department of Neurology, Hospital Universitari Josep Trueta: Mar Castellanos, PhD; David Genís Batlle, MD; Jordi Gich, BPsych; Rosa Meléndez, MD; Albert Molins, MD; Lluís Ramió, MD; Joaquín Serena, PhD; Yolanda Silva, PhD. Dementia Unit, Hospital Santa Caterina: Marta Hernández, BPsych; Saioa Lejarreta, MD; Manuela Lozano, BPsych; Immaculada Pericot, PhD; Joan Vilalta-Franch, PhD; Antoni Turon Estrada, PhD; Josefa Turbau, MD; Jordi Llinás, BPsych; Secundino López-Pousa, PhD. Dementia Unit, Hospital de Palamós: Elisabet Alsina, BSw; Rosa de Eugenio, MD; Erélido Hernández, MD; Margarita Flaqué, BPsych. Neurology Department, Hospital de Figueres: Olga Carmona, MD; Marta Cullell, BPsych; Teresa Osuna, MD; Maria Aguirregomozcorta, MD; Ma del Mar Fernández, MD. Neurology and Geriatrics Departments, Hospital d’Olot: Josep Bisbe, MD; Marta Linares, BPsych; Fabian Marquez Daniel, MD; Natalia Vallmajó, MD. Neurology and Geriatrics Departments, Hospital de Blanes: Fernando Espada, MD; Teresa Casadevall, MD; Héctor Perkal, MD; Marta Viñas, MD. Neurology Department; Hospital de Campdevánol: Isabel Casas, BPsych; Josefa Turbau, MD; Josep Ma Cuy, MD; Anna Manzano, MD. Pharmacy Unit, Healh Region of Girona: Anna Maria Roig, BPharm; Joan Coromina, BPharm; Mónica Palacios Soto, BPharm.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
