Abstract
There is currently no consensus on the nosological position of apathy in clinical practice, although many different articles indicate that apathy is a common, behavioral disturbance in the general Parkinson’s disease (PD) population, often related to severe motor symptoms, hypothesizing that the dysfunction of the nigrostriatal pathway may play an important role in its pathophysiology. However, not all patients with PD become apathetic, indicating that apathy should not entirely be considered a dopamine-dependent syndrome in PD. The aim of this study was to examine the prevalence and clinical correlates of apathy in a representative community-based sample of patients within 2 variants of the PD: akinetic-rigid type and tremor-dominant type. Specifically, we wanted to investigate whether these 2 variants of PD would present with apathy as a primary behavioral disorder and whether apathy could be associated with different cognitive and behavioral disorders. Apathy is present in both the groups but significantly more evident in the akinetic-rigid group associated with frontal impairment but not related to motor impairment or depression. We discuss the results, starting with anatomical and physiological brain studies.
Keywords
Apathy is defined as lack of motivation as manifested by diminished goal-directed behavior, reduced goal-directed cognition, and decreased emotional engagement, 1 a reduced interest and participation in normal purposeful behavior, problems in initiation or sustaining an activity, lack of concern or indifference, and a flattening of affect. 2,3 The prevalence of apathy in Parkinson’s disease (PD) varies between 16.5% and 51%, 1 –6 depending upon the assessment instrument and on the samples examined.
There is currently no consensus on the nosological position of apathy in clinical practice. 7 The clinical significance of negative symptoms such as apathy is increasingly recognized in neurological and psychiatric disorders, particularly those associated with frontal–subcortical dysfunction. 8 Many different articles indicate that apathy is common and may occur as an independent behavioral disturbance in the general PD population. 2 –4,9 It has been hypothesized that there is a relationship between apathy and more severe motor symptoms in PD cohorts, supporting the hypothesis that dysfunction of the nigrostriatal pathway may play an important role in the pathophysiology of apathy in PD. 1 –7 In fact, apathy seems to be independent of disease duration, disability, and severity of parkinsonism and levodopa dose in PD, indicating that the brain changes underlying apathy differ from those associated with motor symptoms. 6 Much more interesting is that not all patients with PD become apathetic, indicating that apathy should not entirely be considered a dopamine-dependent syndrome in PD. 4 Existing evidence suggests that apathy can be related to depression, as it is a key symptom of major depression and also a side effect of antidepressant and antipsychotic drugs. 9,10 However, apathy and depression clearly dissociate in specific motor disorders, such as progressive supranuclear palsy, in which there is a high incidence of apathy but a low incidence of depression. 11 Other authors suggest that apathy might be a consequence of chronic disabling disease and its impact on mobility and opportunity for participation in normal activities. Thus, many authors use the term “premature social aging” to describe the findings that patients with PD, compared with otherwise healthy elderly individuals, have little in the way of interests or social activities, spending more time in solitary activities such as watching television or just sitting doing nothing. 5 If apathy is a primary consequence of physical disability, then similar changes might be predicted for patients with articular/orthopedic impairment. Surprisingly, the osteoarthritis sample population, despite the motor disability, showed no evidence of apathy. 5 It is thus likely that the physiopathology of apathy is a multifaceted entity. In line with this notion, Stuss et al 12 proposed dividing apathetic syndromes into 3 subtypes: “emotional,” “cognitive,” and “behavioral.” Levy and Dubois’ 9 observation of patients leads them to replace the term behavior with the concept of autoactivation deficit. This refers to a fundamental deficit of activation of behavior that is not primarily due to an emotional or a cognitive deficit and can be reversed by external stimulation (heteroactivation). This mechanism is associated with the most severe apathetic states. Each of the 3 groups can be ascribed to lesions of different prefrontal cortex (PFC) basal ganglia territories as follows: “emotional-affective” to the orbital–medial PFC and presumably to its connected region within the striatum, namely the ventral striatum, “cognitive” to the lateral PFC (and to its striatal input, namely the dorsal caudate nuclei) and “autoactivation” to basal ganglia lesions that usually affect both the cognitive and limbic territories (bilateral GPi [internal portion of the globus pallidus] or bilateral paramedian thalamic lesions) and also the dorsal–medial region of the PFC. 9 If a focal destruction within the basal ganglia subregions occurs, the signal deriving from the basal ganglia is diminished, the ongoing behavior is not validated (ie, not amplified) at the level of the cortex and could be difficult to maintain, and the forthcoming one (if it is not reflexive) is not activated. 9 In sum, an autoactivation deficit results from the inability of voluntary thoughts or actions to reach the activation threshold due to a decreased signal-to-noise ratio at the level of the PFC. 9
The aim of this study was to examine the prevalence and clinical correlates of apathy in a representative community-based sample of patients, with a diagnosis of PD, and to be more precise, with an akinetic-rigid type or a tremor-dominant type of PD. Specifically, we wanted to investigate whether 2 variants of PD patients would present with apathy as a primary behavioral disorder, and whether apathy could be associated with different cognitive and behavioral disorders; moreover, we sought to investigate whether apathy is more dominant in 1 of the 2 groups, in relation to their motor performances and/or cognitive results.
Study Design
The study included 103 patients (60 men and 43 women) having idiopathic PD 13 recruited from January 1, 2001 to July 31, 2010. Patients aged from 62 to 70 years.
Patients were divided in 2 groups: patients with PD of an akinetic-rigid type
14,15
(n = 56) and patients with tremor-dominant type PD (n = 47). Patients were evaluated in on-pharmacological states. All the patients responded to
Neuroimaging studies were assessed, including magnetic resonance imaging (in 49 patients, 32 in akinetic type group, and 17 in tremor-dominant type group) and computed tomography scans (in all the patients). Neither signs of normal pressure hydrocephalus nor ischemic infarctions were found in any patients.
Patients were visited by authors P.T. and R.M.A., who divided the patients between 2 groups: akinetic-rigid and tremor-dominant type of PD. After the first visit, the patients were scheduled and tested by the author R.M. who was blinded to the clinical group.
Patients were classified as akinetic-rigid-dominant type when the most salient aspects of the presentation were hypokinesia or bradikinesia, absence or poverty of automatic movements (we refer to this as hypokinesia), manifested by, for example, hypomimia with reduced blinking, or a reduced arm swing during walking (according Unified Parkinson's Disease Rating Scale (UPDRS) part III, a test widely used to assess motor alterations in PD). 14,15,17 Tremor-dominant type has been defined as a patient presenting with resting tremor, characterized by involuntary, rhythmic, and sinusoidal alternating movements of one or more body parts, with a variable amplitude (considering UPDRS part III). 14,15 –17 The study was conducted in accordance with the Declaration of Helsinki and with the Ethics Guidelines of the Institute.
Outcome Measures
All patients were administered complete neurological and neuropsychological examinations. Main outcomes of the study were the Mini-Mental State Examination, 18 Frontal Assessment Battery ([FAB]; subtests similarities, lexical fluency, motor series assessment, conflicting instructions, go/no-go sequences, prehension behavior, or environmental autonomy), 19 Digit Span subtest (digit span forward and backward) and Arithmetic subtest (from Wechsler Adult Intelligent Scale [WAIS] 20 ), Stroop test, 21 global behavioral symptoms, assessed by the NeuroPsychiatric Inventory (NPI), 22 and caregiver stress, assessed by the Relative Stress Scale (RSS). 23 In addition to these main outcome measures, 3 further scales were used: the Cornell Scale for Depression in Dementia, 24 the Behavioral pathology in Alzheimer’s Disease Rating Scale (BEHAVE-AD), 25 and the Clinical Insight Rating Scale (CIR 26 ; which provides a measure of its 4 awareness: disease recognition, cognitive deficit, disease progression, and functional deficit). In order to evaluate apathy, we employed the Clinician/Researcher Rated Version of the Apathy Evaluation Scale (AES-C) and the parallel Self-Report Version of the same instrument (AES-S). 27 Of note, apathy is an individual item on the NPI and the BEHAVE-AD.
Statistical Analyses
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS, version 16.0). Between-group comparisons of changes were tested using the paired samples t test. This was done for the overall scores for each outcome variable. In addition, subanalyses of Spearmann ρ correlation and 2-tailed analyses were performed between behavioral data obtained using the apathy scores (AES-C and -S), the FAB scores, Cornell’s Depression Scores, RSS, CIR, and NPI scores. Results are presented as mean changes from baseline with standard deviations, and P values are presented where appropriate.
Results
All the participants were right-handed (+22.1 ± 1.1) according to the handedness test of Briggs and Nebes. They had an average of 12 ± 4 years of education. All the patients completed all the tests (mean age 64.1 ± 7.3 years, range = 62-70 years; average age at onset = 53.5 ± 1.1 years, range = 45-67 years). The patients had PD for a mean of 2.7 ± 1.15 years and had been treated with dopaminergic preparations (
The akinetic-rigid-dominant group (40 males and 16 women) averaged 65.2 ± 5.1 years; average age at onset was 56.1 ± 3.2 years. Their mean equivalent dosage of
The tremor-dominant type group (23 males and 24 women) averaged 57.7 ± 2.3 years; and the average age at onset was 51.4 ± 1.5 years. Their mean equivalent dosage of
The mean UPDRS part III was 19 ± 3.1.
A synopsis of the cognitive performances obtained by the 2 groups has been reported in Table 1; a synoptic summary of the behavior scores has been reported in Table 2; the differential reappraisal of apathy scores (AES-C and the parallel self-report version of the same instrument AES-S) has been reported in Table 3. Some important cognitive differences exist between the 2 groups: akinetic-rigid patients did worse in Stroop test, performed lower on both the Digit Span and Arithmetic subtests of the WAIS (P < .05 in both scores) on interference subitems (P < .01), and performed worse on all subtests of the FAB (P < .05). When considering the different subitems of the FAB, they did worse in analogies, phonemic fluency, and go/no-go tasks (P < .05).
Cognitive Synoptical Results Obtained by the 2 Groups Studied a
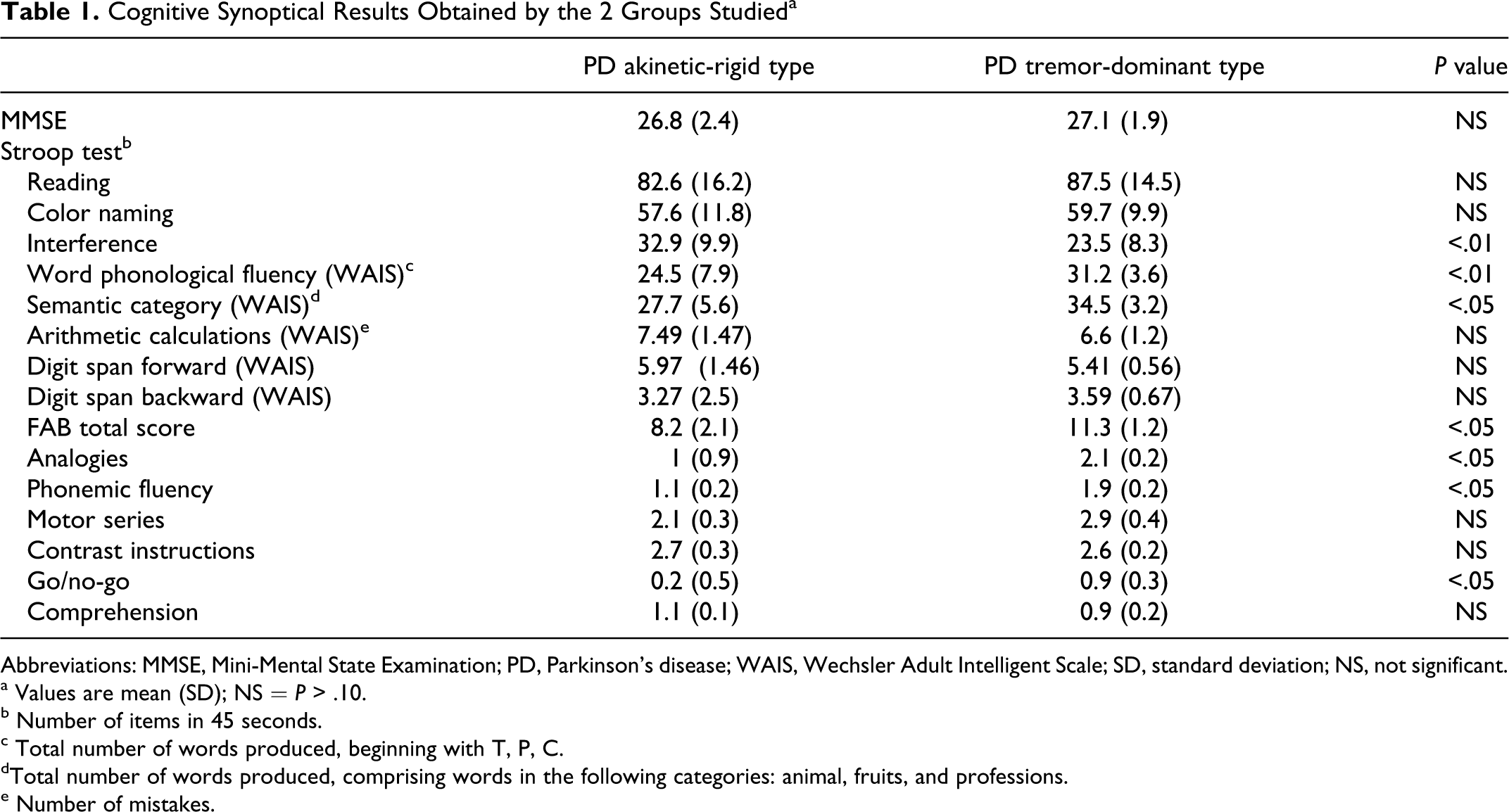
Abbreviations: MMSE, Mini-Mental State Examination; PD, Parkinson’s disease; WAIS, Wechsler Adult Intelligent Scale; SD, standard deviation; NS, not significant.
a Values are mean (SD); NS = P > .10.
b Number of items in 45 seconds.
c Total number of words produced, beginning with T, P, C.
dTotal number of words produced, comprising words in the following categories: animal, fruits, and professions.
e Number of mistakes.
Behavioral Synoptical Results a
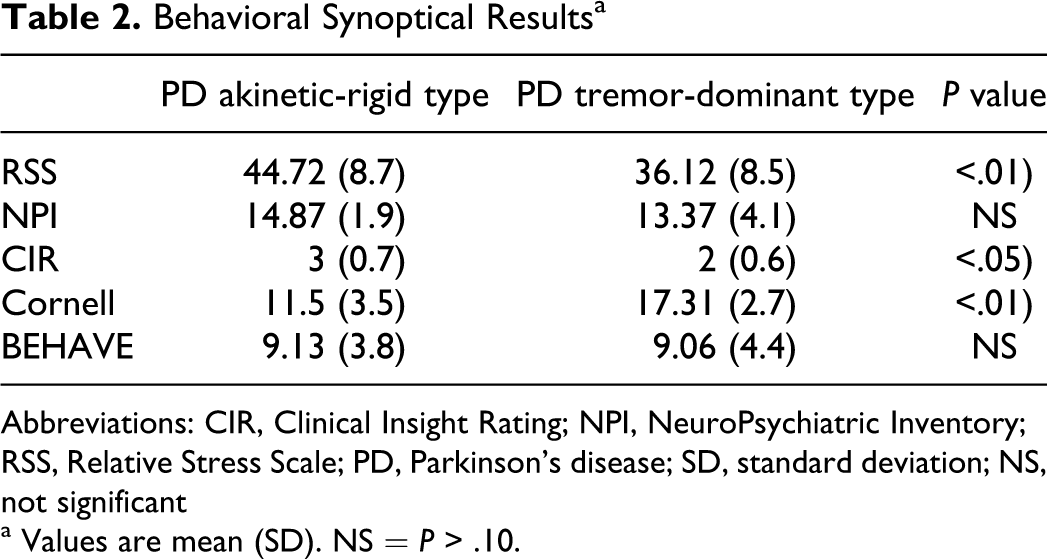
Abbreviations: CIR, Clinical Insight Rating; NPI, NeuroPsychiatric Inventory; RSS, Relative Stress Scale; PD, Parkinson’s disease; SD, standard deviation; NS, not significant
a Values are mean (SD). NS = P > .10.
Apathy Scores: Either by Self-Evaluation (AES-S) or by Researcher Evaluation (AES-C) a
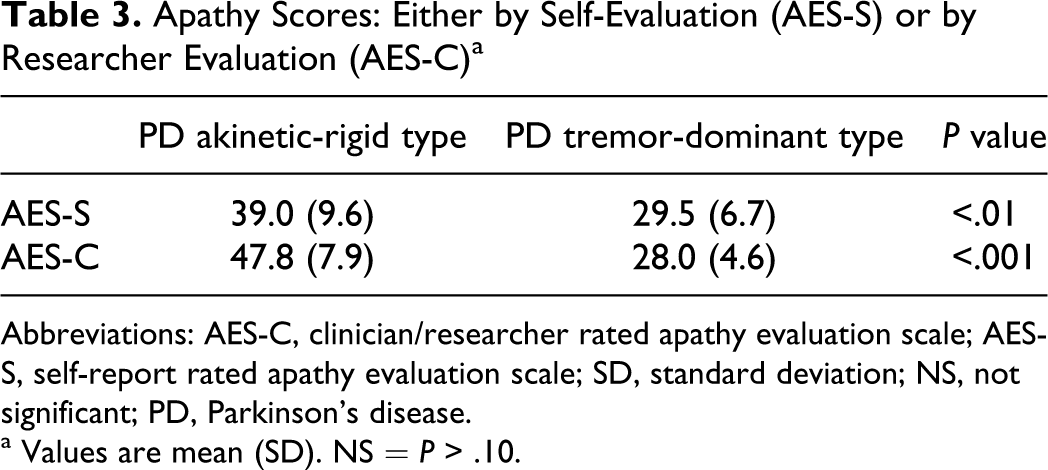
Abbreviations: AES-C, clinician/researcher rated apathy evaluation scale; AES-S, self-report rated apathy evaluation scale; SD, standard deviation; NS, not significant; PD, Parkinson’s disease.
a Values are mean (SD). NS = P > .10.
The akinetic-rigid and tremor-dominant groups did not differ in general behavior aspects (as stated by the 2 major tests, NPI and BEHAVE-AD, as a whole; P > .05). The patients with akinetic-rigid type PD did show more insight into their situation, as tested by CIR (P < .05), and their caregiver’s burden was much worse, as stated by RSS score (P < .01). Patients with tremor-dominant PD endorsed more severe major depression symptoms, according to the Cornell Scale (s < .01).
Very interestingly, patients with akinetic-rigid type PD did manifest overt apathy, which merged from AES-S (P < .01) and even more clearly from the AES-C scores (P < .001).
Spearman rank correlation analyses indicated that there was a significant correlation between the AES-S and RSS (r = .88, P < .01) and AES-C scores and RSS (r = .83, P ≤ .01); AES-S with increased interference mistakes on the Stroop test (r = .69, P < .05) and AES-C with increased interference mistakes on the Stroop test (r = .73, P < .05); there was a negative correlation between AES-S and FAB scores (r = −.83, P < .05) and a negative correlation between AES-C with FAB scores (r = −.81, P < .01); analyzing the subitems, it showed a negative relationship between AES-S and the go/no-go strategies (r = −.71, P < .05), the analogies task (r = −.79, P < .05), and the fluency (r = −.77, P < .05) and a negative relationship between AES-C and the go/no-go strategies (r = −.81, P < .01), the analogies task (r = −.77, P < .05), and the fluency (r = −.74, P < .05). There was no relationship between AES-S and depression nor with insight ratio (CIR) and with UPRDS III scores. There was no relationship between AES-C and depression nor with insight ratio (CIR) and with UPRDS III scores.
Discussion
This study found cognitive differences between patients with PD with a prevalent akinetic variant and patients with PD with a prevalent tremor variant. Akinetic-rigid patients performed more poorly on tasks generally thought to be mediated by the frontal lobe. They showed more insight into their situation, and their caregiver’s burden was much worse. They were also more apathetic than patients with a prevalent tremor. The results of our work seem to provide evidence that apathy is one of the symptoms of PD as the akinetic predominant type of PD seems to clearly exhibit apathy, even though they appear to have awareness of the problem, as demonstrated by the self-report on the specific test (AES-S). The tremor-dominant type of PD showed more depression, less insight, and less apathy than the other group of patients. Generally, these patients did better in cognitive frontal tasks.
In our study, apathy was not related to motor impairment, so it cannot be considered “premature social aging.” 5 On the contrary, our data demonstrate that apathy is tightly related to frontal cognitive impairment, more evident in akinetic-rigid population. Patients with apathy demonstrated impairment in focusing attention, being capable of making decisions, and implementing rapid mental decision scores. These data are in full accordance with data from Levy and Dubois 9 and Schultz et al. 28 Apathy can result from a focal lesion within the striatum, the globus pallidus, or the thalamus: this focal lesion leads to a disruption of a functional circuit, causing a failure in PFC activation. The same result occurs in PD, where there is virtually no direct lesions, but a functional alteration of the same circuitry described above, secondary to the loss of striatal dopamine innervation. Classically, dopamine is associated with reward processing. 9,28 –30 For instance, dopaminergic neurons may signal discrepancies between the predicted reward as a result of a given behavior and the reward that the participant eventually receives. 28 In addition, dopamine may also code uncertainty in reward delivery. 31 Both these mechanisms may suggest a possible role for dopamine signals in attention-based learning and evaluating the odds in decision making, based on the potentially unpredicted rewarding events. As the reward processing circuit involves the orbital–prefrontal–ventral striatum circuit, the influence of dopamine on reward processing may act through a modulation of this circuit via the mesocorticolimbic pathway. Therefore, one may hypothesize that apathy in patients with PD falls within the subtype of “emotional affective” mechanisms and may result from a dysfunction of the orbital–medial prefrontal–ventral striatum circuit. 9 Moreover, our data reflect the fact that apathy is not related to depression but rather to augmentation of caregiver stress. This aspect seems to be in accordance with several data which suggest that apathy in PD is not due to a disruption of emotional-affective processing: apathy is present in PD even at relatively early stages and in the absence of dementia, 2 –10 when the dopaminergic mesocorticolimbic pathway is thought to be relatively spared. 32 Tasks used to assess reward sensitivity and the effect of changes in reward contingencies, such as the gambling 33 and reversal 34 tasks, which are sensitive to lesions of the orbital–medial prefrontal–ventral striatum loop, were not found to be impaired in patients with PD, even in patients tested while “off” levodopa therapy. 35 According to Levy and Dubois, 9 apathy in PD may result from the disruption of “cognitive” processing usually mediated by the dorsolateral PFC-dorsal caudate nucleus circuit. This in agreement with the fact that cognitive dysfunction in PD resembles that of patients with direct dorsolateral PFC lesions 36 and that dopamine denervation of the alternative target, that is, the orbital–medial PFC–ventral striatum circuit, is significantly less severe and not overtly evident. In fact, our study evidences that patients with apathy do have insight into their clinical situation, suggesting that the frontal circuits are not entirely destroyed. Another hypothesis is to consider that apathy is one of several clinical syndromes due to the same elementary dysfunction. This proposal follows the suggestion made by Rolls 37 that “dopamine is a non-specific modulator, that sets the thresholds of firing in striatal neurons regardless of what type of information these neurons carry, as far as the information is relevant for the cortex.” So, apathy can result from the inability of the basal ganglia to validate the relevant signal that is transferred to the PFC. In addition, the overactivity of the γ-aminobutyric acidergic output neurons of the GPi may lead to an excessive and global inhibition of the thalamocortical circuits and thereby hypoactivates the frontal regions involved in the generation of actions based on self-guidance. 9,37 In support to this view, several functional imaging studies showed that while patients with PD were asked to perform a freely selected volitional motor task, at “off” state, an hypometabolism was observed in the medial frontal cortex (mostly in the rostral supplementary motor area) and in the dorsolateral PFC, as compared to normal controls. 38,39 This hypometabolism was reversed by apomorphine, a dopamine agonist, 40 and by internal pallidotomy. 39 Levy and Dubois 9 thus hypothesize that, following striatal dopamine depletion, both defaults in selection and signal amplification contribute to apathy because the output structures can no more disambiguate the relevant signal and this may cause problems in decision making, inducing aborted or delayed responses (see data and literature in Moretti et al 41 ).
Several important questions remain to be elucidated regarding apathy in PD. It has been said that apathy might be considered a predictive factor for dementia in PD. 42 Given that dementia contributes significantly to morbidity and mortality in PD, 43 this finding underlines the need to detect apathy with valid instruments as early as possible and to keep a close eye on the cognitive status of patients with PD in whom apathy is not associated with depression or not-optimal anti-Parkinson treatment. 42,44 Our data seem to demonstrate that there is an independent pathological substrate for apathy (without depression) associated with, not overtly noticeable but real, cognitive impairment in patients with PD. Much more research is necessary to support this theory. It has also been suggested that the presence of an apathy syndrome in PD could be a marker of more rapid or severe cognitive decline in the course of PD: our data do not provide the opportunity to study the follow-up of these patients and additional study in this area is needed. Moreover, data from our study indicate 2 different cognitive and behavioral pathways in 2 distinct clinic variants: akinetic-rigid and tremor-dominant type of PD. Many studies should be done to differentiate the anatomical, biological, and physiological eventual different substrate in the 2 forms, in order to better differentiate them. Another important, and not totally independent question, is the following: should we treat apathy by increased dosing of dopaminergic medication despite optimal motor control in patients with PD or with serotonin/dopaminergic inhibitor reuptake drugs, in order to decrease the caregiver stress, the patient’s burden and, more importantly, a possible cognitive–behavioral involution?
Footnotes
Acknowledgment
The authors wish to thank Andrew Wright Smithson, PhD, for the careful reading and corrections of the manuscript.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
