Abstract
Purpose:
To assess changes in the volumes and spatial locations of tumors and surrounding organs by cone beam computed tomography during treatment for cervical cancer.
Materials and Methods:
Sixteen patients with cervical cancer had intensity-modulated radiotherapy and off-line cone beam computed tomography during chemotherapy and/or radiation therapy. The gross tumor volume (GTV-T) and clinical target volumes (CTVs) were contoured on the planning computed tomography and weekly cone beam computed tomography image, and changes in volumes and spatial locations were evaluated using the volume difference method and Dice similarity coefficients.
Results:
The GTV-T was 79.62 cm3 at prior treatment (0f) and then 20.86 cm3 at the end of external-beam chemoradiation. The clinical target volume changed slightly from 672.59 cm3 to 608.26 cm3, and the uterine volume (CTV-T) changed slightly from 83.72 cm3 to 80.23 cm3. There were significant differences in GTV-T and CTV-T among the different groups (
Conclusion:
The volume, volume change rate, and Dice similarity coefficient of GTV-T were all correlated with increase in radiation treatment. Significant variations in tumor regression and spatial location occurred during radiotherapy for cervical cancer. Adaptive radiotherapy approaches are needed to improve the treatment accuracy for cervical cancer.
Keywords
Introduction
Cervical cancer is the most common female cancer worldwide. Most patients have locally advanced disease in which the standard treatment is chemotherapy with radiation followed by brachytherapy, and expected cure rates range from 30% to 90%, depending on the cancer stage. 1,2 Recent advances in radiotherapy treatment planning and delivery technology have enabled the formulation of highly accurate conformal radiotherapy treatment plans and low damage to organs at risk (OAR). Some studies have found that intensity-modulated radiation therapy (IMRT) is associated with less gastrointestinal and hematologic toxicity, relative to conventional techniques. 3
The IMRT has been increasingly accepted as a treatment for gynecologic cancer. The IMRT enables highly conformal uniform dose delivery to target volumes (eg, the cervix, uterus, parametrium, and pelvic lymph nodes) while reducing doses to OAR (bowel, bladder, rectum, and bone marrow) compared to the conventional 4-field box techniques. 3,4 A retrospective study showed that, with IMRT, late gastrointestinal toxicity was reduced from 50% to 11% for all grades, with favorable tumor control and survival. 5 However, tumor regression and organ motion can affect the dose received regardless of the method of delivery. Some studies have found that the uterus and cervix can move substantially during treatment. 6 –8 Due to motion of the uterus and cervix, changes in the clinical target volume (CTV) can influence the exposure to radiation of adjacent organs, such as the intestine, bladder, and rectum. Therefore, it is necessary to evaluate these changes during radiotherapy to reduce errors.
Image-guided radiotherapy was developed to detect anatomical motion and adjust radiation therapy accordingly. The motion has 2 major components—volumetric changes in targets (ie, tumors) and positional changes of the targets and surrounding organs. In clinical practice, we usually use center of mass or fiducial markers to evaluate target changes and motions. 9 However, this method is not comprehensive enough, so here, we introduce the method of Dice similarity coefficient (DSC) and volume change rate (ΔV) to compare fully the space location of 2 registration image spaces and the volumetric changes for targets.
The purpose of the study was to use cone beam computed tomography (CBCT) to monitor the regression of volumes and changes in spatial location of the GTV-T, uterus (CTV-T), and the CTV in patients with cervical cancer during IMRT.
Methods and Materials
Patients
This prospective study comprised 16 patients with cervical cancers at stages IB2 to IIIB, based on the International Federation of Gynecology and Obstetrics classification. 10 Specifically, 3, 5, 2, and 6 patients had stage IB2, IIB, IIIA, and IIIB cervical cancer, respectively. This study was approved by our hospital institutional review board in September 2014.
Patients underwent IMRT with concurrent cisplatin-based chemotherapy, followed by high-dose-rate intracavitary brachytherapy. Each patient was given an external beam radiation therapy dose of 48.6 Gy in 27 fractions of 1.8 Gy using 7 beams and 6 MV photons to the planning target volume. After this course, each patient received high-dose-rate intracavitary implants once per week that weekly delivered 28 Gy in 4 fractions. All patients received concurrent cisplatin (80 mg/m2) every 3 weeks per cycle (total 2-4 cycles).
Distinguishing Tumors From Normal Tissue
All patients were treated with IMRT, in accordance with the guidelines of the International Commission on Radiation Units and Measurements 50 and 62. The CTV consisted of the regional lymph nodes (common, internal and external iliac, obturator, and presacral), upper vagina, parametrium, cervix, and uterus. The CTV extended from the L4-5 interspace superiorly to 3 cm to the midobturator foramen. The CTV was expanded 0.8 to 1.0 cm uniformly to obtain the pelvic planning target volume.
Cervix and fundus were contoured for each patient, as were bladder, rectum, intestine, and sigmoid. 11 We also contoured the gross tumor volume (GTV) and uterus (CTV-T) to evaluate the regression of the target and the movement of uterus and CTV. The CTV should comprise GTV, cervix, uterus, upper vagina, parametrium, and pelvic nodes (obturator, common, internal, and external iliac). We mainly used CBCT to contour the tumor target and OAR, and magnetic resonance imaging (MRI) and computed tomography (CT) were both taken as only subsidiary references. In order to measure the volume of GTV more precisely, weekly MRI images were obtained during radiotherapy. 12
Imaging and Delivery
Patients in the supine position underwent simulations with a customized vacuum immobilization device using a CT simulator (Big Bore CT, Philips Brilliance Pinnacle) with a 5-mm slice thickness scan. To minimize organ motion, and in accordance with standard practice in our department, patients drank 200 mL of water and emptied their bowels 1 hour before the planning CT scan.
Each patient had a pelvic CT and MRI (Philips Achieva 3.0 T X-Series MRI System) scan before radiotherapy. Each patient also had weekly CBCT scans performed in the treatment position throughout the course of the external-beam radiation treatment. 13
The serial images were acquired at the radiotherapy dose of 9 Gy/5f, 18 Gy/10f, 27 Gy/15f, 36 Gy/20f, and 48.6 Gy/27f in which every 5 fractions were followed by CBCT and once-weekly pelvic MRI scans. Therefore, the total serial images were 80 CBCTs and 80 MRIs. Each CBCT image was set at the same window/level (800/1200). In our daily practice, each CBCT data set was reconstructed and transferred electronically to a Pinnacle3 treatment planning workstation. Then image fusion was achieved by using CT–CT mutual information.
Volumetric Analysis
The CTV-T and GTV-T were separately contoured using diagnostic imaging information and descriptions from the physical examination. The cervix, uterus, lymph nodes, and structures were contoured on each CT scan using the Pinnacle software suite. 14 We used CT–CT normalized mutual information to proceed with fusion (Pinnacle version 9.2s), and the window/level was 400/800. To ensure that the uterus and cervix were accurately differentiated, the uterus volumes were calculated independently. In addition, cervical volumes were contoured based on T2-weighted MRI images.
Positional Analysis and Evaluation Method
To determine the change in position of the cervix during treatment, DSCs were evaluated between the target and spatial position changes. 15 The DSC similarity method has been widely used for deformation in image segmentation and registration assessment and is defined by Equation 1.

Dice similarity coefficients range from 0 to 1, and no overlap indicates agreement. The volumetric changes for targets were calculated using Equation 2.

For Equations 1 and 2,
Statistics Analysis
Paired
Results
Cervical Regression and Evaluation
The cervical contours and measurements of interfraction variation showed dramatic tumor regression and significant positional changes during the course of treatment (Figure 1). The mean GTV-T volume of 79.62 cm3 (range: 42.3-180.3 cm3) at the start of treatment (0f) was reduced to 0.86 cm3 (range: 15.9-31.3 cm3) by the end of treatment (Table 1). On average, the cervical volume was significant different in GTV-T at all treatment points (

Volumetric changes in GTV-T, CTV-T, and clinical target volume (CTV) during therapy.
Tumor Volumes (GTV-T, CTV-T, and CTV) During Therapy (cm3).
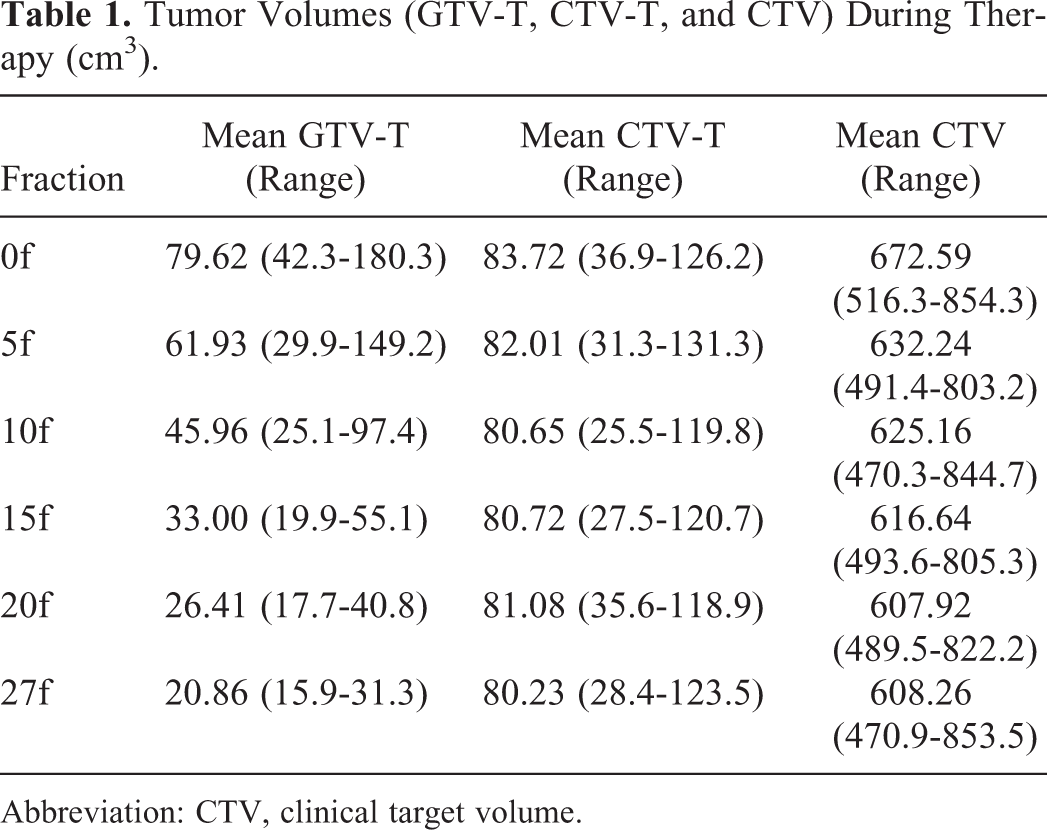
Abbreviation: CTV, clinical target volume.
The CTV and CTV-T decreased slightly over the course of treatment. The mean CTV went from 672.59 cm3 (range: 516.3-854.3 cm3) to 608.26 cm3 (range: 470.9-853.5 cm3); The CTV-T changed slightly from 83.72 cm3 (range: 36.9-126.2 cm3) to 80.23 cm3 (range: 28.4-123.5 cm3). There were significant differences in the CTV-T volume among the different treatment points (
Motion of the Cervix and CTV
The location of the cervix in the pelvis varied greatly during treatment (Tables 2 and 3). The volume change rates of GTV-T and CTV gradually increased throughout the treatment course. The mean volume change rate (ΔV) ranged from 23.05% to 70.85% for the GTV-T, 4.71% to 6.78% for the CTV-T, and 5.84% to 9.59% for the CTV (Figure 2). There were significant differences between different treatment points (
The Volume Change of Target During Therapy.

Abbreviation: CTV, clinical target volume.
DSC Changes of GTV-T, CTV-T, and CTV.

Abbreviations: CTV, clinical target volume; DSC, Dice similarity coefficient.

The volume change rates of GTV-T, CTV-T and CTV during therapy.
Positional Analyses
Changes in position of the cervix during treatment, represented by the DSC of the GTV-T, decreased during treatment from 0.65 to 0.37. The mean DSC of the CTV went from 0.46 to 0.59 and the CTV-T went from 0.84 to 0.87. DSC5f was significantly different from DSC15f, DSC20f, and DSC27f (
There was a significant correlation between volume and volume change rate (DV) of the GTV among different time points during the radiotherapy (

Dice similarity coefficient (DSC) changes of GTV-T, CTV-T, clinical target volume (CTV) during therapy.
Discussion
The MRI and the integration of CT can accurately determine the tumor and adjacent normal tissues and organs while a clinically automatic fusion method can avoid the error between the subjective factors and the operator. In this study, we show that the volume and location of the cervix changed significantly during the course of treatment. These anatomical changes must be taken into account when planning radiation therapy, particularly when image-based inverse planning (image-guided radiotherapy) is used to design highly conformal radiation therapy.
Tumor volume is an important prognostic factor for local control and survival in cervical cancer. The MRI has been reported to be more precise than any other imaging modality for uterine tumors. It has been previously shown that tumor volume can be measured more precisely using MRI, and tumor regression can be accurately evaluated by sequential MRI obtained during radiotherapy.
16
In our study, there was a significant difference in GTV-T volume during the treatment fractions (
Our study, based on the images of serial CT scans, showed cervical regression and motion in patients during treatment. Our results are consistent with the findings of other studies. Lee
Tumor regression during radiotherapy can cause increased uterine activity and dramatic changes in tumor position. 19,20 Most studies showed that the position of cervix shifted less than uterus. In our study, we showed that the volume change rate of GTV-T and CTV gradually increased with the increase in the fraction of radiotherapy.
The DSC was only used for the evaluation of internal positional change in our study. In addition, ΔV was taken as reference for tumor volume change. Concretely, DSC was used to compare volumes of interest in the reference image to the corresponding volumes in the moving image after deformation. The DSC indicates the overlapping ratio between the 2 volumes of interest. It has been shown that the use of the DSC is appropriate for comparing image registrations that range from 0 to 1, the latter indicating overlap and perfect agreement, 21 with MRI studies. This suggests that DSC values >0.70 represent good agreement. 22 If the DSC is smaller, the greater the difference in the original target space position, the less the IMRT isodose line surrounding the target. Therefore, a change in the DSC can be used to predict whether IMRT plans need to be modified, and ΔV can be used to predict tumor volume change.
Our results show that the DSC of GTV gradually decreased and correlated with increasing fractions of radiotherapy. There was a significant correlation between the volume and volume change rate (DSC) of the GTV among different time points during the radiotherapy (
In this study, we observed that tumor regression and spatial location changes were the most obvious during the second and third weeks. This was also observed in previous studies using MRI, which suggests that the best time to modify radiation treatment planning is in the second or third week, 23 and online adaptive radiotherapy might be necessary to manage interfraction motion and volume regression of targets. 24
Conclusion
There are large variations in GTV and CTV position in patients with cervical cancer. Dramatic tumor regression and significant positional changes during the course of treatment were observed. The best time to change the radiotherapy plan is at 2-3 weeks. The primary tumor volume, volume change rate, and DSC correlate with greater treatment fractions. Therefore, our study supports the need to develop adaptive therapy approaches to improve therapeutic efficacy and outcomes of radiotherapy for cervical cancer.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Clinical Key Specialty Construction Program, China, and Key Clinical Specialty Discipline Construction Program of Fujian, China.
