Abstract
Objectives:
To test the feasibility of modified biopsy needles as probes for irreversible electroporation ablation and periprocedural biopsy.
Methods:
Core biopsy needles of 16-G/9-cm were customized to serve as experimental ablation probes. Computed tomography-guided percutaneous irreversible electroporation was performed in in vivo porcine kidneys with pairs of experimental (n = 10) or standard probes (n = 10) using a single parameter set (1667 V/cm, ninety 100 µs pulses). Two biopsy samples were taken immediately following ablation using the experimental probes (n = 20). Ablation outcomes were compared using computed tomography, simulation, and histology. Biopsy and necropsy histology were compared.
Results:
Simulation-suggested ablations with experimental probes were smaller than that with standard electrodes (455.23 vs 543.16 mm2), although both exhibited similar shape. Computed tomography (standard: 556 ± 61 mm2, experimental: 515 ± 67 mm2; P = .25) and histology (standard: 313 ± 77 mm2, experimental: 275 ± 75 mm2; P = .29) indicated ablations with experimental probes were not significantly different from the standard. Histopathology indicated similar morphological changes in both groups. Biopsies from the ablation zone yielded at least 1 core with sufficient tissue for analysis (11 of the 20).
Conclusions:
A combined probe for irreversible electroporation ablation and periprocedural tissue sampling from the ablation zone is feasible. Ablation outcomes are comparable to those of standard electrodes.
Introduction
Percutaneous image-guided ablation is clinically used for the treatment of metastases, as salvage therapy, and more recently for the treatment of small (<3.0 cm) primary tumors. 1 –3 Unlike for surgically resected tumors, little or no tissue is removed during image-guided ablation. Therefore, tissue analysis is not routinely available to assess the treatment efficacy for percutaneous tumor ablation. Instead, postprocedure imaging using contrast-enhanced computed tomography (CT), magnetic resonance imaging, ultrasonography, 4 and most recently positron emission tomography (PET)/CT imaging 5 is performed to confirm adequate coverage of the tumor and to estimate ablation margins. Inadequate margins after thermal ablation as determined using CT imaging have been shown to be an independent predictor of local tumor progression. 6,7 However, both false-negative and false-positive results can be expected when assessing immediate postablation CT images for treatment success. 8 Alternative methods such as split-dose fluorodeoxyglucose PET/CT have been recently investigated to assess immediate postablation treatment outcome and to identify viable tumor tissue within the ablation zone. 5 Compared to histological assessment, imaging may not adequately identify residual viable tumor within the treated region, especially at an immediate and early postablation time point.
Sofocleous et al 9,10 and Snoeren et al 11,12 assessed tissue adherent on the electrode after liver 9 –12 and lung 13 tumor ablation with immunohistochemistry and histology in an effort to assess ablation success and long-term oncologic outcomes. They reported that viable or prolific tumor cells extracted with the ablation probe correlated with a significantly higher risk of local tumor recurrence 9 –13 or ablation failure, as well as actual patient survival. 9,13 Results from this research indicate that there may be clinical value in histology or biomarker analysis of immediate postablation biopsy samples, however, postablation biopsies have not seen wider adoption yet. Extraction of core biopsy samples following ablation would require placement of biopsy needles in addition to the ablation probes. This could increase the bleeding risk when treating tumors in well-perfused organs such as the kidney. A single device capable of delivering ablations and retrieving core biopsies could allow periprocedural tissue sampling in order to confirm that the target lesion was adequately met and to eventually predict treatment outcomes with minimal added risk.
Irreversible electroporation (IRE) is a nonthermal ablation technique, and its vessel and collecting system sparing characteristics render it a potential candidate for renal ablation. 14,15 However, in the kidney, electric field distortions caused by conductivity changes due to tissue architecture or the proximity of the collecting system may be a reason for ablation zone aberrations and incomplete ablation. 16 Patches of viable cells may survive in the center of an IRE ablation zone when IRE is not achieved. 17 Biopsy samples from ablation zones could possibly detect incomplete ablation.
In a first step, ablations performed using a device for both ablation and biopsy have to be proven noninferior to ablations performed with clinically used standard ablation electrodes.
The primary aim of the presented study was to test the feasibility of using modified core biopsy needles as probes for IRE ablation through numerical simulations and an in vivo animal model. We hypothesized that when using similar treatment parameters, a modified biopsy needle electrode could be used to deliver IRE ablations equivalent or noninferior in quality and size to ablations performed with standard electrodes.
Materials and Methods
Animal Model
Under approval by the Institutional Animal Care and Use Committee, 20 percutaneous CT-guided IRE ablations were performed in the kidneys of 9 healthy female Yorkshire pigs (weight range, 30-35 kg; Archer Farms, Darlington, Maryland). The animals were premedicated with tiletamine hydrochloride and zolazepam hydrochloride (Telazol, 4.4 mg per kilogram of body weight; Fort Dodge Animal Health, Fort Dodge, Iowa) administered intramuscularly and after intubation. General anesthesia was maintained with continuous inhalation of 1.5% to 3% isoflurane (Baxter, Illinois) and 100% oxygen on a Penlon Nuffield ventilator (Penlon Ltd., Abingdon, UK). Buprenorphine (0.01 mg/kg) was given via intramuscular injection before the start of each procedure. Rocuronium (1.2 mg/kg) was administered intravenously 5 minutes before applying IRE pulses. All animals were euthanized within 4 hours following treatment with an intravenous injection of pentobarbital sodium (140 mg/kg, Euthasol; Virbac AH, Fort Worth, Texas).
Ablation and Core Biopsy
Clinically used 16-G/9-cm core biopsy needles with a 2-cm throw length (Quick-Core; Cook Medical Inc, Bloomington, Indiana) were modified to function as monopolar experimental electrodes by insulating the cannula with nylon heat shrink tubing (Pebax; Cobalt Polymers, Cloverdale, California) over 6 cm in length from the handle, while leaving the tip and inner stylet exposed and electrically conductive (Figure 1A). Clinically used 19-G monopolar IRE probes (NanoKnife System; Angiodynamics Inc, Queensbury, New York) were used for performing standard ablations. Two commercially available generators (NanoKnife System; Angiodynamics Inc and BTX ECM 830; Harvard Apparatus, Holliston, Massachusetts) were used for delivering square pulses, the former was used with the standard electrodes, and the latter generator was used with the experimental electrodes. Ten ablations were performed with each electrode type, with 1 ablation in the midpole and the other ablation in the lower pole of the animal. For both treatment cohorts, the 2 electrodes were inserted parallel in an axial plane under CT guidance (Lightspeed RTLS; GE, Milwaukee, Wisconsin) by a single operator. Before insertion of the experimental electrode, the plunger was retracted until the biopsy gun was primed. The experimental electrode was then advanced to the border of the designated ablation zone in the kidney with the inner stylet fully retracted. Before pulse application, the inner stylet and specimen notch were carefully exposed to a length of 2 cm while avoiding firing of the cutting cannula (Figure 1B). Following completion of pulse delivery, the modified biopsy gun was fired while maintaining the needle position to obtain a core sample only in the experimental ablation cohort. The biopsy needles were then withdrawn from the ablation zone. One core biopsy sample was taken from each needle position (n = 20, equaling 2 biopsies per ablation zone). No biopsy samples were taken from the ablations performed using the standard electrodes. Both experimental and standard ablations were performed using the same pulse parameter setting. The distance between the 2 parallel electrodes in both treatment groups was adjusted to 15 mm (allowing a deviation of ±1 mm for each electrode) and confirmed by CT measurements. The exposure, which defines the noninsulated distal portion of the needle electrodes, was adjusted to 2 cm for both electrode types. A train of 90 direct current (DC) pulses of 100 µs length was applied using a field strength of 1667 V/cm (equaling 2500 V at an interelectrode distance of 15 mm).

A, Modified core biopsy needle design for ablation and biopsy with exposed inner stylet tip and specimen notch (a) and insulated cannula (6 cm length; b). B, Schematic display of 2 parallel biopsy ablation devices with the inner stylet and specimen notch exposed and expected ablation zone (a, enlarged). The biopsy core is taken from the same location. The cutting cannulas (b) are partially insulated to avoid inadvertent ablation of the tracts. The proximal exposed parts are used to connect the pulse generator cables (+, −). The vertical line indicates the skin surface.
Simulation
Computer-aided design (CAD) models of the modified biopsy electrode, standard needle electrodes, and a cubic volume (10 cm3) representing normal renal parenchyma were constructed using Inventor (Autodesk, San Rafael, California) and imported into Comsol Multiphysics (Comsol, Burlington, Massachusetts). The exposure and needle spacing for both conditions were kept identical, with values of 2.0 and 1.5 cm, respectively. The pair of electrodes and the tissue were discretized into tetrahedral element mesh in Comsol (Figure 2). A static DC model was setup, with boundary conditions on electrode at 2500 V, and the other was set as electrical ground. The boundary of the tissue block was assumed to be electrically insulated. This mesh was used to compute the electric potential over the domain for a single electric pulse by solving the Laplace equation (

Computer-aided design (CAD) model rendering for the experimental core biopsy needle design.
Imaging
Contrast-enhanced (1 mg/kg iohexol 350 mgI/mL; GE Healthcare, Milwaukee, Wisconsin) axial CT images (Lightspeed RTLS; GE) of the kidneys (slice thickness: 1.5 mm, reconstruction interval: 3 mm) were acquired within 1 hour following ablation with both electrode types. Ablation zones were assessed and measured on axial CT planes using an Advantage 4.4 Workstation (GE Healthcare). Two-axis measurements were performed on axial planes parallel and perpendicular to the needle insertion tracts. A cross-sectional area was calculated for each ablation zone assuming an approximately elliptical shape using the formula π × x/2 × y/2.
Histopathology Analysis
Core biopsy samples were grossly inspected, photographed, and immediately fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 4 µm thickness, and stained with hematoxylin and eosin (H&E). Biopsy specimens were histologically assessed for a sufficient amount of tissue to confirm ablation effects (diagnostic adequacy) as well as for findings indicative of cellular necrosis.
Animals were euthanized within 4 hours following treatment, and kidneys were harvested immediately after euthanasia. The organs were examined for needle insertion points to identify ablation zones. The organ was then sectioned along the needle insertion tracts to obtain the largest available cross section of the ablation zone (corresponding to an axial CT imaging plane), and a 3- to 5-mm-thick slice was fixed in 10% neutral-buffered formalin. The tissue slice was consecutively embedded in paraffin, sectioned at 4 μm thickness, stained with H&E, and assessed for morphologic changes in cell death. Histologic measurements were performed using image processing software on photomicrographs of the cross section of the ablation along with adjoining unaffected tissue. The size of the contiguous region of ablated tissue was calculated using the formula detailed in the Imaging section.
Statistics
Student t test was used to determine differences in the mean ablation area calculated from the largest cross-sectional diameters of the ablation zone for the 2 treatment groups. The t test was performed separately for CT and histology measurements. A 95% confidence interval was assumed for significant differences between measurements from the 2 groups.
Results
All ablations in both groups were completed successfully without complications. Despite using a larger sized electrode (16 vs 19 G), there was no difference in bleeding encountered during ablation with the experimental electrode when compared to the standard electrode. Qualitatively, the gross pathology appearance and the shape of ablation zones created with the experimental and standard electrodes were found to be comparable. The shape of ablation zones for both groups was approximately ovoid with shape deformations wherever the collecting system was involved. The ablation zone created with the experimental electrode featured wider needle tract defects due to the lower gauge size of the electrodes and the subsequent core biopsy. Adjacent to the needle trajectories, a central 1- to 2-mm-wide zone of pale tan discoloration suggestive of thermal coagulation could be identified in ablation zones created with the experimental electrodes (Figure 3B). Apart from this difference, the ablation zones from both treatment groups appeared uniformly dark red, suggesting vascular hyperemia and/or hemorrhage (Figure 3B and C). No difference in appearance and shape could be attributed to midpole or lower pole locations apart from the fact that the collecting system was involved in fewer cases in lower pole ablations.

A, Biopsy core taken from the ablation zone containing sufficient tissue for histology evaluation (a), and specimen notch containing blood and debris insufficient for histology assessment (b). Ablation zone created with the combined device (B) and the standard needle electrodes (C). Arrows indicate the needle trajectories.
Histopathology
Histopathology of kidney necropsy specimens indicated successful ablation in all cases of both groups. There was no qualitative difference discernible by the study pathologist between ablations created using the modified biopsy electrode and the standard electrodes. Ablations in both groups presented as regions of cellular changes indicative of cell death (nuclear condensation and cytoplasmic hypereosinophilia) with retention of the extracellular tissue architecture of the cortex, medulla, or pelvis, affecting all functional units of the organ (tubules, glomeruli, blood vessels, and pelvic urothelium). These changes were consistent with an early stage of cell death (Figure 4A, B).

Histological characteristics of samples from the experimental electrode and control electrodes. Samples from the ablation zones of both the control (A) and experimental (B) electrodes manifest similar histological changes. There was evidence of heat-induced damage immediately adjacent to the needle tract of the experimental electrode (D), which was absent in the control electrodes (C). The histological characteristics of 70% of biopsy cores matched that of the ablation zone (E), and only some cores did not give evidence of ablated renal tissue (F).
The affected areas also displayed vascular hyperemia and hemorrhagic tracts due to needle placement. Tissue directly adjacent to these tracts showed streaming of nuclei and hyalinization of stroma consistent with thermal effects, which were not observed in the ablation zone more distant from the electrode tracts (Figure 4C, D).
Average cross-section dimensions were 21.4 and 17.7 mm for the standard electrode and 22.8 and 14.6 mm for the experimental electrodes (P = .264 for the major axis and P = .088 for the minor axis). For both the modified biopsy electrode and the standard electrode, the measurements did not reveal a statistically significant difference in ablation zone size. The cross-sectional axis measurements were used to compute the area of ablation (standard: 313 ± 77 mm2; experimental: 275 ± 75 mm2; P = .292). Within each group, there was no significant difference in ablation zone size dependent on the location in the kidney (midpole vs lower pole, experimental: P = .385 and standard: P = .456). The mean histologic ablation size for each group (modified biopsy needle vs standard electrode) and location (midpole vs lower pole) is given in Table 1 and Figure 5.

Mean (±standard deviation [SD]) cross-sectional area of ablation zones as measured on computed tomography (CT) for the experimental electrodes and control needle electrodes at sample locations in the middle and lower pole in the kidney.
Comparison of Mean (±SD) Cross-Sectional Areas of Ablation Zones (mm2) Calculated From Axial Photomicrograph Measurements of Histological Sections.

Biopsy Core
Biopsy cores were considered adequate if they contained a sufficient amount of renal tissue for histologic assessment of ablation effects (Figure 3A). Adequate amounts of tissue could be retrieved from 65% (13 of the 20) of the single pass biopsies performed after treatment delivery. Thirty-five percent (7 of the 20) of samples did not contain a sufficient tissue core, mostly containing cellular blood components (erythrocytes). Among samples containing a core of tissue, 84.6% (11 of the 13) contained renal tissue from the ablation zone. Of the 13 samples, 2 contained skeletal muscle tissue and capsular fat but not renal tissue and therefore could not be used for assessment of the ablation zone. The histological findings in 72% (8 of the 11) of these biopsy samples were consistent with the findings in the necropsy specimen of the ablation zone from which the biopsy was taken (Figure 4E). In some cases, hyalinization of stroma and streaming of nuclei were consistent with a localized thermal effect in the immediate proximity of the needle tracts. In the remaining 28% of the biopsy samples, a lack of indicative histologic changes in cellular necrosis did not allow to confirm tissue ablation within the yielded biopsy core (Figure 4F).
Imaging
Immediate postprocedure contrast-enhanced CT indicated a similar appearance both for ablation zones created with experimental and standard electrodes. Axial cross sections of ablation zones approximated an ellipsoid shape and were typically composed of a central nonenhancing and a peripheral lightly enhancing zone. Demarcation from the adjacent renal parenchyma was sharp. Size measurements were performed including the central and peripheral zones of ablation. The mean distance between the 2 parallel electrodes as measured on CT was 14.3 ± 0.5 mm (14.2 ± 0.5 mm for midpole and 14.4 ± 0.4 for lower pole locations) both for the experimental electrode (Figure 6A) and the standard IRE electrodes (Figure 6C) and independent from the location within the kidney (data not shown). No statistically significant difference in size was found between ablation zones created in the experimental group and the standard group (standard: 29.3 × 24.1 mm; experimental: 29.1 × 22.3 mm; P = .462 for the major axis and P = .137 for the minor axis), as well as between the middle and lower pole locations for each treatment group (mid pole vs lower pole, experimental: P = .295; standard: P = .136). The cross-sectional axis measurements were used to compute the area of ablation (standard: 556 ± 61 mm2; experimental: 515 ± 67 mm2; P = .246). The mean CT ablation size for each group (experimental electrode vs standard electrode) and location (midpole vs lower pole) is given in Table 1 and Figure 5.
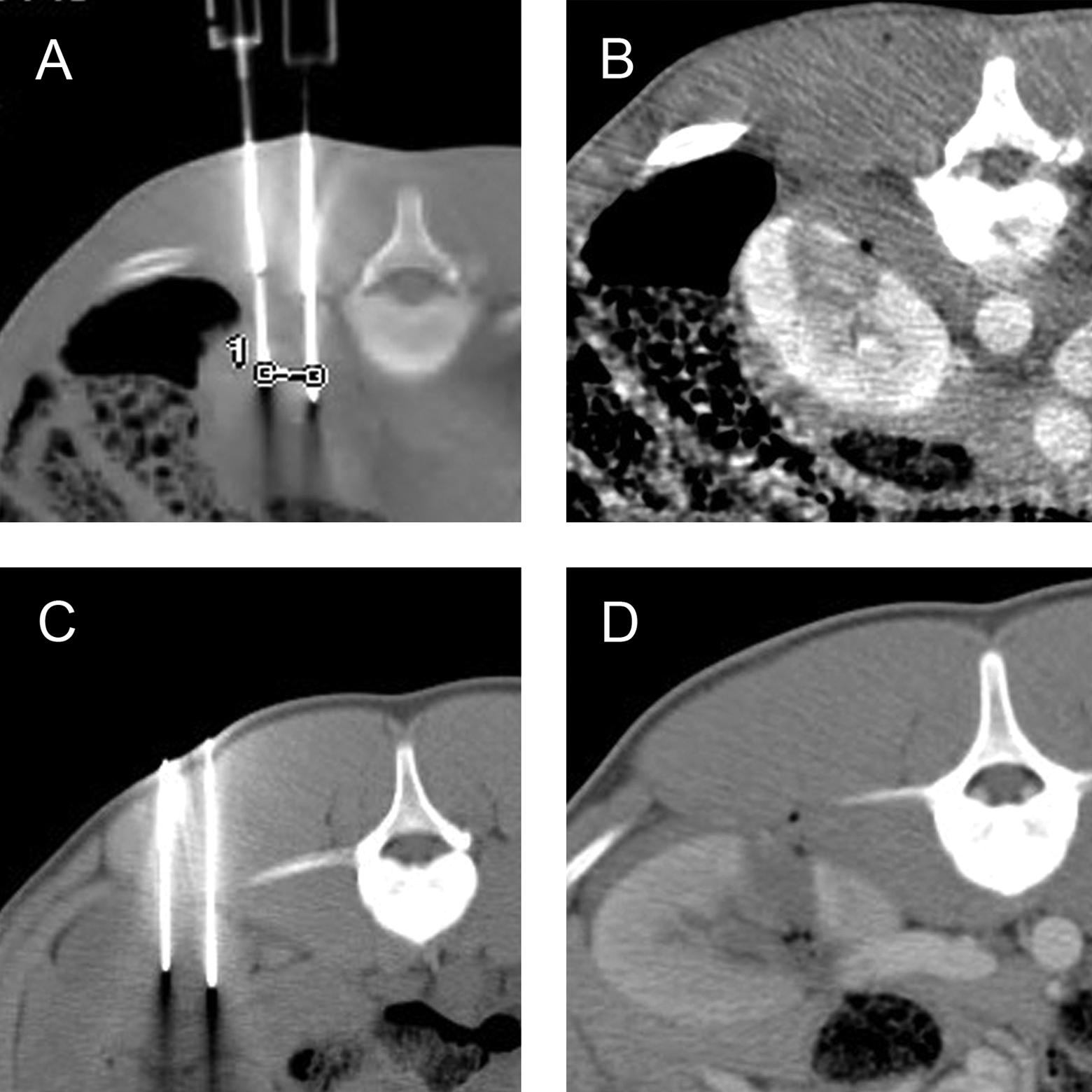
Computed tomography (CT)-guided needle placement using the combined biopsy device (A) and the standard irreversible electroporation (IRE) needle electrodes (C). Contrast-enhanced CT showing an ablation zone created with the experimental electrodes (B) and the standard IRE needle electrodes (D).
Simulation
Results from numerical simulation for the experimental and standard electrodes indicated equivalently sized ablation zones with similar shape at the largest cross section. The simulated zone of IRE extended to the 650 V/cm isoline. A transitional zone of reversible electroporation would be expected in the periphery of the ablation zone between 650 and 400 V/cm isolines. The area beyond 400 V/cm was considered unaffected due to insufficient electric field effects. When compared to the simulated ablation for the standard electrode, the simulated experimental electrode ablation was slightly smaller when measured perpendicular to the electrode axis but was slightly longer when measured along the electrode axis. This finding was consistent with histological measurements and could be attributed to the electrode geometry. However, the sharp edges as well as the notch geometry of the biopsy device used as the experimental electrode did not seem to affect the overall shape of the predicted ablation zone. At the chosen critical threshold of 650 V/cm, simulation results predicted that ablations performed with the experimental electrodes resulted in an area of 455.23 mm2 at the largest cross section (no statistical difference in CT measurements, P = .178) and ablations performed with the standard electrodes resulted in an area of 543.16 mm2 (no statistical difference in CT measurements, P = .751). Histology measurements were significantly smaller than simulation for both the experimental and the control groups (P = .0005 for histology vs experimental group simulation and P < .0001 for histology vs standard simulation).
Discussion
Core needle biopsies and image-guided ablation are 2 common interventional oncology procedures, and a simple device modification allows the combination of these procedures using a single probe. In previous research, modified devices have been used to combine ablation and biopsy procedures, however, they were limited in their scope and goals. Bruners et al 21 reported modifying a core biopsy needle (Trucut, Cardinal Health, IL, USA) to act as a bipolar radiofrequency electrode so that it may be used to ablate the needle tract following biopsy. Similarly, Pritchard et al 22 investigated radiofrequency cauterization using the biopsy introducer needle to reduce postbiopsy hemorrhage. Although these studies indicate that biopsy probes may be adapted to perform limited ablation of the surrounding tissue, there was no further investigation into the feasibility of using biopsy needles for creating larger ablation volumes.
The results from this study indicate that it is feasible to use modified biopsy core needles as IRE ablation probes using a parallel needle pair geometry and to extract periprocedural core biopsies from the ablation zone without repositioning or additional needle placement. Histologic workup of ablated tissue following necropsy demonstrated that the delivery of IRE using this device resulted in successful ablation without significant impact on the quality or the size of ablation when compared to the clinically used standard electrodes.
Minor differences between ablations created with the experimental and standard electrodes were observed in the immediate surroundings of the needle. Apart from a larger defect due to the lower gauge of the experimental electrode and the subsequently taken biopsy, a 1 to 2 mm zone of thermal damage was observed more frequently around the trajectories of the experimental probe than when using the standard needle electrodes. Reasons for this observation may be the lower gauge experimental probe and the sharp edges of the inner stylet and specimen notch, which may have led to a greater heat generation due to localized current concentration. Under certain circumstances, such as a higher field density, thermal effects have been reported previously using IRE. 23 Although, thermal effects need to be considered when placing needles close to structures that may be affected by thermal damage, such as the urinary collecting system, they appear to be restricted to the immediate vicinity of the electrode and do not have an impact on ablation size or shape. In the control group, ablation zones were insignificantly larger in the lower pole region, a difference that may be attributed to a wider parenchymal layer in the axial cross section at this level.
A minimum of 1 in 2 core biopsy samples taken at a single pass from 1 ablation zone using the experimental device was adequate in quantity and quality for histologic assessment. Samples were considered inadequate due to insufficient amounts of collected kidney tissue. In 2 biopsy cases containing perirenal fat and muscle fiber, inadequate sampling may have been caused by needle dislocation.
Early ablation effects could be identified histologically on H&E-stained biopsy samples in the majority of cases. Only in a minority, biopsy cores were not indicative of ablation effects, although ablation was confirmed through subsequent necropsy workup in all cases. Unlike in thermal ablation techniques, IRE is known to take a period of time for completion of ongoing cellular necrosis, and this may prevent early detection of cell death using H&E stains alone. 24,25 This issue could be addressed with viability stains for immediate assessment of earlier stages of cellular death. 16,26,27
The electric field strength and current during IRE is susceptible to local conductivity variations between different compartments and tissues, reducing chances of homogeneous and complete ablation. 17 Although tissue may appear ablated on imaging, showing an ablation zone with sufficient margin creation, 6 imaging cannot rule out residual viable tissue, which can only be identified through pathology 8 using dedicated markers. 26 –28 Core biopsy samples taken from the ablation zone may not only confirm that the target lesion was successfully met but also indicate an incomplete ablation whenever there is evidence of viable tumor tissue in the biopsy sample. Compared to thermal ablation techniques, this may be more relevant in IRE ablations, which may be inhomogeneous even in the center of the ablation zone due to electric field aberrations. 17 Periprocedural histologic workup of ablation zone biopsy samples might identify residual viable tissue and indicate modification of ablation parameters and/or repeat ablation in such cases.
There are several limitations to the concept of periprocedural biopsies in IRE. It may take 4 to 24 hours for IRE ablation effects to completely develop and to result in histopathological changes visible through H&E staining. Therefore, core biopsies taken immediately after ablation and assessed by H&E staining alone may not reflect the final ablation status of the tissue. Analyzing biopsy samples with immunohistochemistry stains and antibodies as described in prior studies 11,12,26 –28 may better reflect the ablation status of the tissue immediately postprocedure than H&E stains alone.
Ablation zones were compared based on 2-dimensional axis measurements, implying that the largest cross section is representative for the ablation zone. Ablation volume measurements would more accurately depict ablation zones in case of asymmetric configurations or spatial deviations of the ablation zone.
Apart from electrode geometry, minor, statistically not significant differences in ablation size may have resulted from the use of different generators for treatment delivery.
In conclusion, the use of a combined device for ablation and periprocedural core biopsy is technically feasible. The study results indicate comparable outcomes in terms of ablation quality and size. Biopsy samples yielded adequate material for analysis, and histology workup could confirm ablation in the majority of cases from periprocedural biopsy samples. Periprocedural tissue sampling may confirm that the ablation target was successfully met and identify residual viable tissue. Further studies will be necessary to evaluate the predictive value of periprocedural core biopsy samples for ablation outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S. B. Solomon is a consultant for Covidien and received a grant from GE Healthcare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The institution of S. B. Solomon received an unrestricted grant and equipment from Angiodynamics Inc.
