Abstract
Objective:
This objective of this study is to investigate the effects of the radiolabeled cyclic peptide 153Sm-DTPA-c(CGRRAGGSC) on MHCC97-H human liver cancer cells in vitro and in vivo.
Methods:
The protein expression levels were examined by Western blot analysis. Biological activity of 153Sm-DTPA-c(CGRRAGGSC) was assessed with the radioligand binding assay and competitive inhibition experiment. Subcellular localization of the cyclic peptide was observed by fluorescence microscopy. Animals were implanted with MHCC97-H cells and administered with 153Sm-DTPA-c(CGRRAGGSC). Hematoxylin and eosin staining, electron microscopy, and immunohistochemistry were performed to evaluate the effects of 153Sm-DTPA-c(CGRRAGGSC) on implanted tumors.
Result:
The expression levels of interleukin 11 receptor were significantly elevated, by 2-to 5-fold, in tumor cell lines, especially for MHCC97-H cells. Characterization of 153Sm-DTPA-c(CGRRAGGSC) showed that the biological activity of the cyclic peptide was not altered after labeling, and the radiolabeled cyclic peptide exhibited sufficient binding affinity to interleukin 11 receptor . The cyclic peptide of c(CGRRAGGSC) was mainly distributed in the cytoplasm and on the cell membrane of MHCC97-H cells. The in vivo experiments showed that the tumor growth was significantly inhibited by the treatment of 153Sm-DTPA-c(CGRRAGGSC). The inhibitory effect of 153Sm-DTPA-c(CGRRAGGSC) on tumor growth was further confirmed by Hematoxylin and eosin staining, electron microscopy, and immunohistochemistry. Moreover, the expression levels of interleukin 11 receptor in implanted tumors were significantly decreased in the treatment groups.
Conclusion:
153Sm-DTPA-c (CGRRAGGSC) could specifically bind to interleukin 11 receptor on MHCC97-H liver tumor cells, inhibiting the cell proliferation and inducing cellular apoptosis. These findings provide experimental evidence for the development of individual treatment of liver cancers, as well as recurrence and metastasis.
Introduction
Liver cancer is one of the most common malignancies with high incidence in China. 1,2 The molecular mechanisms underlying the pathogenesis of liver cancer remain to be fully elucidated, and the treatment of the disease recurrence and metastasis has always been a challenging clinical problem. Previous studies have shown that interleukin 11 (IL-11) and its receptor (IL-11R) are closely related to the tumor growth and metabolism, especially in the bone metastasis of tumors. 3 Interleukin 11 is a cytokine with unique structure, which could be rarely detected in normal tissues. However, the expression levels of IL-11 are significantly elevated during certain viral infection and in tumor tissues. Correspondingly, the expression levels of IL-11R are also dramatically upregulated in common malignancies, including osteosarcoma, prostate cancer, and breast cancer. 4 –7 Therefore, IL-11R could be used as a candidate target for cancer diagnosis and treatment. 8
In recent years, studies have shown that the motif of Cys-Gly-Arg-Arg-Ala-Gly-Gly-Ser-Cys has homology to IL-11, 7 and the cyclic peptide CGRRAGGSC [c(CGRRAGGSC); single letter amino acid code] could specifically bind to the α chain of IL-11R. 4 –9 Thus, investigators have been trying to label the cyclic peptide with radiotracers to develop probes for in vivo molecular imaging. 9 Previous articles from our laboratory have found that radioactive molecular probe99Tcm-diethylenetriaminepenta-aceticacid-t-butylester (DTPA)-c(CGRRAGGSC) could be used as an imaging agent for IL-11R overexpressing tumors and bone metastasis, such as prostate cancer, 10 –13 and 153Sm-DTPA-c(CGRRAGGSC) could exert direct inhibitory effects on PC-3 human prostate cancer cells. 11 However, the properties and effects of the cyclic peptide (IL-11 analogue) in liver cancer have not yet been established.
In this study, the cyclic peptide was labeled with 153Sm to obtain 153Sm-DTPA-c(CGRRAGGSC), and the binding characteristics were analyzed. Moreover, the in vivo targeting property and potential, as well as the therapeutic efficacy, of 153Sm-DTPA-c(CGRRAGGSC) were also investigated in nude mice bearing human hepatocellular carcinoma xenografts.
Materials and Methods
Cell Lines and Animals
MHCC97-H and MHCC97-L human hepatoma cell lines were purchased from the Cell Bank of Chinese Academy of Sciences (Shanghai, China). PC-3 prostate cancer cell line, MDA-MB-231 and MCF-7 breast cancer cell lines, SKOV3 ovarian cancer cell line, HeLa cervical cancer cell line, and HHL-5 normal human liver cell line were all purchased from KeyGEN Biotech (Nanjing, Jiangsu, China). These cells were cultured with Dulbecco modified Eagle medium (Gibco, Grand Island, New York), supplemented with 10% fetal bovine serum (Gibco) in a 37°C, 5% CO2 incubator.
Totally, 20 BALB/c nude mice, half males and half females, 4-week-old, weighing 15 to 19 g, were provided by the Animal Center of the Yangzhou University. All animal experimental procedures were approved by the Animal Ethical Committee of the Guiyang Medical College.
Western Blot Analysis
The expression levels of IL-11R were detected by the Western blot analysis. For cell cultures, cells were lysed on ice with the lysis buffer containing protease inhibitors. For tumor tissues, the tissues were cut into pieces and lysed by the lysis buffer containing protease inhibitors. After centrifugation at 1500 rpm for 5 minutes, the protein concentration in the supernatant was determined by the bicinchoninic acid assay method. Protein sample of 40 μg was separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis, and then electronically transferred onto a nitrocellulose membrane. The blot was blocked with bovine serum albumin (BSA) in triethanolamine-buffered saline with Tween (TBST) at 25°C for 4 hours, and then incubated with rabbit antimouse anti-IL-11R antibody (1:1000 dilution; Santa Cruz, Santa Cruz, California), anti-IL-11 antibody (1:1000 dilution; Santa Cruz), and goat antimouse anti-β-actin antibody (1:5000 dilution; Santa Cruz), respectively, at 4°C for 6 hours. The membrane was then incubated with horseradish peroxidase-conjugated goat antirabbit or rabbit antigoat immunoglobulin G (ZSGB-BIO, Beijing, China) at 25°C for 2 hours and developed with the DAB detection kit (ZSGB-BIO).
Preparation of 153Sm-DTPA-c(CGRRAGGSC)
DTPA-c(CGRRAGGSC) was synthesized according to a published protocol. 7,9 Briefly, 0.5 mL reaction system contained 370 MBq (in 20 μL) 153SmCl3 and 20 μg DTPA-c(CGRRAGGSC; a kind gift from the Molecular Imaging Research Center of the Baylor College of Medicine, Houston, Texas), and the pH value was adjusted to 8.0 with 0.1 mol/L HCl. After 30-minute reaction at room temperature, the products were purified and analyzed with high-performance liquid chromatography, and the stability evaluation was performed as described previously. 11
Saturation Analysis Experiment
The MHCC97-H cells were collected, and 0.5 mL cell suspension (5 × 105 cells/mL) was mixed with 0.1 mL 153Sm-DTPA-c(CGRRAGGSC) at the concentrations from 1 × 10−7 to 10 × 10−7 mol/L in a tube, with the volume adjusting to 1 mL. The tube was then incubated at 4°C for 3 hours, and ice-cold Hank’s buffer was added to stop the reaction. After centrifugation at 3000 rpm at 4°C for 14 minutes, the supernatant was discarded, and the precipitation was mixed with formic acid at 60°C for 30 minutes. The total binding (TB) and nonspecific binding (NSB) events in the precipitation were counted with a radioimmunoassay γ counter (E502; Packard, Meriden, Connecticut), and the specific binding (SB) events were calculated as SB = TB – NSB. The saturation curve was obtained with the concentrations of 153Sm-DTPA-c(CGRRAGGSC) and the radioactive counts per minute. The Scatchard plot was composed of the concentrations of 153Sm-DTPA-c(CGRRAGGSC), and the SB–FB ratios (FB was the radioactive counts per minute for the free ligands, which was calculated as FB = T – TB, where T represents the total amount of radioactivity and TB represents the TB). The equilibrium dissociation constant (Kd) and the maximal binding capacity (Bmax) were obtained with the linear regression.
Competitive Inhibition Experiment
Totally, 250 μL reaction solution contained 70 μL MHCC97-H cell suspension (1 × 109 cells/L), 6 × 10−7 mmol/L 153Sm-DTPA-c(CGRRAGGSC), and unlabeled c(CGRRAGGSC) at the concentrations from 1 × 10−10 to 1 × 10−1 mmol/L. For the NSB group, 70 μL ddH2O, 6 × 10−7 mmol/L 153Sm-DTPA-c(CGRRAGGSC), and unlabeled c(CGRRAGGSC) at the concentrations from 1 × 10−10 to 1 × 10−1 mmol/L were mixed in the tubes. The experimental procedures were the same with the radioligand binding assay. The competitive inhibition curve was obtained with the SB count per minute and the concentrations of unlabeled peptides.
Subcellular Localization Analysis
For the preparation of fluorescein isothiocyanate (FITC)-c(CGRRAGGSC)-DTPA, 1 mg FITC was solved in 2 mL dimethyl sulfoxide in dark, followed by the addition of 0.1 mg c(CGRRAGGSC) and 100 μL 0.1% acetic acid. The solution was vortexed and incubated at 4°C for 8 hours. For staining, MHCC97-H cells in the logarithmic growth phase were fixed with 4% paraformaldehyde for 10 minutes and then blocked with 1% BSA for 30 minutes. Then, the cells were incubated with FITC-c(CGRRAGGSC)-DTPA at 37°C for 1 hour. After washing with PBS, the cells were incubated with 100 μL 4′,6-diamidino-2-phenylindole at room temperature for 2 minutes and then observed with an inverted fluorescence microscope.
Animal Modeling and Grouping
MHCC97-H cells were collected and adjusted to the density of 1 × 107 cells/mL. Totally 0.1 mL cell suspension was injected subcutaneously into each nude mouse under the armpit of the right forelimb. The following experiments were performed when the implanted tumor grew to the size of 0.8 to 1.0 cm in diameter. The mice were divided into the following groups (n = 5 for each group): the control group, in which the model mice was injected with 0.2 mL saline via the tail vein and the low-, middle-, and high-dose groups, in which the model mice were administrated with 153Sm-DTPA-c(CGRRAGGSC) via the tail vein at the doses of 5.5, 11.0, and 22.0 MBq, respectively.
On day 16 after treatment, these mice were killed. The tumors were excised, measured, and weighed, and the tumor volumes were calculated accordingly. The tumor inhibition rate was calculated according to the following equation: the tumor inhibition rate = (W1 − W2)/W1 × 100%, where W1 represents the averaged tumor weight in the control group, and W2 represents the tumor weight for each mouse in the treatment groups.
Hematoxylin and Eosin Staining
The tumor tissues fixed with 10% formaldehyde for 1 day and then embedded in paraffin. The samples were cut into 5-μm sections on a M390006 paraffin slicing machine (Leica, Solms, Germany). Sections were then subjected to hematoxylin and eosin (H&E) staining according to the standard protocol and observed with light microscopy.
Electron Microscopy
Subcellular structure was detected with the electron microscopy (EM). The tumor tissues were fixed with 4% glutaraldehyde for 1 day and then embedded in paraffin. The sample was observed by a transmission electron microscope (Leica).
Immunohistochemistry
The expression of IL-11R, Ki-67, and Bcl-2 was detected by immunohistochemistry. On day 16 after treatment, the tumor was excised, and immunohistochemistry was performed with a commercially available kit (ZSGB-BIO), according to the manufacturer’s instructions.
Statistical Analysis
Data were expressed as mean ± standard deviation. SPSS 14.0 software was used for statistical analysis. The paired t test and analysis of variance were performed for the group comparison, with the least significant difference test for data of homogenous variance, and the Games-Howell test for data of nonhomogeneous variance. P < .05 was considered as statistically significant.
Results
Detection of IL-11R Expression Levels in Various Tumor Cell Lines
To investigate the expression levels of IL-11R in various human tumor cell lines and normal liver cells, the Western blot analysis was performed. Our results showed that, compared with the normal cells, the expression levels of IL-11R in these tumor cell lines were significantly elevated, by 2- to 5-fold (all P < .05; Figure 1). Particularly, the expression level of IL-11R in the MHCC97-H cells with high metastatic potential was dramatically increased (P < .05). The upregulated expression levels of IL-11R in these tumor cell lines might imply the role of IL-11R in the disease pathogenesis.

Interleukin-11 receptor (IL-11R) expression levels in various tumor cell lines and normal cells. A, The expression levels of IL-11R in MHCC97-H and MHCC97-L human hepatoma cell lines, PC-3 prostate cancer cell line, MDA-MB-231 and MCF-7 breast cancer cell lines, SKOV3 ovarian cancer cell line, HeLa cervical cancer cell line, and HHL-5 normal human liver cell line were detected by Western blot analysis. B, Statistical analysis of the expression levels of IL-11R. Compared with the control group, *P < .05.
Characterization of 153Sm-DTPA-c(CGRRAGGSC) in MHCC97-H Cells
The products of 153Sm-DTPA-c(CGRRAGGSC) was first analyzed with HPLC. The quality control results showed that the labeling rate for 153Sm-DTPA-c(CGRRAGGSC) was >85%, the radiochemical purity was >96.4%, and the specific activity was 1.32 × 105 MBq/μmol. The radiochemical purities for 153Sm-DTPA-c(CGRRAGGSC) at 0, 2, 4, 8, 12, 24, 36, and 48 hours after preparation at room temperature were 99.79% ± 0.23%, 99.37% ± 012%, 99.14% ± 0.09%, 98.73% ± 0.26%, 97.81% ± 0.35%, 96.51% ± 0.42%, 94.22% ± 0.69%, and 89.65% ± 0.74%, respectively. The radiopharmaceutic could stay stable for 32 hours at 4°C in saline, with the radiochemical purity of 95.7%. These results suggested that 153Sm-DTPA-c(CGRRAGGSC) was suitable for the following experiments.
To characterize the biological activities of 153Sm-DTPA-c(CGRRAGGSC), the radioligand binding assay and competitive inhibition experiment were performed in MHCC97-H cells. Our results from the radioligand binding assay showed that the SB count per minute was increased along with the increasing concentrations of 153Sm-DTPA-c(CGRRAGGSC); however, the increasing rate was gradually declined, suggesting the saturation trend (Figure 2A). Moreover, the Scatchard plot was essentially linear, revealing the Kd value of 0.38 ± 0.05 nmol/L and the Bmax of 768 ± 52 fmol/mg protein (Figure 2B). In addition, the results from the competitive inhibition experiment showed that along with the increasing concentrations of nonlabeled c(CGRRAGGSC) in the reaction system, the binding of 153Sm-DTPA-c(CGRRAGGSC) to the receptor was gradually decreased, with no significant differences in the binding activities between nonlabeled and labeled ligands (Figure 2C). The 50% inhibiting concentration (IC50 value) was 7.56 ± 0.22 nmol/L, and the Ki value was 3.02 ± 0.19 nmol/L. These results suggest that the biological activity of the cyclic peptide c(CGRRAGGSC) is not significantly altered after labeling, and the radiolabeled peptide exhibits sufficient binding affinity to IL-11R.

Characterization of 153Sm-DTPA-c(CGRRAGGSC) in MHCC97-H cells. The biological activity of prepared 153Sm-DTPA-c(CGRRAGGSC) in MHCC97-H cells was characterized with the radioligand binding assay (A and B) and the competitive inhibition experiment (C). A, The saturation curve. B, The Scatchard plot. C, The competitive inhibition curve. In these curves: FB = T − TB, and SB = TB − -NSB (T, total amount of radioactivity; TB, total binding; SB, specific binding; NSB, non-specific binding; FB, free ligands).
Localization of Cyclic Peptide c(CGRRAGGSC) in MHCC97-H Cells
The localization of DTPA-c(CGRRAGGSC) labeled with FITC in MHCC97-H cells was then observed with fluorescence microscopy. Our results showed that FITC-c(CGRRAGGSC)-DTPA was mainly distributed in the cytoplasm and on the cell membrane of MHCC97H cells (Figure 3). However, no obvious staining was observed in the nuclei. Moreover, HHL-5 normal human liver cells did not show any positive staining for FITC-c(CGRRAGGSC)-DTPA (Figure 3). These results suggest that the cyclic peptides might target on the IL-11R, which is abundantly expressed in the cytoplasm and on the cell membrane of MHCC97-H cells.
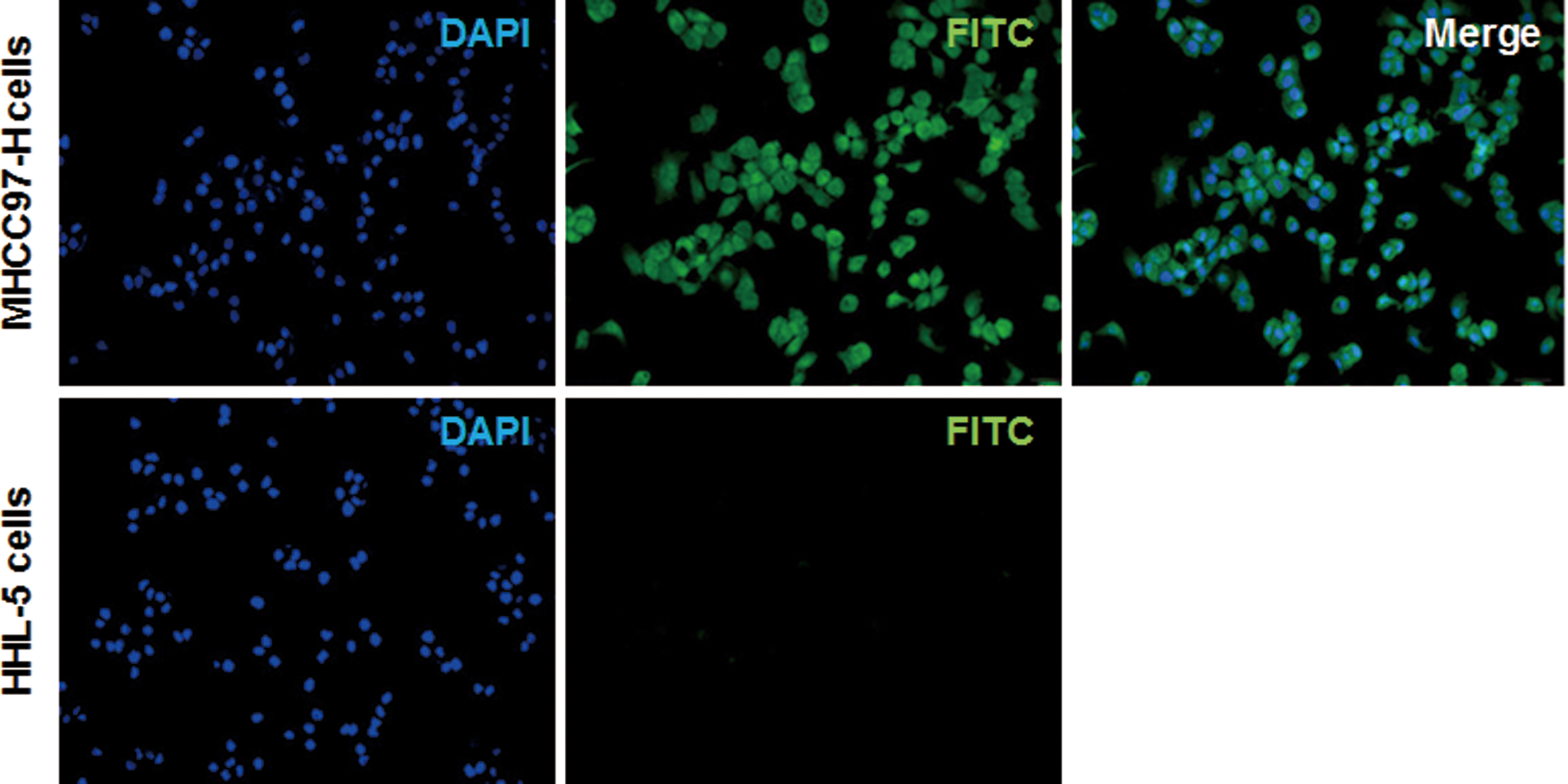
Localization of cyclic peptide c(CGRRAGGSC) in MHCC97-H cells. The binding and localization of DTPA-c(CGRRAGGSC) labeled with fluorescein isothiocyanate (FITC; green) in MHCC97-H cells, and control cells were observed with fluorescence microscopy (×1000). Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI; blue).
Effects of 153Sm-DTPA-c(CGRRAGGSC) on Implanted Tumors in Nude Mice
The effects of 153Sm-DTPA-c(CGRRAGGSC) on implanted MHCC97-H tumor cells in nude mice were next investigated. Totally 1 × 106 MHCC97-H cells were subcutaneously implanted into each nude mouse under the armpit of the right forelimb, and the radiopharmaceutic was administered when the implanted tumors grew to 0.8-1.0 cm in diameter. After drug administration, the health status of these mice was assessed every day. No significant abnormality was observed in the mouse performance. Results from the visual observation showed that, in the treatment groups, at the early stage, necrosis and crust formation were noted in the tumors, and at the later stage, the peripheral tumor tissue continued to grow, which was, however, much slower than that in the control group. These mice were then killed on day 16 after drug treatment, and the tumor sizes and weights were measured. As shown in Figure 4, our results showed that, compared with the control group, the tumor growth was significantly inhibited in the treatment groups. In particular, the most obvious inhibiting effects were observed in the high-dose treatment group. These results showed that 153Sm-DTPA-c(CGRRAGGSC) could significantly inhibit the in vivo tumor growth.

Effects of 153Sm-DTPA-c(CGRRAGGSC) on in vivo tumor growth. Totally 1 × 106 MHCC97-H cells were subcutaneously implanted into each nude mouse under the armpit of the right forelimb, and 153Sm-DTPA-c(CGRRAGGSC) was administered when the implanted tumors grew to 0.8-1.0 cm in diameter. These mice were killed on day 16 after drug treatment. The tumor sizes (A) and weights (B) were measured, and the inhibitory rates (C) were calculated accordingly. Compared with the control group, *P < .05; compared with the previous group, # P < .05.
Histological Characterization of Implanted Tumors in Nude Mice Treated With 153Sm-DTPA-c(CGRRAGGSC)
Tumor tissues in the control and treatment groups were then assessed with H&E staining, EM, and immunohistochemical staining. In order to minimize interference of the radiolabeled cyclic peptide on the physiological activities of these model mice, only the tumor tissues from the low-dose group were analyzed. Our results from the H&E staining showed that, in the control group, the implanted tumors grew vigorously, and the tumor cells were tightly packed, distributed in cords and nests. However, in the low-dose treatment group, the tumor cells were loosely arranged, with large areas of coagulation necrosis and fibrous connective tissue (Figure 5A). Results from EM showed that, in the control group, the nucleoli were apparent, with evident karyokinesis. However, in the low-dose treatment group, intracellular vacuoles were noted, and the karyokinesis was not evident, while karyopyknosis, karyorrhexis, and karyolysis were commonly observed (Figure 5B). In addition, the immunohistochemical staining showed that IL-11R was mainly expressed on the cell membrane of MHCC97-H cells, which a little expression in the cytoplasm (Figure 5C and D). In the control group, the positive rate for IL-11R in the tumor cells was 84.13% ± 5.71%, while in the low-dose treatment group, the positive rate for IL-11R was 61.57% ± 5.98% (P < .05; Figure 5C and D). Moreover, the expression levels of Ki67 and Bcl-2 were both significantly decreased in the low-dose treatment group, compared to the control group (Figure 5C and D). Taken together, these results further confirm that the implanted tumors would be inhibited by the treatment of 153Sm-DTPA-c(CGRRAGGSC).

Histological characterization of implanted tumors treated with 153Sm-DTPA-c(CGRRAGGSC). A and B, Implanted tumor tissues in the control and low-dose treatment group were assessed with hematoxylin and eosin (H&E) staining (A; ×100) and electron microscopy (EM; B; ×5000). C, Implanted tumor tissues in the control and low-dose treatment group were assessed with the immunohistochemical staining (×200). Compared with the control group, *P < .05.
Effects of 153Sm-DTPA-c(CGRRAGGSC) on IL-11R Expression in Implanted Tumors
To investigate the mechanism through which 153Sm-DTPA-c(CGRRAGGSC) exert the inhibitory effect on implanted tumors in nude mice, the expression level of IL-11R in the tumors was detected by Western blot analysis. Our results showed that, compared with the control group, the expression levels of IL-11R were significantly decreased in the treatment groups (all P < .05; Figure 6), with more obviously declined expression levels in the higher dose group. These results suggest that 153Sm-DTPA-c(CGRRAGGSC) would target on IL-11R to inhibit the proliferation of MHCC97-H tumor cells in nude mice.
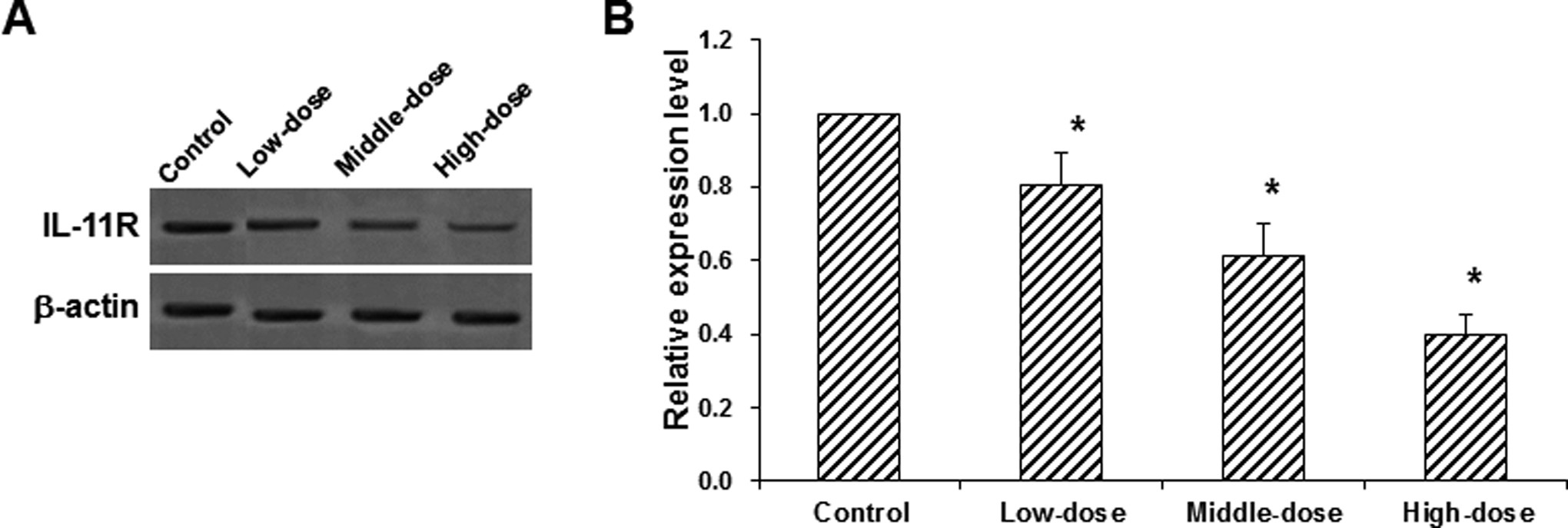
Effects of 153Sm-DTPA-c(CGRRAGGSC) on interleukin-11 receptor (IL-11R) expression in implanted tumors. A, The expression levels of IL-11R in the implanted tumors in nude mice treated with 153Sm-DTPA-c(CGRRAGGSC) were detected with Western blot analysis. B, Statistical analysis of the expression levels of IL-11R. Compared with the control group, *P < .05.
Discussion
It has been reported that the expression levels of IL-11 and its receptor IL-11R are elevated in various tumors. 3 –7 However, the roles of IL-11 and IL-11R in the pathogenesis of liver cancer have not yet been fully addressed. Pathogenesis and development of liver cancer could be attributed to a variety of pathways and factors. Specifically, infection of hepatitis B virus (HBV) or hepatitis C virus (HCV) plays a significant role in the disease initiation and progression. The HBV- and HCV-related liver cancer cells are also prone to metastasis. The MHCC97-H human liver cancer cell line is characterized by high metastatic potential and stable biological activity and represents an ideal model for the investigation of tumor recurrence and metastasis. In the present study, the expression levels of IL-11R in various tumor cell lines and normal cells were detected by Western blot analysis. Our results showed that the MHCC97-H cells exhibited the highest expression level of IL-11R, which was 5-fold higher than that in the normal cells.
The in vivo targeting specificity of radiopharmaceutic is important to ensure the treatment efficacy for liver cancer metastasis and recurrence. Since the radionuclide 153Sm was readily available and suitable for both γ-ray diagnostic imaging and β-ray radiation therapy, it was selected in this study to label the cyclic peptide c(CGRRAGGSC). The binding specificity of 153Sm-DTPA-c(CGRRAGGSC) to MHCC97-H cells and the biological activity of the peptide were first assessed. Our results showed that the biological activity of the peptide was not significantly altered after labeling, and the Kd and Bmax values revealed the sufficient affinity of 153Sm-DTPA-c(CGRRAGGSC) to IL-11R. Next, the FITC-c(CGRRAGGSC)-DTPA probe was constructed. The fluorescence microscopy indicated that the probe could specifically bind to MHCC97-H cells, without binding to HHL-5 normal cells, further implying the affinity of c(CGRRAGGSC) to MHCC97-H cell with high expression of IL-11R.
For the in vivo experiments, due to the overall toxicity that would be induced by the high dosage and to minimize the interference on the physiological activities of model mice, the tumor tissues from the low-dose group were analyzed herein. Our in vivo experiments showed that the rapid tumor growth was observed in the control group and low-dose group, while the tumor growth in the middle- and high-dose groups was much slower, with statistical significance. Moreover, significant differences were observed in the tumor size on day 16 after treatment between the 153Sm-DTPA-c(CGRRAGGSC)-treated groups and the control group. These results indicate the high affinity of 153Sm-DTPA-c(CGRRAGGSC) to the implanted MHCC97-H tumor cells, and the radiopharmaceutic injected via the tail vein at different dosages could rapidly and effectively concentrate in the tumor tissues, other than in the blood circulation and other organs.
Light and EM showed that, in the low-dose group, tumor cells were loosely arranged, with large areas of coagulation necrosis and fibrous connective tissue. In the remaining foci, the tumor cell volume was enlarged, and the cell membrane was damaged, with intracellular vacuoles. The karyokinesis was not evident, while karyopyknosis, karyorrhexis, and karyolysis were commonly observed. Along with the increasing tumor inhibitory effects, more significant necrosis would be noted. Moreover, the results from the Western blot analysis showed that the IL-11R expression level was changed in tumor tissues after the radioactive drug intervention. These results suggested that 153Sm-DTPA-c(CGRRAGGSC) could specifically bind to IL-11R in tumors, eliminating the tumor cells. Significant relationship was observed between the tumor inhibitory effect and the drug concentration. On the other hand, the normal in vivo cells with little or no expression of IL-11R would escape from the killing effects of the radiopharmaceutic. Based on these results, 153Sm-DTPA-c(CGRRAGGSC) could achieve high tumor specificity in vivo.
Ki67 is a nuclear antigen closely related to cell proliferation, which is used as an important marker to evaluate the tumor cell proliferation. 14 Our results from the immunohistochemistry showed that the positive rate for Ki67 was significantly decreased in the low-dose treatment group compared with the control group, suggesting that 153Sm-DTPA-c(CGRRAGGSC) could inhibit the proliferation of tumor cells. Moreover, the downregulated expression levels of IL-11R in the treatment groups suggested that 153Sm-DTPA-c(CGRRAGGSC) could target on MHCC97-H tumor cells and inhibit the cell proliferation. Bcl-2 could specifically inhibit cellular apoptosis and protect cells against apoptosis induced by a variety of pathological factors. It has been shown that the expression levels of Bcl-2 are significantly decreased in apoptotic tumor cells. 15,16 Our previous study has shown that 153Sm-DTPA-c(CGRRAGGSC) could not only directly kill tumor cells but also increase the apoptosis rate of these cells and induce the cell cycle arrest in the G and/or G1 phase. 11 In this study, our results showed that the Bcl-2 expression rates in the treatment groups were significantly lower than in the control group, suggesting that the inhibitory effects of 153Sm-DTPA-c(CGRRAGGSC) on tumor cells would be mediated by multiple mechanisms, which might be associated with the synergistic effects of the apoptosis-enhancing activity and the cell killing effect.
In conclusion, our results showed that 153Sm-DTPA-c (CGRRAGGSC) could specifically bind to IL-11R on MHCC97-H human liver tumor cells, inhibiting the cell proliferation and inducing cellular apoptosis. These findings provide experimental evidence for the development of individual treatment of liver cancers, as well as recurrence and metastasis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Joint Research Project from the Hospital Management Center of Wuxi City (No. YGZX-1213) and the National Natural Science Foundation Western Plan (No. 81560297).
