Abstract
Proton therapy dose is affected by relative biological effectiveness differently than X-ray therapies. The current clinically accepted weighting factor is 1.1 at all positions along the depth–dose profile. However, the relative biological effectiveness correlates with the linear energy transfer, cell or tissue type, and the dose per fraction causing variation of relative biological effectiveness along the depth–dose profile. In this article, we present a simple relative biological effectiveness-weighted treatment planning risk assessment algorithm in 2-dimensions and compare the results with those derived using the standard relative biological effectiveness of 1.1. The isodose distribution profiles for beams were accomplished using matrices that represent coplanar intersecting beams. These matrices were combined and contoured using MATLAB to achieve the distribution of dose. There are some important differences in dose distribution between the dose profiles resulting from the use of relative biological effectiveness = 1.1 and the empirically derived depth-dependent values of relative biological effectiveness. Significant hot spots of up to twice the intended dose are indicated in some beam configurations. This simple and rapid risk analysis could quickly evaluate the safety of various dose delivery schema.
Keywords
Introduction
The advantage of proton beam therapy over conventional radiotherapy is an improvement in the conformality of the energy deposition inside the patient. This improvement arises from Bragg peak at the end of the proton range. 1 -3 High-density ionization events along with low energy proton tracks result in increased irreparable DNA damage that is the physical origin of an enhanced biological efficiency or relative biological effectiveness (RBE). 4 The RBE can be measured by cell survival experiments or derived by biophysical models. 5 Proton radiation has been shown to be more biologically effective for human cell killing compared with X-rays 6,1 and so the standard of practice employs a dosimetric weighting factor between 1.1 and 1.2. 7 -9 However because RBE correlates with track structure, it increases near the end of range, at the Bragg peak. Relative biological effectiveness is derived from several Bragg peaks at several depths weighted to produce a flat physical dose with each peak contributing a pocket of densely ionizing track structures.
Several published studies support this intuition that from the midpoint to the distal side of spread out Bragg peak (SOBP), the RBE increases to a maximum of as much as 3 just
10
-15
beyond the distal dose falloff (DDF). The RBE value increases from 1.1 at the absorber entrance to as much as 1.6 at the midpoint of the SOBP
11
and to as much as 2.9 in the DDF.12 Carabe
Variation in RBE over the SOBP could result in variations in biological dose that are not apparent when a uniform RBE of 1.1 is used to calculate the treatment plan. This article presents a simple technique to enable visualization of proton biological dose in 2-dimensions and compares that with standard proton dose using a uniform RBE of 1.1. The isodose distribution profiles were accomplished using matrices that represent coplanar intersecting beams. These matrices were combined and contoured to represent the distribution of dose using standard RBE weighting values (
Materials and Methods
Excel and MATLAB programs were used to construct biological isodose distributions of an artificial proton beam using either
The data for this test of the methodology were mathematically derived. The RBE values were scaled using a relative depth within the 30 mm SOBP. The published data were used to establish percentage depth positions of RBE data.
The scaled position of each point of interest within the SOBP (X(n)) is calculated:

where
Two dosimetry profiles were constructed for comparison. In addition to the isodose profile generated using the standard weighting factor
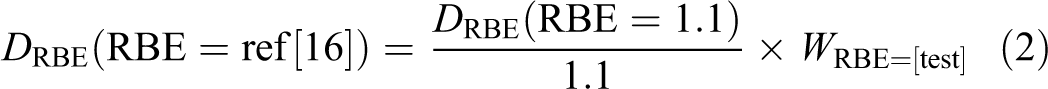
where
Microsoft Excel 2013 was used to develop a matrix. Each column of the matrix contains the values of the X-ray dose present in y for the transverse section of the treatment object and each row contains the values of the X-ray dose present in x for the transverse section. Each element in the matrix represents the value of the fractional dose taken from the digital data of the dose-depth profile.
The transverse section of the treatment object is assumed to be a 9 cm diameter circle, and the planning target volume (PTV) is a concentric circle with a 30 mm diameter. Thus, the matrix transverse section is 89 × 89 mm2. The width of the modeled proton beam is 30 mm and it is constructed within the matrix with the planned orientation. The matrix is filled manually for beam angles of 0°, 90°, 180°, 225°, 270°, and 315°, for
Results
A simple visualization of biological dose was created using MatLab to construct a circular PTV within a homogeneous circular treatment target volume (TTV) in 2-dimensional space. The standard RBE of 1.1 was assumed throughout an SOBP that was adjusted to cover the 3.0 cm diameter PTV. This model was used to compare a variety of beam configurations designed to provide an even dose distribution at RBE = 1.1, planned to the 90% isodose over the PTV. These plans were then compared with isodose profiles that would result from applying the depth dependent, 10% survival RBE values reported by Britten

The 30 mm spread out Bragg peak (SOBP) depth–dose profiles with
The modeled plan represents a transverse section of a 9 cm diameter circular TTV with a concentric PTV of 3 cm diameter, intersected by proton beams (30 mm width, 80% to 80%). Two, 3, 4, 5, and 6 beams, with 45°, 90°, 135°, and 180° separation, intersect the PTV such that their DDF is at the distal side of the PTV with an SOBP of 30 mm (90% to 90%). The dose distributions for these beams were first calculated using the standard uniform
When the

Isodose distribution profiles of 2 proton beams with 45° of separation (0° and 315°) range = 60 mm, spread out Bragg peak (SOBP) = 30 mm. A,

Isodose distribution profile of parallel opposed proton beams at 0° and 180°. A,
If a third field is added to the parallel opposed plan such that the separation is 90° between the beams, the influence of the increased RBE over the second half of the SOBP of the third beam can be visualized readily in the increased dose throughout the upper half of the box. The difference between the calculated dose distributions indicates both an increase in the excess dose over that produced by 2 opposing beams at the distal periphery of the 3 beams and increased dose to more than half of the treatment volume. For 3 beams with a 45° angle between them, the hot spot of the corrected
When the angle between 3 modeled beams is 90°, the hot spot visualized when using the corrected
Adding a fourth field to the 45° offsets reduces the area encompassed by the 30% dose excess, but the hot spots created by incorporating the corrected

Isodose distribution profile of 4 proton beams at 0°, 90°, 180°, and 270°. A,
Additional fields are not effective at reducing either the distal or central hot spots. The configurations for 5 and 6 beam distributions (Figure 5) display corrected

Isodose distribution profile of 6 proton beams with 0°, 90°, 180°, 225°, 270°, and 315°. A,
Discussion and Conclusion
The current standard of practice in proton therapy clinics is to use a biological effectiveness correction factor based on empirical evidence
8
to account for the increased RBE of proton radiation over X-ray radiation. This weighting factor,
The application of weighting factors not equal to 1.1 indicates hot or cold spots in the dose distribution within the target or excessive dose to healthy tissue. The techniques developed herein disclose some significant differences between the clinically applied
The variation in biological dose depends on the number of proton fields and the angles between them because the increased
What is presented is a simple method for evaluating potential risk. This method can be used to rapidly assess the risk of any clinically planned treatment. For argument’s sake, suppose that a maximum increase in biological dose of 25% greater than the dose determined using the standard software
Important factors that cannot be considered in this work because of their complexity include the anatomical placement and extent of the hot spot. For example, a hot spot aligned with a particularly susceptible tissue mass may result in higher risk. Several factors are patient and organ specific and lie outside the scope of this work. Mitigation could be accomplished by slightly reducing the incident beam energy for some number of fractions as suggested by Buchsbaum
Nonetheless, this work and the work of others 8,14,17,30,35 make it clear that although we have an excellent understanding of the dose deposition physics of charged particle irradiation, we do not have sufficient understanding of the biological responses to that energy absorption. Because it is the biological response that is driving the clinical prognosis, this uncertainty is quite compelling.
In time, this methodology could be optimized and placed into treatment planning scenarios much like we use raw dose in the current process. Treatment planning systems (TPSs) currently are beginning to look at biologic optimization. What we envision as this project’s ultimate use is the real-time generation of biologic dose in a TPS that can then be modulated via the allowance of nonlinear SOBP shape so as to make biologic dose more homogeneous. This, in essence, means inverting the current process of optimizing physical dose and accepting biologic dose to optimize biological dose and accepting physical dose. This would have to done over time as the work to date in clinical radiation oncology is completely based on physical dose. It would also require a major change in how we define beams as we would be very unlikely to use SOBP’s designed to have a flat physical dose as we currently do. Ultimately, the delivery of proton treatment plans would be made up of series of unique mixtures of pristine Bragg peaks rather than SOBPs. In the end, the visualization of biologic dose is crucial in providing the safest possible treatment plans to patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
