Abstract
Lung cancer is one of the most common causes of cancer-related deaths worldwide. Although great efforts and progressions have been made in the study of the lung cancer in the recent decades, the mechanism of lung cancer formation remains elusive. To establish effective therapeutic methods, new targets implied in lung cancer processes have to be identified. Tripartite motif-containing 25 has been associated with ovarian and breast cancer and is thought to positively promote cell growth by targeting the cell cycle. However, whether tripartite motif-containing 25 has a function in lung cancer development remains unknown. In this study, we found that tripartite motif-containing 25 was overexpressed in human lung cancer tissues. Expression of tripartite motif-containing 25 in lung cancer cells is important for cell proliferation and migration. Knockdown of tripartite motif-containing 25 markedly reduced proliferation of lung cancer cells both
Introduction
Lung cancer is the leading cause of cancer deaths worldwide among both men and women. 1 Lung cancers develop through a multistep process involving development of multiple genetic and epigenetic alterations, particularly activation of growth-promoting pathways and inhibition of tumor suppressor pathways. 2,3 Greater understanding of the multiple biochemical pathways involved in the molecular pathogenesis of lung cancer is crucial to the development of treatment strategies that can target molecular aberrations and their downstream activated pathways. 4,5 Identification of the molecular markers determining the risk of occurrence and progression and approaches for therapeutic treatment of lung cancer are the most significantly important problems in molecular oncology.
The tripartite motif-containing (TRIM) proteins are characterized by an N-terminal zinc finger really interesting new gene (RING) domain, 1 or 2 Bbox domains, and a coiled-coil domain (CCD). The RING domain confers ubiquitin E3 ligase activity, the function of the B-box domain is largely unknown, and the CCD is implicated in multimerization of TRIM proteins. 6,7 The TRIM25 was originally identified as an estrogen-responsive gene and plays a key role in the development of the uterus. 8 The TRIM25 has been identified as a key component of the retinoicacidinduciblegene-1 (RIG-I) signaling pathway. 9 The TRIM25 acts as a positive regulator of the type I interferon (IFN) production pathway by activating RIG-I in the initial stages of viral infection. 9 More recently expression of TRIM25 has also been associated with ovarian and breast cancer and is thought to positively promote cell growth by targeting the cell cycle regulator 14-3-3 sigma for proteasomal degradation. 10 -12 Therefore, TRIM25 may act as a tumor promoter, but whether TRIM25 has a function in lung cancer development remains unknown.
This study showed that TRIM25 expression was increased in lung cancer tissues and lung cancer cell lines. The TRIM25 is important for proliferation and migration of lung cancer cell. Knockdown of TRIM25 markedly reduced
Material and Methods
Chemicals and Antibodies
Lipofectamine 2000 transfection and TRIZOL LS Reagents were purchased from Invitrogen (Grand Island, New York). Antibodies against TRIM25, p53, and MDM2 were purchased from Abcam (Cambridge, Massachusetts). β-Actin antibody was from Cell Signaling Technology (Danvers, Massachusetts). Unless otherwise noted, all other chemicals were from Sigma (Shanghai, China).
Patients and Specimens
Paraffin-embedded, archived tissue samples were obtained from 9 normal lung tissues and 38 patients diagnosed with lung cancer between January 2012 and October 2013 at the Department of Pathology, Affiliated Hospital, Shandong University. The histologic characterization and clinicopathologic staging of the samples were determined according to the World Health Organization criteria and current International Union Against Cancer tumor node metastasis Classification.
Histological and Immunohistochemical Analysis
The normal human lung tissues and human lung tumor tissues were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS) overnight and subsequently embedded in paraffin wax. Sections cut at a thickness of 4 μm were stained with hematoxylin and eosin for histological analysis. Immunohistochemical (IHC) analysis was performed for different markers in these arrays as described previously. The proportion of stained cells was semiquantitatively determined following published protocols.
Cell Culture
Lung cancer cell lines (ATCC, Manassas, Virginia) were cultured under the following conditions: H1650, A549, H1299, H460, A427, PAa, and 95D cell lines were cultured using 10% fetal bovine serum (Cat#10099-141; Invitrogen, Carlsbad, California) in RPMI-1640 (Cat#C11875; Invitrogen). BEAS2B cell line was cultured using 10% fetal bovine serum (Invitrogen) in Dulbecco modified Eagle medium (DMEM, Cat#C11965; Invitrogen). Cell culture was according to manufacturer’s protocol. All the cell lines were grown at 37°C in a 5% CO2/95% air atmosphere and were revived every 3 to 4 months.
Knockdown of TRIM25 Gene With Small-Interfering RNA and Establishment of TRIM25 Stable Knockdown Cell Lines
Cells were seeded into 6-well plates at 2 × 105 cells per well, grown for 24 hours in complete medium, and then transfected with ON-TARGET plus Smart pool small interfering RNA (siRNA) specific to TRIM25 or scrambled siRNA-negative control (Dharmacon, Chicago, Illinois) at a final concentration of 100 nmol/L using Hyperfect transfection reagent (Qiagen, Courtaboeuf Cedex, France) according to the manufacturer’s directions. Note that 2 different siRNAs against nonoverlapping messenger RNA (mRNA) sequence of TRIM25 were applied. To examine RNAi efficiency, cells were harvested 2 days after siRNA transfection and analyzed by Western blots. For establishment of TRIM25 stable knockdown cell lines, retroviral construct containing pSuper with short hairpin RNA (shRNA) against human TRIM25 and pSuper were prepared as described previously. The generation of retrovirus supernatants and transfection of lung cancer cells were conducted as described previously. The expression of TRIM25 was confirmed by Western blotting analysis.
Colony Formation Assay
Cells were trypsinized and plated (400 cells/well) in 6-well tissue culture plates. Cells were incubated in 5% CO2 at 37°C for a total of 3 weeks, and colonies were washed, fixed, and stained with 0.005% violet in methanol. Numbers of colonies were manually counted.
Cell Invasion and Motility Assay
Invasion of cells was measured in Matrigel (BD, Franklin Lakes, New Jersey)-coated Transwell inserts (6.5 mm; Costar, Manassas, Virginia) containing polycarbonate filters with 8-μm pores as detailed previously. 13 The inserts were coated with 50 μL of 1 mg/mL Matrigel matrix according to the manufacturer’s recommendations. The 2 × 105 cells in 200 μL of serum-free medium were plated in the upper chamber, whereas 600 μL of medium with 10% fetal bovine serum was added to lower well. After 24 hours incubation, cells that migrated to the lower surface of the membrane were fixed and stained. For each membrane, 5 random fields were counted at 10× magnification. Motility assays were similar to Matrigel invasion assay except that the Transwell insert was not coated with Matrigel.
Cell Death Detection
Following treatments, cells were harvested with trypsin/EDTA, suspended in PBS, and mixed with an equal amount of 0.4% trypan blue stain (Invitrogen). Viable cells maintained membrane integrity and did not take up trypan blue. Cells with compromised cell membranes took up trypan blue and were counted as dead. Cells were counted in 5 different fields, and the number of dead cells was calculated as percentage of the total cell population.
Apoptosis Fluorescence-Activated Cell Sorting Analysis
After treatment, cells were washed in PBS and fixed with ice-cold 70% ethanol overnight. Cells were then suspended in PBS containing RNase A (100 mg/mL), propidium iodide (50 mg/mL), Annexin V-FITC (50 mg/mL), and 0.1% Triton X-100, and incubated in the dark for 1 hour. Fluorescence-activated cell sorting (FACS) was performed to quantify the apoptotic population showed by Annexin V staining.
Western Blotting
Cells were lysed in lysis buffer, and total protein contents were determined by the Bradford method. The 30 μg of lysis were separated by reducing sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and probed with specific antibodies. Blots were washed and probed with respective secondary peroxidase-conjugated antibodies, and the bands visualized by chemoluminescence (Amersham Biosciences, Shanghai, China).
Apoptosis ELISA Assay
Apoptosis in lung cancer cells was also measured using the Cell Death Detection ELISA PLUS kit (Roche Diagnostics, Indianapolis, Indiana), which detects histone-complexed DNA fragments in cell cytoplasm. 14 Cells were seeded into a 96-well dish at a density of 1 × 104 cells/ well. Following applied treatments, cell lysates were prepared following the manufacturer’s procedure and incubated with an antihistone, biotin-conjugated antibody in a microplate with streptavidin-coated walls. After washing and incubation with peroxidase substrate, histone-complexed DNA fragments in the samples were quantified by reading the plate at 405 nm on a BenchMark Plus microplate spectrophotometer (Bio-Rad, Nanjing, China).
Bromodeoxyuridine Incorporation Assay
Lung cancer cells (1 × 105 cells/ well) were seeded in 0.5 mL DMEM containing 10% FBS onto the 48-well tissue culture plates. After culture for 72 hours , the cell proliferation was assessed using bromodeoxyuridine (BrdU) incorporation though the BrdU ELISA colorimetric assays (Roche) according to the manufacturer’s protocol. The ELISA optical density value was recorded as indicator of cell proliferation. Each condition was tested in triplicate.
Coimmunoprecipitation
Aliquots of 1000 mg of proteins (in 1 mL of lysis buffer) of lung cancer tissues or in lung cancer cells were precleared by incubation with 30 mL of protein A/G Sepharose (beads; Sigma) for 1 hour at 4°C. The precleared samples were incubated with the anti-TRIM25 (1 mg/mL) in lysis buffer overnight at 4°C. Thereafter, 30 mL of protein A/G beads were added, and the samples were incubated for 2 hours at 4°C. The beads were washed 5 times with PBS (4°C) and once with the lysis buffer, boiled, separated by 10% SDS-PAGE, and transferred onto a polyvinylidene fluoride membrane.
Xenografted Tumor Model In Vivo
BALB/c nude mice (4-5 weeks of age, 18-20 g) were purchased from the Center of Experimental Animal of Shandong University and were housed in barrier facilities on a 12-hour light–dark cycle. All experimental procedures were approved by the Institutional Animal Care and Use Committee of Shandong University. The BALB/c nude mice were randomly divided into 2 groups (n = 5/group). One group of mice were inoculated subcutaneously with A427-pSuper cells (1 × 106, suspended in 100 μL sterile PBS) per mouse in the right oxter as control group. The other group were inoculated with A427-shTRIM25 cells (1 × 106, suspended in 100 μL sterile PBS), respectively. Tumor volume was calculated using the equation (L ×W2)/2.
Statistical Analysis
The results were analyzed using SPSS 18.0 software (Chicago, Illinois). Each experiment was repeated a minimum of 3 times. A 2-tailed
Results
TRIM25 is Overexpressed in Human Malignant Lungs
To test whether TRIM25 may function as a tumor promoter in lung cancer, we first examined the expression of TRIM25 in 9 human normal lung tissues and lung cancer tissues via Western blot. As shown in Figure 1A and B, in all human lung cancer tissues, expression of TRIM25 was significantly higher than that in surrounding normal tissues. We then analyzed TRIM25 expression in more lung cancer tissue samples (n = 38) by IHC, and we also found that expression of TRIM25 was significantly increased in lung cancer tissues (Figure 1C). These results indicated that TRIM25 may play a possible role in the development or progression of lung cancer.

The TRIM25 is overexpressed in human lung cancer. The TRIM25 and β-actin (the loading control) expression in 9 human lung cancer tissues (“T”) or in surrounded normal tissues (“N”) was tested by Western blots (A), relative TRIM25 expression (vs β-actin, 9 clinical tissues for each group) was quantified (B). Representative immunohistochemical (IHC) images showed TRIM25 protein expression in human lung cancer tissues or in normal tissue (C). Data were presented as mean ± standard deviation (SD). **
Small Interfering RNA Knockdown of TRIM25 Inhibits Lung Cancer Cell Proliferation In Vitro
Above-mentioned results clearly demonstrated that TRIM25 is overexpressed in human lung cancer tissues. To study the potential role of TRIM25 in lung cancer cells, siRNA strategy was then applied. First, we measured the expression levels of TRIM25 in 7 lung cancer cell lines (A549, H1299, A427, H460, PAa, 95D, and H1650) and 1 normal lung cell line (BEAS2B) by Western blot. The results show that levels of TRIM25 were significantly increased in nearly all tumor cell lines compared with the BEAS2B cell (Figure 2A). We used siRNA to generate TRIM25 knockdown in the A427 cell line. Two TRIM25 siRNAs, each corresponding to nonoverlapping sequence of TRIM25 mRNA, were transfected into A427 cells to selectively knockdown TRIM25. Western blot results in Figure 2B demonstrated that both siRNAs dramatically downregulated expression of TRIM25 in A427 cells. A427 cells proliferation, tested by simple cell counting (Figure 2C), clonogenicity assay (Figure 2D and E), and BrdU incorporation assay (Figure 2F), was significantly inhibited by the 2 TRIM25 siRNAs.

The siRNA knockdown of TRIM25 inhibits lung cancer cell proliferation
Short Hairpin RNA Knockdown of TRIM25 Inhibits Lung Cancer Cell Tumorigenesis In Vivo
To extend our
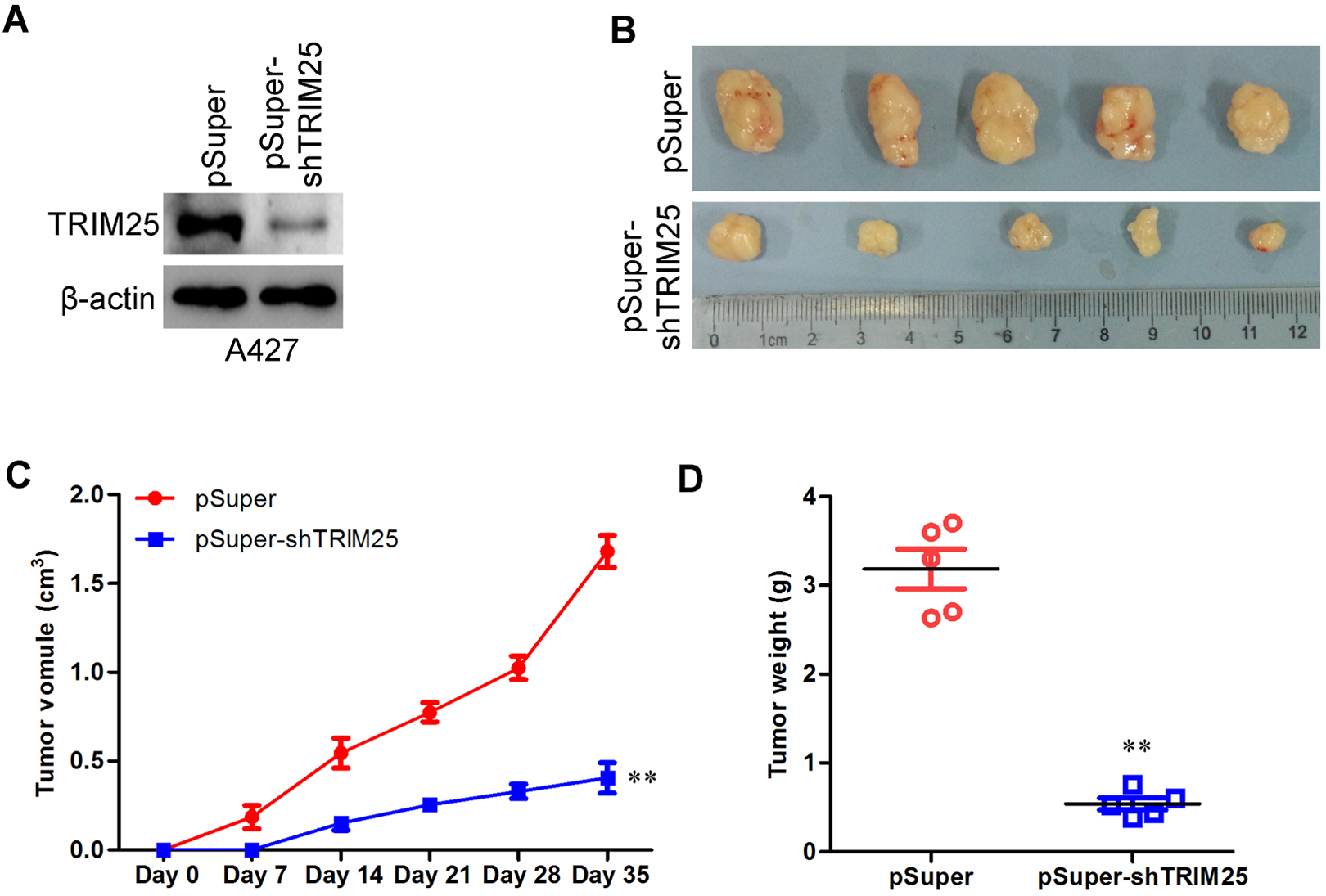
The shRNA knockdown of TRIM25 inhibits lung cancer cell tumorigenesis
Small Interfering RNA Knockdown of TRIM25 Inhibits Lung Cancer Cell Migration and Invasion In Vitro
We next assessed whether TRIM25 could affect the ability of lung cancer cells to migrate and invade. The effect of TRIM25 on lung cancer cell migration was first assessed by wound healing assay. Silencing TRIM25 dramatically reduced the closure of wound area compared to their control cells (Figure 4A and B). This result was confirmed by Boyden chamber assay (Figure 4C and D). Moreover, silencing TRIM25 cells showed a lesser degree of invasion through Matrigel (Figure 4C and D). These results indicate that TRIM25 promotes migratory and invasive behaviors in lung cancer cells.

The siRNA knockdown of TRIM25 inhibits lung cancer cell migration and invasion
Small Interfering RNA Knockdown of TRIM25 Increases Doxorubicin Sensitivity
The efficiency of the current standard doxorubicin or doxorubicin-based regimens is moderate, probably due to preexisting or acquired resistance of the lung cancer cells. We next tested whether TRIM25 played a role in doxorubicin resistance. As shown in Figure 5A and B, temozolomide (10 nmol/L) alone only induced minor cell death (tested by trypan blue staining assay) and apoptosis (tested by Annexin V FACS assay) in A427 cells, which were both significantly aggravated by TRIM25 silencing (Figure 5A and B). Histone-DNA apoptosis ELISA assay further confirmed the apoptosis-sensitization effect by TRIM25 knockdown in A427 cells (Figure 5C). These results suggest that TRIM25 might be important for doxorubicin resistance in lung cancer cells, and knockdown of TRIM25 could increase doxorubicin sensitivity.

The siRNA knockdown of TRIM25 increases doxorubicin sensitivity. Untransfected control A427 cells (“NO siRNA”) or A427 cells transfected with indicated siRNA (100 nmol/L each, 48 hours) were treated with temozolomide (TMZ; 100 mmol/L), cells were further cultured, cell death was tested by trypan blue staining assay (A, 72 hours); cell apoptosis was tested by Annexin V FACS assay (B, 48 hours) or histone-DNA ELISA plus assay (C, 48 hours). siRNA indicates small interfering RNA; TRIM25, tripartite motif-containing 25.
TRIM25 Forms a Complex With p53
Above-mentioned results indicate that TRIM25 is important for lung cancer cell progression. Thus, we studied the potential underlying mechanisms. P53 is an important tumor suppressor that, upon activation, induces growth arrest and cell death. Control of p53 is thus of prime importance for proliferating cells but also for cancer therapy, where p53 activity contributes to the eradication of tumors. MDM2 functionally inhibits p53 and targets the tumor suppressor protein for degradation. Coimmunoprecipitation results showed that TRIM25 is associated with regular p53 in human lung cancer tissues (Figure 6A) as well as in lung cancer cell lines (Figure 6B). This association appeared important for p53 degradation, as knockdown of TRIM25 by siRNA increased the expression of p53 in A427 cells (Figure 6C). To recognize any clinical correlation of TRIM25 and p53, we analyzed expression of p53 in the same human lung cancer tissues. Highly inverse correlation between TRIM25 and p53 expression was drawn (Figure 6D). Thus, these results show that TRIM25 forms a complex with p53, which is relevant to p53 degradation.

The TRIM25 forms a complex with p53, required for p53 degradation. The TRIM25, p53, and MDM2 association in 5 human lung cancer tissues (A) and in A427, A549, and H1650 cells (B) was tested by coimmunoprecipitation (Co-IP) assay. Expression of p53in untransfected control A427 cells (“NO siRNA”) or A427 cells transfected with indicated siRNA (100 nmol/L each, 48 hours) was measured by Western blot (C). Highly inverse correlation between TRIM25 and p53 expression in lung cancer tissues was drawn (D). siRNA indicates small interfering RNA; TRIM25, tripartite motif-containing 25.
Discussion
Lung cancer is the leading cause of cancer-related deaths worldwide, due to its high incidence, malignant behavior, and lack of effective treatment strategy. 1 Although platinum-based combination chemotherapy is the first-line treatment for patients with advanced lung cancer, the response rates vary among patients, ranging from 26% to 60%. 1,7 Recent studies indicate that the discovery and application of specific prognostic biomarkers can improve the treatment and outcome of lung cancer. 15 Despite a considerable amount of research, very few stable biomarkers have been identified for risk assessment or predication of clinical outcome and further investigations are necessary.
Tripartite motif-containing 25, also known as estrogen-responsive finger protein, plays an essential role in cell proliferation and innate immunity.
16
-18
Its overexpression correlates with progression of several cancers, thus representing a marker of poor prognosis.
8,14,19
In this current study, we propose that TRIM25 might be an important oncogene for lung cancer. Our evidences include that TRIM25 is overexpressed in malignant lung tissues, and its expression is low in surrounding normal lung tissues. The expression of this molecular gene in lung cancer cells regulates cell proliferation and migration, and knocking down of TRIM25 by targeted siRNAs suppressed lung cancer cells proliferation and
Expression of TRIM25 in breast cancer is positively associated with lymph node metastasis and worse patient survival, and functional studies have demonstrated that TRIM25 plays an oncogenic role in breast cancer at least by targeting proteolysis of 14-3-3 sigma, a tumor suppressor that suppresses cell cycle progression. 8,20 In antivirial response of human cells, IFN upregulates TRIM25 and induces its conjugation with the ubiquitin-like protein ISG15, and TRIM25 is indeed an ISG15 E3 ligase for 14-3-3 sigma and its ISGylation enzyme activity is RING domain dependent. 16,21 Very little is known about the mechanisms of TRIM25-mediated cell migration. An interesting finding in this study is that TRIM25 was in the complex with p53 and Mdm2 in both human lung cancer tissues and in lung cancer cells, and siRNA knockdown of TRIM25 increased p53 expression in lung cancer cells. Highly inverse correlation between TRIM25 and p53 expression was drawn in the human lung cancer tissues. Thus, TRIM25 mediates malignant behaviors of lung cells probably through regulating p53 expression.
Significantly, we found that TRIM25, besides regulating cell migration, was also important for lung cell proliferation. Our observations are in line with very recent studies showing that required of TRIM25 for proliferation of carcinoma cells. 11 Another important finding of this study is that TRIM25 also played a role in doxorubicin resistance. Although the detailed mechanisms need further characterizations, these studies should provide valuable information for possibly improving doxorubicin activity in lung cancer.
In summary, the present findings highlight several new aspects of the role of TRIM25 in the lung cancer progression. Our results suggest that expression of TRIM25 might be critical for lung cancer cell migration, proliferation as well as doxorubicin resistance. Although much is yet to be learned regarding the clinical relevance of its function, we suggest that TRIM25 could be a promising new therapeutic target in antilung cancer therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
