Abstract
Background:
The patterns of intracranial failure in patients with brain metastasis from pulmonary neuroendocrine carcinoma (PNEC) remain unknown.
Methods:
From 1998 to 2013, 29 patients with the diagnosis of PNEC were treated for brain metastasis: 16 patients (55%) underwent whole-brain radiation therapy (WBRT), 5 (17%) patients underwent WBRT with a stereotactic radiosurgery (SRS) boost, and 8 (28%) patients underwent primary SRS alone.
Results:
The median age at treatment was 61 years (range: 44-84 years) and the median follow-up was 9.6 months (0-157.4 months). Of the patients treated with SRS alone, 1 patient had radiographic local progression of disease and 1 patient had a distant intracranial failure. Of the patients treated with WBRT with or without an SRS boost, 9 patients developed intracranial progression, including 1 local failure. No differences in rates of intracranial progression or local failure between the 2 groups (P = .94 and P = .44, respectively) were observed. The actuarial rates of distant intracranial failure at 12 months were 32.9% (95% confidence interval [95% CI] 8.9%-56.8%) and 25% (95% CI 0.0%-67.4%) in patients undergoing primary WBRT or SRS, respectively (P = .31). The median overall survival was 15.8 months in patients treated with WBRT and 20.4 months in patients treated with primary SRS (P = .78).
Conclusion:
Patients with brain metastasis from PNECs can be effectively treated with either WBRT or SRS alone, with a pattern of failure more consistent with non-small cell lung cancer than small cell lung cancer. In this series, there was not a statistically significant increased risk of distant intracranial failure when patients were treated with primary SRS.
Keywords
Introduction
The incidence of pulmonary neuroendocrine tumors is increasing worldwide due to improvements in pathological analysis, development of the international World Health Organization (WHO) classification system, and heightened awareness of the histological subtype itself. 1 Large cell neuroendocrine carcinomas (LCNECs) of the lung represent a rare subtype of primary lung cancer accounting for only 2.9% of cases. 2 The primary management for pulmonary LCNECs typically involves a form of local therapy, such as surgical resection or radiation therapy. Moreover, given their neuroendocrine differentiation and aggressive biological nature, there have been retrospective and prospective studies investigating the response rates for LCNECs to the combination cisplatin-based chemotherapy regimens typically prescribed to patients with small cell lung cancer (SCLC) and the overall efficacy of multimodality treatment. 3,4
Even after definitive therapy for their primary disease, patients with LCNECs are at risk of local, regional, and distant failure, including development of brain metastases. The standard radiotherapeutic management options in patients with brain metastasis include whole-brain radiation therapy (WBRT) with or without a stereotactic radiosurgery (SRS) boost or primary SRS. In comparison to WBRT, primary SRS may lead to improvements in local control, reduced rates of cognitive neurotoxicity, and improvements in quality of life. 5 Nevertheless, a potential disadvantage with the use of a local modality alone in the upfront setting is the possibility of subclinical micrometastatic disease, which may lead to early distant intracranial failure. Although the concern for this risk is higher in neuroendocrine carcinomas, the incidence is not well known. Therefore, the objective of the present study was to report the clinical outcomes and patterns of failure in patients with pulmonary neuroendocrine carcinoma (PNEC) whose brain metastasis was treated with either WBRT or primary SRS.
Materials and Methods
We used the institutional review board (IRB)-approved Burkhardt Brain Tumor and Neuro-oncology Center and Department of Radiation Oncology databases to identify 29 patients who, between 1998 to 2013, underwent radiation treatment for a brain metastasis with a histologic diagnosis of non-small cell carcinoma of the lung (NSCLC) with neuroendocrine differentiation, features (9 patients), or LCNEC (20 patients). Patients were treated with WBRT in the supine position with use of a thermoplastic mask for immobilization. The WBRT treatment was delivered over 1 to 4 weeks using megavoltage (MV) linear accelerators with photon beam energies of 6 MV delivered via parallel opposed lateral fields to ensure coverage of the entire cranial contents. The dose was specified at the central axis at midplane of the brain. Patients with SRS treated with Gamma Knife (Elekta Instruments AB, Stockholm, Sweden) radiosurgery prior to 2007 were treated with a 201-source 60Co Gamma Knife system. After this date, patients were treated with a 192-source unit (Perfexion). All radiosurgery patients received a local anesthetic agent and an intravenous sedative/anxiolytic medication. A rigid Leksell stereotactic coordinate frame was applied to the patient’s head, and high-resolution (1 mm slice) contrast-enhanced magnetic resonance images and computed tomography images were acquired. For radiosurgical planning, the dose prescribed to the peripheral margin was typically chosen based on the lesion size according to the Radiation Therapy Oncology Group 90-05 protocol. 6
Clinical patient information including age, recursive partitioning analysis (RPA), disease-specific graded prognostic assessment (DS-GPA), performance status (Karnofsky performance score [KPS]), gender, primary tumor-type histology, presenting symptoms, treatment technique, radiographic response, and overall survival were retrospectively obtained. 7 –9 Radiation therapy treatment data were abstracted including the total dose and fractionation pattern for those patients undergoing WBRT, and the dose delivered to the periphery of the tumor volume, maximum dose, heterogeneity index (HI), conformality index (CI), and gradient index (GI) for those patients treated with SRS.
The standard follow-up schedule consisted of appointments after the completion of radiation therapy, typically at 3-month intervals. Follow-up examinations consisted of an assessment of any new neurologic or systemic symptoms, neurologic physical examination, and magnetic resonance imaging (MRI) scans. For patients with recurrences diagnosed on follow-up MRI scans, the site of the recurrence was specifically analyzed as a local failure (same site as previous treatment) or distant failure (different site or presence of leptomeningeal metastases). The cause of death was coded for all patients, when available, and the Social Security Death Index was used to verify dates of death when necessary. Patients were considered to have suffered an intracranial death if they had stable systemic disease and progressive neurologic dysfunction, rapidly progressive systemic disease and advancing neurologic dysfunction, or severe neurologic disability at the time of death. 10 Approval from our IRB was obtained prior to performing this study.
Actuarial likelihood of local control and survival estimates were calculated according to the product-limit estimate (Kaplan-Meier) method. Data were collected on a Microsoft Excel database, and data analysis was performed using SAS version 9.2 (SAS Institute, Cary, North Carolina). The threshold of statistical significance for differences was set at P < .05.
Results
The clinical features of the 29 patients in this study are presented in Table 1. In total, 5 (17%) patients underwent WBRT with an SRS boost, 8 (28%) patients underwent SRS alone, and 16 (55%) patients underwent WBRT alone. Patients treated with an SRS boost were decided to have combined modality treatment at the time of diagnosis (as opposed to treatment response), based on the discretion of the treating physician. The median age was 61 years (range [R]: 44-84 years) and a majority of patients had a favorable performance status with a median KPS of 80 (50-100), 90% with an RPA of 1 to 2, and 69% with a DS-GPA of 2.5 to 4.0. Seventeen (59%) patients were treated with chemotherapy: 10 (34%) patients were treated with a platinum agent combined with etoposide, 3 (10%) patients were treated with carboplatin and a taxol agent, 1 (3%) patient was treated with carboplatin and pemetrexed, 2 (7%) patients were treated with carboplatin and gemcitabine with or without addition of a taxol agent, and the chemotherapy treatment for 1 patient was not available in the shared medical record. The median number of cycles of chemotherapy received across all patients was 4 cycles (R: 1-6 cycles). Seven patients received chemotherapy after treatment of their brain metastasis, 2 had previously undergone treatment with SRS alone, and the remainder had received WBRT.
Patient Characteristics.

Abbreviations: WBRT, whole-brain radiation therapy; SRS, stereotactic radiosurgery; yrs, years; m, months; KPS, Karnofsky performance score; DS-GPA, disease-specific graded prognostic assessment; RPA, recursive partitioning analysis.
Twelve (41%) patients were diagnosed with brain metastasis at initial presentation of their disease and 6 (21%) patients were diagnosed with concurrent extracranial metastases based on a review of systemic imaging studies. Of the patients diagnosed with brain metastasis at initial diagnosis, 6 patients underwent WBRT, 2 patients were treated with WBRT and an SRS boost, and 4 patients were treated with SRS alone. The median time from initial diagnosis to development of the first brain metastasis was 4.7 months (0-77.7 months). A majority of patients (62%) treated with WBRT underwent treatment of a single lesion, with a maximum of 5 lesions treated in 1 patient. Half of the patients undergoing treatment with SRS alone had a single brain metastasis, 3 patients were treated to 2 lesions, and 1 patient was treated to 4 lesions. Eight (28%) patients underwent surgical resection of at least 1 brain metastasis. Of these patients, 7 were treated with adjuvant WBRT with or without the addition of an SRS boost to the resection cavity and 1 patient underwent SRS alone to the resection bed.
The median WBRT dose was 3750 cGy (1800-4000 cGy) prescribed over 15 fractions (5-20 fractions). For patients undergoing an SRS boost, the median lesion volume was 0.815 cm3 (0.14-45.54 cm3), the maximum linear size was 1.3 cm (0.7-5.1 cm), and the median prescription marginal dose was 2400 cGy (1800-2500 cGy). For patients treated with SRS alone, the median lesion volume was 4.2 cm3 (0.08-27.20 cm3), the maximum linear size was 2.4 cm (0.64-4.83 cm), and the median prescription marginal dose was 2000 cGy prescribed to the 53% isodose line (1500-2400 cGy). For patients treated with SRS alone, the median CI, HI, and GI were 1.42 (1.18-1.80), 1.90 (1.70-2.02), and 2.91 (2.41-4.33), respectively.
Of the patients treated with WBRT, 6 (29%) patients continued to have extracranial progression of their disease, whereas all of the patients treated with SRS alone continued to have controlled primary disease. Only a single (5%) patient treated with WBRT and 1 (13%) patient treated with SRS alone experienced progression of a previously treated brain metastasis. Nine (43%) patients treated with WBRT developed distant intracranial brain metastases, whereas only 1 (13%) patient treated with SRS alone experienced distant intracranial failure. The 6-month rates of distant intracranial failure were 25.4% (95% confidence interval [95% CI]: 3.7-47.1) and 0% for patients treated with WBRT and SRS, respectively. The 12-month rates of distant intracranial failure were 32.9% (95% CI: 8.9-56.8) and 25.0% (95% CI: 0-67.4%) for patients treated with WBRT and SRS, respectively (P = .3327; see Figure 1). Salvage treatments for the 9 patients who continued to have intracranial progression after WBRT consisted of SRS in 5 patients and repeat WBRT in 1 patient. The remaining 3 patients did not receive any further therapy. Salvage treatments for the 2 patients who continued to have intracranial progression after primary SRS consisted of repeat SRS in 1 patient and surgical resection in another.
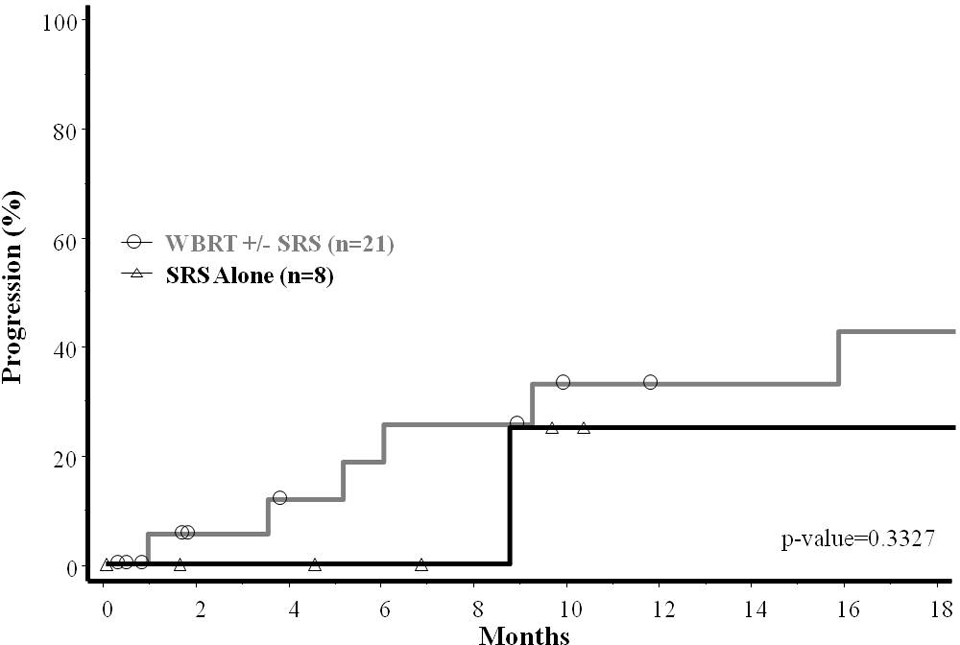
Kaplan-Meier curve of distant intracranial failure estimates (progression outside a previously treated brain metastasis) for patients treated with either whole-brain radiation therapy (WBRT) with or without a stereotactic radiosurgery (SRS) boost or primary SRS.
At a median clinical and radiographic follow-up of 9.6 months, 6 (21%) patients remained alive, and the cause of death was attributed to progression of the patient’s non-central nervous system (CNS) disease in 17 (59%) patients. The median survivals for patients in RPA class I, II, and III were 26.1 months, 16.6 months, and 7.1 months, respectively. Similarly stratified, the median survivals for patients with DS-GPA 0-1.0, 1.5-2.0, 2.5-3.0, and 3.5-4.0 were 2.4 months, 13.0 months, 16.6 months, and 26.1 months, respectively. Based on the last medical record entry, 11 (52%) patients treated with WBRT and 2 (25%) patients treated with SRS alone had an intracranial death. The median survival from the time of treatment was 15.8 months in patients treated with WBRT compared to 20.4 months in patients treated with SRS alone (P = .779).
Discussion
The PNECs represent a range of histopathological entities including typical carcinoid, atypical carcinoid, LCNEC, and SCLC. Although LCNEC and SCLC are both classified as high-grade neuroendocrine carcinomas due to similarities in their histopathological analysis, there are distinct differences in their morphology, gene expression profiles, and immunophenotype. 11,12 The biological behavior and clinical outcomes between these 2 tumor types are also thought to be distinctive. For example, in 1 study of 140 patients who underwent complete resection of a high-grade NEC, multivariate analysis revealed that larger tumor diameter and lymphatic permeation were strong unique prognostic factors in all stages of LCNECs, factors that were not found to be prognostic in patients diagnosed with SCLC. 13 On the other hand, these factors have been shown to be prognostic in patients diagnosed with primary adenocarcinoma of the lung. 14 In another study of 28 patients with PNECs measuring less than 3.0 cm undergoing surgical resection, important differences were found between patients with LCNEC and SCLC, including the incidence of occult lymphatic involvement, recurrence rates after surgery, and overall survival. 15 At the same time, it is important to note that LCNECs are associated with a more clinically aggressive course of disease with more frequent locoregional lymph node recurrences and lower survival rates compared to staged-matched patients with NSCLC. 16 Taken together, this pathologic information combined with correlative clinical studies suggests that the behavior patterns of LCNEC and other NSCLC subtypes with neuroendocrine differentiation differ substantially from SCLC and lay the foundation for the rationale for this study.
The findings of the present study demonstrate that patients with brain metastases from PNECs can be effectively treated with a primary local modality, such as SRS, with high rates of local control and modest rates of distant intracranial failure. Moreover, the results of this study also indicate that the addition of WBRT to a local modality or the use of WBRT alone in the upfront setting may not provide any significant benefit with regard to local control, distant intracranial control, or overall survival although the patient numbers in this series are few. To the best of our knowledge, these results represent the first published report regarding the biological behavior, clinical outcomes, and patterns of failure in patients with brain metastasis from this rare histologic subtype of lung cancer.
With regard to brain metastases, multiple nomograms and prognostic assessment scales have been developed, most notably the RPA and DS-GPA. Previously performed research at our institution has explored the use of the RPA classification system in patients with NSCLC and validated its use in patients with SCLC. 17,18 In the present study, the survival estimates for patients with brain metastases from PNECs appeared to be more favorable than the corresponding RPA-matched SCLC groups (see Table 2). Although the RPA classification system has been validated in multiple data sets, the DS-GPA has the advantage of being diagnosis specific and reflects a more contemporary cohort of patients who received modern systemic agents. Despite the small numbers of patients included in this study, the DS-GPA also stratified patients into distinct prognostic groups. It is important that these clinical details are able to stratify patients into separate prognostic categories, as this helps clinicians to select appropriate treatments, but continuing research should explore the addition of biomarkers into these stratification systems.
RPA and DS-GPA Classifications and Comparative Survival Estimates for Patients With Brain Metastasis From Lung Cancer.

Abbreviations: RPA, recursive partitioning analysis; DS-GPA, disease-specific graded prognostic assessment, NSCLC, non-small cell lung cancer; SCLC, small cell lung cancer; m, months.
Multiple randomized controlled trials have demonstrated that patients with brain metastases treated with SRS alone have comparable overall survival rates to those treated with SRS and WBRT. 19,20 The results of the present study also demonstrate favorable local control and survival outcomes in patients with brain metastasis from PNECs treated with SRS alone compared to those treated with WBRT or a combined modality approach. These results are also timely and relevant, given the increasing awareness of these specific histological subtypes and the uncertainty regarding the appropriate management of these patients if they develop brain metastases. There is considerable controversy regarding the optimal management of patients with brain metastasis, especially regarding the role and timing of WBRT. 21 In the short term, WBRT is associated with a decline in learning and memory function, and in the long term, survivors are at increased risk of white matter changes. 20,22 Furthermore, Kondziolka and colleagues reported significantly higher incidences of patient-reported fatigue, short-term and long-term memory changes, concentration problems, and depression in patients treated with WBRT compared to those treated with SRS. 23 In light of this evidence of the detrimental side effects of WBRT, the results of the present study suggest that select patients may be appropriately treated with primary SRS.
One concern with the use of primary SRS in patients with LCNECs is the possibility for distant intracranial failure, given their neuroendocrine differentiation and the known intracranial failure patterns in patients with SCLC treated with a local primary modality. Kuremsky and colleagues reported their institutional experience of patients with lung cancer of all histologies treated with SRS at first presentation of brain metastasis and included 31 patients with SCLC, 26 of whom had previously undergone prophylactic cranial irradiation (PCI). 24 In this cohort, the freedom from distant brain failure at 1 year was 37%, and the distant control rate at 2 years was only 23%. On multivariate analysis, SCLC histology was associated with an increased likelihood of local failure and distant failure. Wegner and colleagues reported that in 44 patients with SCLC treated with SRS alone, after PCI, after WBRT, or in a planned combined modality approach, new distant intracranial lesions developed in 61% of patients at a median of 7 months. 25 Moreover, the median overall survival was only 6 months in patients who were treated with primary SRS compared to 14 months for those treated with the addition of WBRT. In the present study, the 12-month distant failure rates for patients with neuroendocrine carcinomas treated with WBRT and SRS were both quite similar (32.9% and 25%, respectively) and more favorable than the reported rates of distant failure in patients with SCLC treated with local therapies. It is important to note, however, that a majority of patients who received chemotherapy after treatment of brain metastasis (thereby possibly helping to control microscopic intracranial disease) were in the WBRT cohort. Therefore, addition of post-CNS treatment chemotherapy did not affect the distant intracranial recurrence patterns. Taken together, these findings uphold the current standard practice of upfront WBRT rather than a primary radiosurgical approach in patients with SCLC. Alternatively, the results of the present study suggest that primary SRS may be sufficient for select patients with other subtypes of neuroendocrine differentiation, but further investigation is warranted.
A majority of patients (69%) in this study were specifically diagnosed with LCNEC and the remaining patients with NSCLC with neuroendocrine differentiation or features. One of the difficulties in studying patients with these tumor types is the difficulty in accurate diagnosis. This difficulty in histological classification was underscored in 1 study of 170 cases of PNECs evaluated by pulmonary pathology specialists, which showed that unanimity of diagnosis was observed in only 20 cases, a majority consensus in another 115 cases, and no consensus in 35 cases (κ = 0.40). 26 Therefore, it is important to note that even studies of patients with LCNEC, classified by the published WHO classification system, are subject to considerable variability and heterogeneity. This complexity was also observed in our institutional experience. One patient in this series was initially diagnosed with SCLC but additional review at our institution with immunohistochemical staining and morphological analysis was in fact consistent with a diagnosis of NSCLC with neuroendocrine differentiation. Therefore, one of the potential drawbacks of the current series is that 6 of the patients in this study had their pathology diagnosed at an outside institution.
The low risk of distant intracranial failure in patients treated with SRS may reflect good selection bias (biological factors, clinical factors, and patient characteristics) as the modality for brain metastasis treatment was decided at the time of diagnosis at the discretion of the treating physician. When considering the effect of chemotherapy, although more patients treated with SRS received chemotherapy as well, more patients who were treated with WBRT received chemotherapy after brain metastasis treatment. Therefore, in well-selected patients, primary SRS allows for effective treatment of brain metastasis without a substantial risk of distant intracranial failure. Given the rarity of this subtype of lung cancer and the retrospective nature of this data set, we consider the results of this study to be hypothesis generating. We consider that these results should be confirmed in a larger multi-institutional setting.
Conclusion
Neuroendocrine carcinomas of the lung represent a rare but increasingly recognized subtype of lung cancer. Given their neuroendocrine differentiation and previously understudied patterns of presentation and failure in patients who develop brain metastasis from these tumor subtypes, radiotherapy treatments at our institution were previously inconsistent. To help corroborate the findings of our retrospective study, multi-institutional studies should be performed to pool the outcomes of patients with these histological subtypes in order to better understand the clinical characteristics and biological behavior of these tumors. Moreover, additional studies in all tumor subtypes should be performed to determine prognostic factors that may identify the groups of patients who would be at lowest risk of distant intracranial failure and could be appropriately treated with primary SRS.
Footnotes
Authors’ Note
This study was presented in part at the ASTRO 56th Annual Meeting in San Francisco, CA, from September 14-17, 2014.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.S. Ahluwalia is consulting and/or speaking for Monteris Medical, Inc and PrIME Oncology, Inc. J.H. Suh is consulting for Varian Medical Systems MA. Vogelbaum is consulting and/or speaking for Merck & Co, Inc, Royalty payments, equity, and fiduciary role in Infuseon Therapeutics, Inc. G.H. Barnett is consulting and/or speaking for Varian Medical Systems, Monteris Medical, Inc, royalty payments: Mako Surgical Corp, Roche (Hoffman-La Roche, Ltd.). S.T. Chao is speaking for bureau for Varian Medical Systems (honorarium).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
