Abstract
This study was performed to observe the effect of the combination of nimotuzumab with radiation or gemcitabine-based chemoradiation on antipancreatic cancer cell therapy. Pancreatic cancer cells (PANC-1) were treated with nimotuzumab alone or combined with radiation (2, 4, or 8 Gy), which was either with or without gemcitabine chemotherapy. Cell proliferation, cell cycle distribution, and apoptosis were observed. The inhibition rate, the percentage of G2/M phase arrest, and the apoptosis rate of the combined nimotuzumab with radiation group was significantly higher than the group without nimotuzumab (P < .001). The inhibition rate, the percentage of G2/M phase, and the apoptosis rate of the nimotuzumab therapy combined with gemcitabine-based chemoradiation group were obviously higher than that in gemcitabine-based chemoradiation group (P < .001). In conclusion, nimotuzumab could enhance the anticancer effect of radiation and gemcitabine-based chemoradiation in PANC-1 cancer cells because of the enhancement of cell cycle arrest and apoptosis.
Keywords
Introduction
Due to the atypical clinical symptoms of pancreatic cancer, most patients lose the opportunity of surgery to remove the cancer. Radiotherapy and/or chemotherapy is the common method used for the management of patients with local advanced disease to prolong survival time. In the current oncological practice, the antimetabolic drug gemcitabine (G) is considered as standard for chemotherapy although its efficacy is limited. Radiotherapy is the treatment of choice for pancreatic cancer, but the effectiveness of treatment is limited by radiation resistance. On the other hand, the dose limit of the surrounding normal tissue makes it impossible to accept the curable radiation dose. Novel therapeutic approaches are urged to develop and are currently under clinical investigation in an attempt to treat this fatal disease. 1
Epidermal growth factor receptor (EGFR) is a member of the receptors of the ErbB family, which is universally distributed on the surface of multimalignant tumors. The ErbB family of proteins contains 4 receptor tyrosine kinases that are structurally related to the EGFR, its first discovered member. In humans, the family includes Her1 (EGFR and ErbB1), Her2 (Neu and ErbB2), Her3 (ErbB3), and Her4 (ErbB4). The gene symbol, ErbB, is derived from the name of a viral oncogene to which these receptors are homologous, erythroblastic leukemia viral oncogene. Epidermal growth factor receptor can promote proliferation; inhibit apoptosis; and increase the ability of motility, adhesion, and invasion of tumor cells. Therefore, EGFR enhances the chance of tumor invasion and metastasis. Cancer cells with high level of EGFR expression often resist chemotherapy and radiotherapy with poor prognosis. 2 –4 It has been reported that high expression of EGFR is found in 30% to 60% of pancreatic cancers. 5,6 In this study, we hypothesized that inhibiting EGFR could improve the therapeutic effect of pancreatic cancer. We tested our hypothesis in pancreatic cancer cell line PANC-1 in which the high level of EGFR expression had been evident. 7,8
Nimotuzumab (h-R3) is an anti-EGFR monoclonal antibody that binds to EGFR on the surface of cancer cells and blocks the downstream signaling transduction pathway mediated by EGFR. Nimotuzumab can inhibit the proliferation of tumor cells and play an antitumor effect. 9 This is a continuous study of our previous work, and we have recently reported that h-R3 could enhance the anticancer effect of radiation in PANC-1 cancer cells because of the enhancement of apoptosis. 10 In this study, we further evaluated the impact of h-R3 on radiation or G-based chemoradiation in EGFR-overexpressed pancreatic cancer cell lines PANC-1.
Materials and Methods
Cancer Cell Culture
Human pancreatic cancer cell line PANC-1 was generously provided by the Center Laboratory of the First Affiliated Hospital of Harbin Medical University (Harbin, China), and the PANC-1 cells were cultured in Dulbecco's modified Eagle medium supplemented with 10% fetal bovine serum (Hyclone Company, Logan, USA), 1% glutamine, and 1% antibiotic mixture. Cells were grown in humidified incubator at 37°C in air atmosphere containing 5% carbon dioxide. Media were replaced every 2 or 3 days. Exponentially growing cells were harvested with 0.25% (w/v) Trypsin-0.53 mmol/L EDTA solution, washed, and suspended in phosphate-buffered saline (PBS).
Drugs and Reagents
G was product of Eli Lilly (Indianapolis, USA), and h-R3 was the product of Biotech Pharmaceutical Co (Beijing, China). CCK-8 (Cell Counting Kit-8) was purchased from Dojindo Co (Japan). The cell cycle and apoptosis kits were purchased from Beyotime Co (China).
Experiment Design
There were 6 experimental groups: the blank control group (C), cells were cultured for 72 hours in the normal cultural condition; the h-R3 alone group (h-R3), cells were cultured for 48 hours and then treated with h-R3 for 24 hours; the radiation alone group (R), after cultured for 48 hours, the cells were exposed to 6 MV-X-ray radiation and continued to culture for 24 hours; the combination group of h-R3 and radiation (h-R3 + R), cells were cultured for 24 hours, treated with h-R3 for 24 hours, exposed to 6 MV-X-ray radiation, and continued to culture for 24 hours; the combination group of G and radiation (G + R), cells were cultured for 24 hours, treated with G for 24 hours, exposed to 6 MV-X-ray radiation, and continued to culture for 24 hours; and the combination group of h-R3, G, and radiation (h-R3 + G + R), cells treated with h-R3 were cultured for 24 hours, treated with G for 24 hours, exposed to 6 MV-X-ray radiation, and then continued to culture for 24 hours eventually. The concentration of intervention of h-R3 was 10 μg/mL; the concentration of G was 0.1 μg/mL; and the doses of radiation were 2, 4, and 8 Gy (flow cytometry detected the cells exposed to 4 Gy radiation). Dulbecco's modified Eagle medium was added to the blank C group and the R group.
Irradiation
The culture plates were covered with 1.5 cm tissue materials when 6 MV-X-ray radiation from a high-energy linear accelerator was used. The irradiating field was 10 × 10 cm2 field at 100 cm source skin distance, and radiation method was isocenter radiation. The radiation doses were 2, 4, and 8 Gy, respectively.
Assay of Cell Growth Curve
The PANC-1 cells were obtained during the exponential growth phase and adjusted to the concentration of 1.5 × 104 cells/mL by counting plate. Cells were inoculated into 96-well plates at 100 µL/well. Counting from the time that cells adhered to the edge, the optical density (OD) value of each group of cells was assayed every 24 hours in the following 7 days by CCK-8 method, and the cell growth curve was plotted using OD value as the vertical axis.
Proliferation of PANC-1 Cells by CCK-8
The PANC-1 cells were inoculated into a 96-well plate at density of 5 × 104 cells/mL during the exponential growth phase and were treated according to the experimental group. CCK-8 (10 μL) was added into each well 72 hours after cell seeding and then continued to culture for 1 hour. The OD value was assayed at wavelength of 490 nm with an enzyme-linked immunosorbent assay, and the inhibition rate was calculated using the following formula: inhibition rate = 1 − (the average OD value in the experimental group/the average OD value in the C) × 100%. Assay of each group was reproduced 3 times.
Determination of PANC-1 Cell Cycle Distribution and Apoptosis by Flow Cytometry Machine
The PANC-1 cells in the exponential growth phase were inoculated into 6-well plate at 2 × 104 cells in 2 mL of medium. Cells were incubated for 24 hours when cancer cells attached to plate wall. After an inoculation time of 24 hours, all the cells were totally adherent to the edges, and then the cells were treated according to the experimental groups. For analysis of cell cycle distribution, the samples were prepared as single cell suspension, fixed with 70% ethanol, and preserved at −20°C. Propidium iodide (PI) staining solution and RNA enzyme were added after washing with cold PBS. After 30 minutes staining at 4°C, cell cycle distribution was analyzed by flow cytometry machines (FCMs). Analysis of each group was repeated 3 times. For determination of apoptosis, the cells were washed with PBS after harvesting by the centrifugation, labeled by Annexin V-fluorescein isothiocyante and PI, and finally the determination of cells apoptosis was carried out by FCM. Determination of each group was repeated 3 times.
Statistical Analysis
All data were represented as mean ± standard deviation
Results
Effect of Combination of h-R3 With Radiation or G-Based Chemoradiation on PANC-1 Cells
The OD value could reflect the number of the surviving cells indirectly, and we could calculate the inhibition rate using it. The inhibition rate of cells in each group is shown in Figure 1A: H-R3 group (2.36 ± 2.08) did not have significant inhibition to the cells, while the inhibition rate of h-R3 + R group (51.19 ± 16.26) was obviously higher than that in R group (27.03 ± 13.53; P < .001) and the inhibition rate of the h-R3 + G + R group (60.81 ± 15.53) was obviously higher than that in G + R group (41.48 ± 14.76; P = .001). In the same experimental group, with the increase in radiation dose, the inhibition rate gradually increased (P = .001; Figure 1B).

A, Inhibition rate of each group. Nonparametric Kruskal-Wallis test was used and results are given as means ± SDs (n = 18). *P < .001; **P = .001. B, Inhibition rate of the same group with different radiation dose. In the same experimental group that received radiation, the radiation doses were 2, 4, and 8 Gy, respectively. With the increase in radiation dose, the inhibition rate was gradually increased, nonparametric Kruskal-Wallis test, results are expressed as means ± SDs (n = 6), P = .001. SD, standard deviation.
Enhancement of G2/M Phase Arrest in PANC-1 Cells by Combination of h-R3 and Radiation or G-Based Chemoradiation
Distribution of each cell cycle phase (G0, G1, S, G2, and M) was analyzed by FCM. We computed the percentage of G0/G1, S, and G2/M phase, and the statistical differences between percentages of cells in G2/M phase of the 2 groups are summarized in Figure 2. When compared to the control group (11.13 ± 0.15), there was no significant difference in the h-R3-treated group (12.25 ± 0.22; P = .058), however, R group (18.57 ± 0.51) and G + R group (30.93 ± 0.64) had significantly higher G2/M phase arrest (P < .001). The percentage of G2/M phase in h-R3 + R group (34.11±1.09) was significantly higher than that in R group (P < .001). The percentage of G2/M phase arrest in the h-R3 + G + R group (40.95 ± 0.81) was significantly higher than that in G + R group (P < .001).

The percentage of cell cycle distribution (%) and comparison of G2/M phase. Results are means ± standard deviations (SDs; n = 3). NS, no statistical differences; *, P < .001.
Cell cycle distribution in untreated control group cells (Figure 3A) was mainly in G0/G1 phase and S phase, less in G2/M phase. Cell cycle distribution of h-R3-treated cells (Figure 3B) was similar to the untreated control cells, but the percentage of cells in G2/M phase of R group (Figure 3C) increased. The ratio of G2/M phase of G + R group (Figure 3D), h-R3 + R (Figure 3E), and h-R3 + G + R group (Figure 3F) continuously increased, respectively, indicating the combination of multiple therapies enhanced G2/M phase arrest.
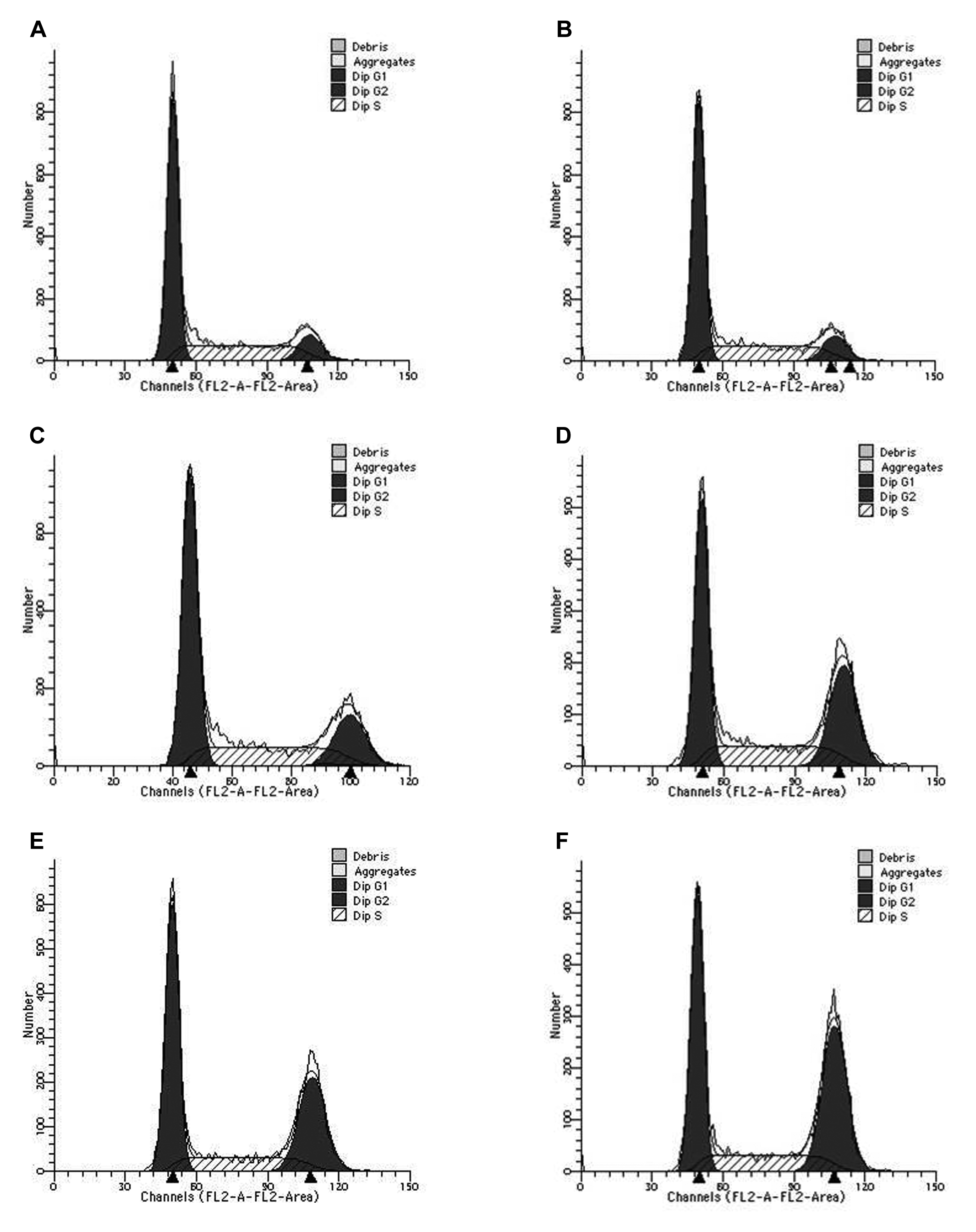
Scattergram of cell cycle. (A), The blank control group (C), (B) the h-R3 alone group (h-R3), (C) the radiation alone group (R), (D) the combination group of G and radiation (G + R), (E) the combination group of h-R3 and radiation (h-R3 + R), and (F) the combination group of h-R3, G and radiation (h-R3 + G + R).
Evidence of Enhanced Apoptosis in PANC-1 Cells by Combination of h-R3 and R or G-Based Chemoradiation
The results of detection of cell apoptosis in each group are shown in Figure 4: PANC-1 cells in the untreated control group had the lowest rate of apoptosis (1.12% ± 0.08%), and with the increase in intervention factors, the apoptosis rate increased gradually. The apoptosis rate in h-R3 group (3.94% ± 0.16%) was higher than that in the control group (P < .001), the apoptosis rate in h-R3 + R (8.81% ± 0.09%) group was higher than that in the R group (5.55% ± 0.16%; P < .001), and the apoptosis rate in h-R3 + G + R group (11.58% ± 0.40%) was significantly higher than that in the G + R group (6.47% ± 0.31%; P < .001). Accordingly, h-R3 enhanced the apoptosis of the R and chemoradiation groups.

Apoptosis rate of each group. Results are given as means ± standard deviations (SDs; n = 3). *P < .001.
Discussion and Conclusion
The majority of patients with pancreatic cancer are diagnosed at advanced stage with inoperable locally advanced tumors or metastatic disease. 1 Each year almost 200 000 people die of pancreatic cancer. 11 Pancreatic cancer is an incredibly challenging disease due to its high rate of resistance to traditional chemotherapy and radiotherapy. Published literatures did not show a better survival benefit on G-based chemoradiation.
During the past decade, EGFR was paid close attention to, however, EGFR study mainly concentrated on head and neck cancer, lung cancer, and colon cancer. 12,13 There were only a few of studies that focused on pancreatic cancer. Bloomston et al found that EGFR has an expression of 69% in pancreatic cancer. 14 The expressions of EGFR messenger RNA and protein were found in pancreatic cancer cell lines Miapaca-2, PANC-1, BXPC-3, CaPan-1, CaPan-2, ASPC-l, SW1990, Hs766T, CFPAC, All65, HPAC, and HPAF-II. 15
In our present study, pancreatic cancer cell line PANC-1 was chosen to investigate the biological effects of combinations of anti-EGFR drug with cytotoxic drug and radiation. Our results indicated that the anti-EGFR drug h-R3 with a dose of 10 μg/mL alone did not show an obvious depressant effect of proliferation, but when combined with radiation, the therapeutic effect significantly increased. The inhibition rate of tumor growth in the h-R3 +R group was higher than the R alone group. A similar result was also found when h-R3 was added to the combined group of G + R. Therefore, anti-EGFR drug h-R3 can enhance the inhibition and proliferation of G and R on pancreatic cancer cells PANC-1. Some studies showed that combination of cetuximab (C225; a kind of monoclonal antibody of EGFR) and radiotherapy or chemotherapy can improve the efficacy of radiotherapy. 16 –18 Wang et al reported that h-R3 had a synergy effect with chemoradiation in the treatment of glioma. 19
Ueda et al studied the relationship between EGFR expression and tumor invasion and metastasis, suggesting that the cytoplasmic EGFR may play an important role in invasion and metastasis of pancreatic cancer. 20 Akashi et al reported that h-R3 enhanced the antitumor efficacy of radiation in certain human non-small-cell lung cancer cell lines in vitro and in vivo, and this effect may be related to the level of EGFR expression on the cell surface rather than to EGFR mutation. Overexpression or mutation of EGFR is closely related to occurrence and development of a great variety of tumors including pancreatic cancer and also has relationship with chemotherapy or radiotherapy resistance and survival. 21
We found that the combination of h-R3 +G +R can significantly increase the proportion of cells within G2/M phase and reduce the proportion of cells within S phase through detecting cell cycle distribution by FCM. Interference of the combination of the 3 factors caused blocking of more cells in the G2/M phase. Radiation and cytotoxic drugs not only resulted in inhibition of proliferation and death of tumor cells but also caused blocking of the remaining living cells in the G2/M phase of the cell cycle. The G2/M phase plays a very important role in the synthesis and repair of DNA; the cells blocked in this phase are hard to proliferate and differentiate, and on the other hand, the cells are unable to repair the DNA damage induced by chemotherapy and radiation. 22,23 Meanwhile, the rate of apoptosis was evaluated by FCM, and the results showed that compared with the control group, h-R3 alone group increased in the apoptosis rate of cells, and when h-R3 was combined with R or combined with G-based chemoradiation, the rate of apoptosis increased. H-R3 could induce cell apoptosis and enhance apoptosis induced by R and G. Perhaps h-R3 exerts its biological effects by promoting apoptosis and blocking cell cycle in the G2/M phase so that cells cannot enter the next cycle to inhibit cell proliferation, which may also be part of the mechanism of the synergistic action of h-R3 combined with chemoradiation.
In conclusion, h-R3 could promote apoptosis of pancreatic cancer cells PANC-1 and enhance the growth suppression effect of cancer cells when combined with R and G-based chemoradiation, which may be due to the enhancement of cell cycle arrest and apoptosis by h-R3.
Footnotes
Acknowledgments
We thank Dr Fenglin Cao and all the other colleagues in the Blood Cancer Center Laboratory for providing experimental support, thank all the doctors in the Cancer Radiotherapy Center for irradiation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
