Abstract
Lung cancer is a common malignant tumor with high morbidity and mortality. Here we compared the effects and outcome between central and peripheral stage I lung cancer using image-guided stereotactic body radiotherapy. From June 2011 to July 2013, a total of 33 patients with stage I lung cancer were enrolled. A total of 50 Gy in 10 fractions or 60 Gy in 10 fractions was delivered in the central arm (n = 18), while 50 Gy in 5 fractions in the peripheral arm (n = 15). Statistical analyses were performed using logistic regression analysis and Kaplan-Meier method. The mean follow-up time was 38.1 months. Three-month, 1-, 2-, and 3-year overall response rates were 66.7%, 83.3%, 61.1%, and 72.2% and 66.7%, 80%, 80%, and 80% in the central and peripheral arms, respectively. Three-year local control rates (94.4% vs 93.3%, P = .854), regional control rates (94.4% vs 86.7%, P = .412), and distant control rates (64.2% vs 61.7%, P = .509) had no differences between the central and the peripheral arms. Grade 2 radiation pneumonitis was observed in 6 of 18 patients in the central arm and in 1 of 15 patients in the peripheral arm (P = .92). Grade 2 radiation esophagitis was 5.7% in the central arm, while none occurred in the peripheral arm (P = .008). Five (15.1%) of all patients felt slight fatigue during radiotherapy. Other major complications were not observed. In conclusion, helical image-guided stereotactic body radiotherapy for central stage I lung cancer is safe and effective compared to peripheral stage I lung cancer.
Introduction
Lung cancer is a common malignant tumor with high morbidity and mortality. With computed tomography (CT) examination and more common use of positron emission tomography (PET)/CT, the detection rates of early-stage lung cancer have improved. Pulmonary lobectomy is the standard treatment of early stage lung cancer, with excellent local control rate (LCR) of more than 90% and 5-year survival of more than 50%. 1 Some elderly patients with early stage lung cancer are not amenable with surgery for medical reasons of coexisting illnesses. More and more inoperable patients underwent radiotherapy (RT). However, compared with surgery, the efficacies of conventionally fractionated RT in patients with early stage lung cancer were unsatisfactory. Conventional RT fails to control the primary tumor in 60% to 70% patients, and over half of the patients ultimately die from progressive lung cancer. 2
Stereotactic body radiotherapy (SBRT) is of the nature of high fractionated dose, and it could shorten the treatment course and elevate radiation dose compared with traditional regimens. Stereotactic body radiotherapy has been an excellent treatment option for early stage lung cancer because it can make the maximum dose concentrate in gross tumor volume (GTV) and dose decline steeply in the normal tissue around GTV. Tumor LCR and survival of SBRT in inoperable patients with early stage lung cancer are not significantly different from that of surgery. 3 –5 Stereotactic body radiotherapy for lung tumors located in the pulmonary hilar region might be tolerable and acceptable when irradiated volumes were carefully taken into consideration. 6
Compared with intensity-modulated RT (IMRT), single-arc intensity-modulated arc therapy (IMAT), 3-dimensional conformal RT (3D-CRT), volumetric-modulated arc therapy (VMAT), and helical tomotherapy (HT) may be superior to organs at risk (OARs) sparing and to providing higher target dose uniformity. 7 –10 Recently, HT appears to be superior to VMAT in OAR sparing mainly in central lung cancer that requires conformal dose avoidance of multiple immediately adjacent OARs. 11 However, most studies focused on either central lung cancer or peripheral lung cancer in the early stage. In this study, therefore, we compared the effects and safety on central and peripheral stage I lung cancer using IG-SBRT via HT.
Materials and Methods
Characteristics of Patients
This retrospective analysis was based on 33 patients treated with HT-based SBRT from June 2011 to July 2013 for stage I lung cancer. Patient demographics and clinical characteristics are summarized in Table 1. Patients had a performance status with Eastern Cooperative Oncology Group of 0 to 2. These patients were medically inoperable or they refused to undergo surgery. The median age was 73 years (range, 44-87 years). Lung cancer stage was classified based on the tumor, node, metastasis (TNM) seventh edition by American Joint Committee on Cancer. Eleven patients were biopsy confirmed, comprising squamous cell cancer (n = 4) and adenocarcinoma (n = 7). Twenty-two patients were diagnosed by more than 3 doctors from radiology, pulmonary, and RT departments. Clinical diagnosis of lung cancer depended on consecutive increase in tumor size in CT scans or pathological uptake of 18F-fluorodeoxyglucose (18F-FDG) in the lesion in PET scan. Staging of negative N and M status was based on contrast-enhanced CT scans or FDG-PET-CT in all patients. All patients signed informed consent in accordance with the policy of the hospital’s ethical institutional review board.
Characteristics of Patients.
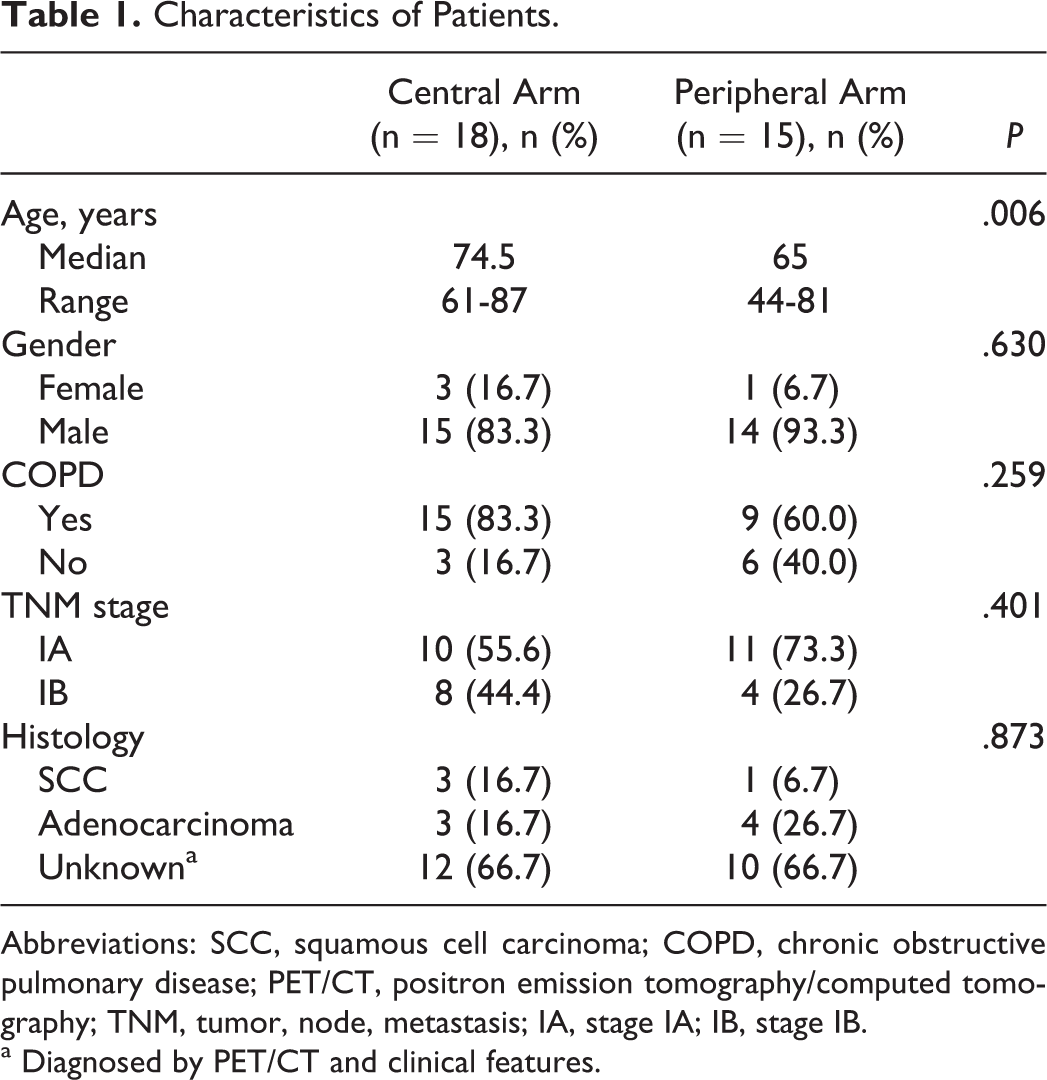
Abbreviations: SCC, squamous cell carcinoma; COPD, chronic obstructive pulmonary disease; PET/CT, positron emission tomography/computed tomography; TNM, tumor, node, metastasis; IA, stage IA; IB, stage IB.
a Diagnosed by PET/CT and clinical features.
Treatment Planning and Delivery
All patients were positioned by body fixation and simulated with application of respiration-correlated 4-dimensional CT (4D-CT). The scan range was from the inferior margin of mandible to 5 cm below the costophrenic angle. Slice thickness was 3 mm, and hatch spacing was 3 mm. The 4D-CT scans were obtained to track the motion of the tumors and other internal organs during free breathing using the real-time position management system. The primary tumor in the enhanced CT or FDG-PET/CT was delineated as GTV. Gross tumor volume was delineated at the lung window level. The clinical target volume was equal to GTV. Internal target volumes (ITVs) which combined the extension of GTVs at the 4 phases of the respiratory cycle on the 4D-CT scan were created. The planning target volume (PTV) extended a margin of 0.3 cm from the ITV for patients treated with HT to account for daily setup errors. The 18FDG-PET-guided RT planning was optional. The tumor region was scanned daily and the patient was repositioned based on adaptive RT (ART) principles. Target delineation was performed in the Pinnacle treatment planning system (Philips Medical Systems, Bothell, Washington). Afterward, each patient’s planning CT scans and the contours were transferred into the HT Hi-Art II planning system (TomoTherapy, Madison, Wisconsin) for treatment planning.
The dose fractionation schedules, 50 Gy/5 fractions were delivered on consecutive days for peripheral lung cancer (biological effective dose [BED] = 100 Gy) and 50Gy/10 fractions or 60 Gy/10 fractions were delivered once daily, 5 days per week for central lung cancer (BED = 75 Gy or 96 Gy). Among the patients with central tumors, 9 patients aged more than 75 years received 50 Gy/10 fractions for their serious chronic obstructive pulmonary disease combined with cardiovascular disease, smaller tumor size. And the remaining 9 patients with central tumors received 60 Gy/10 fractions.
All plans were assessed comprehensively by dose volume histogram (DVH) of Treatment Planning System (TPS) and isodose line graph. Optimized indicators for all plans were as follows: (1) The dose of OARs should not exceed the tolerance dose to the normal tissues, (2) 90% isodose line covered 100% of the PTV, (3) with respect to target-dose inhomogeneity, the prescribed dose should correspond with the 80% isodose line located at the edge of the PTV within lung tissue, which forces a hot spot (≥100% of the prescription dose) to occur within the central region of the tumor, (4) high-dose volume should be contained in the vicinity of the tumor, and the ratio of the volume of the prescription isodose to the volume of PTV should be <1.2, (5) OARs around PTV received doses were no more than their own tolerance dose, (6) total and contralateral pulmonary dose should be as small as possible.
Following Up and Statistics
During treatment, all patients were monitored daily for acute treatment-related toxicity. Tumor size was measured during each follow-up using the standard of Response Evaluation Criteria in Solid Tumors (RECIST) 1.1: a complete response (CR) is total tumor disappearance, a partial response (PR) is a decrease of ≥30% in the longest tumor diameter or total tumor diameters; a stable disease (SD) is a decrease of <30% or an increase of ≤20% in the longest tumor diameter or total tumor diameters; a progress disease (PD) is an increase of >20% in the longest tumor diameter or total tumor diameters, or with new lesion. Both CR and PR stand for the overall response rate.
Local failure, regional failure, distant failure, and survival were all measured from the start of SBRT. Local failure was defined as abnormalities in enhanced CT scan that were confirmed by progressive and incremental increases in the standardized uptake values of a lesion in serial PET/CT imaging in the radiation field, with or without biopsy. Tumor progression in the hilum, mediastinum, or supraclavicular lymph nodes was considered regional failure. Tumor progression in the contralateral lung, distant organs, and tissues or bones was regarded as distant failure. Overall survival (OS) was calculated from the start of SBRT to the date of death of any causes.
Toxicities were graded using the radiation therapy oncology group (RTOG). Acute radiation toxicity occurred between day 1 and day 90 from the start of radiation treatment and late radiation toxicity occurred after day 90.
Patients were assessed after completion of SBRT at 4 to 6 weeks and then were followed up every 3 months for the first and second year. Thereafter, follow-up was carried out in 6-month intervals. The last follow-up was March 1, 2015. Clinical examinations for grading of toxicity and chest CT were performed at every follow-up. The PET/CT was optional and mainly performed when tumor relapse was suspected.
Relation to clinical response was examined with logistic regression analysis. Survival time and time to other end points were calculated using the Kaplan-Meier method. For overall survival, any death was defined as an event. Patients who died from other diseases without tumor progression at that time were considered as censored. All P were 2-sided and P < .05 was regarded statistically significant. Statistical analyses were performed using the SPSS 16.0 software (SPSS Inc Chicago, Illinois).
Results
Dosimetric Values
The dosimetric information of total lung DVHs and nearby OARs were stored, and summary of DVH-based analysis is shown in Table 2. Median lung doses (MLD), V2.5, V5, V10, and V20 of lung for all the patients in this study were 0.87 Gy, 39%, 23%, 13%, and 5%, respectively. Median V35, V50 of esophagus, and V30 of heart were all almost 0. Among these parameters, V2.5 (P = .006), V5 (P = .011), V10 (P = .004), and V20 (P = .028) of lung were all significantly different between the central arm and the peripheral arm (Table 2).
Dosimetric Factors.

Abbreviations: GTV, gross target volume; V20, volume of both lungs (minus the gross tumor volume) receiving 20 Gy; SC, spine cord; SD, standard deviation.
Response Evaluation
The overall response of 3-month, 1-, 2-, and 3-year in the central and the peripheral arms were 66.7%, 83.3%, 61.1%, and 72.2% and 66.7%, 80%, 80%, and 80%, respectively, without significant difference. In the third year, the CR was 27.7% and 26.7%, and the PR was 44.5% and 53.3% in the central and the peripheral arms, respectively. The rates of progression free of the 1-, 2-, and 3-year in the central and the peripheral arms were 94.4%, 83.3%, and 77.7% and 86.7%, 86.7%, and 80%, respectively.
Toxicity
Despite several patients with advanced age and high comorbidity rates, HT-based SBRT was well tolerated. None required drug intervention for toxicity during the SBRT. Adverse events in this study were mild. Three (16.7%) patients in the central arm and 1 (6.7%) patient in the peripheral arm had acute radiation pneumonitis.
No one had grade 3 or higher toxicity. Four (22.2%) patients with central lesions and 1 (6.7%) patient with peripheral lesion reported or were found to have acute grade 1 radiation esophagitis, and no one was found to have grade 2 radiation esophagitis. No grade 2 or higher radiation esophagitis was observed in the peripheral arm, compared to 1 patient with grade 2 radiation esophagitis in the central arm (P = .008). Four patients (22.2%) with late grade 2 pneumonitis were observed in the central arm. Five (15.1%) of all patients felt slight fatigue during RT. No patient reported chest pain, rib fracture, and hematological toxic effects. Blood routine tests were normal before and after SBRT. Hematology toxicity and skin injury were not observed.
Failure Pattern
One case of local metastasis and 1 contralateral lung metastasis were observed in the peripheral arm. In the central group, 1 patient had local metastasis, 2 had contralateral lung metastasis, and 1 had adrenal gland metastasis. All relapses were confirmed by a combination of CT and PET/CT. Six patients died during the period of observation after treatment, including 4 patients in the peripheral arm and 2 patients in the central arm. Only 1 patient died of cancer progression in the 2 arms.
Survival
All patients completed therapy without interruption. No patient was lost to follow-up. Mean follow-up time was 38.1 months (95% CI: 33.5 to 42.6) for all the people, and the mean OS was 42.8 months (95% confidence interval [CI]: 37.1-48.6 months) and 36.8 months (95% CI: 27.8-45.8 months) in central and peripheral arms (P = .516), respectively. The median survival was 47.7 months in the central arm and 46.1 months in the peripheral arm (Figure 1). The 3-year OS rates for patients with central versus peripheral tumors were 75.6% and 72.0%, respectively (P = .516). The median time to tumor progression was 40.0 months (95% CI: 22.4-57.6 months) in the peripheral arm and was not reached in the central arm.

Overall survival for the central and peripheral stage I lung tumors after the start of stereotactic body radiotherapy (SBRT).
No significant differences were observed in 3-year LCRs (94.4% vs 93.3%, P = .854), 3-year regional control rates (94.4% vs 86.7%, P = .412), 3-year distant control rates (64.2% vs 61.7%, P = .509), and 3-year disease-free survival rates (60.6% vs 59.8%, P = .755) between the central and the peripheral arms.
Discussion
Overall response rate (ORR = CR + PR) is another evaluation of the short-term treatment efficacy. In this study, 3-year ORR in the central and peripheral arms was 72.2% (CR 27.7%, PR 44.5%) and 80% (CR 26.7%, PR 53.3%), respectively. Timmerman et al 12 reported that ORR was 89% (CR 51%, PR 38%) for 55 patients with early stage non–small-cell lung cancer (NSCLC) at 3-year after the completion of SBRT. In a prospective phase II clinical trial, 39 patients with early stage lung cancer underwent SBRT using daily online CBCT image guidance, and the ORR at 6 weeks, 4 months, and 1 year was 46% (CR 3%, PR 43%), 53% (CR 15%, PR38%), and 91% (CR 27%, PR 64%), respectively. 13 The ORR they reported was found to be increasing with the follow-up to some extent. Long-term ORR reported in our study was lower than those of the above-mentioned researches that may be of insufficient samples.
It is important to protect the surrounding normal tissues during RT. The toxicity of SBRT to peripheral lung cancer is minimal. However, SBRT might induce irreversible damages to esophagus, trachea, or major blood vessels for central lung cancer. Hence, more attention should be paid to central NSCLC radiation therapy. In the present study, 18 patients in the central group accepted moderate dose (9 patients received 50 Gy/10fractions and 9 patients received 60 Gy/10fractions) and 3 patients had acute radiation pneumonitis. The symptom was cough, without sputum and fever. The radiation therapy was not interrupted. The other 1 patient with pulmonary emphysema had acute radiation esophagitis. He had a pain during feeding, and semi-liquid diet was applied. After 2 weeks postradiation therapy, the pain was relieved. The tumor of this patient was located near the left main bronchus. No obvious side effect was observed in the peripheral arm. Grade 3 or higher RP and grade 2 or higher other toxicities were not observed. Other researches also demonstrated that SBRT had minimal side effects. Timmerman et al 12 reported that among 55 patients with inoperable early peripheral NSCLC underwent IMRT/VAMT-based SBRT. The dose was 18 Gy/3 times, and the side effect was acceptable.
The rate of protocol-specified treatment-related grade 2, 3, and 4 adverse events was 31%, 24%, and 2%, respectively, and no grade 5 treatment-related adverse events was reported. 12 Xia et al 14 reported the toxicities of 43 patients with inoperable stage I/II NSCLC who underwent body gamma-knife radiosurgery treatment prospectively were as follows: both acute grade 2 and grade 3 pneumonitis were 2.3% (1 of 43). It is more difficult for HT to deliver beams to a restricted area compared with noncoplanar treatment methods because HT produces continuously rotating arc-type beams. Helical tomotherapy was inferior to IMRT in sparing low-dose (below 20 Gy) volume regions (P < .001) and in minimizing the integral dose to the entire thorax for patients with stage III NSCLC. 15 However, in the previous studies, HT was better in protection of surrounding tissues than VMRT and IMAT. 10,11 Therefore, whether the low-dose irradiation of large lung volumes or the high-dose irradiation of small lung volumes may be the main cause for pneumonitis is still uncertain. It is still controversial that whether low dose with high volume radiation or high dose with low volume radiation induces pneumonitis. With regard to lung dose constraints of HT-based SBRT, for no deviation, the V20 must be less than 10%. For a minor protocol deviation, the volume of lung receiving 20 Gy (V20) was required to be restricted to 15% according to the RTOG 0236 protocol constraints of IMRT-based SBRT. In this study, MLD, V2.5, V5, V10, V20, V30, V40, V50, and V60 were 0.87 Gy, 39%, 23%, 13%, 5%, 2.5%, 1%, 0, and 0, respectively. The V20 (5%) in this study was within the lung dose constraints of HT-based SBRT.
Our results of toxicity were similar to the outcomes of Xia et al’s. 14 It may suggest that HT-based SBRT were as capable of reducing pneumonitis as body λ-knife radiosurgery.
Metastasis is the main failure pattern in early-stage lung cancer. 16,17 Stereotactic body radiotherapy for early-stage lung cancer is reported for providing excellent LCRs (93%-98%). 12,18,19 In our study, no significant difference was observed in tumor LCR, regional progression-free rate, and metastasis-free rate between these 2 groups, and the 3-year LCRs were high. These results were coincident with those of others 12,18,19 and also suggested that the main reason of therapy failure was distant metastasis. Regional failure rate in the hilum and mediastinum was quite low, which might result from immunoreaction after SBRT. Hypofractionated RT induces an abscopal effect, which was defined as a significant growth inhibition of the tumor outside the field. 20 For example, intercellular adhesion molecule-1 (ICAM-1) and retinoic acid early inducible-1 (Rae-1), one of the natural killer cell group 2D (NKG2D) ligands, regulatory T cells, were increased by radiation. 21,22 The complex sequence of inflammatory events can cause further cell and tissue damage to eliminate potential invaders and can lead to cytoprotective responses that would help to limit the spread of damage and wound healing through tissue regeneration or replacement ultimately. 23,24
The accuracy and precise of initial staging pre-RT might impact on the efficacy. Two patients were diagnosed with early stage lung cancer based on contrast-enhanced CT without PET/CT examination. After SBRT, the size of tumor increased and mediastinal lymph node enlargement with high uptake of FDG were found. It might be assumed that occult tumors at diagnosis were harbored in those patients, which may be interpreted that not all the patients in this study had received PET/CT scan at the initial staging. It reveals that PET/CT scan should be suggested when patients are in a high risk of occult tumors.
Helical tomotherapy is a technology that delivers fan-beam IMRT under megavoltage computed tomography (MVCT). The MVCT can be used to measure the degradation of tumor daily. Weekly used ART technique can reduce the volume of lung receiving 20 Gy (V20). Helical tomotherapy is a safe technique for treating moving target and hypofractionation, which has been proved by University of Virginia. 25 Helical tomotherapy-based SBRT showed excellent safety, efficiency, and mild toxicity, although most of the previous trials about HT-based SBRT were insufficient of samples. 26 –28
Although SBRT for central tumors may be queried or even prohibitive for its risks, VU University Medical Center recently published a systemic review which suggested SBRT can offer a safe and effective curative treatment for patients with central tumors who were unfit for surgery. 29 Similar to these results, 50Gy/10Fx (BED = 75 Gy) or 60Gy/10Fx (BED = 96 Gy) was delivered in central lung cancer in this study; the median V20 for lungs in central arm was 6.75% (range, 2%-17%), which was markedly below 15%; MLD for lungs was 0.87 Gy (range 0.23-15.4 Gy). Lower V20 and MLD may contribute to the protection of lung with mild toxicity. This study proved that a total dose of 50 Gy in 10 fractions or 60 Gy in 10 fractions was feasible of HT in SBRT for central stage I lung cancer.
Conclusion
Delivery of IG-SBRT with 50Gy/10fx via HT in patients with stage I central lung cancer is safe, well tolerated, and results in an excellent LCR as well as 50Gy/5fx in patients with peripheral stage I lung cancer.
Footnotes
Acknowledgments
We thank Dr Cheng Yang, Department of Urology, Zhongshan Hospital, Fudan University for his assistant in manuscript revising.
Authors’ Note
ZZC organized the study and helped to write the manuscript. HJ treated the most of patients with lung cancer, organized and performed the study, statistical analysis, and drafted the manuscript. HY contributed to collect the clinical data and performed the statistical analysis. SSM and HY performed the follow-up the most of patients with lung cancer. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded Supported by the Medical Guidance Program of the Science and Technology Commission of Shanghai Municipality, (No. 14411970100 to Dr. Jian He).
