Abstract
We investigated the dose uncertainty caused by errors in real-time tracking intensity-modulated radiation therapy (IMRT) using the CyberKnife Synchrony Respiratory Tracking System (SRTS). Twenty lung tumors that had been treated with non-IMRT real-time tracking using CyberKnife SRTS were used for this study. After validating the tracking error in each case, we did 40 IMRT planning using 8 different collimator sizes for the 20 patients. The collimator size was determined for each planning target volume (PTV); smaller ones were one-half, and larger ones three-quarters, of the PTV diameter. The planned dose was 45 Gy in 4 fractions prescribed at 95% volume border of the PTV. Thereafter, the tracking error in each case was substituted into calculation software developed in house and randomly added in the setting of each beam. The IMRT planning incorporating tracking errors was simulated 1000 times, and various dose data on the clinical target volume (CTV) were compared with the original data. The same simulation was carried out by changing the fraction number from 1 to 6 in each IMRT plan. Finally, a total of 240 000 plans were analyzed. With 4 fractions, the change in the CTV maximum and minimum doses was within 3.0% (median) for each collimator. The change in D99 and D95 was within 2.0%. With decreases in the fraction number, the CTV coverage rate and the minimum dose decreased and varied greatly. The accuracy of real-time tracking IMRT delivered in 4 fractions using CyberKnife SRTS was considered to be clinically acceptable.
Introduction
Stereotactic body radiotherapy (SBRT) has been spreading worldwide as a new treatment modality for stage I nonsmall-cell lung cancer. 1,2 To improve its safety and efficiency, reducing the internal target volume and planning target volume (PTV) margins and dose escalation is desirable. To reduce the margins, compensation for respiratory tumor movement needs to be considered. 3,4 The breath-hold method, however, has limitations for elderly patients and patients with severe pulmonary disease. The treatment time is prolonged and residual motion is always present for breath-hold and gating techniques compared with the tracking method. 5 As for tracking, the CyberKnife Synchrony Respiratory Tracking System (SRTS, Accuray, Sunnyvale, California) is a new technology that can make real-time adjustments for changes in tumor position. 6 In CyberKnife planning, both isocentric and conformal nonisocentric intensity-modulated radiation therapy (IMRT) plans are available. 7 Both SRTS and conformal nonisocentric IMRT planning can reduce margin expansion and decrease doses to normal tissue, so a combination of these techniques should be employed whenever possible. However, the SRTS has inherent problems in terms of tracking errors between the target and the center of the gantry and the phase difference between the target and the external marker. 8,9 In IMRT for a mobile target, the interplay effect between patient’s respiratory motion and complex collimator movement can compromise the dose accuracy, creating hot and cold spots within the target, which cannot be resolved by adding margins. 10 –12 The purpose of this study was to evaluate the dose uncertainty caused by tracking errors in real-time tracking IMRT using CyberKnife SRTS.
Methods
Patient Eligibility and Characteristics
Twenty lung tumors that had been treated with real-time tracking non-IMRT SBRT using CyberKnife SRTS between February 2011 and April 2012 were used for this study. The eligibility criteria were as follows: (1) histologically confirmed primary nonsmall-cell lung cancer staged as T1a-T2aN0M0 (TNM7th) or lung metastasis with ≤5 cm diameter; (2) dose constraints of the organs at risk within acceptable ranges; (3) no previous radiotherapy around the lesion; (4) age >20 years; (5) Eastern Cooperative Oncology Group performance status ≤2; (6) treatment with the tracking system feasible; (7) forced expiratory volume 1.0 ≥700 mL and Pa
Patient and Tumor Characteristics.

Abbreviation: IMRT, intensity-modulated radiation therapy.
aMedian (range).
bAccording to the Eastern Cooperative Oncology Group.
cSmoking history (+) indicates a Brinkman Index of ≥400.
Intensity-Modulated Radiation Therapy Planning Using SRTS
Radiation treatment was planned using a computed tomography (CT)-based 3-dimensional treatment planning system (Multiplan; Accuray) as described in detail previously. 14 Two-mm thick CT (40-slice, SOMATOM Definition AS; Siemens Medical Solutions, Munich, Germany) images were taken during end-exhalation phases. Then, 4-dimensional (4D) CT (Respiratory Gating System AZ-733V; Anzai Medical Co Ltd, Tokyo, Japan) was performed under normal breathing controlled by an audio guidance system. This was an adopted phase-based approach and detected 11 phases (end inspiration to end inspiration in 10% increments; 0%-100%). The lesions, including spiculae under the lung window, were taken as the gross tumor volume (GTV). The clinical target volume (CTV) was equal to the GTV. The PTV was defined as the CTV plus a tracking error in each patient and a setup margin of 2 mm in all directions. Our method for estimating tracking errors has been described in detail previously. 15 We have developed a system for obtaining pretreatment statistical measurements of the CyberKnife tracking error by using beam’s eye view (BEV) images to enable the prediction of the patient-specific accuracy. Briefly, respiratory motion data of the internal tumor position (ITP) and the external marker position (EMP) were derived from the cine magnetic resonance imaging (MRI) images obtained from each patient. The dynamic motion phantom was used to reproduce both the ITP and the EMP motions of each patient. The CyberKnife was subsequently operated with the SRTS, with a charge-coupled device (CCD) camera mounted on the head of the linear accelerator. The BEV images from the CCD camera were recorded during the tracking of a ball target by the linear accelerator. The ball was stationary at the center of the BEV image when tracking was error free (no residual motion) but would be shifted from the center of the BEV image when the tracking was not error free. The tracking error was defined as the distance from the center of the BEV image to the center of the ball in the BEV images. The tracking error was measured at 15 Hz using VideoCapture (software developed in-house by author HS for analyzing the tracking error). The software used a 320 × 240-pixel image with a resolution of about 0.5 mm at the distance of the ball. We calculated the tracking error with which the target could be tracked with a probability of over 95% (Ep95) for each beam direction. The detection accuracy of the tumor position (determined from cine MRI images) was <0.2 mm. Moreover, the detection accuracy of the tracking error when using the BEV images was <0.2 mm. These 2 detection accuracies were derived from our measurement system and were not obtained from the SRTS. After validating the tracking error in each case, we made 40 IMRT plans using 10 different collimator sizes for the 20 patients (sizes: 10, 12.5, 15, 20, 25, 30, 35, 40, 50, and 60 mm). The collimator size was determined for each PTV diameter; smaller ones were one-half of the PTV in diameter and larger ones were three-quarters (Figure 1). The planned dose was 45 Gy in 4 fractions prescribed at 95% volume border of the PTV (prescribed isodose line ≥70%). The calculated model was the Ray tracing. For each IMRT planning, the number of beams was adjusted so that treatment was finished in about 30 minutes, simulating actual clinical treatment. The numbers of beams were 100 to 150 for the smaller collimator and 50 to 110 for the larger one.
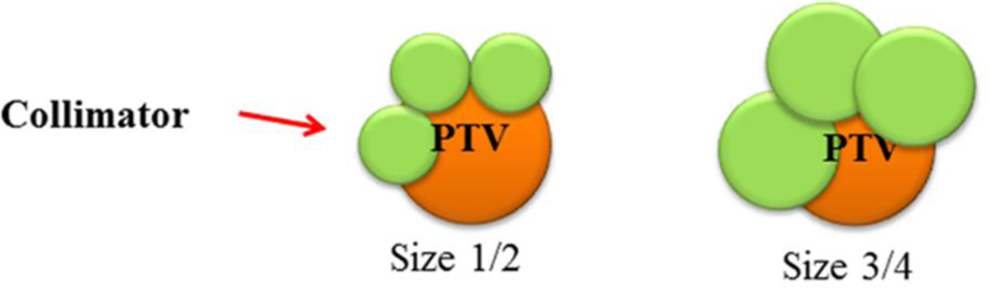
Tracking intensity-modulated radiation therapy (IMRT) planning. The collimator size was determined for each planning target volume (PTV) diameter.
Simulation of Dose Planning and Analysis of Dose–Volume Histograms
The tracking error in each case was substituted into the calculation software (ShioRIS, software developed in-house by author HS) and randomly added using a normal distribution with σ = tracking error in the setting for each beam. The dose rate for each beam was set at infinity since the influence of the interplay effect was assumed to be at the worst state. Therefore, the tracking error was added to each beam. A series of IMRT plans incorporating tracking errors was simulated 1000 times. In the 40 000 plans, the maximum, minimum, and coverage (D99 and D95) dose data on the CTV (tumor) were compared with the original data. Thereafter, the same simulation was carried out by changing the fraction number from 1 to 6 in each IMRT plan. This was also a simulation in which the same 45 Gy was simply divided into 1 to 6 fractions, and the beam number in each fraction did not change. Finally, a total of 240 000 plans were analyzed.
Results
The median actual tracking errors of each patient were 1, 1, and 1.5 mm (range: 1-2.5, 1-2.7, and 1-3.5, interquartile range: 1-1, 1-1.5, and 1.5-2) for the lateral, anterior–posterior, and superior–inferior directions, respectively. The median PTV volumes were 19.9 cm3 (range: 5.6-65.5). The mean homogeneity and conformity indices and the conformity number of the original planning were 1.26, 1.14, and 0.78, respectively, for the smaller collimator (1/2 size), and 1.29, 1.11, and 0.81, respectively, for the larger one (3/4 size). Figure 2 shows the changes in CTV maximum and minimum doses, and dose coverage diagrams for each collimator size in the 4-fraction plans. The CTV maximum doses of the original data were higher than those of the planning data in the overall sample cases. In contrast, the CTV minimum doses of the planning data were higher than those of the original planning data. In the larger collimator, however, a decrease in the CTV minimum dose was observed in 9 cases. Figure 3 shows the worst examples of the CTV maximum and minimum doses and dose coverage scatter diagrams for each collimator size, respectively. The mean homogeneity and conformity indices and the conformity number of the simulated planning were 1.22, 1.09, and 0.77, respectively, for the smaller collimator and 1.24, 1.07, and 0.79, respectively, for the larger one. The change in dose coverage was small (95% confidence intervals were about 3% or less). However, the change in dose coverage was relatively large in the case with a large tracking error. In the worst case, the actual tracking error was 1.8, 2, and 2.5 mm for the 3 directions, respectively. Table 2 shows the changes in the CTV dose data and coverage according to the fractionation schedule. With decreases in the fraction number, the CTV coverage and the minimum dose decreased for each collimator. Moreover, the 95% confidence intervals varied greatly.

The 4-fraction simulation results showing the change in clinical target volume (CTV) maximum, minimum, and coverage doses for each collimator. The mean and 95% confidence interval values are shown.

The worst example of the frequency distribution graph that shows the change in CTV maximum, minimum, and coverage dose when the tracking error is added in all cases. The red line shows the CTV dose of the original planning data. The increase in the minimum dose and dose coverage is shown in the smaller collimator size of this example and just the opposite occurred in the larger collimator size. CTV indicates clinical target volume.
Changes in the CTV Dose Data and Coverage According to the Fractionation.a

Abbreviations: CTV, clinical target volume; D99, 99% volume border of the CTV; D95, 95% volume border of the CTV.
aThe values indicate mean and the 95% confidence interval.
Discussion
It is well known that the influence of the interplay effect may decrease as a result of blurring of errors when IMRT is delivered with 30 or more fractions. 10,16 There have been some reports about IMRT using TomoDirect (Accuray) for upper lung tumors, mediastinal lesions, and breast cancers. 17,18 The IMRT using SRTS-hypofractionated irradiation similar to that used in this study has been carried out all over the world. 19,20 In fact, however, clinical use of hypofractionated IMRT using SRTS has expanded, while the relationship between tracking errors and interplay effects have not been well studied. 21 The 5 to 7 mm margins have been uniformly added to the CTV in all directions in most institutions without even analyzing the tracking error of each patient. Pepin et al 8 warned of the need for suitable margins that would provide 95% point coverage for a target, considering a magnitude of 2σ from the mean of the Gaussian in each direction from the data log files produced by the SRTS model. In 4 fraction cases in this study, if the tracking errors were within 2 mm in all directions, the difference in CTV dose data and coverage was within 2% to 3% and was considered to be clinically acceptable. Therefore, this study suggested that hypofractionated IMRT using SRTS for the central lesion or peripheral lesion with a smaller tracking error may be acceptable, and it seems that it is particularly effective for the central lesion with various adjacent OARs. On the other hand, with decreases in the fraction number, the CTV coverage and the minimum dose decreased and varied greatly. Particularly, for 3 or fewer fractions, the error of the dose coverage became more than 3% and the dose to a tumor target may not be guaranteed. The IMRT using SRTS in which the fraction number is large may be possible even if the tracking error is large.
One of the limitations of this study is that only virtual planning was performed. For example, if the tracking error is large in cases with a peripheral lesion in the lower lobe, elderly patients with an unstable respiratory curve or patients with severe pulmonary disease, SRTS using IMRT may not be acceptable because of the increase in the uncertainty of tracking accuracy due to the respiratory phase difference and instability of the baseline. 22,23 Indeed, the CTV minimum doses of planning data were lower than those of original planning data in the 9 larger collimator (3/4 size) cases. The tracking errors of these plans were relatively high (approximately 2 mm or more in all directions). Further investigations systematically sorted out by the magnitude of tracking errors are warranted with more patients. Moreover, we did not add the tumor deformation during treatment. Webb 24 indicated that the tumor deformation observed in the CT study may not represent the reality during treatment and an additional margin could be required. Moreover, we could not evaluate the 4D dose data of organs at risk using this in-house software because organs at risk themselves move with respiration and anatomical volume changes in real time. Hence, this study only investigated target dose distribution. Therefore, 4D planning or other additional analysis may be needed. 25 The maximum and minimum dose values are particularly sensitive to single voxel hot/cold spots. Further investigation including a dose index about organs at risk using a 4D planning software are warranted in the future.
The factor of treatment time is very important in irradiation using a technique such as tracking or gating. It is necessary to ensure respiratory stability, including the baseline shift and phase control, to guarantee the correlation between them during irradiation. 24 –26 Malinowski et al 27 reported that both tumor motion and the relationship between tumor and respiratory surrogate displacements change in most treatment fractions with patient in-room time of 30 minutes. In our facility, we have used an audio guidance system and continuous positive airway pressure to stabilize the correlation mentioned previously as well as doing IMRT planning using 1 type of collimator so that the irradiation time is within 30 minutes. Therefore, further investigation of the proper use of respiratory motion guidance and IMRT planning with various collimators and the formulation of guidelines are warranted in the future.
Conclusion
Although the interplay effect was detected in this simulation study, the accuracy of real-time tracking IMRT delivered in 4 fractions using CyberKnife SRTS was considered to be clinically acceptable. Using a smaller collimator and 4 or more fractionation led to a decrease in uncertainty. In the case of tracking IMRT, however, it is necessary to evaluate the interplay effect for every case in order to provide safe treatment.
Footnotes
Abbreviations
Acknowledgments
The authors are grateful to the members of the Yokohama CyberKnife Center including Mr Kosaku Inada, Mr Manabu Senda, and Ms Kumiko Ogawa for their valuable help in this research.
Authors’ Note
All authors certify that this article has not been published in whole or in part nor is it being considered for publication elsewhere.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
