Abstract
In recent years, increasing attention has been directed toward the mechanical microenvironment, which has become a major research focus in elucidating the mechanisms underlying tumor progression. On one hand, endogenous mechanical cues, such as matrix stiffness, fluid shear stress(FSS), and mechanical strain, have been shown to promote tumor cell malignant phenotypes, including proliferation, migration, 4 invasion, and drug resistance. These activities are primarily through mechano-transduction pathways involving integrin–FAK and YAP/TAZ. Meanwhile, mechanical microenvironment was also found to influence immune cell activity and tumor immune evasion. On the other hand, exogenous mechanical stimuli, such as magnetic fields and ultrasound, can be harnessed for therapeutic purposes by altering the tumor mechanical microenvironment in situ to enhance drug delivery or directly induce cell apoptosis. Furthermore, mechanical interventions exhibit synergistic effects when combined with conventional therapies like radiotherapy and chemotherapy, thereby amplifying antitumor efficacy. Inspired by these impressive outcomes, recent advances and mechanism of mechanical microenvironment dependent tumor growth interventions are reviewed in current work, along with outlooks of the translation challenges of this emerging strategy. As an effort to achieve precise and effective tumor control while overcoming current limitations, attempts such as multi-omics and artificial intelligence should be considered to empower personalized mechanical intervention.
Keywords
1. Introduction
According to the latest data released by the International Agency for Research on Cancer (IARC), the global cancer burden continues to increase. In 2022, there were approximately 20 million new cancer cases and about 9.7 million cancer-related deaths worldwide. Current statistics show that lung cancer, breast cancer, and colorectal cancer rank as the three most commonly diagnosed malignancies. Based on current trends, the IARC projects that by 2050 the annual number of new cancer cases will exceed 35 million globally, with cancer-related deaths potentially approaching a doubling, underscoring a further intensification of the global cancer burden. Conventional treatments including surgical resection, radiotherapy, and chemotherapy remain the primary clinical options along with unsatisified medical outcomes. 1 For example, surgical interventions carry significant risks and potential complications post-surgery 2 ; radiotherapy may induce systemic dysfunction and radiation-related injuries due to cumulative dose effects 3 ; and chemotherapy potentially disrupt the growth and proliferation of local healthy cells, leading to severe adverse effects. 4 With biotechnology advancement, emerging strategies such as immunotherapy, CAR-T cell therapy, and targeted therapy are being progressively applied into multimodal clinical practice, paving the possibility of personalized, precision, and effective therapies for tumor control. 5 However, immunotherapy is challenged by substantial interpatient variability in therapeutic response and high costs, while CAR-T therapy exhibits tumor-type specificity to limit its applicability. 6 Consequently, exploration of effective, economically viable, and precise new strategies remains a pivotal focus in tumor intervention research.
Advances in tumor mechanobiology have made it feasible to exploit the mechanical microenvironment attributes of tumors to elucidate pathogenic mechanisms and develop novel diagnostic and therapeutic strategies. For example, researchers demonstrated that mechanical forces can be transmitted via actin filaments and the cytoskeleton, triggering mechano-transduction pathways that activate transcription factors and ultimately alter cellular function and phenotype. 7 These findings imply a fundamental relationship between mechanical signals and tumor cell activities, including metabolism, proliferation, and other malignant behaviors.
The tumor microenvironment (TME) undergoes significant biomechanical remodeling during cancer progression, characterized by increased extracellular matrix(ECM) stiffness, interstitial fluid shear stress (FSS), elevated fluid pressure, and solid stress.
8
The conversion of mechanical signals into biochemical signals in tumor cells depends on a functional mechanical linkage network formed by motor proteins, the cytoskeleton, and transmembrane adhesion molecules (such as integrins). This continuous mechanical transmission system enables external mechanical stimuli to be sensed by cells and converted into specific downstream signaling responses. The cytoskeleton plays a central hub role in the intracellular transmission and spatial distribution of mechanical forces. These mechanotransduction processes ultimately remodel tumor cell phenotypes and promote malignant behaviors such as proliferation, invasion, and metastasis(Figure 1).8,9 Furthermore, these changes disrupt tissue architecture, compromise drug delivery, promote treatment resistance, and inhibit immune cell recruitment and effector functions. Besides, changes in the biomechanical properties of ECM causes the malignant transformation of epithelial cells, while fibroblast-driven ECM stiffening and structural remodeling are pivotal factors in cancer progression.
7
Increased tumor stiffness and solid stress modulate pro-tumorigenic signaling via mechano-transduction, influencing tumor cells proliferation and invasion. For example, sustained proliferation of tumor cells within restricted spaces results in pronounced tissue pressure and cell stretching, thereby triggering the activation of stretch-gated Piezo channels.
10
Notably, mechanical cues within the tumor microenvironment usually operate as a highly coupled network rather than as isolated, independently changing variables.
11
Therefore, research on the tumor microenvironment and its clinical translation should shift from traditional single-factor approaches toward a multidimensional and dynamically integrated systems analysis framework. When tumor cells metastasize, the cells are affected by various mechanical factors from the surrounding microenvironment
9

Mechanical interventions (such as reducing matrix stiffness and applying dynamic mechanical stress) have been proven to improve the TME, enhance drug delivery efficiency, and even directly suppress the malignant phenotype of tumor cells(Figure 2).
12
Moreover, integrating mechanical interventions with other treatment modalities can potentially lead to synergistic therapeutic outcomes. For instance, mechanical preconditioning can radiosensitize tumor cells by disrupting DNA repair pathways.
13
This strategy may permit dose reduction, decrease adverse effects, and help overcome radiation resistance. These advances thereby inspire the overview of current progress of mechanical microenvironment-dependent tumor growth intervention. Currently, research in tumor mechanobiology is still largely focused on the fundamental mechanisms underlying its influence on tumor initiation and progression, whereas systematic summaries and discussions regarding its clinical translational potential remain relatively limited.8,9 In this context, previous reviews have comprehensively summarized the mechanistic roles of various mechanical factors in tumor progression as well as related fundamental studies. Building upon these studies, this review further outlines the potential therapeutic applications of mechanical factors in cancer treatment and the key challenges they face from a clinical translational perspective. In addition, we briefly discuss potential future directions involving the integration of artificial intelligence and computational modeling to enable personalized mechanical intervention therapies, with the aim of facilitating the clinical translation of mechanoregulation-based strategies. Mechanical cues in TME and therapeutic strategies. Schematic of mechanical cues in TME, mechanotransduction pathways, cellular function impacts, and combination therapy strategies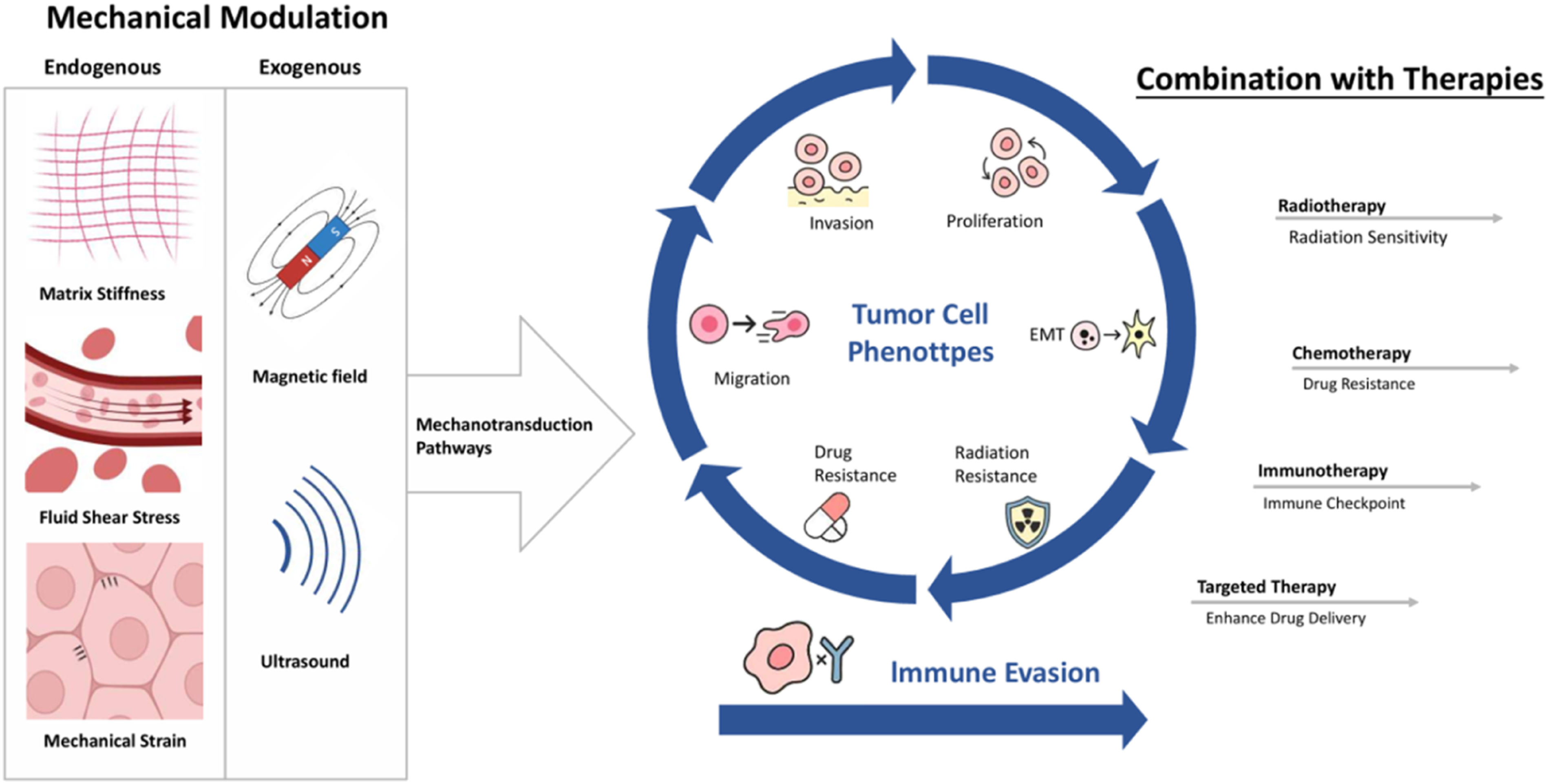
In conclusion, this review synthesizes current advances in understanding how mechanical stresses shape tumor cell fate and modulate therapeutic sensitivity. We discuss emerging mechanotransductive pathways, preclinical interventions, and translational barriers, emphasizing the need for standardized modeling and quantitative frameworks. By integrating molecular insights with preclinical and emerging clinical evidence, we aim to delineate the path toward a unified mechanobiological framework for oncology—transforming mechanical modulation from a conceptual paradigm into a clinically actionable strategy.
2. Effects of Endogenous Mechanical Microenvironment on Tumor Tissue and Its Therapeutic Applications
2.1. Matrix Stiffness
Matrix stiffness plays a pivotal and highly regulatory role in driving tumor progression. Relative to normal tissues, tumor tissue stiffness is markedly elevated, typically rising by 5- to 20-fold. For instance, normal breast tissue matrix stiffness ranges from 0.2 to 1.83 kPa, whereas in malignant breast tissue it rises to 4–10 kPa.
14
With malignant progression, tumor cells together with cancer-associated fibroblasts (CAFs) contribute to increased matrix stiffness through lysyl oxidase (LOX)-mediated collagen cross-linking, elastin deposition, and additional remodeling pathways (Figure 3). During this process, actin filaments (F-actin) serve as key mediators that enable cells to detect and respond to variations in matrix stiffness.
8
In low stiffness microenvironments, F-actin polymerization remains minimal, leading to limited formation of stress fibers. However, as tumor progression advances and matrix stiffness increases, actin polymerization is markedly enhanced, promoting the formation of robust and stable stress fiber structures. Concurrently, the activity of F-actin–associated capping and severing proteins, such as Cofilin, is reduced.
15
This reduction stabilizes cytoskeletal architecture and facilitates the nuclear translocation of YAP/TAZ, thereby activating transcriptional programs that promote cellproliferation and suppress apoptosis. Consequently, a series of signaling cascades are activated, and through secreted factors such as TGF-β and EGF, collagen deposition is promoted, creating a positive feedback loop that sustains tumor-associated phenotypes including proliferation, migration, invasion, metastasis, therapy resistance, stemness, angiogenesis, and immune escape.8,14,16 Stiffness: on soft substrates, tumor cells spread less and have short actin filaments (the white fibrous structures distributed around the cells in the figure above). However, on stiff substrates tumor cells undertake a polarized morphology and develop stress fibers
Recent studies have further elucidated the multidimensional molecular mechanisms of this vicious cycle: at the mechanosensing level, the integrin-FAK axis serves as a core signaling pathway (e. g. , 16 kPa matrix significantly enhances FAK phosphorylation in hepatocellular carcinoma models, promoting proliferation and drug efflux); at the transcriptional regulation level, high stiffness remodels tumor behavior via the Hippo-YAP/TAZ pathway (e. g. , YAP nuclear localization in breast cancer activates the Wnt/β-catenin pathway, increasing the proportion of CD44+ cancer stem cells); and at the metabolic adaptation level, stiffness changes affect cellular metabolism (e. g. , in soft matrices, hepatocellular carcinoma cells induce mitochondrial dysfunction via the ROS/JNK pathway, upregulating glycolytic enzymes and ATP production, leading to chemotherapy resistance and stemness acquisition).17-19 YAP/TAZ are pivotal effectors of mechanotransduction that drive malignant transformation. Their aberrant activation represents a critical downstream event across diverse tumor types, where they orchestrate context-dependent transcriptional programs: in hepatocellular carcinoma, promoting inflammation-to-malignancy transition; in lung cancer, enhancing invasion and migration. 18 Mechanistically, matrix stiffening activates integrin–FAK signaling, which potentiates PI3K/Akt survival pathways and MAPK/ERK proliferation signals. YAP/TAZ also synergize with β-catenin to amplify Wnt transcriptional output, collectively promoting EMT and tumor progression. 20 During tumor progression, cells sense and transduce external mechanical signals through matrix viscoelasticity, a key mechanical platform, thereby modulating their responses to matrix stiffness and influencing cellular behavior. Recent studies have shown that matrices with rapid stress relaxation are more likely to promote cancer cell invasion, whereas slow relaxation may inhibit this process. 21 This indicates that “stiffness” in the tumor microenvironment is not a single physical parameter but rather a coupled system of stiffness and viscoelasticity. Understanding this coupling mechanism is crucial for developing therapeutic strategies targeting matrix remodeling.
The dense and highly cross-linked architecture of tumors presents a major barrier to efficient nanodelivery. Radiation can enhance tumor vascular permeability by activating hypoxia-inducible factor 1 (HIF1) or upregulating vascular endothelial growth factor (VEGF) expression through MAPK–dependent pathways. The synergistic effect of radiation and nanodelivery may kill tumors while increasing drug efficiency to reduce matrix stiffness and remodel the TME. 22 Beyond its established role in regulating chemotherapy sensitivity via mechanical modulation of tumor cell phenotypes, matrix stiffness is also recognized as a critical determinant of radio-resistance. For instance, in pancreatic cancer models, elevated matrix stiffness mechanically activates YAP/TAZ-mediated DNA repair pathways and suppresses apoptosis-related mechanotransduction, collectively promoting radiotherapy resistance. 23
In spite of directly modulating tumor cell phenotypes, matrix stiffness regulates additional cellular behaviors and tissue remodeling through mechanosensing-transduction systems, thereby influencing tumor growth. Notably, high matrix stiffness usually arises from the deposition of highly cross-linked collagen fibers, forming a dense stromal architecture and increasing tumor interstitial fluid pressure (IFP). 24 Further analysis of tumor specimens from patients with triple-negative breast cancer (TNBC) revealed that the number of tumor-infiltrating CD8+ T cells was significantly reduced in breast tumors with high collagen fiber density. 25 This observation suggests that a dense collagenous matrix may act as a physical barrier and increase the mechanical stiffness of the tumor microenvironment, thereby restricting the migration and infiltration of effector immune cells into the tumor core and ultimately contributing to the establishment of an immunosuppressive tumor microenvironment. 26 Meanwhile, increased matrix stiffness can further promote immune evasion by upregulating PD-L1 expression on the surface of tumor cells. 27 The above process is referred to as immune checkpoint blockade, during which multiple key signaling pathways—including PD-1/PD-L1, Wnt/β-catenin, PI3K/Akt/mTOR, and TGF-β/Smad—facilitate immune evasion primarily by modulating T cell activity.
Clinical studies in colorectal cancer have demonstrated that administration of the TGF-β pathway inhibitor LY3200882 decreases ECM accumulation and stromal stiffness, leading to TME remodeling and enhanced immune cell infiltration. Matrix softening strategies can remodel the tumor mechanical barrier, thereby improving drug penetration within tumor tissues and promoting immune cell infiltration.
Building on insights into this vicious cycle and its underlying mechanisms, therapeutic strategies have been developed to modulate tumor matrix stiffness through pharmacological targeting, enzymatic degradation, and physical modulation, aiming to disrupt critical molecules and signaling pathways and achieve targeted intervention within the mechano-biological regulatory network of the TME. Specifically, molecular targeting approaches function by selectively inhibiting critical enzymes including LOX and LOXL2, resulting in direct intervention in collagen cross-linking processes. Clinical evidence shows that the LOX inhibitor β-aminopropionitrile (BAPN) suppresses collagen crosslinking by inhibiting LOX activity, achieving a >2-fold softening of tissue stiffness and increasing drug penetration at tumor sites by 2.5-fold. 28 Mechano-transduction regulation entails blocking mechanosignaling cascades to influence matrix stiffness. Pharmacological strategies targeting the balance between ECM deposition and remodeling are also effective. 16 TGF-β inhibitors, including the clinically approved Pirfenidone, attenuate matrix stiffness by suppressing cancer-associated fibroblast (CAF) activation and collagen accumulation, offering a clinically translatable strategy for modulating the tumor mechanical microenvironment.29,30 Regulation of extracellular matrix stiffness enables the reprogramming of immune function to improve treatment outcomes. As matrix stiffness in the tumor microenvironment increases, the YAP/TAZ mechanotransduction pathway becomes activated and subsequently upregulates PD-L1 expression, forming the so-called “mechanical–immune checkpoint axis”. This regulatory axis promotes T-cell functional exhaustion by increasing PD-L1 levels on tumor cell surfaces and inducing an immunosuppressive microenvironment. 31 Targeting this mechanical axis can significantly enhance the efficacy of anti–PD-1 blockade therapy and suppress tumor growth. Previous studies have shown that demonstrates that combinatorial therapy with immune checkpoint inhibitors (e.g., PD-1/PD-L1 and CTLA-4 inhibitors) suppresses stromal fibrosis, leading to matrix softening and potentiation of T cell activity. 32 When combined with anti–PD-L1 immune checkpoint blockade, this strategy can significantly enhance antitumor efficacy. 33 Therefore, the combined application of matrix mechanical modulation and immune checkpoint inhibition is considered a promising synergistic therapeutic strategy. 32 In addition, enzymatic and physical modulation techniques have also been reported to reduce matrix stiffness, such as intra-tumoral injection of hyaluronidase and collagenase to degrade the collagen network and reduce matrix stiffness, thereby enhancing chemotherapeutic efficacy. Nanoparticle-based mechanical modulation is a core strategy to remodel the TME: collagenase or hyaluronidase-modified nanocarriers (e.g., gold nanoparticles, dextran) degrade ECM components (collagen, hyaluronic acid), which directly reduces matrix stiffness and relieves interstitial solid stress. This mechanical normalization enhances drug penetration as a secondary effect, while primarily sensitizing tumor cells to therapy via disrupted mechanotransduction (e.g., integrin-FAK-YAP axis). 34 Meanwhile, ultrasound, through cavitation and thermal effects, enables delivered nanoparticles to disrupt the cytoskeletal structure, reduce matrix stiffness, and thereby enhance drug permeability.35,36
Clinical studies indicate that matrix stiffness modulation has potential therapeutic efficacy in cancer treatment. For example, the TGF-β inhibitor LY3200882 has achieved positive outcomes in a phase II clinical trial for pancreatic cancer. 37 Nevertheless, the full biological consequences of over-softening or poorly controlled ECM modulation remain unclear, theoretically encompassing disruption of tissue barrier integrity and heightened risk of tumor spread. 38 Nevertheless, the full biological consequences of over-softening or poorly controlled ECM modulation remain unclear, theoretically encompassing disruption oi-f tissue barrier integrity and heightened risk of tumor spread. 28 Additionally, pharmacological interventions may cause off-target effects and systemic toxicity. Therefore, future clinical strategies should prioritize precise and selective matrix remodeling and enhance safety through dose and delivery optimization.
2.2. Fluid Shear Stress (FSS)
FSS is defined as the tangential force generated by interstitial fluid flow in the TME and by blood circulation acting on tumor cells. 39 Upon exposure to FSS, tumor cells respond via mechano-transduction and activation of associated signaling pathways. Briefly, FSS exerts multiple effects by regulating tumor cell proliferation, metastasis, invasion, and EMT processes, influencing the survival of circulating tumor cells (CTCs), and contributing to tumor cell dissemination by affecting tumor cell migration and intravasation. 8
Emerging evidence indicates that FSS modulates tumor cell behavior via diverse signaling mechanisms and essential biological processes. Within the 0.5–5 dyn/cm2 range, FSS induced rapid activation of the ROCK–LIMK–cofilin to YAP1–TEAD cascade promotes cell movement; while at higher FSS, TRPM7-mediated Ca2+ influx engages the RhoA/myosin-II and IQGAP1/Cdc42 axes, resulting in migration reversal and diminished intravasation efficiency.40,41 Cells with elevated TRPM7 activity demonstrate higher shear sensitivity, reduced intravasation efficiency, and less aggressive metastatic lesions. 41 Research into FSS-driven mechanisms of CTC invasion and metastasis reveals that various tumor cell lines exploit activation of the PRSS3/PAR2 signaling axis to promote invasion and metastasis. 42 Furthermore, high shear stress (HSS) enhances the expression of BMP ligands and integrin receptors, modulates cell cycle and ROS levels, and drives upregulation of p53, PGC-1α, NRF-1, and cytochrome c, which collectively impair mitochondrial function and trigger caspase-dependent tumor cell apoptosis. 43
Regarding increasingly validation of FSS in tumor progress, it can also be applied as therapeutic target. Existing evidence indicates that FSS can remodel tumor cell phenotypes at multiple levels. Accordingly, preclinical studies are leveraging these mechanisms to test various feasible regulatory strategies in vivo. 44 On one hand, mechanosensitive ion channel antagonists (e.g., TRPM7, Piezo1 antagonists) can weaken cellular responses to FSS by blocking mechanosensitive ion channels or signaling pathways. For example, the Piezo1 antagonist GsMTx4 reduces tumor cell migration by inhibiting nuclear deformation in CTCs. 45 On the other hand, a study explored an extracorporeal circulation device for CTC removal in a mouse model: blood was withdrawn via a venous catheter, exposed to high fluid shear stress (HSS) within a pump-driven system, and then reinfused into the body to eliminate circulating tumor cells (CTCs). 46 Moreover, delivering drugs in HSS regions or applying localized pulsed HSS via catheters can enhance drug penetration in tumor regions and improve tumor cell killing efficacy(e.g., in prostate cancer studies, drug penetration improved 9-fold and tumoricidal efficacy rose by approximately 5.2-fold). 47
Additionally, modulating FSS can indirectly influence other cell behaviors and affect tumor progression. For example, studies on FSS have shown that it can upregulate cancer-associated fibroblast (CAF)–mediated PD-L1 expression via the YAP/TAZ signaling pathway, thereby suppressing T-cell infiltration. Accordingly, investigators employed verteporfin (a YAP inhibitor) in combination with an anti–PD-1 antibody for synergistic therapy, achieving ∼50% inhibition of PD-1 expression while simultaneously blocking PD-1/PD-L1 interactions on tumor and immune cell surfaces. 48 Following treatment, CD8+ T-cell infiltration increased by ∼2.8-fold, mean tumor volume decreased by 68%, and tumor growth was almost completely suppressed, demonstrating pronounced antitumor activity and synergistic immune enhancement.49,50 These observations thereby imply current studies have found that FSS affects tumor cell phenotypes in multiple aspects. Clinically, the mechanisms of FSS on tumor cells can now be utilized to regulate FSS itself or the cells’ perception of FSS for therapeutic purposes.
Although modulation of FSS (FSS) has inspired multiple promising therapeutic strategies in preclinical cancer studies, clinical translation remains limited. Principal barriers include the lack of standardized in vitro FSS exposure models and quantitative dosimetric frameworks, as well as unresolved safety and off-target risks associated with targeting mechanosensitive ion channels. 51 Mechanopharmacology reviews highlight concerns regarding systemic inhibition of channels like Piezo or TRP, which may lead to unintended effects. Future efforts should focus on developing robust, standardized FSS exposure systems, defining safe and effective dosing paradigms, and exploring locally targeted mechanobiological therapies (e.g., via delivery of Piezo or TRPM inhibitors) in combination with immunotherapy and pharmacotherapy to maximize efficacy while minimizing risk. 52
2.3. Mechanical Strain
Mechanical strain, including both mechanical stretching and compressive stress, exerts complex effects on tumor cell behavior. During tumor growth, excessive ECM deposition, tumor overgrowth-induced tumor interstitial pressure (TIP) elevation, inflammatory cell aggregation, and vascular damage or tissue edema caused by therapeutic interventions remodel the mechanical microenvironment, exerting solid stress on tumor cells. Under the influence of solid stress, tissues undergo physical deformation, leading to remodeling of cell morphology and cytoskeletal structure, and this deformation is considered an important trigger for cellular sensing of external mechanical stimuli. 53 Compressive stress in the tumor core (10–30 kPa) is converted into circumferential tension by the surrounding matrix, promoting edge invasion. A representative example is provided by cancer-associated fibroblasts (CAFs), which secrete collagen and fibronectin; their integration reinforces ECM stiffness and produces persistent tensile stress. 54 In the early stages of tumor development, compressive stress suppresses proliferation by inducing cytoplasmic relocalization of YAP, attenuating YAP/TAZ transcriptional activity, and altering vascular structure to restrict nutrient supply. 55 However, when stress is alleviated or at the tumor periphery, activation of the TRPV4–PI3K/AKT axis promotes cell cycle progression and enhances tumor cell survival. 56
Mechanistically, compressive stress facilities tumor cells metastasis and invasion by activating MAPK/ERK and PI3K/AKT signaling, upregulating migration-associated genes, inducing cytoskeletal remodeling, and stimulating protease-mediated extracellular matrix degradation. 57 This mechanosignaling extensively cross-talks with PI3K/Akt and MAPK pathways via integrin-FAK, forming a positive feedback loop to amplify oncogenic output. In addition, activation of the TGF-β/Smad pathway induces epithelial–mesenchymal transition (EMT), further promoting tumor progression.8,58-60 Mechanical stress also synergizes with Wnt/β-catenin signaling by enhancing β-catenin nuclear accumulation, synergistically driving EMT and stemness. 28 In advanced tumors, compressive stresses activate YAP/TAZ nuclear translocation via actin stress fibers, promoting proliferation. 61 As an alternative, mechanical stretching forces typically activate the YAP/TAZ pathway to overcome contact inhibition and promote cell proliferation. 62 Their roles in metastasis and invasion may involve altering cell morphology and polarity, activating Rho/ROCK pathway, and enhancing extracellular matrix-degrading enzyme secretion. 63 For example, in breast cancer, tensile loading markedly activates the TGF-β/Smad pathway, leading to pronounced shifts in EMT markers (≈60% reduction in E-cadherin and a 3-fold increase in N-cadherin/Vimentin). This directly induces EMT in epithelial cells, thereby increasing their invasive and metastatic capacity by 3–5-fold. 64 Consequently, the invasive and metastatic capabilities of tumor cells are enhanced.8,58,60,64
Targeting mechano-signaling cascades with pharmacological agents reduces tumor cell responsiveness to mechanical stress, thereby inhibiting tumor growth and migration. For example, in mesothelioma and ovarian cancer with elevated YAP expression, YAP/TAZ inhibitors block YAP–TEAD binding and enhance tumor cell sensitivity to chemotherapeutic agents. 65 Likewise, in lung cancer, the Rho/ROCK inhibitor Fasudil, administered orally, reduces tumor mechano-sensitivity, suppresses pseudopod formation, and limits pulmonary metastasis. 66 In addition, intratumoral injection of matrix metalloproteinases (MMPs) or hyaluronidase to selectively degrade collagen, hyaluronan, and other tumor ECM components can reduce TIP and improve the mechanical microenvironment. 28 In clinical trials involving patients with pancreatic cancer, investigators used PEG–hyaluronidase (PEGPH20) to reduce TIP, effectively increasing drug penetration, as evidenced by tumor perfusion rising to 1.8-fold. 67 Concurrently, a 2.3-fold increase in pO2 demonstrates that this mechano-therapeutic intervention can markedly enhance the cytotoxic efficacy of radiotherapy. 68 Moreover, reducing compressive stress within tumor tissues has been shown to enhance the efficacy of immunotherapy. In an ICB-resistant metastatic breast cancer mouse model, angiotensin receptor blockers alleviated tumor mechanical compressive stress by inhibiting CAF activation, thereby enhancing T cell–mediated antitumor activity. 69
Furthermore, the regulation of tensile/compressive forces can be achieved by applying exogenous forces such as high-intensity focused ultrasound or magnetic forces, thereby intervening tumor growth. 70 For instance, tumor cells labeled with magnetic nanoparticles (e.g., Fe3O4) and subjected to alternating magnetic fields undergo periodic stretching, which activates Piezo1 channels, induces Ca2+ overload, and triggers apoptosis.71,72 In addition, genetic approaches such as silencing mechanosensitive genes (e.g., Piezo1, YAP) or biophysical strategies like implanting slowly degradable flexible scaffolds have been shown to regulate tumor-associated stretching/compressive stress and enhance the efficacy of chemotherapy. 73 Mechanistically, periodic mechanical stretching activates mechanosensitive ion channels, leading to Ca2+ influx, NADPH oxidase activation, and elevated ROS levels, thereby amplifying oxidative stress damage induced by radiotherapy. 58 These approaches represent early-stage strategies that warrant further investigation as potential standalone or combination therapies.
However, these mechanical stress–based intervention strategies still face certain safety risks and translational barriers. For example, drugs targeting mechanotransduction pathways may cause systemic toxicities, off-target effects, and long-term adaptive risks; although tumor TIP–lowering interventions can enhance drug penetration, they may also induce tumor cell escape or disrupt immune barriers, potentially accelerating tumor progression. To enable an effective transition from experimental studies to clinical practice, it is urgent to establish a standardized mechano-dose framework that defines action thresholds, frequencies, and exposure windows for different forms of mechanical stress, thereby optimizing the balance between safety and therapeutic effect. Concurrently, building clinically actionable biomechanical platforms capable of precisely controlling stimulation parameters will facilitate translation toward prospective clinical trials.
3. Effects of Exogenous Mechanical Stimulation on Tumors and Therapeutic Applications
3.1. Magnetic Fields
Magnetic fields can be broadly categorized according to their physical properties into static magnetic fields (SMFs) and electromagnetic fields (EMFs), such as alternating, pulsed, and rotating forms. 74 The biological impact of these fields on tumor cells varies significantly depending on parameters such as intensity, frequency, and exposure duration. 75 Early studies suggested that chronic exposure to high-frequency magnetic fields increased the risk of neurological disorders and cancer, while also promoting drug resistance and impairing therapeutic efficacy.76,77 However, with the recognition of their therapeutic potential in oncology, recent research has shifted toward elucidating the mechanisms by which different magnetic field types affect tumor biology and toward defining practice strategy for clinical application.75-78
Studies have demonstrated that magnetic fields with specific frequencies and intensities, such as extremely low-frequency oscillating fields, can induce hyperpolarization of tumor cell membranes by modulating transmembrane ion fluxes (Ca2+, K+, Na+).
79
These changes disrupt tumor cell metabolism and angiogenesis, thereby suppressing proliferation.
80
In addition, theoretical and computational studies have proposed that magnetic fields might perturb DNA hydrogen bonding and proton transfer dynamics within base pairs, potentially affecting genomic stability; however, this remains a hypothesis requiring further experimental validation.
81
By contrast, more consistently reported indirect mechanisms include alterations in membrane permeability or activation of calcium channels, leading to Ca2+ influx, stimulation of endonuclease activity, and apoptosis (Figure 4).
82
Furthermore, high-intensity magnetic fields strand breaks, and promote mitochondrial apoptosis by upregulating p53 and Bax, downregulating Bcl-2, and activating Caspase-3/9 signaling.83,84 The mechanism of exogenous mechanical stimulation on tumors: (a) Ultrasound - mediated sonoporation induces cell membrane injury; (b) magnetic field - induced hyperthermia causes DNA damage, ultimately triggering apoptosis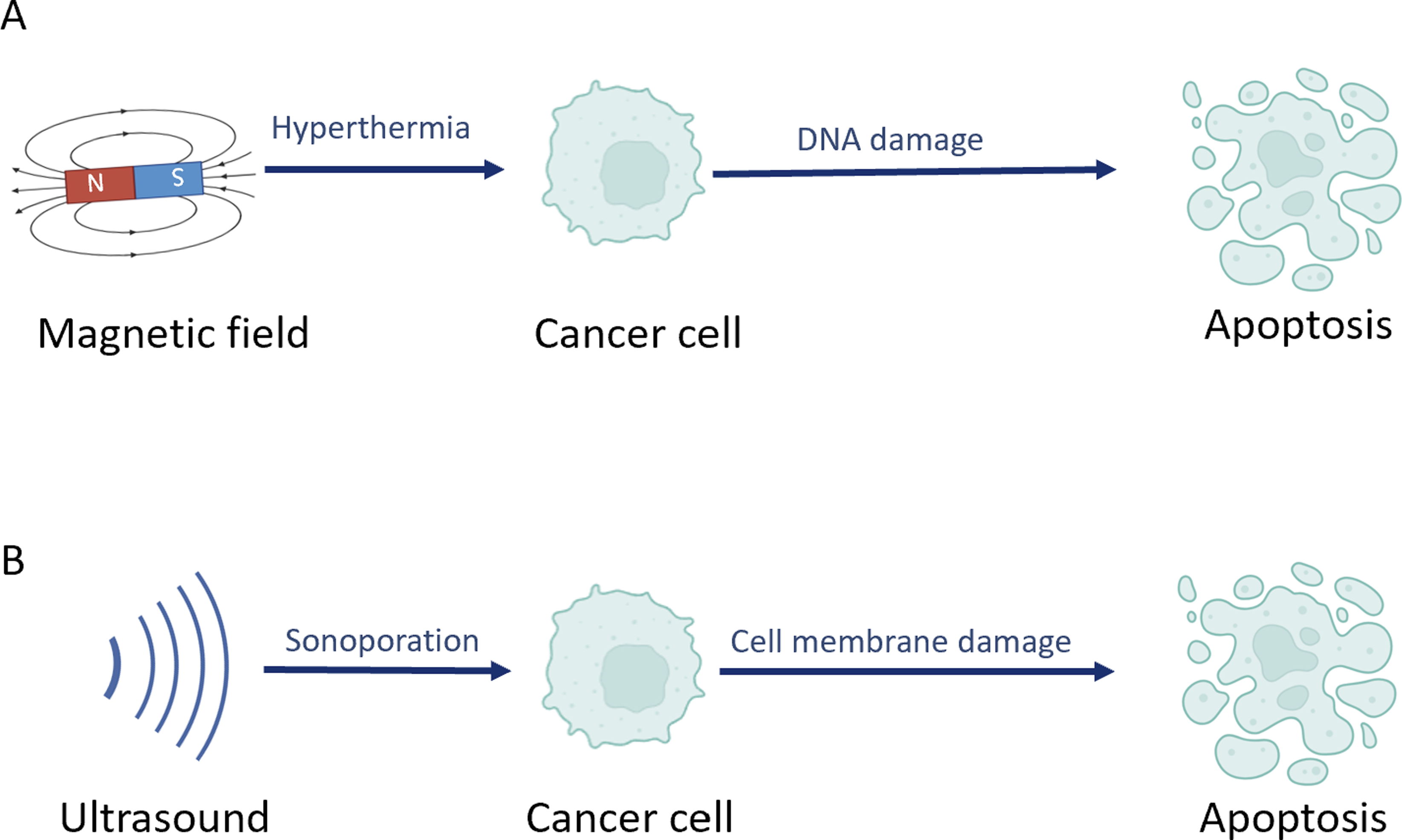
In addition to directly regulating tumor cell behavior, magnetic fields can suppress angiogenesis and modulate immune responses, thereby exerting antitumor effects. High static magnetic fields (>500 mT) reduce vascular density and erythrocyte flow velocity, induce endothelial cell swelling and vascular occlusion, limit oxygen and nutrient delivery, and inhibit glycolysis-driven ATP production, ultimately restricting tumor proliferation. 85 Pulsed magnetic fields further disrupt tumor metabolism by inducing mitochondrial edema, impairing energy production, and damaging rough endoplasmic reticulum structure, thereby interfering with protein synthesis. 86
From an immunological perspective, alternating and high-gradient magnetic fields have been shown to activate macrophages and T cells, while also enhancing the activity of T cells, NK cells, and macrophages, leading to increased inflammatory infiltration and oncolytic activity. 87 Previous research indicates that magnetic field therapy demonstrates antitumor efficacy in vitro and in animal models across multiple cancer types (including breast, liver, and brain cancers), with potential synergistic interactions when combined with conventional therapies such as chemotherapy, radiotherapy, or ultrasound.88,89 Preclinical studies have applied magnetically guided microneedle arrays to deliver controllable mechanical stimulation to tumor sites in breast cancer patients and, when combined with a single external 6 Gy fraction of radiotherapy, markedly enhanced tumor suppression (tumor volume inhibition increased from 62% with radiotherapy alone to 84%), further validating the synergistic potential between magnetic mechanoregulation and radiotherapy.90-92 Collectively, these findings underscore the therapeutic potential and clinical feasibility of integrating magnetic-based combinatorial approaches in future cancer treatments.93,94
Despite the notable efficacy of magnetic field therapy—particularly alternating magnetic fields—across multiple tumor models, translational barriers persist. There is a lack of standardization regarding the safety limits, frequencies, and optimal treatment windows across various magnetic field types; moreover, the heterogeneous intratumoral distribution of magnetoresponsive nanoparticles undermines the precision of local thermal dosing and drug delivery. 95 These limitations are especially pronounced in stroma-dense pancreatic and breast cancers, where energy transmission and magnetic responsiveness are markedly constrained. 96 In addition, the synergistic mechanisms between magnetic fields and radiochemotherapy, immunotherapy, and mechanobiological therapies remain insufficiently validated in a systematic manner. 97 Future efforts should prioritize establishing unified dosimetric standards, developing highly selective magneto-responsive materials and controllable magnetic therapy platforms, and delineating the molecular bases and optimal sequencing of multimodal synergies, thereby advancing standardization and clinical translation of magnetic field therapy.
3.2. Ultrasound
Ultrasound, a mechanical wave with frequencies above 20 kHz, exerts unique therapeutic effects in oncology through cavitation and mechanical mechanisms. Through cavitation, ultrasound generates microbubbles that expand and collapse in liquid environments, producing mechanical forces that damage tumor cell membranes and cause cytoplasmic leakage (Figure 4), while simultaneously enhancing drug and gene delivery efficiency. 80 The mechanical effects of ultrasound further destabilize the cytoskeleton, disrupt cellular morphology, and inhibit tumor cell proliferation and migration.
Building on these mechanisms, a range of ultrasound-based therapeutic strategies has been developed. For example, sonodynamic therapy (SDT) employs ultrasound to activate sonosensitizers within tumor and endothelial cells, generating singlet oxygen that damages tumor structures, inhibits blood supply, and stimulates immune responses. 98 Ultrasound exerts direct mechanical modulation to enhance chemotherapy efficacy: its cavitation effect mechanically disrupts the cytoskeleton and increases membrane permeability, promoting liposomal drug release as a consequence of mechanical cell membrane perturbation. Anti-angiogenic ultrasound uses low-frequency mechanical waves to remodel tumor vasculature, which not only restricts nutrient supply but also normalizes the mechanical microenvironment (reduced interstitial pressure) to synergize with drug action. 99 Innovative immunotherapy strategies include engineering contrast agent shells functionalized with anti-PD-L1 peptides and IL-15 mRNA, which, upon ultrasound exposure, induce immunogenic tumor cell death and activate NK cells and cytotoxic T cells, ultimately eliciting systemic antitumor immunity. 100 Clinically, a phase II trial in non-small cell lung cancer demonstrated that ultrasound elastography–guided low-frequency mechanical vibration combined with stereotactic body radiotherapy (SBRT) significantly improved local tumor control and reduced the incidence of radiation pneumonitis compared with SBRT alone. 101 A phase II clinical trial in patients with non–small cell lung cancer evaluated ultrasound-elastography–guided low-frequency mechanical vibration combined with stereotactic body radiotherapy (SBRT). The results showed that the combination arm achieved a significantly higher local tumor control rate (up to 92%) than SBRT alone, while the incidence of radiation pneumonitis was lower, with late grade ≥ 3 toxicity decreasing to approximately 19%. 102
Taken together, ultrasound exhibits multifaceted strengths, including cavitation in cancer therapy, mechanical effects, and has yielded encouraging results in multiple studies combining it with chemo-radiotherapy. However, broader clinical adoption remains limited by insufficient standardization of acoustic parameters and modes of action, variability in energy focusing and tissue penetration, and a lack of systematic validation of long-term safety. 103 Future research should prioritize addressing the above challenges to advance the clinical translation of ultrasound in cancer therapy.
4. Summary and Prospects
Comparative Summary Analysis of Distinct Mechanical Factors in Cancer Therapy
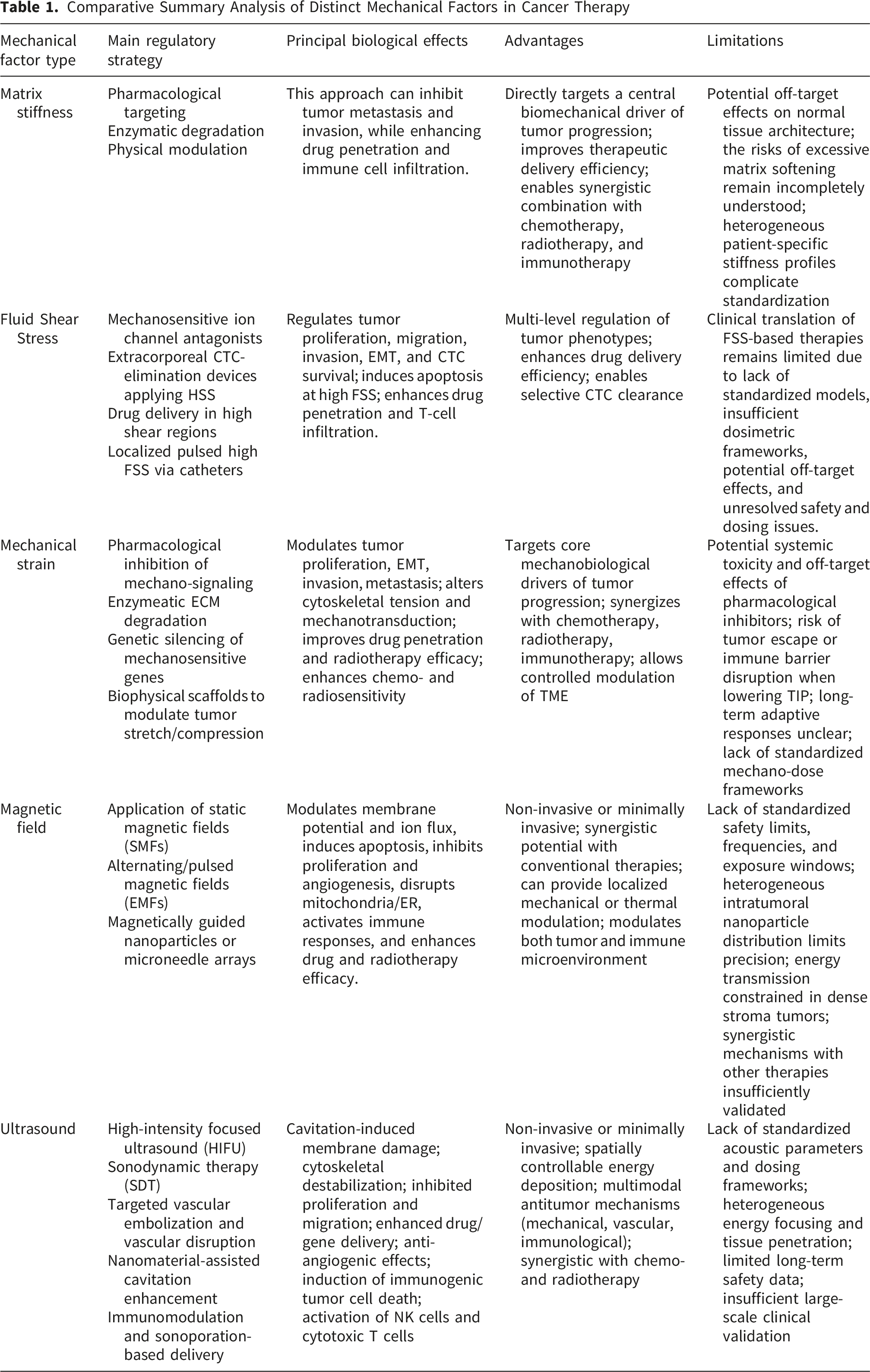
With the progressive maturation of New Approach Methodologies (NAMs)—such as organ-on-chip platforms, artificial intelligence (AI) models, and organoid technologies—these approaches, owing to their superior human biological relevance, reduced cost, and fewer ethical concerns, are gradually supplanting traditional animal experiments, which have long suffered from interspecies differences and high failure rates.94,102,106,107 Recently, the U.S. Food and Drug Administration (FDA) announced the gradual removal of mandatory animal testing requirements for drug development, allowing NAMs such as AI-based modeling and organoids to serve as substitutes. 108 This landmark shift underscores the inevitable trend toward a more precise and efficient paradigm in new medical products/strategy discovery and development. Mainstream platforms integrate multidisciplinary approaches—including microfluidic technology, 3D biomaterials, and artificial intelligence—to dynamically reproduce endogenous mechanical cues (e.g., matrix stiffness, FSS) and exogenous interventions (e.g., ultrasound, magnetic fields), thereby providing robust systems for mechanistic exploration and drug screening. Microfluidic chip models regulate matrix stiffness and FSS through multi-channel systems.109,110 For example, tumor-vessel chips can simulate the behavior of circulating tumor cells under different shear stresses, while multi-organ chips help evaluate the effects of exogenous mechanical interventions. 110 Hydrogel-based 3D cultures recreate tumor matrices of varying stiffness, as demonstrated in pancreatic cancer organoids that confirm stiffness-dependent chemosensitivity and spheroid models that visualize strain-induced invasion. 111 Mechano-biological coupling models integrated with biosensors quantitatively evaluate cellular responses to magnetic fields, ultrasound, and other mechanical stimuli, offering critical data for treatment optimization. 112 Importantly, AI-assisted digital models combine mechanical and biological datasets from preclinical systems with machine learning algorithms to predict tumor responses, such as stiffness–drug resistance correlations in 3D cultures, thereby informing personalized therapeutic strategies. 113 This deep integration with artificial intelligence not only improves the predictive accuracy of the models but also promotes the transition of mechanical regulation strategies from empirical exploration to quantitative precision design. For example, Wang et al developed a graph convolutional neural network that simultaneously processed atomic force microscopy (AFM)-derived stiffness maps, single-cell RNA sequencing (scRNA-seq) data, and dynamic contrast-enhanced MRI features from breast cancer organoids. 114 This integration demonstrates how AI-driven analysis links tumor mechanics with clinical translation. By correlating mechanical signatures (AFM stiffness) with molecular programs(scRNA-seq) and imaging features (MRI), such models enable non-invasive prediction of tumor mechanical phenotypes and therapeutic responses, thereby guiding personalized mechanotherapy. Collectively, these advanced models overcome the limitations of traditional 2D systems, faithfully reproduce the mechano-biological coupling of the TME, and provide indispensable platforms for elucidating mechanistic regulation and identifying effective therapeutic strategies.
Summary of Basic, Preclinical, and Clinical Progress of Mechanical-Based Cancer Therapies

In this context, establishing standardized, multimodal evaluation frameworks that integrate mechanical, functional, and molecular readouts will be indispensable for advancing mechanotherapeutic applications. 119 Consistent with recent FDA guidance, a further priority is the development of in vitro models that accurately recapitulate the mechanical microenvironment, enabling parallel drug screening and tailored mechanotherapeutic optimization. Equally important is the translation of optimized in vitro mechanobiological conditions to individual patients. Advances in computational oncology have facilitated the development of patient-specific digital twins that integrate multimodal imaging with biomechanical modeling. 120 For example, Liu et al incorporated preoperative MR elastography into a finite element model of glioblastoma to optimize ultrasound-mediated drug delivery parameters, subsequently applied in a compassionate-use setting with improved drug penetration and tumor regression. Collectively, such frameworks provide a rational basis for personalized mechanical interventions and strengthen the clinical translatability of in vitro findings. 121
Concludingly, future research directions may include the following aspects: 1) leveraging spatial transcriptomics and single-cell sequencing to map the molecular networks underlying mechanical regulation and establishing a “mechanogenomics” database; 2) integrating artificial intelligence to predict patient-specific responses to mechanical interventions and tailor personalized treatment plans; 3) investigating the synergistic mechanisms between mechanical modulation and immunotherapy or targeted therapy to optimize combination strategies (e.g., mechanical preconditioning to enhance immune cell infiltration); 4) developing standardized evaluation frameworks and high-precision integrated devices (e.g., synchronized mechanical-radiation systems).119-123 By integrating advances in engineering, biology, and medicine, therapeutic strategies based on mechanical regulation are progressively transitioning from empirical interventions toward more quantitative and precision-oriented approaches. While preclinical data are promising, further large-scale clinical trials are required to substantiate their efficacy and safety in diverse patient populations.
5. Conclusion
In summary, this review synthesizes current advances in understanding how the tumor mechanical microenvironment regulates cancer progression and therapeutic response. Endogenous biomechanical cues, including matrix stiffness, FSS (FSS), and mechanical strain, actively promote malignant progression through coordinated mechanotransductive mechanisms encompassing integrin–FAK signaling cascades, cytoskeletal remodeling, mechanosensitive ion channel regulation, and YAP/TAZ pathway activation. Beyond mechanistic insights, emerging exogenous interventions, including ultrasound- and magnetic field-based strategies, demonstrate potential to reprogram biomechanical states and enhance therapeutic efficacy. By integrating endogenous cues with exogenous modulation and providing comparative analysis of different modalities, this work positions mechanical regulation as a clinically actionable axis for precision oncology. Future translation requires standardized mechano-dosimetry and patient stratification based on tumor-specific mechanical signatures.
Footnotes
Acknowledgements
We gratefully acknowledge Dr. Lei Yang (Hebei Key Laboratory of Biomaterials and Smart Theranostics, School of Health Sciences and Biomedical Engineering, Hebei University of Technology, Tianjin, China) for his valuable guidance, academic mentorship, and continuous support. We also thank the research platform and resources provided by his group, which facilitated the completion of this review.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (U23A6008), Jiangsu Provincial Medical Key Discipline Construction Unit (Oncology Therapeutics (Radiotherapy)) (No. JSDW202237), and Heye Health Technology Chong Ming Project (HYCMP-2026002).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have read and approved the submitted manuscript. There are no conflicts of interest. The manuscript has not been submitted elsewhere nor published elsewhere in whole or in part. All relevant ethical safeguards had been met.
Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this work, the author used ChatGPT in order to improve language readability. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
