Abstract
Objective
This study aimed to assess renal cell carcinoma (RCC) complexity using the R.E.N.A.L. nephrometry score and evaluate the feasibility and safety of the non-clamping sutureless technique combined with a radiofrequency ultrasound scalpel (RFUS) for low-complexity RCC.
Methods
This retrospective, multi-center cohort study categorized patients into three groups: Group I (suture laparoscopic partial nephrectomy [LPN] with clamping renal artery), Group II (sutureless LPN without renal artery clamping, combined with monopolar electrocoagulation), and Group Ⅲ (sutureless laparoscopic nephron-sparing surgery [LNSS] without renal artery clamping combined with RFUS). Key outcomes included operative time, intraoperative blood loss, postoperative hospital stays, serum creatinine (Scr) levels, and the estimated glomerular filtration rate (eGFR) of the affected kidney. Post hoc power analysis was used to evaluate eGFR variation across groups, and a multiple linear regression model was employed to analyze factors influencing postoperative eGFR alterations.
Results
The study included 60 patients with exophytic and low-complexity RCC. Blood loss in Group III was comparable to that in Group I but less than that in Group II (P = 0.035). Operative time and postoperative hospital stays were significantly shorter in Group III than in the other groups (P < 0.01). A statistically significant increase in Scr levels was observed during the early postoperative period, continuing through the 6-month follow-up assessment compared with preoperative levels (P < 0.01). The 6-month postoperative eGFR of the affected kidney in Group III was significantly higher than that in the other groups (P < 0.01). The decrease in preoperative and 6-month postoperative eGFR values was greatest in Group I (P < 0.01), followed closely by Group II (P < 0.01), while no significant reduction was observed in Group III. The post hoc power analysis revealed high detection power (0.912 or 1.0). Warm ischemia time emerged as the sole significant eGFR predictor (R2 = 0.814, P < 0.05).
Conclusion
The non-clamping sutureless technique combined with RFUS for RCC effectively alleviates renal ischemia–reperfusion injury, offering superior renal function protection while achieving comparable oncological outcomes.
Keywords
Introduction
Renal cell carcinoma (RCC) is a malignant tumor that originates from the renal tubular epithelial system of the renal parenchyma. 1 Male patients have an RCC incidence rate 1.8 times higher than that of their female counterparts. 2 The morbidity rate in urban regions is 4.3-fold greater than that in rural ones, and people aged 50–70 years exhibit a higher morbidity rate.2,3 RCC accounts for approximately 3% of all cancer cases and predominantly occurs in developed countries. 4 The R.E.N.A.L. nephrometry score (RNS), proposed by Kutikov and Uzzo, is based on five key anatomical attributes of renal malignancies. 5 They argued that the anatomical structures of renal masses are quantified using the RNS, a standardized and reproducible scoring system. Previous studies have demonstrated that the RNS places considerable value on surgical planning and is a key determinant influencing perioperative outcomes.6,7
Surgery remains the most prominent treatment for RCC. 8 Partial nephrectomy (PN) results in improved renal function and higher disease-free survival rates. 9 Laparoscopic partial nephrectomy (LPN) and laparoscopic nephron-sparing surgery (LNSS) are currently popular clinical techniques. LNSS has significantly advanced, characterized by decreased blood loss, reduced trauma to patients, quicker postoperative convalescence, and nephron preservation.10,11 Clamping the renal vessel may impede blood supply, facilitating more accurate tumor excision with a cold knife. However, this technique may exacerbate renal ischemia-reperfusion injury and increase the risk of kidney damage.12,13 Therefore, shortening renal ischemia time (IT) within a limited duration has become a primary goal of modified LNSS. 12 LNSS without renal artery clamping still poses a challenge for tumor removal, hemostasis, and suturing. 14 For low-complexity RCC, sutureless PN has potential advantages in preserving renal function while yielding comparable perioperative results, rendering it a superior option for treating RCC. 14
This study utilized the RNS to evaluate RCC complexity. Ultimately, it aimed to assess the impact of a non-clamping sutureless technique on the perioperative phase and prognosis of patients with low-complexity RCC, rather than the non-clamping technique.
Materials and Methods
Clinical Data
The reporting of this retrospective, multi-center cohort study adheres to the “Strengthening the Reporting of Observational Studies in Epidemiology” guidelines. 15 The study consecutively enrolled 60 patients with T1-stage RCC from the urology departments of four participating hospitals —the First Affiliated Hospital of Guizhou University of Traditional Chinese Medicine, the Affiliated Hospital of Guizhou Medical University, Liupanshui People's Hospital of Guizhou Province, and Zhijin County People's Hospital—retrospectively from July 2021 to December 2023. Professor Tang performed all surgical procedures. Clinical staging was conducted in accordance with the Union for International Cancer Control Tumor–Node–Metastasis classification system, incorporating relevant diagnostic and radiological imaging findings. Participants were chronologically assigned to one of three surgical intervention groups: Group I (suture LPN with renal artery clamping, n = 20), Group II (sutureless LPN without clamping combined with monopolar electrocoagulation, n = 20), and Group III (sutureless LNSS without clamping combined with a radiofrequency ultrasonic scalpel [RFUS], n = 20). The inclusion criteria were as follows: confirmed RCC, age 30–82 years, an RNS of 4–6 points, the presence of a unilateral renal tumor, and assignment to one of the three surgical groups. The following exclusion criteria were applied: preoperative renal dysfunction, bilateral renal malignancies, severe primary diseases, mental illness, complicated cardiovascular and cerebrovascular diseases, liver, kidney, or hematopoietic systems. Patients or their family members who declined to provide written informed consent before participating in the trial were excluded. All patient data were de-identified to ensure anonymity and protect privacy. This study was conducted in accordance with the principles of the Helsinki Declaration of 1975, as revised in 2024. 16 The protocol was approved by the Ethics Committee of the Affiliated Hospital of Guizhou Medical University (Approval No. 2023-375) on April 26, 2023. Written informed consent was obtained from all individual participants included in the study.
Medical Indices and RNS Assessment
Enhanced computed tomography (CT) analysis of the urinary system was performed on 60 patients to determine renal parenchymal thickness, renal vessel location, and tumor size and location. Each patient presented normal results for serum creatinine (Scr), estimated glomerular filtration rate (eGFR) of the affected kidney and renal dynamic imaging, blood, urine, and stool routine, liver and kidney function, electrolytes, coagulation tests, and electrocardiogram. By reviewing CT scans, we evaluated RCC complexity using the RNS. All data were independently assessed by two urologists. In cases of uncertainty, a third doctor was consulted for further evaluation. According to Kutikov and Uzzo, 5 the RNS encompasses the radius (maximal diameter in centimeters), exophytic/endophytic properties, proximity of the tumor to the collecting system or sinus (in millimeters), anterior/posterior orientation, and location relative to the polar lines. RNS-based complexity is classified as low (4-6 points), moderate (7-9 points), and high (10-12 points). The primary endpoint was the change in renal function of the affected kidney over a 6-month follow-up period. The following key outcomes including operative time, blood loss, postoperative hospital stay, Scr, eGFR, drainage time, ambulation time, and gastrointestinal recovery time marked the secondary endpoints.
Operation
Establishing a Surgical Channel
All procedures were initiated with the patient positioned in the standard lateral decubitus position. The operative table was flexed to elevate the waist brige. Following the induction of general anesthesia, a 1–2 cm vertical incision was made inferior to the 12th rib along the posterior axillary line. The muscle layers and thoracolumbar fascia were bluntly dissected using a long curved hemostat to access the retroperitoneal space. 17 A self-made balloon dilator was inserted and insufflated with 500–700 mL of air to create the working space. Subsequently, three laparoscopic ports were placed under direct visualization: a 12-mm port at the initial incision site, a 5-mm port inferior to the costal margin along the anterior axillary line, and a 10-mm port superior to the iliac crest along the mid-axillary line. A CO2 pneumoperitoneum was established and maintained at a pressure of 12–15 mm Hg. The entire procedure was conducted using a 30° laparoscope.
Group Ⅰ: Suture LPN with Renal Artery Clamping
After gaining access, the renal artery and its branches were meticulously dissected and clamped using a laparoscopic bulldog clamp. The renal tumor was resected with a minimum 0.5-cm margin of healthy parenchyma using endoscopic scissors. Renorrhaphy and reconstruction of the collecting system were performed using a 3–0 Vicryl suture in a double-layer, continuous manner to achieve hemostasis and closure of the renal defect (Figure 1a). 18

Surgical procedures conducted on patients across three groups. a: sutureLPN with renal artery clamping; b: sutureless LPN without renal artery clamping combined with monopolar electrocoagulation; c: sutureless LNSS without renal artery clamping combined with an RFUS. LPN: laparoscopic partial nephrectomy; LNSS: laparoscopic nephron-sparing surgery.
Group Ⅱ: Sutureless LPN Without Renal Artery Clamping Combined with Monopolar Electrocoagulation
Gerota's fascia was incised utilizing a combination of blunt and sharp dissection. Perirenal fat was carefully dissected along the renal capsule to completely expose the tumor, without routinely dissecting the renal hilum. The tumor, along with a 0.5-cm margin of normal tissue, was excised using both blunt dissection with suction-irrigation and cold endoscopic scissors. Hemostasis was achieved by systematically applying monopolar electrocoagulation (set at 30 W in pure-cut mode) to all bleeding points and the entire resection bed until a uniform eschar was established (Figure 1b). 19
Group Ⅲ: Sutureless LNSS Without Renal Artery Clamping Combined with RFUS
Surgical exposure in this group was identical to that in Group II. The RFUS device was employed to resect the tumor with a 0.5-cm parenchymal margin. Dynamic hemostatic control was maintained by immediately sealing active bleeding points using the targeted bipolar coagulation mode of the scalpel, followed by systematic fulguration of the entire tumor bed to ensure hemostatic competence (Figure 1c).
Conclusion of Operation and Specimen Handling
Following tumor excision, vascular clamps (in Group I) were released, and the operative field was inspected for at least 5 min under reduced pneumoperitoneum pressure (8 mm Hg) to confirm secure hemostasis. All resected specimens were retrieved in an endoscopic bag and sent for intraoperative frozen-section and final pathological analyses. An 18-French closed-suction drain was placed adjacent to the resection site. The pneumoperitoneum was subsequently released, ports were removed under direct visualization, and fascial defects at port sites > 10 mm were closed with absorbable sutures.
Postoperative Management and Definitions
On postoperative day 1, patients were assisted in standing at the bedside but were not permitted to ambulate. Perirenal drain output was quantified every 24 h. The drain was removed using a two-step protocol: if the 24-h output was < 10 mL, the drain was retracted by 2 cm; if the output remained below 10 mL over the subsequent 24 h, the drain was removed entirely. The drain site was left unsutured to facilitate healing by secondary intention. Patients were advised to delay full ambulation until the drain site was completely epithelialized, typically within 1–3 days.
Ambulation time was defined as the postoperative period (in hours) during which the patient was first able to stand and engage in guided physical activity beyond the bedside, as assessed and approved by the medical team.
Observational Indicators and Statistical Processing
Patient demographics and perioperative data were analyzed using descriptive statistical methods. The following key outcome measures were evaluated: operative time, blood loss, postoperative hospital stay, Scr, eGFR, drainage time, ambulation time, gastrointestinal recovery time, and renal function parameters. Data distribution normality was assessed using the Shapiro–Wilk test. One-way analysis of variance (ANOVA) was utilized to compare normally distributed continuous variables across multiple groups with homogeneity of variance, while Welch ANOVA was employed when the variance was heterogeneous. To analyze differential variables within each group, the paired t-test and Wilcoxon signed-rank test were applied to normally and non-normally distributed data, respectively. Normally distributed continuous variables are presented as the mean ± standard deviation. Categorical variables were analyzed using the Chi-square test.
Post hoc power calculations were conducted to confirm adequate statistical power for detecting clinically significant differences, utilizing the observed eGFR variations from preoperative to 6 months postoperative. A multiple linear regression model was constructed to evaluate potential predictors of eGFR changes following renal surgery, incorporating the following covariates: patient age, preoperative eGFR, maximum tumor diameter, intraoperative blood loss (IBL), warm ischemia time (WIT), RNS, and operative time. Statistical significance was set at P < 0.05 for all analyses, which were performed using SPSS (version 25.0).
Results
The primary endpoint was the change in renal function of the affected kidney over a 6-month follow-up period. The incidence of serious adverse events during the postoperative phase and peri-procedural events marked the secondary endpoints. In total, 60 patients with exophytic and low-complexity RCC, including 32 men and 28 women, were enrolled in this retrospective cohort study. In total, 30 cases of right tumors and 30 of left tumors were noted. Among the patients with pathological data, 41 presented with T1a stage tumors, while the remaining patients had T1b stage tumors. Pathological examination revealed that the tumor, the external capsule, and a surrounding 0.5–1.0 cm of healthy parenchyma were successfully excised. The surgical margin status was negative (R0 resection) in all 60 cases across the three groups.
Group Ⅲ experienced shorter operative times and postoperative hospital stays than the other two groups (P < 0.01, Figure 2a-b). Blood loss in Group Ⅲ was less than that in Group Ⅱ (P = 0.035, Figure 2c); however, it was comparable to that in Group Ⅰ. In 40 cases, renal ischemia-reperfusion damage was alleviated as the renal artery was not clamped during surgery. None of the cases reported secondary infections, hemorrhage, or organ damage. The drainage tube was removed 1–6 days postoperatively. Gastrointestinal recovery occurred within 8–48 h, and ambulation was attained within 3–7 days. No significant differences were noted among these variables (Table 1).

Comparison of four variables among the three groups. a: operative time; b: blood loss; c: postoperative hospital stay; d: the 6-month postoperative eGFR of the affected kidney; e: decrease in the eGFR of the affected kidney before and after surgery among the three groups; f: eGFR changes in the affected kidney between the T1a and T1b stages. eGFR: estimated glomerular filtration rate.
The Comparison of Variables in Three Surgical Methods.

RNS: R.E.N.A.L. nephrometry score; Scr: Serum creatinine; eGFR: Estimated glomerular filtration rate; SD: standard deviation.
Statistical analysis yielded no significant differences in the preoperative eGFR or Scr (preoperative, postoperative, and 6-month postoperative) across the three groups (Table 2). However, the postoperative and 6-month postoperative Scr levels in each group were significantly higher than the preoperative levels (P < 0.01). The 6-month postoperative eGFR of the affected kidney in Group Ⅲ exceeded that in the other two groups (P < 0.01, Figure 2d). The decline in the eGFR from preoperative to 6-month postoperative assessments was most pronounced in Group Ⅰ (P < 0.01), followed closely by that in Group Ⅱ (P < 0.01). Conversely, this decline was not significant in Group Ⅲ (Figure 2e). T1a and T1b stages exhibited a similar trend (Figure 2f).
The Decrease of eGFR of the Affected Kidney among Three Groups, T1a and T1b Stages.
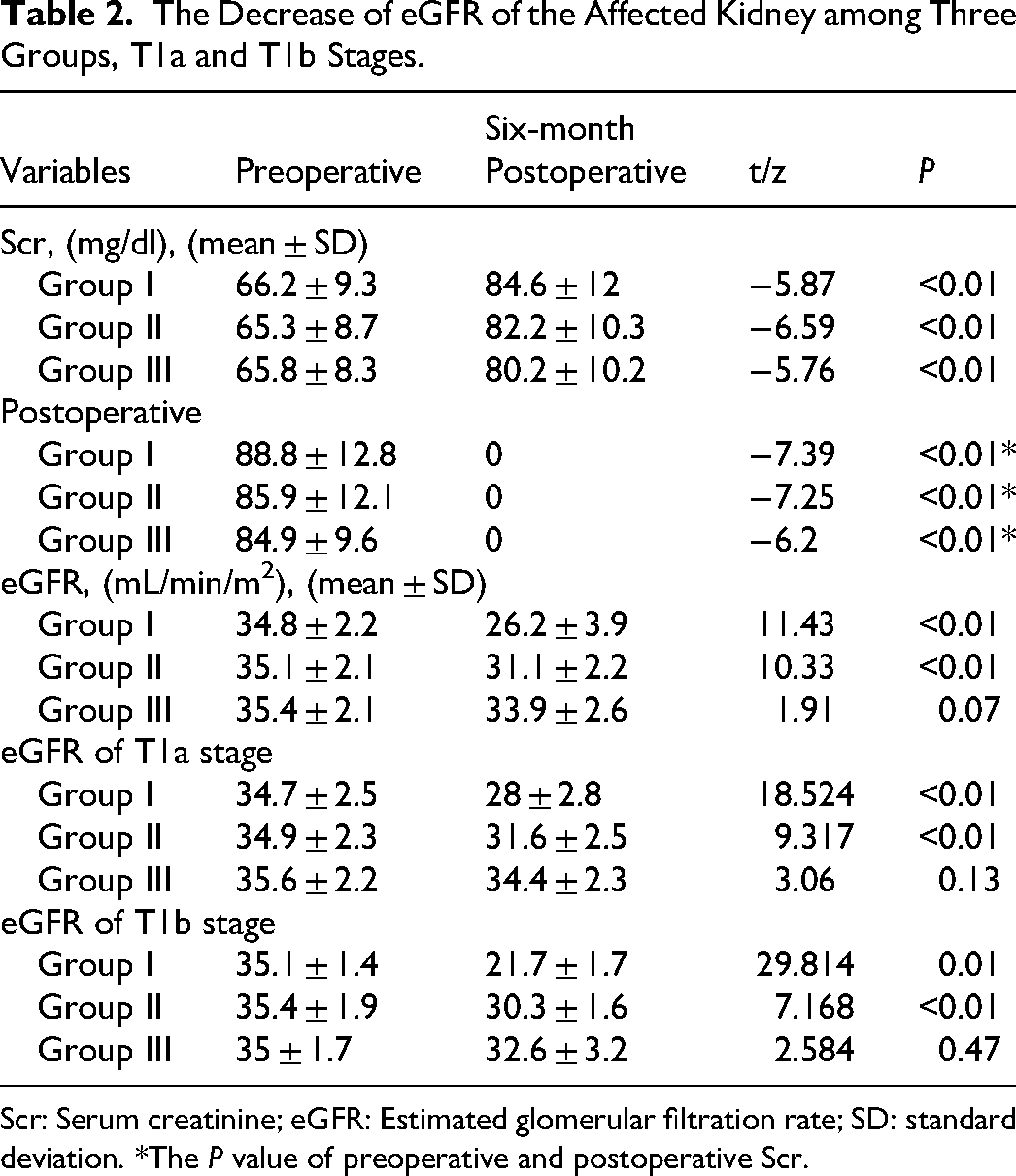
Scr: Serum creatinine; eGFR: Estimated glomerular filtration rate; SD: standard deviation. *The P value of preoperative and postoperative Scr.
The post hoc power analysis indicated a statistical power of 0.912 for comparisons between Groups Ⅲ and Ⅱ and 1.0 for comparisons between Groups Ⅲ and Ⅰ (both P < 0.05). The multiple linear regression model displayed a good fit (R2 = 0.814), indicating that 81.4% of the variance in eGFR changes from preoperative baseline to 6-month follow-up could be collectively explained by the clinical parameters assessed. Multivariate analyses revealed that WIT was a significant predictive factor for the percentage change in the postoperative eGFR at 6 months (B = 0.305, P < 0.05). In contrast, conventional predictors—namely, patient age, preoperative eGFR, maximum tumor diameter, IBL, RNS, operative time, and ischemia type—did not demonstrate statistically significant associations (B = −0.45, P = 0.107; B = 0.125, P = 0.258; B = 0.417, P = 0.06; B = 0.01, P = 0.331; B = 0.121, P = 0.774; B = 0.047, P = 0.79; and B = 1.017, P = 0.57, respectively; Table 3).
A Multivariate Linear Regression Model Concerning Identifing Independent Predictors of eGFR Between Preoperative eGFR and six-Month Follow-up.

B unstandardized coefficient, CI confidence interval, eGFR Estimated glomerular filtration rate, RNS R.E.N.A.L nephrometry score.
Discussion
The consistent documentation of renal tumor size, location, and depth is critical for surgical planning, as these characteristics strongly correlate with prognostic outcomes. 20 RNS, since its inception in 2009, is an objective tool for quantifying renal tumor morphology, enabling the preoperative prediction of surgical complexity and early postoperative functional outcomes. 21 Partial nephrectomy (PN) achieves oncological outcomes comparable to radical nephrectomy (RN) while conferring superior renal functional preservation—an essential advantage linked to reduced cardiovascular morbidity and all-cause mortality. 22 Nephron-sparing surgery (NSS) has emerged as the gold standard for T1a and select T1b RCC. 23 Three pivotal determinants govern postoperative renal function in PN: preoperative nephron mass, intraoperative ischemic injury, and functional residual nephron volume. 24 The relationship between WIT and renal functional impairment represents a critical focus in the refinement of PN. Current evidence strongly recommends maintaining a WIT value below the 25-min threshold, as renal function demonstrates a progressive decline with each additional minute of ischemia beyond this point. 25 In the study known as “every minute counts” by Thomson et al yielded an inverse relationship between WIT and functional recovery, suggesting that even minor reductions in ischemic time may yield substantial benefits for renal preservation. 26 Consequently, several surgical techniques have been developed to minimize renal ischemia, including arterial selective clamping, early unclamping, and, ultimately, zero-ischemia or off-clamp procedures. While clamping facilitates tumor excision, it also carries significant pathophysiological consequences. 27 IT is strongly associated with acute kidney injury (AKI) and short-term functional decline, with this relationship following a non-linear pattern that is further influenced by baseline renal function. These insights have advanced the refinement of surgical strategies, advocating for clampless NSS techniques to mitigate short-term renal dysfunction and prevent AKI. 28 Furthermore, the clamping technique itself adds procedural complexity and potentially elevates the risk of postoperative complications. While the present study employs a clamping strategy for comparison with sutureless clampless techniques, the paradigm in NSS continues to evolve towards eliminating global renal ischemia. This aligns with growing evidence supporting purely off-clamp approaches that aim to entirely avoid vascular occlusion and its potential impact on renal function. 29 This strategy is further supported by recent studies demonstrating that off-clamp PN may be safely and effectively performed, even in cases presenting high anatomical complexity, as evidenced by a propensity score-matched analysis reporting comparable perioperative outcomes and superior functional preservation in tumors with a high RNS. 30 Notably, a study focused on purely off-clamp robotic NSS for totally endophytic tumors demonstrated excellent mid-term oncological and functional outcomes, underscoring the potential of zero-ischemia approaches to expand the limits of NSS. 31 Our sutureless technique contributes to this paradigm by demonstrating effective hemostasis without renal artery clamping, albeit using RFUS energy-based sealing modalities rather than a purely off-clamp dissection technique.
The conventional PN technique necessitates a two-layer closure involving vascular and collecting system repair, followed by parenchymal suturing. 32 This approach carries inherent risks, including vascular injury, parenchymal volume loss, prolonged WIT, and subsequent renal functional impairment. 33 In contrast, contemporary sutureless techniques significantly enhance surgical safety by improving operational efficiency—eliminating layered suturing reduces WIT by 30%–40%—while better preserving renal parenchymal volume.34–36 Although the existing evidence base for clampless techniques is predominantly derived from robot-assisted and LPN surgery, rendering NSS data scarce and more challenging to interpret.37–40 A consensus exists that clampless procedures potentially pose a risk of increased intraoperative hemorrhage. Our findings demonstrate that clampless and sutureless NSS techniques may achieve an average IBL comparable to that reported in major series.13,41 Our sutureless technique yielded superior perioperative outcomes—including a shorter operative time, a shorter WIT, and superior renal functional preservation—compared to conventional approaches. To alleviate the risk of rebleeding related to active bleeding during nephrectomy, unipolar electrocoagulation hemostasis is utilized in interventional therapy. The tumor bed is not sutured after tumor resection, thereby minimizing the number of surgical steps required and mitigating the difficulty of the surgery. Nevertheless, this technique was ultimately abandoned owing to its association with increased bleeding and potential damage to the remaining renal unit. The study by Carbonara et al shows that purely off-clamp and sutureless robot-assisted partial nephrectomy (RAPN) carried out through a transperitoneal approach does not affect oncological and renal functional outcomes in selected tumor cases. 42
Current analyses of renal ischemia in PN often overlook the critical influence of parenchymal resection and reconstruction techniques on functional outcomes. Important factors in postoperative renal recovery include the quantity and quality of the preserved parenchyma. Our study applys this concept to the retroperitoneal approach in LNSS, aligning with the evolving paradigm in NSS that emphasizes reducing ischemic injury and better preserving renal functional outcomes in low-complexity T1 RCC, thereby achieving comparable oncological and renal functional outcomes. The sutureless and clampless technique is preferentially employed when anatomically feasible, aiming to mitigate procedure-related complications, including iatrogenic arterial transection and pseudoaneurysm formation. Our findings align with those reported by Tanaka et al, in which none of the 74 patients undergoing clampless–sutureless PN developed a renal artery pseudoaneurysm. 43 Collectively, these findings further confirm the notable vascular safety associated with this approach. A previous meta-analysis indicated a higher incidence of positive surgical margin (PSM) following zero-ischemia techniques than that associated with cold or warm ischemia approaches. 13 Indeed, PSM is an established risk factor for increased rates of local recurrence, distant metastasis, overall disease relapse, cancer-specific mortality, and all-cause mortality. 44 Consequently, patients with PSM are often recommended to undergo intensified management strategies, which may include additional surgical interventions, adjuvant therapies, and enhanced surveillance protocols. In the present study, we successfully achieved negative surgical margins in all cases while maximizing functional preservation via parenchymal-sparing techniques. Notably, no local recurrences were observed during the follow-up period. Our findings suggest that this approach potentially yields superior oncological and functional outcomes, leveraging and advancing beyond the benchmarks set by prior techniques.
Our clinical series demonstrates that conventional electrocautery induces tissue hemostasis through high-temperature carbonization, resulting in structurally fragile eschar formation and an elevated risk of secondary hemorrhage. In contrast, RFUS employs precisely controlled thermomodulation to achieve vascular sealing via collagen denaturation while preserving the integrity of tissue architecture. This advanced energy platform integrates simultaneous cutting and coagulation capabilities via dual-mode energy delivery—ultrasonic vibration for tissue division and bipolar radiofrequency for hemostasis—facilitating bloodless parenchymal transection and reliable sealing of vessels ≤ 5 mm in diameter. RFUS-assisted clampless LPN enhances intraoperative safety, minimizing concern for delayed bleeding and promoting superior renal functional preservation. 45 Mounting clinical evidence corroborates zero-ischemia sutureless LNSS as the preferred surgical approach for low-complexity renal masses, demonstrating superior perioperative outcomes, including significantly reduced IBL, complete avoidance of warm ischemic injury, and enhanced renal functional preservation. These advantages emanate from the synergistic combination of advanced energy-based parenchymal sealing and the elimination of suture-related parenchymal compression.36,46
We analyzed a cohort of 60 patients with low-complexity RCC treated with LPN and LNSS across three medical institutions. Our findings indicated a sequential increase in preoperative Scr levels postoperatively and at 6 months in all three groups. Nonetheless, the mean Scr level remained within the normal range. We addressed this paradox through three mechanistic explanations. Prior imaging studies have confirmed that preoperative magnetic resonance imaging-based renal parenchymal volumetry accurately predicts 3-month Scr stability, indicating that preserved nephrons achieve functional compensation through an increased single-nephron eGFR. 47 Longitudinal analysis revealed remarkable stability in Scr levels post-nephrectomy, despite documented compensatory hypertrophy and a 24% decline in the total eGFR. This dissociation between Scr and eGFR trends validates the limited sensitivity of Scr in detecting early renal functional deterioration and demonstrates the remarkable adaptive capacity of remnant nephrons via hyperfiltration and structural remodeling. 48 A long-term study by Ibrahim on living kidney donors revealed that contralateral renal compensation maintains Scr stability despite an approximate 30% reduction in the measured eGFR following uninephrectomy. 49 According to the risk stratification protocol outlined in the 2019 European Society for Medical Oncology (ESMO) guidelines, localized patients with RCC received risk-stratified follow-up, with low-risk T1a cases undergoing annual imaging. 50 The 6-month follow-up intervals in the current study aligned with safety monitoring and ESMO low-risk recommendations. Post hoc analysis confirmed sufficient statistical power; however, larger sample sizes could enhance reliability.
To accurately assess the impact of diverse surgical approaches on renal function in patients with RCC, we also observed a decline in the eGFR of the affected kidney preoperatively and 6 months after surgery. Notably, Group Ⅲ demonstrated favorable outcomes owing to the enhanced preservation of renal parenchyma. This trend was observed in the T1a and T1b stages, attributable to the optimization of the surgical procedure. These data suggest that zero-ischemia, sutureless techniques confer advantages in LNSS surgery, resulting in superior postoperative outcomes. Post hoc power analysis confirmed robust statistical power, ensuring adequate sensitivity to detect clinically meaningful differences in renal functional outcomes within Group III. While the power satisfied the standard threshold, a larger sample size could enhance the reliability of the results where necessary.
The high R2 value indicates that our model captures key physiological determinants of postoperative eGFR decline. However, only WIT emerged as a statistically and clinically significant independent predictor, mechanistically aligning with the pathophysiology of ischemia-reperfusion injury. The maximal tumor diameter did not exhibit a significant association with functional outcomes, likely due to the predominance of small tumors in the cohort, wherein tumor-related parenchymal volume loss was clinically negligible. 51 Furthermore, RNS demonstrated no correlation with renal function preservation, aligning with its original design to predict surgical complexity, including blood loss and operative time, rather than functional outcomes. 52 We prioritized clinical validity over this statistically unstable fit, suggesting the presence of unmeasured confounders or collinearity effects.
While we acknowledge the inherent limitations of this retrospective, non-randomized, multi-center study, which features a relatively small cohort, we implemented several measures to ensure methodological rigor. Most notably, all surgical procedures were conducted by a single senior surgeon, Professor Tang, who possessed extensive experience—exceeding 200 cases—in both clampless and sutureless LPN prior to this study. This high-volume background ensured technical consistency and proficiency across all cases, thereby effectively controlling for operator-related variability and minimizing the potential influence of the learning curve on procedural outcomes. In addition, our department has established a consortium of urological specialists across tertiary medical centers to facilitate a prospective, multi-center, randomized controlled trial with a larger cohort. This pilot study demonstrates the initial safety and feasibility of the non-clamping sutureless technique combined with RFUS, thus providing a foundation for subsequent large-scale clinical validation. While the 6-month follow-up data assess perioperative safety parameters, extended surveillance beyond 24 months remains imperative to comprehensively evaluate long-term oncological outcomes and renal functional preservation. A prospective international registry would provide real-world evidence on long-term functional outcomes across diverse surgical settings.
Conclusion
The non-clamping sutureless technique, combined with RFUS for RCC effectively alleviates renal ischemia-reperfusion injury, offering superior renal function protection while achieving comparable oncological outcomes. This method is feasible in LNSS without renal artery clamping for treating low-complexity T1 RCC. To further explore the viability of this LNSS technique, we plan to apply this procedure to more cases in the future.
Footnotes
Abbreviations
Acknowledgements
We sincerely thank all participating patients for their contributions to this study. This work was supported by grants from the First Affiliated Hospital of Guizhou University of Traditional Chinese Medicine, Guizhou Provincial Science and Technology projects, and Guizhou Health Commission research funding.
Ethical Statement
The informed consent was obtained from each patient involved this study approved by the Ethics Committee of the Affiliated Hospital of Guizhou Medical University (Ethical approval number: 2023-375). And the study involved routinely gathered medical data that were managed anonymously at the whole process, concerning the data removing and statistical analysis process. All experiments were performed in accordance with relevant guidelines and regulations.
Author Contribution
The idea was put forward by KT. Relevant data collection, sorting and pictures were carried out by SX and YM. The project was designed by KT and SX, statistically analyzed and written by KT and SY. Authors mentioned above participated in the thesis and checked repeatedly. All authors (SY, SX, YM, BC, JZ and KT) approved and submitted the version finally.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the High-level Talent Project of the First Affiliated Hospital of Guizhou University of Traditional Chinese Medicine (GYZYYFY-BS-2023 (14)), the Science and Technology Plan Project Of Guizhou Province in 2024 ([2024]405), the Science and Technology Fund Project of the Guizhou Health Commission (grant no. gzwkj2021-211), and Graduate Innovative Plan Project of Guizhou Province in 2024 (No. 2024YJSKYJJ361).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The article and supplymentary materials are the original presentation of the study. For further information, please contact the corresponding author.
