Abstract
Objective
This work introduces MamoRef, an innovative whole-field, near infrared spectroscopy based device for adjunctive breast examination, aiming to help classify benign and malignant lesions in women. Utilizing low-power, non-ionizing red and near-infrared lasers, it provides metabolic information to aid physicians in characterizing lesions in BI-RADS II to IV patients, offering a non-invasive screening alternative.
Approach
Clinical studies were conducted, benchmarking MamoRef against conventional imaging and core biopsies. The device generates 2D maps of relative oxyhemoglobin, deoxyhemoglobin, and oxygen saturation. NIRS-specialized professionals, with basic clinical training, independently scored MamoRef images using a 6-point scale analog to BI-RADS. Scores were averaged and normalized for biopsy comparison.
Main results
The studied clinical cases show promising outcomes. For neoproliferative lesions, MamoRef images reveals high deoxygenated hemoglobin and diffuse high oxygenated/total hemoglobin, suggesting neovascularization around necrotic tissue. Preliminary receiver operating characteristic analysis yielded an area under the curve of 0.77. At a 0.6 threshold, MamoRef showed 70% accuracy and 74% specificity.
Significance
Preliminary results suggest MamoRef can potentially differentiate benign from malignant lesions detected by standard imaging. Trained clinicians might detect and characterize lesions using these metabolic maps. Further larger-scale studies are needed to validate these findings and improve the technology, positioning MamoRef as a potential low-cost, accessible adjunctive screening tool.
Introduction
Breast cancer remains the second most prevalent cancer (12%) worldwide, and the fourth leading cause of cancer death globally. Incidence is higher in countries with a higher Human Development Index (HDI), but mortality is almost 20% lower than in countries with a lower HDI. Although breast cancer mortality has been declining since 2002, this disease continues to represent the most frequent cause of cancer death in women. 1
The goal of breast cancer screening is to reduce morbidity and mortality, both overall and from breast cancer, with a reasonable balance of risks.2,3 Mammography is an invaluable screening and diagnostic tool for breast cancer. To standardize mammography across the countries, the American College of Radiology (ACR) developed the Breast Imaging-Reporting and Data System (BI-RADS). The main shortcoming of conventional mammography is a decrease in sensitivity with the overall sensitivity measured at approximately 70%. 4 The standard 2-view digital mammogram does not detect all cancers with the sensitivity decreasing as the density of breast tissue increases. In women with dense breast tissue, 76% of cancers are missed. 4
None of the guidelines recommend magnetic resonance imaging (MRI) or ultrasonography (US) as the first-line screening method in asymptomatic, average-risk women. 2 The debate centers on the benefits of screening with a focus on the appropriate age to start with it versus any harm from overdiagnosis, false-positive test results, and exposure to unnecessary radiation. 4 The current US Task Force recommendation for breast cancer screening for women at average risk is one mammogram every two years at ages 40-74 years. 5
There is insufficient evidence to assess the risk-benefit balance for the adjunctive use of breast US or MRI in women with dense breasts on a negative mammogram. 5 MRI as a supplement to mammography improves the screening for breast cancer carriers aged 50 and older. 4 Contrast-enhanced MRI creates a blood flow map, that allows detecting tumor neovascularity and peritumoral inflammation; this explains its high sensitivity 6 ; meta-analyses show that for contrast-enhanced MRI a sensitivity of 77% was reported, with a specificity of 86%.7,8 Although these vascular-based technologies offer diagnostic advantages and are well integrated into clinical practice, they require intravenous injections, and their use may be limited by cost, availability, and accessibility. 3
Most current clinical modalities for breast cancer diagnosis rely on the observation of morphological changes in tissue; stronger approaches are needed to examine or even determine metabolic changes quantitatively. 3 As a complement to these well established techniques, near-infrared (NIR) spectroscopy can measure, in a completely non-invasive way, local metabolic signals by combining blood volume and oxy- and deoxyhemoglobin measurements that signal angiogenesis in hypermetabolic tissues, as well as determining the concentrations of lipids and water in tissue.9,10 These parameters have proven useful for discriminating malignant and benign tissue in the breast, and several studies have demonstrated the efficacy of these NIR techniques in exploring malignant changes during neoadjuvant therapies.10–12
It should be noted that NIR techniques allow access to biochemical and physiological data that are not obtained through other methods. 9 Cancer tissue differs from normal tissue in its metabolic activity and density: malignant lesions tend to be surrounded by neovascularization and oxygen-rich blood that feeds its growth. 13 In addition, when tumors grow rapidly or reach a certain size, their central areas may not receive enough oxygen to support further growth. This can lead to necrotic lesions rich in deoxygenated blood, even when neovascularisation is present in the peripheral region. Previous studies show unique absorption signatures associated with dysplastic, malignant, and benign tissue transformations.10,14,15 There are also clinical studies that have evaluated the findings in case series and even in larger prospective cohorts.9,16–18
A recent work describes and validates an innovative continuous-wave (CW), wide field diffuse reflectance device named MamoRef, which uses NIR radiation to detect, characterize and follow the evolution of neoplastic lesions of the breast. 3 Previous studies described different aspects that were evaluated and implemented to improve the ability to differentiate between normal and pathological tissues, enhance image quality, and identify the most suitable materials for the clinical prototype.19–22
MamoRef is a low-cost, easy-to-maintain device that can be implemented in a variety of contexts without the need for specialized personnel. Beyond screening recommendations, several new techniques are being developed to improve early detection of breast cancer, reduce the number of unnecessary biopsies, and improve early treatment protocols.23,24 This study aims to describe the findings of MamoRef images in patients who required core biopsy for suspected cancer on standard breast nodule evaluation images, and to assess its potential to differentiate malignant and benign lesions by comparing its performance with established imaging modalities.
At its current stage of development, MamoRef is intended to be a complementary diagnostic modality. Its goal is to use the unique functional and biochemical information that near infrared spectroscopy (NIRS) provides to supplement the structural information from traditional imaging techniques. We believe this has the potential improve overall diagnostic performance and (by providing the physicians with an additional criteria to) aid in malignancy discrimination.
To validate its potential for this role, we must first establish its baseline performance as a standalone technology. Therefore, this work compares MamoRef against the established imaging techniques. While we believe MamoRef may have future potential as a screening tool, this would require more extensive validation with a larger patient cohort.
Materials and Methods
Study Design and Setting
A prospective exploratory study was carried out between April 2023 and November 2023 at a recognized hospital in Argentina: Hospital Privado de la Comunidad (HPC) located in Mar del Plata, Argentina . It is a high-complexity hospital which since 2018 has a mastology unit accredited by the Sociedad Argentina de Mastología (Argentine Society of Mastology). The hospital performs 460 breast surgeries per year, and it has a patient cohort who receives all health care procedures within the facilities of this hospital (clinical, laboratory, and imaging services) with complete follow-up. The study was approved on date 2021-11-02 by the hospital’s institutional review board (Consejo Institucional de Revisión de Estudios de Investigación - CIREI) and was conducted following the Helsinki Declaration. It is reported according to the STROBE guidelines for reporting observational studies. 25
Population
Patients were prospectively included using simple consecutive random sampling. This included all patients evaluated over 18 years of age who agreed to participate and who required a biopsy due to their mammography findings or, if this was not possible, MRI or breast ultrasound. Patients who were unable to perform the study due to technical reasons (for example: inability to assume the correct position on the stretcher), acute inflammatory lesions, or any other condition that, in the opinion of the physicians, could alter the NIR signal like the presence of tattoos, piercings, or similar non-natural pigmentation on the breast surface were excluded. Table 1 shows the demographic information of the patients included in this study.
Demographic information of the patients that were included in this study.

Procedures and Variables
All patients were evaluated using standard imaging and medical strategies. Those who had a suspicious finding that required a core biopsy were invited to participate in the study.
Figure 1 outlines the resulting patient circuit when MamoRef is introduced in the diagnostic pipeline. These circuit is in accordance with the design target of the proposed device and was well received by both the physicians and the patients.
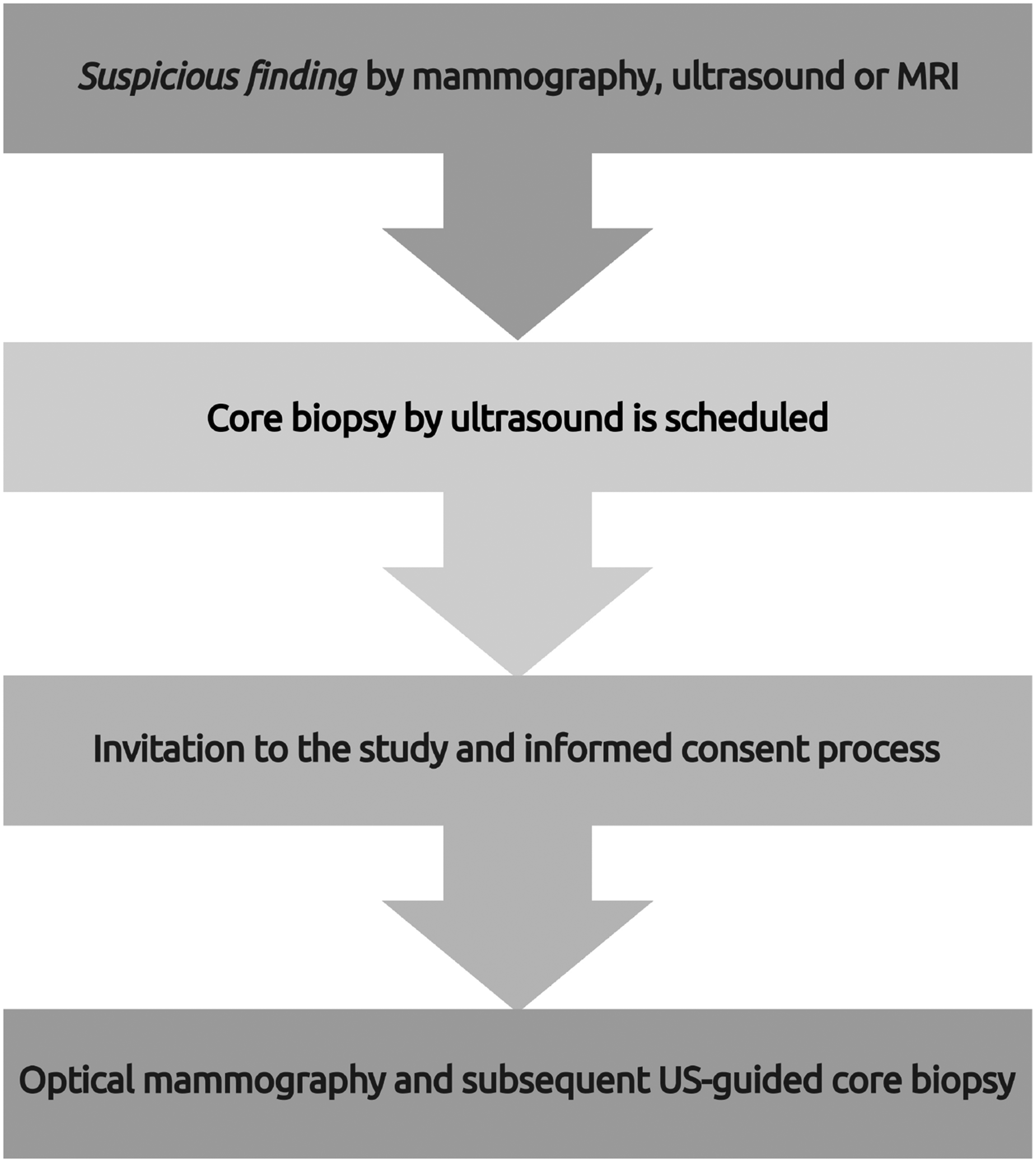
Diagram of the patient’s circuit. If a possible lesion is found by a standard technique (x-ray mammography, US or MRI), an US-guided core biopsy is scheduled; at that stage, the patient is invited to participate in the MamoRef study (for which a signed consent is required).
Prior to the MamoRef measurement, several key patient data points were recorded as part of the informed consent process. These included age, weight, body mass index, heart rate, blood pressure, skin color classified via the von Luschan scale, hematocrit, blood hemoglobin concentration, and the presence of tattoos or piercings in the thoracic area.
Optical Mammography
To begin the measurement procedure, the patient climbs onto a stretcher that has a transparent (acrylic) window, below which the optoelectronic instruments are located (see Reference Carbone et al. 3 for details and Figure 2 for a simplified schematic representation of MamoRef). Then, she gently rests her breast on the window (one at a time), while she places her arms in the most comfortable position. A typical study takes about three to five minutes per breast, and during this period, the patient is asked to rest as still as possible to avoid acquiring low-quality or blurry images.

Schematic of MamoRef. A set of three laser beams (emitting at
The whole process is completely painless, harmless, and safe, since the sources used emit low-power, non-ionizing light. Due to the diffusive propagation of light inside the soft tissue, every image is acquired in the so-called reflectance geometry, which allows to avoid the mechanical compression of the breasts used in X-Ray mammography, which suffer no other pressure than the one exerted by the body weight, making the optical mammography completely comfortable.
The objective of MamoRef is to find and characterize the metabolic behavior of possible inhomogeneities in the breast tissue, which may result from several neoplastic pathologies. To this end, and because the molar absorption coefficients of tissue compounds, such as oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR), strongly depend on light wavelength, it is possible to obtain maps of oxy- and deoxygenated blood using at least two wavelengths; these maps are directly relatable to metabolic function and, as mentioned before, could in principle allow the differentiation of distinct types of inhomogeneities.
Thus, the MamoRef approach relies on acquiring, by a CCD camera, diffusely reflected light images of the breast under study, which may contain lesions (benign or malignant) with optical properties, i.e. scattering and absorption coefficients, that differ from those of the healthy background tissue.
Compared to X-ray-based modalities, diffuse reflectance imaging exchanges penetrating power and resolution for metabolic information. While it has limitations regarding resolving power and information extraction from deep structures, it has been shown that, for example, a 10 mm lesion with twice the absorption of the background medium can be detected at depths of up to 3 cm. 26 It is also important to note that this depth refers to the breast in its compressed state during imaging, and that the detected region represents the entire metabolically active area, not only the solid tumor.
The exposed breast is illuminated by low-power (less than 5 mW) continuous-wave lasers of three discrete wavelengths: 660 nm, 785 nm and 830 nm. The scanning is done following a grid pattern with around 50 positions using a galvo-scanner. Using focused beams instead of expanded sources allows for deeper light penetration, thus allowing the device to explore up to 3-4 cm in depth. More technical details of these aspects of MamoRef were described previously. 3
The raw data obtained by MamoRef are a high number of reflectance images, for each wavelength and each laser position. A reconstruction algorithm converts the raw data into maps of relative oxyhemoglobin (HbO) and deoxyhemoglobin (HbR) concentrations. The process begins by pre-processing the raw images: they are cleaned of background noise and normalized against a composite image of the entire scanned area to enhance the contrast of potential inhomogeneities. This yields a relative attenuation map,
MamoRef thus generates three different 2D maps: relative oxyhemoglobin concentration, relative deoxyhemoglobin concentration, and relative oxygen saturation, the latter derived from the first two. Since continuous-wave laser radiation only allows for the measurement of changes with respect to the background, these maps represent relative changes compared to the mean concentration across the entire tissue under examination. Since during the measurement, the patient’s breast is positioned in a prone posture against the transparent measurement window, ensuring a flattened surface for optimal contact, the resulting maps provide an anterior-posterior 2D projection of absorption across the entire breast tissue.
These functional maps, when combined with structural imaging modalities such as ultrasound and X-ray mammography, offer significant potential for characterizing lesion malignancy. As mentioned before, in general, malignant lesions exhibit increased metabolic activity, often accompanied by neoangiogenesis, which manifests as regions with elevated relative oxyhemoglobin concentration, extending beyond the tumor’s physical boundaries. Additionally, some malignant tumors develop central necrosis, which can appear on the maps as a localized increase in relative deoxyhemoglobin concentration.
Ultrasound-Guided Core Biopsy
While the patient lies in a supine position, previous images (mammography, US, MRI) are reviewed and the image to be biopsied is found using ultrasound. Once the image is located, the most appropriate approach site is selected and the area is cleaned with povidone iodine. Using a sterile technique, a small incision is made in the skin with a scalpel; then an introducer is directed to the nodule and with a 14G core needle already mounted on a biopsy gun (Histo®), 3 to 6 samples are taken. The samples are sent to the pathology department in jars with 10% buffered formalin, labeled with patient data and the specific sample collection site. Once the sample collection is finished, the coaxial introducer is extracted and compression is applied to the area with ice for a few minutes. Hypoallergenic Microporous tape (Hypafix®) is placed on the skin over the incision area and a compressive elastic bandage is applied for 24-48 hours. Pain medication is administered if the patient or the physician performing the biopsy deems it necessary.
Imaging
Mammography, MRI, and breast ultrasound are described according to the ACR BI-RADS descriptors. 27 Regarding the optical mammography procedure, an experienced radiologist reported the MamoRef findings in the context of available conventional imaging (mammography, ultrasound, and MRI) and the pathologic anatomy of each patient.
Pathology
The results are described according to their immunohistochemistry (estrogen and progesterone receptors, HER2 and Ki67) that characterize the behavior of the tumor and the histopathological report of the biopsied lesions. 28
Covariables
Some covariates that could modify the indices evaluated in NIRS were recorded, as it is the case of different baseline characteristics such as skin color, age and body mass index (BMI). Skin color was recorded using the Von Luschan valid scale and then patients were classified according to Fitzpatrick 29 phototypes; the presence of chromatic lesions such as nevi or tattoos was also recorded. Physiological variables were recorded during the optical mammography procedure: respiratory rate (RR), heart rate (bpm), blood pressure (BP-BP), and hematocrit. 30
Skin color is of particular interest as it can potentially affect light propagation in tissue. Table 2 has the Von Luschan scale for each patient of the studied cohort. It is important to note that this distribution is demographic dependent and specific to the region in which the study was carried out. It is our priority for future work to have a more diverse cohort.
BI-RADS for each patient included in this study according to different imaging techniques (columns two to four), as well as the corresponding pathology findings (last column), compared to the optical mammography finding (fifth column).

Patient number 2 was excluded because it was not possible to analyze her pathology. Also shown is the skin color of each patient following the von Luschan scale.
Statistics
A data collection form was created to systematize the capture and minimize errors in Redcap®, and it was entered by a trained and data-entry-expert operator. The numerical variables are expressed as median or mean with their corresponding dispersion measure (25-75 % percentiles or standard deviation) as appropriate. The variables are checked for normality using the Kolmogorov-Smirnov test. 31 Categorical variables are expressed as absolute frequency (n) and percentage (%). The agreement between the results is described when both results are in the same direction (negative-negative or positive-positive) for both optical mammography vs. pathology and for optical mammography vs. conventional mammography. The data was analyzed using Stata/IC 15.1 for Mac.
In our study, 21 patients were included, of which 17 had mammography, all of them had breast ultrasound with core biopsy, and 4 patients had MRI in addition to mammography or breast ultrasound. Finally, patient number 2 was excluded from the final analysis due to the inability to perform the core biopsy adequately, as the size of the lesion prevented it. All patients were of Hispanic-Latino ethnicity, 16 were postmenopausal, and none had skin lesions, nevi, or tattoos that could interfere with the images obtained by MamoRef. The most frequent phototypes were IV and V.
The contralateral breast was imaged in all cases of unilateral lesions. These breasts typically had a low BI-RADS score on conventional imaging and therefore did not undergo biopsy. Consequently, for the purpose of our analysis, these contralateral breasts were assigned a ground truth status of ’negative for malignancy’.
Performance Metrics
We conducted a classification test to preliminarily evaluate MamoRef’s performance as a semi-quantitative classifier for breast lesion malignancy. For this test, 5 professionals specialized in NIRS, with basic previous clinical training regarding breast cancer anatomy, independently scored the images generated by MamoRef.
A structured 6-point scoring scale, modeled conceptually after the ACR BI-RADS assessment categories, was developed for this study. Using a familiar framework like BI-RADS aims to provide a standardized structure for assessment and potentially facilitate future clinical translation.32,33
The scale was defined as follows:
Score 0: The image cannot be assessed due to artifacts or other technical issues. Score 1: The image shows normal breast tissue. Score 2: The image displays abnormalities that are likely benign. Score 3: The image displays abnormalities with a low probability of malignancy. Score 4: The image displays abnormalities possibly compatible with malignant lesions. Score 5: The image displays abnormalities highly suggestive of malignancy.
In the interest of avoiding bias, the professionals only had access to the images produced by MamoRef and were instructed not to discuss their scoring criteria with each other. Before scoring, all participants received training on the underlying physics of MamoRef images and the expected appearance of physiological variations (e.g., nipple, effects of pressure, known benign features) in the different chromophore maps, but no specific guidance on clinical interpretation was provided.
The evaluators were provided with the complete set of images for each case (HbO, HbR, total hemoglobin, and oxygen saturation maps). Their assessment was guided by established physiological principles from the literature, which link elevated HbO concentrations to metabolic activity and neovascularization, and localized HbR concentrations to hypoxia or necrosis. An initial validation of this assessment is one of the main objectives of this work.
Cases where half or more of the assigned scores were
Results
At the time of the study, the vital signs were stable without anemia (see Table 1). All patients with BI-RADS below III had breast US or MRI. The findings of each patient according to BI-RADS are described in Table 2. Of all the biopsies performed, only seven were cancer, two of them in situ, as described in Table 3.
Results and specifications of the core biopsies performed.

NA: not available. The hyphens indicate that no biomarker corresponds to the core biopsy performed. Patient number 2 was removed from further analysis due to the impossibility of correctly assessing her histopathology by a regular core biopsy. “Yes/No” results for malignancy and its description are transcribed from the histopathology report obtained from the patient’s biopsy. The abbreviations used are: ER - Estrogen Receptor, PR - Progesterone Receptor, HER2 - Human Epidermal growth factor Receptor 2, Ki67 - Ki-67 Protein, IHC - Immunohistochemistry.
Figures 3 to 6 show examples of the relative concentration maps of hemoglobins (deoxygenated, oxygenated and total), as well as the oxygen saturation map, retrieved by MamoRef, for patients 8 and 12 whose diagnosis found by core biopsy is cancer, and for patient 10, who has no evidence of cancer, respectively. The units of maps from one to three are micro molar (
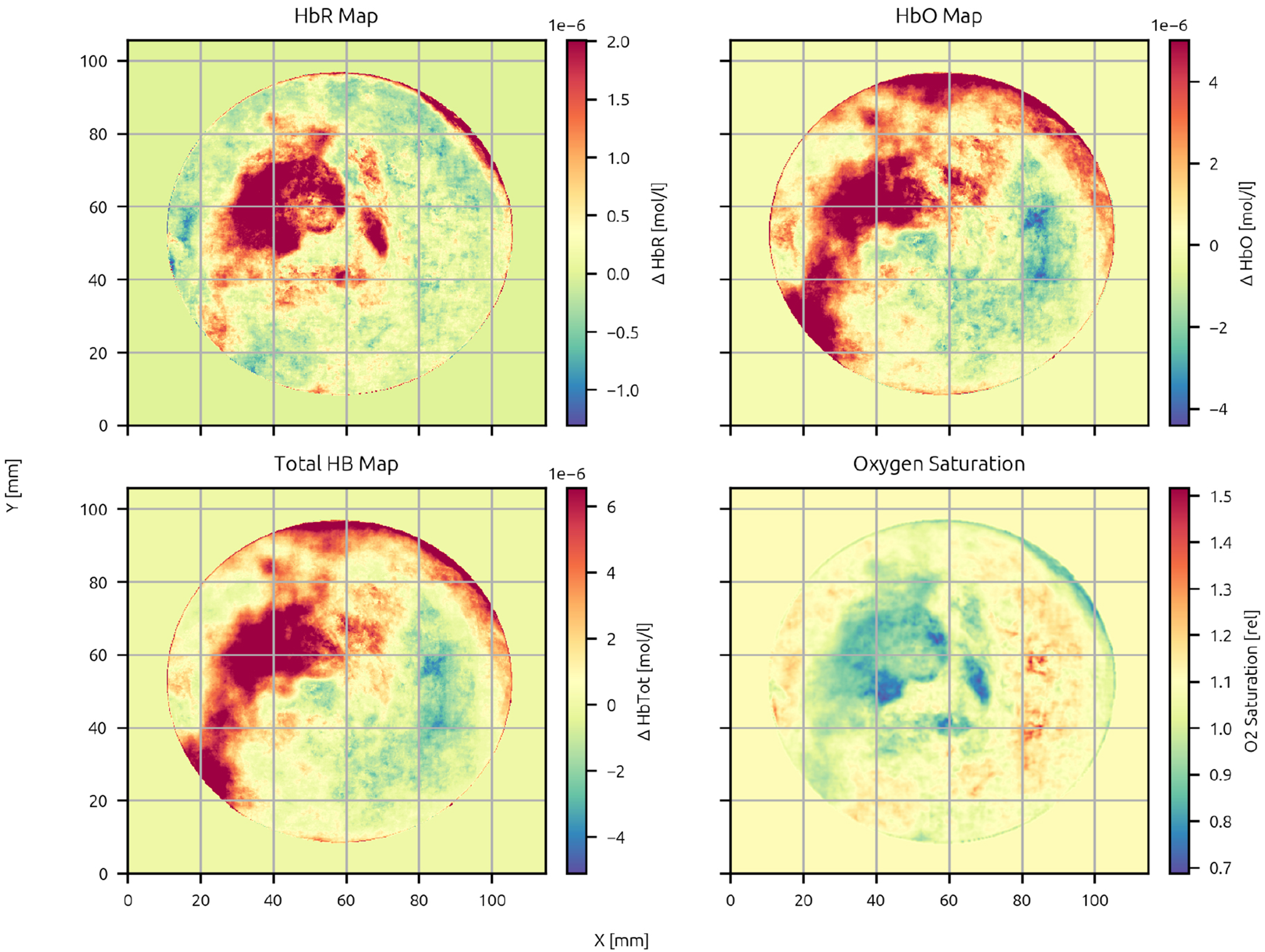
MamoRef’s reconstructed maps for patient 8 in Table 4: Deoxygenated hemoglobin relative concentration map (upper-left plot), oxygenated hemoglobin relative concentration map (upper-right plot), total hemoglobin relative concentration (lower-left plot) and relative oxygen saturation map (lower-right plot).

MamoRef’s reconstructed maps for patient 12 in Table 4: Deoxygenated hemoglobin relative concentration map (upper-left plot), oxygenated hemoglobin relative concentration map (upper-right plot), total hemoglobin relative concentration (lower-left plot) and relative oxygen saturation map (lower-right plot).

MamoRef’s reconstructed maps for the contralateral breast of patient 12 in Table 4: Deoxygenated hemoglobin relative concentration map (upper-left plot), oxygenated hemoglobin relative concentration map (upper-right plot), total hemoglobin relative concentration (lower-left plot) and relative oxygen saturation map (lower-right plot).
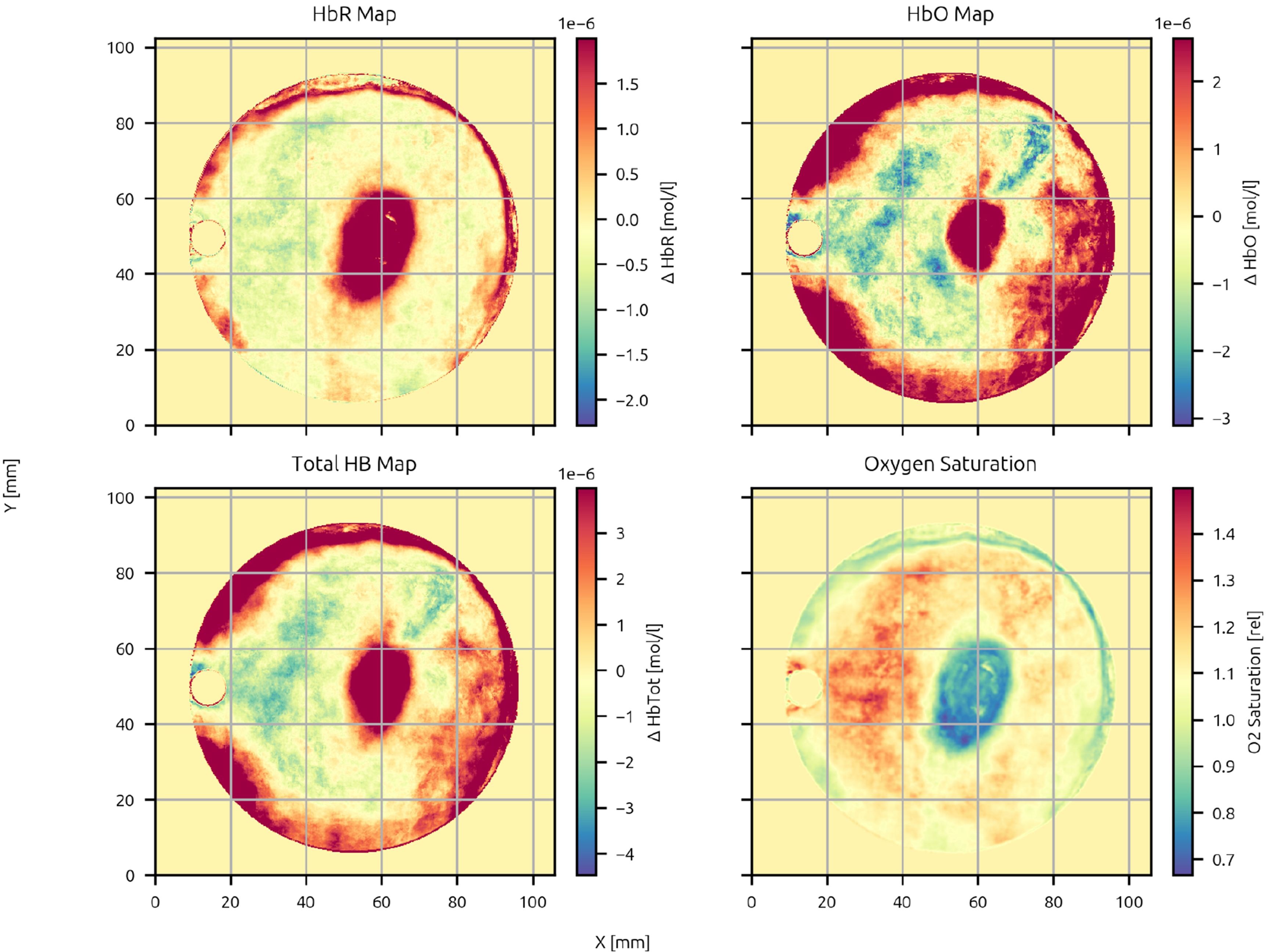
MamoRef’s reconstructed maps for patient 10 in Table 4: Deoxygenated hemoglobin relative concentration map (upper-left plot), oxygenated hemoglobin relative concentration map (upper-right plot), total hemoglobin relative concentration (lower-left plot) and relative oxygen saturation map (lower-right plot). No region is compatible with a malignant lesion.
Analysis of the four different 2D maps obtained by MamoRef.

These results are to be compared with those given in Table 3 and with the Figures available in the supplementary material. The patient number 2 was removed from this analysis due to the impossibility of correctly assessing her histopathology by a regular core biopsy.
Table 4 show descriptive findings provided by medical personnel who participated in the study. They were instructed to produce a descriptive report based solely on the MamoRef images, without having seen the results from other modalities or the final histopathology report.
The physicians were briefed on the working principles of MamoRef and were shown the HbO, HbR, total hemoglobin, and oxygen saturation maps for each case. Based on this information and their own clinical expertise, they described their findings as they would for any other imaging modality. To accurately reflect their independent interpretations, the descriptions were recorded verbatim.
After this analysis was done, the participating physicians correlated the general location of the lesions identified by MamoRef with their location as reported in standard modalities. While exact 3D coordinate comparisons are not possible due to differences in tissue compression and the functional nature of the NIRS data, the general anatomical locations were compared. In all cases, the physicians confirmed that the findings were spatially coincident within the expected margins of error.
In all MamoRef images, the nipple area is prominently visible. This region can be correlated with reference images of the measured breast and used to spatially reference other potential findings. In Figure 3, the nipple is located at
In Figure 4 another case of ductal carcinoma is shown. In this case, a region of increased concentration in HbR, HbO and total hemoglobin is seen between
Figure 5 shows the contra-lateral breast of the patient shown in Figure 4. A notable artery is visible below and to the left of the nipple (around hour 7), which was also visible in natural light due to its shallowness. The images otherwise show a relatively homogeneous nature with no areas of uptake other than the artery and the nipple that are present in HbO and HbR maps.
On the other hand, in Figure 6 a case of no malignant lesion is shown. There, it can be seen that no correlation is found between the zone of high relative HbO concentration located at the right contour (hours 3 to 5) and the HbR relative concentration map. In addition, no notorious decrease in O
Performance Metrics
Figure 7 presents the Receiver Operating Characteristic (ROC) curve derived from the analysis detailed in Procedures and variables, demonstrating an area under the curve (AUC) of 0.77, indicative of good predictive performance.

Receiver Operating Characteristic (ROC) curve. The black slashed line represents the random guess.
Considering a 0.6 threshold, obtained from the ROC curve by identifying the point that maximizes the Youden’s J statistic, MamoRef reaches an accuracy of 70% (90% CI: 55%-85%), sensitivity (true positive rate): 62% (90% CI: 33.33%, 87.50%), and a specificity (true negative rate) of 74% (90% CI: 56%-90%). Confidence intervals (CIs) for performance metrics were calculated using a non-parametric bootstrapping approach. The dataset was resampled with replacement 1000 times to generate bootstrap samples, and the metrics were recalculated for each sample. The CIs were derived from the empirical distribution of the bootstrapped metrics by determining the 2.5th and 97.5th percentiles for 95% CIs. It is important to note that, due to the relatively low number of patients, the 95% CI range is quite large. We expect this to improve as the number of patients in the protocol increases. Moreover, choosing an appropriate threshold involves balancing sensitivity and specificity, which can vary depending on the desired emphasis. Table 5 shows a comparison of these results with standard imaging modalities.
Comparison of statistical metrics for MamoRef with standard imaging modalities.

Note the wide spread of values among different studies, highlighting the difficulty in establishing definitive quantitative statistical values.
To quantify the inter-rater agreement among the evaluators, we computed the Fleiss’ kappa statistic. 42 The resulting value was 0.253 (95% CI: 0.136-0.354), which can be interpreted as slight to fair agreement. As with the previously discussed metrics, the limitations of the current sample size should be taken into account. We anticipate that inter-rater agreement will improve with more clinical studies and as the evaluators gain more familiarity with the image characteristics produced by MamoRef.
Discussion
A preliminary quantitative statistical analysis was carried out to determine the potential ability of MamoRef to correctly assess breast tumor malignancy. It is important to note that this study has an exploratory intention only and it has to be taken carefully. Further statistical analysis will be done in future works when more clinical data is available.
Results show that the comparison between the concentration maps obtained by MamoRef and the corresponding pathology analyses (core biopsies) yield an agreement of nearly 80%, and 6 of the remaining 17 evaluable set of concentration maps do not show any evidence of lesion; in these cases, the corresponding core biopsy showed that there are no detectable signs of cancer. MamoRef was designed with patient comfort, portability, affordability, and diagnostic efficiency as its primary goals.
As a drawback, four MamoRef studies had to be discarded from the final analysis due to artifacts. The most common reasons for them are incomplete or inhomogeneous support of the breast in the measurement window and high intensity reflections due to the laser hitting a damaged (scratched or spotted) portion of the transparent acrylic of the measurement window. There was also one specific case of artifacts due to patient movement while the measurement was being done. These measurement errors generate numerical instabilities in the inversion process that are difficult to distinguish from real anatomical data. For this reason, in the cases in which these artifacts were detected, the whole measurement was discarded.
This work should be contextualized within the broader field of optical breast imaging. While advanced modalities like functional photoacoustic tomography and time-domain or frequency-domain NIRS systems have demonstrated high-resolution capabilities, published clinical validation is often limited to small-scale studies or case reports.43–47 These systems, while technically powerful, also tend to be complex and expensive. In contrast, our study validates a simpler, more accessible continuous-wave approach on a comparatively larger patient cohort, providing a statistical analysis of its performance and demonstrating its clinical potential feasibility in a broader setting.
In this sense, while it is not the first near-infrared (NIR)-based mammography device to reach clinical feasibility testing, 48 MamoRef stands out due to its relative simplicity and accessibility. Specifically, compared to many other diffuse optical tomography (DOT) systems, (i) uses low-cost, off-the-shelf continuous-wave lasers instead of time-resolved sources; (ii) is able to take a measurement of the whole breast in a single, relatively fast acquisition; and (iii) its cost-effectiveness makes it accessible for medical institutions in less developed countries or for primary care facilities. These qualities make it a potential candidate for early-stage screening, treatment monitoring, and general breast diagnostics, especially in cases where other techniques are contraindicated (e.g. pregnant women; women under the age of 40) or unavailable due to accessibility limitations.
Thus, a comprehensive analysis of the initial results in real patients is crucial for the device’s continued development. As this study represents an initial feasibility assessment, the results should be interpreted within the appropriate context. Several challenges typical of early-stage medical hardware development were encountered, including:
A limited number of patients due to stringent enrollment criteria and the availability of only a few prototypes, constrained by financial limitations. Technical challenges inherent to early-stage prototypes, added to the relatively untrained personnel unfamiliar with the novel technique. Notably, four studies had to be excluded due to artifacts caused by improper breast positioning. Difficulties in avoiding bias during image evaluation as physicians, engineers, and physicists shared and recognized emerging patterns as part of their training.
Despite these limitations, they do not preclude the possibility of reaching meaningful conclusions. The comparison between MamoRef’s concentration maps and pathology analyses (core biopsies) showed an agreement of nearly 80%. Additionally, the quantitative performance metrics, though limited by the small sample size and high margin of error, yielded results similar to those of gold-standard screening devices.23,49
As mentioned before, these results, although rigorously obtained, lack of high statistical significance because of the small sample considered. However, we will present a much more reliable statistical analysis in a future paper. It is important to take into account that these values were obtained on a new imaging technique, in its first clinical trial, using first-generation clinical prototypes and with physicians who had none prior experience interpreting MamoRef (or NIR) images and/or physicists with minimal medical training.
This last point is significant. As with any image-based diagnostic technique, proficiency in interpreting results improves with experience. The predictive power of such techniques increases not only with advances in hardware and software but also as users become more acquainted in distinguishing the sometimes subtle differences between healthy and pathological tissues in the images.
Although other optical mammography devices have been considered before, and are currently under study, they usually show a number of differences with ours; for example, there exist several handheld devices that are intended to probe the breast in a similar way that ultrasound does.50–52 This approach is painless, compact, and portable, but as a drawback, it is operator-dependent and cannot image the breast completely at once. A different type of optical mammography devices uses a set of fiber bundles to both emit and detect the pulsed light transmitted through the breast43,53 in combination with time-domain detecting systems. Although this method is able to retrieve absolute values of the optical properties of the breast, instead of relative ones, it is significantly more expensive and cumbersome and needs to compress the breast as in the traditional X-ray mammography. Finally, other devices rely on the use of contrast agents such as indocyanine green (ICG).43,54,55 Although this method can improve the contrast between the neurovascularized lesion and the surrounding healthy tissue, it lacks the non-invasiveness of MamoRef.
There are, however, relatively few clinical studies conducted with devices using NIRS, highlighting the importance of this contribution. The other studies utilizing clinical data are either outdated 56 or employ much more complex devices based on tomography geometries. 57 On the other hand, while thermography may seem related to NIRs and has been explored in numerous clinical studies as a potential tool for breast screening,58,59 it represents a fundamentally different technology with distinct limitations and challenges compared to NIRs. Thermography relies on detecting surface temperature variations, which can be influenced by numerous confounding factors, limiting its sensitivity and specificity for deep tissue characterization. Consequently, the findings of these studies suggest that thermography may not be suitable as a reliable standalone method for breast screening. 60
Future research on MamoRef should focus on several key areas:
Expanding patient studies: Conducting larger-scale studies with proper blinding to generate robust statistical data. Further exploring the effect of skin color: While we aimed for a broad patient cohort, due to the demographic characteristics of the local population, individuals with very light or very dark skin tones could not be recruited for this study. While the method is expected to be robust (as it relies on differential absorption across wavelengths, and melanin has a relatively flat spectral absorption in the NIR window, meaning its contribution is largely canceled out) this hypothesis must be properly validated in future works. Improving hardware: Iteratively designing and building improved prototypes to reduce instances of improperly acquired images. Training personnel: Developing training protocols to familiarize physicians with the unique imaging characteristics of MamoRef. Improved reconstruction algorithm: MamoRef generates hundreds of raw diffuse reflectance images from various source positions and wavelengths, which are synthesized into human-readable 2D chromophore concentration maps (and their derivatives). This process is detailed in our previous works.
3
Future developments include the integration of AI-based image-to-image algorithms capable of providing depth resolution, faster reconstruction speeds, and reduced artifacts. Integrating with existing screening technologies: In the present contribution, MamoRef images were analyzed independently of other diagnostic modalities. However, in real-world scenarios, it is expected that physicians will compare MamoRef data with X-ray mammography and ultrasound to arrive at a diagnosis. Furthermore, AI classification algorithms, which are already being developed for lesion classification in mammograms, could be enhanced by incorporating the metabolic data provided by MamoRef.
While there is still significant work to be done, the initial results are encouraging. MamoRef’s potential as a low-cost (expected to be below
Our current reconstruction algorithm is a model-based analytical approach that converts raw absorption maps into maps of HbO and HbR concentrations. 3 To improve this process, we are developing an AI-based, image-to-image reconstruction pipeline. The intended approach is a supervised learning strategy utilizing convolutional neural networks (CNNs), such as a U-Net architecture. We plan to train the model on a large dataset generated from Monte Carlo simulations. In this strategy, simulated absorption maps will be the input, and the known ground-truth hemoglobin distributions used to generate them will serve as the target output. This methodology has shown preliminary potential to reduce artifacts, improve signal-to-noise ratio and, importantly, to retrieve depth information in the final reconstructed HbO and HbR maps.
Beyond its technical capabilities, it is important to contextualize MamoRef’s potential role within the current breast cancer diagnostic pathway. The results suggest that its most immediate clinical application could be as a non-invasive secondary assessment tool. For instance, for lesions classified as BI-RADS 3 (probably benign) or 4a (finding with a low likelihood of being cancer) by mammography, which have a low to moderate suspicion of malignancy, MamoRef could provide supplementary metabolic information to help assesing risk more accurately. A finding of benign-like metabolic activity could strengthen the case for a follow-up imaging surveillance plan, potentially reducing the number of immediate core biopsies. This would not only alleviate patient anxiety and discomfort associated with invasive procedures but also reduce healthcare costs. Furthermore, by providing functional data on tumor vascularity and oxygenation, MamoRef could enhance the diagnostic confidence of clinicians when integrated with the structural details provided by mammography and ultrasound, leading to more informed decision-making.
Conclusion
Although further clinical studies are needed to accomplish a more statistically significant comparison, our preliminary results show that MamoRef has the potential to differentiate between benign and malignant lesions, once they have been detected by conventional imaging techniques such as X-ray mammography, US or MRI. Trained clinicians could help not only telling apart the malignancy of a lesion, but also detecting them by careful inspection of each map.
In future work, we propose to train an AI algorithm that could automatically locate and classify different breast lesions. Further improvements include the reconfiguration of MamoRef’s structure for the sake of comfort in pregnant women or in women with reduced mobility that find our device uncomfortable.
As an additional improvement, in future iterations, we plan to add visual cues to the MamoRef GUI that highlight to the operator the presence of artifacts as it would be desirable that they make sure that the patient’s breast is fully supported on the window. Also, the acrylic of the measurement window is considered disposable. We plan to recommend using a new plastic sheet for each patient instead of cleaning and sterilizing it as it was done in this study.
Footnotes
Abbreviations
Acknowledgements
The authors acknowledge the use of large language models such as ChatGPT for selective grammar clean-up of some paragraphs. In all cases, the text was written by the authors, and the LLM was instructed not to change the tone or the general language of the text.
Ethics Approval
This work involves the use of clinical data gathered on human subjects. This clinical study was conducted at the Hospital Privado de Comunidad and was approved on 2021-11-02 by the HPC’s institutional review board (Consejo Institucional de Revisión de Estudios de Investigación - CIREI). An amendment was further approved by the same review board on 2023-03-30. This study was authorized by the Ministry of Health of the Province of Buenos Aires, Argentina, under number: DISPO-2022-28-GDEBA-DPEGSFFMSALGP.
The consent was for participation in the clinical study and the use of their medical data for research purposes, as detailed in the approved informed consent form. This study was conducted according to the Declaration of Helsinki and Good Clinical Practice.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Bionirs Arg SA provided financial and material support for the development of the MamoRef device and to carry out the clinical trial. The authors affiliated with this institution only participated in their capacity as scientific researchers and have no direct financial interest in the results obtained.
Data Availability
In the interests of preserving patient anonymity, the data used for the present contribution is not uploaded to a public repository. However, it can be shared if formally requested to ensure reproducibility of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
