Abstract
Early detection of skin cancer is crucial for effective treatment and improved patient outcomes. Recent advancements in oncologic imaging, particularly molecular imaging techniques, have revolutionized cancer diagnostics and treatment by enabling the visualization of tumors and cellular activities at the molecular level. These techniques facilitate the identification of early-stage cancers that might remain undetectable through traditional imaging methods. Innovative technologies such as reflectance confocal microscopy (RCM) and optical coherence tomography (OCT) which visualize skin at near-histologic detail and skin fluorescent imaging (SFI), which targets αvβ3 integrin expression, are promising for non-invasive early detection of melanoma. By integrating in vivo molecular imaging with tumor biomarkers, clinicians can gain more precise insights into processes integral to cancer biology, leading to improved diagnosis, prognosis and the development of personalized treatment strategies. This review explores imaging modalities used in skin cancer diagnosis, highlighting their advantages and limitations, with an emphasis on molecular imaging, stressing its potential to improve early detection, personalize treatment and monitor therapeutic responses.
Introduction
Early cancer detection is crucial. It enables less aggressive treatment, preserves healthy tissue, improves quality of life, lowers costs and reduces mortality through timely intervention. 1 Compared with standard biopsy and histology, imaging is less invasive and more effective for tumor analysis, offering a comprehensive, dynamic view of the tumor and its microenvironment rather than a static snapshot.
Over the past century, medical imaging has evolved dramatically; from the early use of x-rays for basic structural visualization, to the development of computed tomography (CT), magnetic resonance imaging (MRI) and positron emission tomography (PET), which have allowed increasingly detailed insights into both anatomy and function. Today, the field is entering a new era of precision, where imaging is no longer limited to structure or metabolism and can extend into molecular characterization of disease. Emerging modalities and hybrid platforms now offer the ability to visualize specific molecular targets, cellular processes and dynamic interactions within the stromal tumor microenvironment (STE). This convergence of imaging and biomarker science holds transformative potential for early diagnosis, therapeutic monitoring and personalized treatment strategies, underscoring the need for an up-to-date, integrative review of recent advances and future directions.
Cancer diagnostics and treatment are moving toward personalized medicine, with a focus on precise radiotracer targeting and theranostics – the integration of diagnostic testing with targeted therapy – to enhance imaging sensitivity and specificity and drug delivery/treatment. 2 Advancements in resolution and novel imaging probes hold considerable promise for the future.
Molecular imaging offers focused, in vivo visualization of tumor heterogeneity, angiogenesis and treatment monitoring by focusing on proteins, receptors and metabolic processes.3,4 By targeting molecular rather than structural changes, this form of imaging offers the ability to non-invasively identify molecular changes and provides a deeper understanding of disease mechanisms, particularly important during early disease stages.5–7
Molecular imaging technologies in oncology face significant challenges, including spatial limitations, interpretative variability and concerns over cost and accessibility. 7 In terms of the latter, expensive and complex imaging modalities like PET/MRI are in stark contrast to more affordable and widely available options like ultrasound, optical imaging and SFI.
Tumor heterogeneity can significantly impact the accuracy of molecular imaging since it relies on specific biomarkers, which are often differentially expressed throughout a tumor. 8 This can lead to discrepancies in assessments of tumor size and extent, potentially causing false negatives or underestimating spread. Consequently, this can affect treatment as imaging may only show the destruction of cells with the target biomarker, while resistant, non-targeted cell populations continue to thrive and grow.
This review summarizes key imaging modalities currently used in skin cancers in its various stages, highlighting their clinical applications and comparative advantages. For more detailed technical discussions, readers are referred to comprehensive texts and specialized reviews.
Imaging modalities (Tables 1 and 2)
Whole Body/Deep-Tissue Imaging
Magnetic Resonance Imaging (MRI) provides high-resolution in vivo anatomical imaging, making it a valuable tool for assessing tumor size, location, tissue cellularity and perfusion.18–24 It is particularly valuable in assessing the extent of skin tumor invasion in the head and neck regions, where accurate delineation of tumor boundaries is crucial for surgical planning. 32
Representative Images from various Imaging Modalities.

In Vivo Imaging Techniques.
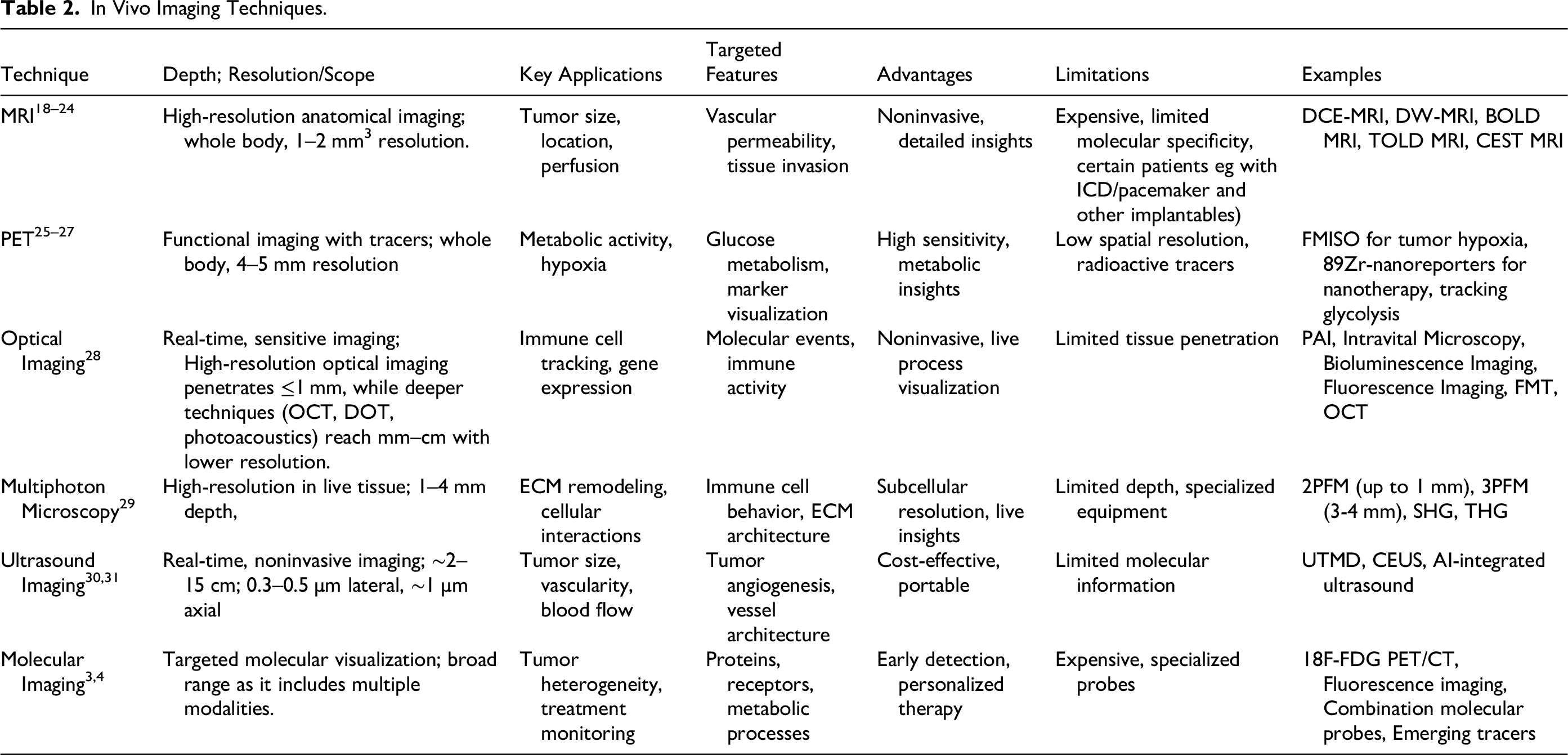
MRI uses strong magnetic fields and radiofrequency pulses to detect signals from hydrogen nuclei in water molecules, generating detailed images of soft tissues. It is particularly effective in evaluating vascular permeability and surrounding tissue invasion. Its noninvasive nature allows for detailed structural and functional insights, though it is expensive and has limited molecular specificity. Specialized techniques such as Dynamic Contrast-Enhanced MRI (DCE-MRI), Diffusion-Weighted MRI (DW-MRI), Blood Oxygen Level-Dependent (BOLD) MRI, Tissue Oxygen Level-Dependent (TOLD) MRI and Chemical Exchange Saturation Transfer (CEST) MRI enhance its application in tumor characterization. Typical resolution is approximately 1–2 mm3. MRI can detect tumors as small as 6.9 mm (mean size for ductal carcinoma in situ) and 9.3 mm (mean size for invasive cancers). 33 MRI has limitations, including safety concerns for patients with metallic implants like pacemakers and reduced effectiveness in imaging tumors with low water content or calcified structures, such as certain bone or mucinous tumors. 34 It provides structural and anatomical information, not metabolic or other cellular activity.
Positron Emission Tomography (PET) provides functional in vivo imaging using radiolabeled tracers to assess metabolic activity, hypoxia and receptor expression.25–27 PET imaging is utilized to detect and monitor metastatic melanoma, particularly in stages III and IV, where it helps identify metastases and assess treatment response. 35
PET detects gamma rays emitted indirectly by a radioactive tracer that accumulates in metabolically active tissues, producing functional images of biological processes. By enabling visualization of radiolabeled tracers that target specific markers such as glucose metabolism, receptor expression, proliferation, hypoxia, or immune activity. PET offers high sensitivity for metabolic and molecular insights, though it requires radioactivity and has low spatial resolution, making it potentially harmful to health and less precise in pinpointing the exact location of abnormalities.
PET is useful for tracking nanotherapy biodistribution, predicting nanotherapy response using zirconium-89 nanoreporters, imaging tumor hypoxia with FMISO, measuring tumor vasculature and permeability and visualizing metabolic processes like glycolysis and choline-phospholipid metabolism. Typical spatial resolution is approximately 4–5 mm and PET can detect tumors as small as 7 mm, corresponding to the detection of tumors with a volume of 0.2 ml in a 5:1 tumor-to-background ratio. 36 The most widely used molecular imaging modality overall, 18F-fluorodeoxyglucose (FDG) PET/CT assesses cancer metabolism by tracking uptake of the 18F-FDG radiotracer. Other notable advances in molecular imaging include fluorescence imaging with targeted fluorescent probes for tissue visualization; dual-modality probes that combine radioisotopes and fluorescence for enhanced imaging; and emerging radiotracers beyond 18F-FDG, such as 68Ga-DOTATATE for neuroendocrine tumors, 89Zr-huJ591 and 18F-DCFPyL for prostate-specific membrane antigen (PSMA), and 18F-alfatide for assessing angiogenesis. Finally, there are new PET scans using radiolabeled tags identifying CD8 cells to differentiate tumor progression from pseudoprogression in the context of immunotherapy. 37
Intermediate to Deep Tissue Imaging (∼2-15 cm)
Ultrasound imaging is an in vivo, noninvasive method for assessing tumor size, vascularity and blood flow, particularly useful in evaluating tumor angiogenesis and vessel architecture.30,31 It is used in skin cancer to non-invasively assess tumor thickness, delineate margins, evaluate regional lymph nodes and monitor for recurrence, providing structural and procedural guidance complementary to molecular or histologic imaging modalities. 38
The ultrasound probe sends high-frequency sound waves into the body and detects echoes reflected from tissue interfaces to create real-time images of internal structures. As a cost-effective and portable technique, ultrasound enables dynamic imaging, though it provides limited molecular information, limited visualization in certain tissues (bone, air-filled areas), lower sensitivity for small or early-stage cancers and low specificity for malignant tissue. Advancements such as ultrasound-targeted microbubble destruction enhance drug delivery and contrast-enhanced ultrasound improves visualization of blood flow and tissue perfusion. Typical resolution for conventional ultrasound is approximately 1–2 mm; High-frequency ultrasound approximately 30–100 µm. Super-resolution ultrasound imaging (SRI) has demonstrated the capability to visualize microvasculature, aiding in the differential diagnosis of breast masses. 39
Shallow to Intermediate Depth Imaging (∼1-2 cm)
Optical imaging enables in vivo, highly sensitive imaging of immune cell tracking, gene expression and target distribution, providing crucial insights into cellular and molecular events as well as immune activity. 28 Optical imaging modalities provide non-invasive, high-resolution visualization of skin architecture, enabling early detection, margin assessment and monitoring of malignant lesions while reducing unnecessary biopsies.
This technology uses visible or near-infrared light to visualize tissues labeled with fluorescent or bioluminescent markers, enabling high-sensitivity imaging of surface or shallow structures. Its noninvasive nature allows for real-time visualization of live processes, yet its limitation lies in restricted tissue penetration. Optical imaging comprises multiple different modalities including photoacoustic imaging (research; clinical trials ongoing), intravital microscopy (research; preclinical trials ongoing), bioluminescence imaging (research; preclinical, genetically modified cells required), fluorescence imaging (used in clinical setting, specifically intraoperatively), fluorescence molecular tomography (research; preclinical) and optical coherence tomography (widely used in ophthalmology clinics). Photoacoustic imaging visualizes tumor vasculature and measures tissue oxygenation. Intravital microscopy provides subcellular-resolution imaging of STE in living organisms. Bioluminescence imaging is used to track cancer cell movement and monitor tumor-stroma interactions. Fluorescence imaging can detect single cells and visualize dynamic interactions between cancer cells. Fluorescence molecular tomography enables 3D imaging of molecular targets, such as matrix metalloproteinases (MMPs) and tumor vasculature. Lastly, optical coherence tomography (OCT) offers label-free imaging of tissue structures and vascular networks with micrometer-scale resolution. Typical resolution is approximately 10 µm. Optical imaging can detect tumors with an average imaging depth of 0.65 mm and a resolution of 10 µm. 7
Superficial Imaging (≤1 mm Depth)
Reflectance confocal microscopy (RCM) is a non-invasive imaging technique which provides high-resolution, in vivo visualization of the skin at a cellular level. Utilizing a low-power laser (usually with a wavelength of 830 nm) to provide high-resolution, label-free imaging by detecting variations in tissue refractive indices and the principle of light reflectance, RCM captures detailed images of the epidermis and superficial dermis without the need for a biopsy. It offers lateral resolution of approximately 0.5–1.0 micrometers and axial resolution around 3–5 micrometers. RCM aids in diagnosing skin cancers, evaluating various inflammatory skin conditions, and monitoring treatment response, offering a rapid and accurate alternative to more invasive methods. 40
Multiphoton microscopy enables high-resolution imaging in live tissues, facilitating the study of cellular interactions and extracellular matrix (ECM) remodeling. 29 It can be used in vivo (especially in intravital microscopy with implanted imaging windows), though usually limited to superficial tissues (so far available in research and preclinical trials in humans) or small animal models due to shallow imaging depth. Multiphoton microscopy employs near-infrared lasers to excite fluorescent molecules via simultaneous absorption of two or more photons, allowing deep, high-resolution imaging of living tissues. Two-photon fluorescence microscopy allows imaging up to 1 mm deep in mouse brains for detailed cellular structure studies, three-photon fluorescence microscopy extends imaging depth to 3–4 mm with enhanced contrast and reduced background, second harmonic generation microscopy provides label-free detection of non-centrosymmetric structures such as collagen and myosin, while third harmonic generation microscopy contrasts optical inhomogeneities within tissues. Typical resolution is approximately 0.3–0.5 µm lateral, ∼1 µm axial). Multiphoton microscopy enables high-resolution imaging of tumor and normal tissue characteristics, allowing for the visualization of cellular morphology and microstructures. 41
Molecular Imaging of the Stromal Tumor Environment (STE) – Focus on Fluorescence
Molecular imaging techniques use probes that target specific markers within the STE to visualize proteins, receptors and metabolic processes. These methods provide valuable insights into tumor heterogeneity and help identify potential therapeutic targets. They deepen our understanding of tumor biology, angiogenesis and treatment response, thereby enhancing early detection, enabling personalized treatment and allowing real-time monitoring of therapeutic outcomes.42–44 Advances in molecular imaging and a deeper understanding of the STE hold great promise for transforming cancer diagnosis and treatment, particularly for melanoma, one of the most aggressive forms of skin cancer.45,46 Melanoma's accessibility and visible traits make it an ideal model for studying the STE's role in cancer development. 47
Molecular imaging allows for visualizing stromal remodeling, providing a powerful tool for early cancer detection (Table 3). Methodologies include MRI (visualizing tumor vasculature, stromal cells and ECM), PET (targeting CAFs, immune cells, tumor vasculature and angiogenesis), fluorescence imaging (including Intravital Microscopy, Fluorescence Lifetime Imaging Microscopy (FLIM) and probes targeting stromal elements), photoacoustic imaging (PAI; imaging tumor vasculature and angiogenesis, melanin content), Mass Spectrometry Imaging (MSI), Ultrasound and Optical Coherence Tomography (OCT).
Molecular Imaging Modalities and Biomarkers/Targets for Melanoma Stroma.

Our understanding of the STE, also known as the tumor microenvironment (TME), in cancer initiation and progression has evolved significantly. 55 Despite its critical role in cancer development, the STE is often overlooked. The STE is a complex ecosystem comprising the extracellular matrix (ECM), cancer-associated fibroblasts (CAFs), the tumor vasculature and a variety of immune cells. These shape melanoma progression, drive therapy resistance and promote the spread of cancer to distant sites.56–58 Rather than viewing cancer solely as uncontrolled cell growth, research now highlights the complex interactions between various cell types and the physicochemical components of the STE, particularly the ECM and neoangiogenesis. 55
Stromal cells, through integrin signaling and microenvironment interactions, drive key processes such as migration, survival, angiogenesis and proliferation.58,59 By fostering tumor growth, immune evasion and heterogeneity, the STE contributes to therapy resistance, making it a crucial focus in cancer research. Investigating this intricate network could enable earlier cancer detection, potentially before angiogenesis even begins.
Fluorescence imaging is a versatile technique which offers high molecular specificity when combined with targeted fluorescent probes. It also allows for real-time visualization of biological processes, making it valuable for surgical guidance. However, a key limitation of fluorescence imaging in vivo is its limited penetration depth in tissues due to light scattering and absorption (making it ideal for skin), as well as the potential for interference from autofluorescence, which is the natural fluorescence emitted by biological tissues.
A new molecular fluorescence technology for examining the STE is Skin Fluorescent Imaging (SFI), which leverages molecular imaging techniques to assess alterations in integrin expression within the STE. 17 The SFI system represents an advancement in melanoma detection, offering a noninvasive, accurate and cost-effective alternative to traditional diagnostic methods. SFI uses an amino acid target tagged with a fluorescent molecular dye – arginine-glycine-aspartic acid (RGD) – to non-invasively detect tissue remodeling in melanoma, a tumor biomarker targeting αvβ3 integrin receptor expressed on blood vessels yet absent in normal tissues. 60 Its fluorescent tag is topically applied, providing a safer and more convenient option than PET imaging, which requires injected radiotracers. One of SFI's most compelling features is its ability to deliver immediate results, reducing patient anxiety and eliminating unnecessary follow-up appointments. With 100% sensitivity and 95.7% specificity for melanoma, 17 it significantly minimizes false negatives and unnecessary biopsies, improving diagnostic accuracy. Unnecessary biopsies introduce risks, including infection, pain and scarring. Early detection capability enhances treatment outcomes and increases quality-adjusted life years (QALYs) for patients, while also lowering healthcare costs by reducing histopathological examinations and associated complications.61,62 Radiolabeled integrin-binding RGD peptides have been used for imaging solid cancers via i.v. administration 63 in melanoma, 64 breast cancer 65 and other advanced solid tumors. 66 In melanoma, αvβ3 integrin receptor expression has been associated with aggressive disease. 67
Targeted Cancer Imaging – Future Directions and Limitations
Appropriate selection of imaging modality is dependent on tumor type, patient demographics and underlying cancer biology. The most effective imaging tool is often specific to the cancer's location and characteristics; for instance, CT scans are frequently used for lung cancer, while MRI is the preferred modality for breast cancer due to its superior soft tissue contrast. 68 Beyond tumor type, patient demographics can influence modality choice and patient satisfaction. Studies have shown that satisfaction with imaging varies by age, with the highest satisfaction rates among patients aged 70–79 and the lowest among those aged 20–29. Choices can also be influenced by factors like patient age, sex, overall health and personal preference. 69
Morphology-based tools such as RCM, multiphoton microscopy and OCT achieve high sensitivity for detecting histopathological abnormalities in tissue architecture yet show low specificity, leading to false positives and unnecessary biopsies. Deeper imaging methods such as MRI, PET and ultrasound are valuable for staging or assessing lesion depth, yet they are costly and lack specificity at the level of the lesion itself (in contrast to metastases). By contrast, the SFI System leverages αvβ3 integrin as a biomarker of angiogenesis to increase specificity for biologically aggressive lesions while maintaining sensitivity for early melanoma detection. While SFI promises a superior balance of accuracy and cost-effectiveness, multi-site validation will be essential to confirm its performance across clinical contexts.
Autofluorescence, motion artifacts and probe variability impact the performance of various imaging modalities. Autofluorescence affects optical imaging techniques like RCM and OCT, where the natural fluorescence of tissue can be used as a diagnostic signal for changes in tissue composition indicative of cancer, though it can also be a source of background noise that interferes with molecular probes. 70 In deep-tissue imaging such as PET, CT and MRI, motion artifacts are a significant challenge, as involuntary movements from breathing or a heartbeat can cause image blurring, and inaccurate tumor quantification. 71 To address this, these modalities often employ motion correction techniques like respiratory and cardiac gating. Finally, probe variability is a key consideration for molecular imaging modalities like PET, which rely on injected agents to create an image. Inconsistencies in a probe's binding affinity, specificity, and clearance from non-target tissues can lead to false-positive results and limit the reliability and clinical application of the technology.
SFI offers exceptional cost-effectiveness by providing accurate, point-of-care melanoma risk assessment, potentially reducing unnecessary procedures among the >5 million skin biopsies performed annually in the U.S., most of which prove benign. Unlike high-cost modalities such as MRI and PET, or advanced optical tools like multiphoton microscopy, RCM and OCT, which require expensive equipment and specialist interpretation yet cannot significantly cut biopsy rates, SFI is low-cost and easy to use. Even ultrasound, though inexpensive, lacks the molecular specificity to meaningfully reduce unnecessary procedures.
Artificial intelligence (AI) is transforming biomedical imaging across a wide range of modalities by enhancing diagnostic accuracy, speeding up workflows and making advanced analysis more accessible. 72 From high-resolution techniques like OCT, 73 multiphoton microscopy 74 and mass spectrometry imaging 75 to widely used platforms like MRI, 76 PET, 77 ultrasound 78 and optical imaging, 79 AI enables faster reconstruction, automated lesion detection and nuanced tissue characterization. These advances not only improve clinical outcomes, such as non-invasive melanoma screening with RCM 80 or early tumor detection with PET and OCT, 73 they also expand imaging access to underserved settings through real-time guidance and mobile diagnostics. Collectively, AI accelerates the shift toward more precise, scalable and patient-centered care.
Notably, among the imaging modalities surveyed, PET 81 and MRI 82 are the most effective and widely used for detecting metastases, offering whole-body metabolic insights and high-resolution soft tissue imaging, respectively. Ultrasound, 83 PAI 84 and MSI85,86 serve more limited or emerging roles, while modalities like RCM, OCT and SFI are not suitable for metastasis detection due to their localized or surface-level focus.
Sensitivity and specificity across different imaging modalities also depend on whether cancers are in early or late stages. For whole body and deep-tissue imaging, modalities like PET and MRI can detect small, early-stage tumors and are also highly effective for late-stage cancers and metastasis detection.87,88 CT scans have low sensitivity for diagnosing early-stage colon cancer. 89 For intermediate depth imaging, ultrasound is cost-effective and portable for breast cancer detection. 90 In shallow and superficial imaging, RCM and OCT have high sensitivity for early-stage histopathological abnormalities yet are not suitable for late-stage cancers because they cannot penetrate deep into the body.91,92 Molecular imaging such as SFI, shows exceptional sensitivity and high specificity for early melanoma detection. 17
Imaging methodologies raise important ethical considerations, including informed consent, radiation exposure and incidental findings. Patients undergoing procedures such as CT and PET face a small risk of developing cancer later in life due to ionizing radiation, 93 while modalities using injected agents, like MRI and PET, carry potential side effects such as allergic reactions or toxicity in sensitive individuals. Advanced imaging also increases the likelihood of incidental findings – unexpected discoveries unrelated to the original reason for the scan – which can create ethical dilemmas about patient communication and follow-up. These issues may lead to patient anxiety, financial strain and overdiagnosis of slow-growing tumors. Ensuring patients are fully informed of the risks and benefits before procedures is therefore critical.
Conclusion
The integration of molecular imaging represents a significant advancement in the early detection and personalized treatment of cancer. These technologies provide detailed insights into tumor biology, enabling clinicians to identify early-stage cancers which might be missed by traditional methods. Various imaging modalities, including MRI, PET, optical imaging, multiphoton microscopy and ultrasound, each offer unique advantages in visualizing tumors and cellular activities. Molecular imaging, particularly techniques like SFI, offers non-invasive, accurate and real-time visualization of cancer-associated molecular changes, enhancing diagnostic precision and treatment monitoring. By focusing on the STE and leveraging specific biomarkers, these approaches improve early detection and allow for personalization of therapeutic strategies and monitoring of treatment efficacy. The continued development and clinical validation of these innovative technologies hold great promise for transforming cancer care, with the potential to improve patient outcomes and reduce healthcare costs.
Footnotes
Acknowledgements
Dr Yoav Litvin and Anne Callahan assisted in preparing and reviewing the manuscript.
Ethics Statement
N/A
Funding
Orlucent, Inc.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
