Abstract
Purpose
In our institute, stereotactic radiosurgery (SRS) and fractionated stereotactic radiotherapy (SRT) were performed by Cyberknife-S7 (CK-S7) which provided the selection of different collimators. This study aimed to compare critical plan qualities including conformality, high-dose area, dose fallout, and treatment efficiency between InCise™ multileaf collimator (MLC) based and Iris™ variable aperture collimators (Iris) based treatment plans.
Methods
Twenty-five patients with intracranial tumors treated with CK-S7 were retrospectively analyzed. The Precision 3.3.0.0 with VOLO™ optimizer and GPU-accelerated Monte Carlo dose calculation algorithm was used for treatment planning. The new conformality index (nCI), homogeneity index (HI), high-dose ratio (HDR) and distribution inside plan target volume (PTV), dose gradient distance (DGD) outside PTV, organs at risk (OARs) sparing, and treatment efficiency were compared between MLC based and Iris based plans.
Results
MLC plan achieved higher nCI, higher HDRs from 135% prescription dose (PD) to 110%PD and trended to form more centralized and gathered high-dose distribution inside PTV, while no statistical difference was found in HI. Iris possessed better dose-engraving ability around the target boundary especially when it close to OARs with strict dose constraints. MLC plan showed shorter DGD from 90%PD to 20%PD. MLC plan achieved less MUs (−67.14%), less nodes (−41.5%), less beams (−74.06%) and shorter treatment time (−51.64%). There were positive correlations between the effective radius of PTV (rPTV) and DGD from 90%PD to 10%PD both in MLC and Iris plans.
Conclusions
MLC plan achieved comparable conformality, higher HDRs, more gathered high-dose distribution, faster dose fallout and more efficient treatment which proved it an excellent SRS/SRT choice for intracranial tumors treated with CK-S7. MLC might take an important advantage for the uncompliant and painful patients. However, Iris showed a better dose-engraving ability, it might be taken into consideration especially when the tumor was close to OARs with strict dose constraints.
Keywords
Introduction
Stereotactic radiosurgery (SRS) and fractionated stereotactic radiotherapy (SRT) have become crucial treatment modalities for patients with brain metastases or benign intracranial disease according to the excellent local control and survival.1–4 The CyberKnife system (CK) (Accuray, Sunnyvale, CA, USA) is a 6-axis robot manipulator device with a linear accelerator mounted on. This system provides a steep dose gradient through noncoplanar beams of various size from 3D workspace to minimize dose to the normal brain tissue, and employs a pair of orthogonal kV x-ray imagers as target tracking system that ensures sub-millimeter targeting accuracy,5–7 which make it an excellent choice for intracranial single or multiple-targets SRS/SRT.
CK features three types of secondary collimator: Fixed collimator, Iris™ variable aperture collimator (Iris), and the InCise™ multileaf collimator (MLC) which was released in CK-M6 and later versions. MLC of the latest CK-S7 version provides a wider field size maximum to 115 mm × 100 mm and is able to form multiple irregular segments under the same beam. 8 Historically, the conformity index (CI) and dose gradient are the key components of SRS/SRT,9–11 and the treatment efficiency get more attention recently as for a wider application of the CK device. What's more, the intracranial tumor size and its location in different brain lobes also decide the beam selection and affect the plan qualities. CK-S7 with Accuray Precision Version 3.3.0.0 treatment plan system, adopts inverse VOLO™ optimizer that takes treatment time as a parameter into the cost function to provide more efficient treatment pathways, and the GPU-accelerated Monte Carlo dose calculation algorithm to provide higher quality random number sequences and larger interaction track libraries. 12 Some researches studied the dosimetric characters of MLC based on CK-M6 which adopted sequential or VOLO™ optimizer, and adopted Finite-Sized Pencil-Beam (FSPB) algorithm for dose calculation.13,14 As for the different leave width, leave number, optimizer and dose calculation algorithm, the discussion is still on-going in the literatures on the differences between MLC and Iris collimators of CK-S7 with the latest inverse VOLO™ optimizer and Monte Carlo dose calculation in terms of dosimetric characters, plan qualities and treatment efficiency. Such researches will provide broader support for the selection of collimators in the further clinical scenarios.
The inhomogeneous dose inside PTV is another specific dose distributions of SRS/SRT compared with the conventional fractionated radiotherapy.15,16 SRS/SRT may offer an additional, indirect mode of cell death which involves causing profound alterations in the tumor microenvironment by means of substantial local oncovascular damage, whereas the conventional fractionated radiotherapy is predicated by means of lethal DNA damage from double-stranded DNA breaks. 16 A high maximum dose inside the tumor is desired because it may improve local control and recurrence. 17 Nevertheless, there is no uniform limit to the maximum dose in SRS/SRT presently and the discussion of the high-dose area inside PTV for SRS/SRT is also lacking in the historical literatures. One goal of this study is to evaluate the high-dose ratio (HDR) and the high-dose distribution inside PTV in both MLC plan and Iris plan since these two parameters may own vital significances for the patient's long-term prognosis of antitumoral inflammatory response and immunotherapies.
In this study, we compared the dosimetric, geometric and delivery parameters between MLC based plan and Iris based plan of CK-S7 as for single or multiple intracranial tumors located in different brain lobes, and found the effects of tumor size to plan qualities. We also assessed the characters of HDR as well as high-dose distribution inside PTV for MLC and Iris collimators, respectively. All these MLC and Iris plans were evaluated by the experienced radiation oncologists to decide which used for clinical treatment then to suggest the selection of collimators under different clinical situations.
Methods and Materials
Cyberknife Device
The CK-S7 is a 6-axis robot manipulator device with a linear accelerator mounted on. The robotic manipulator moves within a 3D workspace which comprised of pre-assigned point-in-space, called nodes, from which the linear accelerator produces 6MV flattening filter free (FFF) photons at dose rate 1000 Mu/min at multiple beam angles. The device employs a pair of orthogonal kV x-ray imagers to localize targets during beam deliver, ensuring sub-millimeter accuracy.
Three collimator types are provided for CK-S7: (1) Fixed collimator (Fix) with 12 circular field sizes ranging from 5 mm to 60 mm in diameter at 800 mm source-axis distance (SAD). (2) Iris™ variable aperture collimators (Iris) created by two banks of six tungsten segments. The two banks are offset by 30°, resulting in a dodecahedral aperture (virtually circular) with the same sizes of Fix. 5 (3) InCise™ mutileaf collimator (MLC) with 2 banks of 26 tungsten leaves, each leaf is 3.85 mm wide at 800 mm SSD and 90 mm thick with full interdigitation and overtravel. The maximum open field size of MLC is 115 mm (leaf motion direction) × 100 mm, and the minimum field size is limited to 7.6 mm × 7.7 mm according to a ‘at least two open leaves’ setting. 5
Patient Characteristics
Twenty-five patients with single or multiple intracranial lesions treated with CK-S7 at our institute were retrospectively analyzed, all patients’ details were de-identified. The median volume of all lesions in this study was 6.05 cm3 with a range from 2.04 cm3 to 34.36 cm3, among them, 17 patients had a signal lesion with the volume range from 2.04 cm3 to 34.36 cm3, 8 patients had multiple lesions with the total volume range from 2.62 cm3 to 32.90 cm3. The locations of these lesions included the frontal lobe, the parietal lobe, the temporal lobe, the occipital lobe, close to the brain stem or optic chiasm with strict dose constraint. The prescription dose (PD) varied from 12 Gy to 30 Gy with 1 to 5 fractions. The detailed size and location of the intracranial tumors were shown in Table 1.
Prescription Dose, Fraction, the Size and Location of Each Case.

Treatment Planning
Computed tomography (CT) images of 1 mm slice thickness and magnetic resonance images of 1 mm slice thickness were co-registered for ensuring the target area. An experienced radiation oncologist contoured the gross target volume (GTV) as well as the organs at risk (OARs) following standard contouring protocols on the CT images which used for primary planning. GTV to PTV was symmetrical 1 mm. The OARs included brain stem, optical chiasm, optical nerve L&R, lens L&R, Eye L&R, as well as normal Brain.
All treatment plans were generated using Precision version 3.3.0.0 (Accuray, Sunnyvale, CA, USA). The processes of creating treatment plans and parameters comparison between Iris and MLC was shown in Figure 1. Iris and MLC plans selected ‘head_Iris-fixed’, ‘head_mlc’ as the treatment anatomy, respectively, and both plans adopted ‘Skull’ as the synchrony method and ‘Full_Path’ as the template path set. Eye L&R were set as ‘never’ which meant the beams not entering or exiting through. The radiation oncologist also contoured the organs that might move involuntarily such as the mandible, oral cavity and larynx, those organs were set as ‘beam exit only’ in order to minimize the dose uncertainty from organ moving in each intrafraction. Four shells of 2 mm, 5 mm, 8 mm, 20 mm isotropic margin from PTV were created for dose fallout. The inverse VOLO™ algorithm5,18,19 was used for beam optimization and the GPU-accelerated Monte Carlo algorithm was used for dose calculation, with a high resolution and 1% uncertainty. As for the dosimetric comparison, the same dose limit to the four shells and the same dose constraints to PTV and OARs were used in the MLC and Iris plans for the same patient. Such method minimized any subjective issues in order to reflect the differences mainly from collimator types. The dose coverage to PTV of the two compared plans were normalized that 100% prescription dose covered 98% PTV volume. The OAR dose constraints for different fractions were evaluated according to the constraints suggested in the Timmerman Sheet. 20 The reporting of this study conformed to STROBE guidelines. 21

Processes of Creating Treatment Plans and Comparisons Between Iris and MLC.
Parameters for Plan Evaluation
The impact of size of the PTV was evaluated by the effective radius of PTV (rPTV), which calculated by equation (1)
Multiple parameters were used for comparing the plan quality of MLC and Iris plans, including new conformality index (nCI), homogeneity index (HI),
18
high-dose ratio (HDR), dose gradient distance (DGD), and intermediate-dose spillage,22,23 defined as equations (2-7).


The above parameters were focused on the evaluation to the target. As for SRS/SRT, the dose fallout outside the target was also a vital issue. DGD represented the required effective distance from PD to %PD, as shown in equations (5 and 6).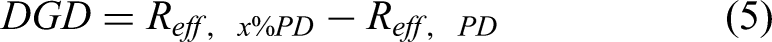

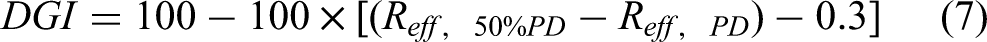
As for the treatment delivery efficiency, the numbers of MU, node, beam, the treatment time per fraction were compared between MLC and Iris plans.
Statistical Analysis
The paired t-test was used to examine the parameters between MLC and Iris plans. The linear multiple regression analysis was performed between the rPTV, nCI and other dosimetric, geometric parameters as well as treatment efficiency. The IBM SPSS Statistics 26 (IBM Corp., Armonk, N.Y., USA) was used, and the threshold for statistical significance was set to p < .05.
Results
Dosimetric Parameters
The dosimetric comparison between MLC and Iris plans in Table 2 were summarized by mean with standard deviation as well as median with range (minimal - maximum). Table 2 showed that MLC plan had a higher nCI, both for the mean and for the median. As for the upper limit for maximum dose in our institute, HI of all plans were lower than 1.4. The ratios of HI higher than 1.36, 1.37, 1.38, 1.39 were 92%, 88%, 84%, 60% for Iris plans, respectively, and 100%, 96%, 96%,76% for MLC plans, respectively. However, HI had no statistical difference between MLC and Iris plans. MLC plan achieved higher HDRs of 135%PD, 130%PD, 120%PD, and 110%PD compared to Iris plan, which reflected that MLC plan achieved more high-dose volume under the same PTV coverage.
Comparison of Dosimetric Parameters, Dose Fallout, Dose Sparing of Organs at Risk and Treatment Efficiency Between Iris-Based and MLC-Based Plans.

nCI: new conformality index, HI: homogeneity index, HDR: high-dose ratio, PD: prescription dose
DGD: dose gradient distance, PD: prescription dose, R2cm: maximum dose to any point 2 cm away from the PTV surface.
D cm3: dose to cm3 of organ at risk, D %: dose to % of the volume of organ at risk, V24: volume receiving 24Gy
Figure 2 compared the distribution of high-dose (dose more than 135%PD, 130%PD, 120%PD, and 110%PD) between MLC and Iris plans in three typical cases: (a) single target located in different brain lobes, (b) multiple targets, (c) target close to OARs. It showed that MLC trended to form more centralized and gathered high-dose distribution inside the target, while on the opposite, the high-dose distribution was more dispersive with Iris. Figure 1 (a) (b) showed that the area of 135%PD, 130%PD isodose were isolated in Iris plan. However, if the target was close to OARs with strict dose constraints, like the optical chiasm in Figure 1 (c), Iris plan achieved better high-dose distribution as it selected more nodes to meet the dose constraint of optical chiasm.

Distribution of High-dose (Purple: 135%PD Isodose Lline, Pink: 130%PD Isodose Line, Orange: 120%PD Isodose Line, and yYellow: 110% PD Iisodose Line) between Iris-based and MLC-based Plans in Three Typical Situations: (a) Single Target Located in Different Brain Lobes, (b) Multiple Targets, (c) Target Close to OARs. PD: Prescription Dose.
Dose Fallout
Table 2 illustrated the dose fallout from the boundary of PTV for the MLC and Iris plans, which were characterized by DGD and R2 cm. As for the DGD from 90%PD to 20%PD with interval 10%PD, MLC plan achieved shorter distance which meant faster dose fallout. However, DGD of PD10% had no significant difference. The DGD of 50%PD were transferred to DGI comparing with the recommends, 24 results showed that all plans satisfied the recommended guidelines. MLC plan had a lower R2cm, the difference was more than 10% in five patients with the maximum up to 29.9%.
Dose to Critical Organs
Regarding sparing of OARs, the volume-dose of brain stem, optical chiasm, optical nerve L&R of all patients were lower than the dose constraints of different fractions suggested in the Timmerman Sheet. 20 Since eye L&R were set as ‘never’ with no beam entering or exiting through, both len L&R and eye L&R achieved quite low dose as shown in Table 2. The lens L D1%,lens R D1% of one patient with tumor located in the right of frontal lobe achieved 144cGy, 163cGy, respectively, while for the other 24 patients, lens L&R D1% were all below 50cGy. And the eye L D50%, eye R D50% of all patients had a maximum of 53cGy according to beam scattering. However, Table 2 displayed no significant difference in sparing the brain stem, optical chiasm, optical nerve L&R, lens R and eye L&R between MLC and Iris plans. Previous studies had concluded that V12Gy, V24Gy of normal brain was the significant predictor of radionecrosis in SRS, SRT, respectively.25–27 As for the only one SRS patient, V12 of normal brain was 0.4 cm3 both for MLC and Iris plan; and for the other 24 SRT patients, V24 of normal brain also showed no significant difference.
Treatment Efficiency
The indicators of treatment efficiency summarized in Table 2 represented that the MLC plan achieved less MUs per fraction, less nodes, less beams as well as shorter treatment time when comparing to the Iris plan. The MLC plan decreased the average number of MUs per fraction, nodes, beams, and the treatment time by 67.14%, 41.5%, 74.06%, 51.64%, respectively.
The Effect of Target Size
Table 3 summarized the results of the linear multiple regression analysis on the effect of rPTV and nCI on parameters between MLC and Iris plans. The regression coefficient, abbreviated as r, indicated that rPTV was positively correlated with DGD from 90% to 10%, R2cm, and normal brain V24 both for MLC and Iris plans with strong influences noted for DGD from 90% to 10% (MLC r ≥ 0.708; Iris r ≥ 0.772), and for normal brain V24 (MLC r = 0.932; Iris r = 0.923). Figure 3 displayed the positively relationship between rPTV and DGD from 90% to 10%. No significant relationships were found between rPTV and HDRs in MLC plans, and also for Iris plans expect 110% isodose line (r = 0.495). There were no significant relationships between nCI and all DGDs for both MLC and Iris plans. As for nCI to HDRs of 135%PD, 130%PD, 120%PD, negative relationships were found in MLC plans while not significant in Iris plans; as for nCI to HDRs of 110%PD, positive relationships were showed with middle influences in both plans (MLC r = 0.442; Iris r = 0.402). When it considered the treatment efficiency, rPTV was not significantly related to number of nodes, beams, MUs/f, and treatment time in both MLC and Iris plans. The nCI in Iris plans showed negative relationships with number of nodes, beams, and treatment time, while in MLC plans not.

Relationship Between the Effective Radius of PTV (rPTV) and Dose Gradient Distance (DGD) from 90% to 10% in Iris-based and MLC-based Plans.
The Linear Multiple Regression Analysis of Effective Radius of PTV (rPTV) and Conformality index (nCI) on Parameters in (a) Iris-Based Plans and (b) MLC-Based Plans.

Discussion
This study compared the MLC plan and Iris plan for twenty-five patients with intracranial tumors treated by CK-S7, and also evaluated the correlation between tumor size and other dosimetric parameters in both plans. The nCIs of each patient's MLC and Iris plans were highly acceptable according to the guidelines proposed by the Radiation Therapy Oncology Group (RTOG). 10 MLC plan had a higher nCI both for the mean (Δ=0.04) and for the median(Δ=0.06) which represented larger volume of normal brain received the prescription dose. Such results were in agreement with Si Young Jang and Anna Ianiro which were both based on CK-M6,13,14 while Jang SY adopted the sequential optimizer and FSPB dose calculation, 13 and Ianiro A adopted VOLO™ optimizer and FSPB dose calculation. 14 It seemed that the MLC showed a higher nCI compared to Iris in both CK versions with different optimizers and dose calculation algorithms.
The prescription isodose around the PTV boundary was seemed like ‘dose-engraving’ which adjected the conformable extent that the collimator could provide. This adjective ‘dose-engraving ability’ was closely related to the nCI in terms of mathematical statistics quantitatively. And when it came to the isodose line, better dose-engraving ability meant Iris could make the prescription isodose line closer to the OARs with strict dose constraints when they were next to PTV. Compared with Iris collimator, the wider penumbra of MLC collimator led to a weaker dose-engraving ability around the PTV surface, especially for target with irregular boundary, or close to the OARs. In this study, two of the maximum nCIs (1.17, 1.2 in Iris plans; 1.17, 1.33 in MLC plans) both appeared when PTV was close to optical chiasm with strict dose constraint, one was shown in Figure 1 (c). No significant relationship was showed between rPTV and nCI in both MLC and Iris plans, the PTV size might not affect the dose-engraving ability with the same collimator.
Previous studies had concluded that V12Gy of normal brain was the significant predictor of radionecrosis in SRS,25,26 but few literatures had gave the predictor of radionecrosis to SRT with different fractions. Minniti 27 had concluded that V24Gy of normal brain was the most significant predictor of radionecrosis in SRT (9Gy × 3fractions), with a cumulative risk of 14% for volumes >16.8 cm3 and 4% for volumes ≤16.8 cm3, so we took V24Gy of normal brain as an indicator for SRT in this study. But no significant relationship was found between nCI and V24Gy of normal brain. The PD given by the radiation oncologist was according to the classification, grading, and staging of the tumor disease, so the PDs were not the same for different patients, which meant the ratio of 24Gy to the PD not equal. For example, if PD was 27Gy in 3 fractions, then V24Gy implied the volume of normal brain covered by 88.9%PD which was mainly decided by the dose fallout outside the target instead of nCI. Ianiro A also did not find the correlation between nCI and V12Gy, since V12Gy referred to a dose that was around 60% of their 21 Gy PD and the study considered such a low isodose would not contribute to nCI. 14 Therefore, whether a higher nCI was associated with a clinically biological difference needed to be further evaluated. However, rPTV was positively correlated to V24Gy of normal brain with strong influences (Iris r = 0.923; MLC r = 0.932), the tumor size might have more influence on V24Gy of normal brain than nCI. All MLC plans in this study achieved V24Gy lower than 16.8 cm3, only V24Gy of one Iris plan was more than 16.8 cm3 (19.78 cm3) and it was not used for that patient's treatment.
One crucial component of SRS/SRT was forming steep dose gradient in order to achieve better OARs sparing, it made the high-dose region exit inside the PTV which was different from the dose heterogeneity of conventional fractionated radiotherapy. Historically, researches mostly focused on the fractionated modes and the predictor for radionecrosis.25–28 Tateishi 17 had found that a high maximum dose within the tumor was desired because it might improve not only local control, but also cancer-specific death and overall survival rates. The high-dose distribution inside PTV of SRS/SRT might be vital not only for fully evaluating the plan quality but also for providing more detailed dosimetric parameters to the patient's long-term prognosis of antitumoral inflammatory response and immunotherapies. This study introduced the evaluation for high-dose area by comparing both the HDR and the high-dose distribution between MLC and Iris plans. Results showed that MLC plan had higher HDRs of 135%PD, 130%PD, 120%PD, and 110%PD compared to Iris plan, which reflected that MLC plan achieved more high-dose volume under the same PTV coverage.
The node and angle of beam were generated automatically by the inverse VOLO™ algorithm, the lesions in this study located in different brain lobes, single or multiple, and close to OARs which compared the high-dose distribution in more situations. Results showed that MLC trended to form more centralized and gathered high-dose distribution, while the high-dose distribution was more dispersive with Iris, as displayed in Figure 1 (a, b). D'Souza 29 had referred that a wider beam margin might produce more homogeneous dose distributions, but the research had not evaluated the concrete high-dose distribution within the target. We found that in all Iris plans, the number of beams was more than the number of nodes, which meant that there existed different beam angles in at least one node, in other words, Iris plans in this study were non-isocenter. On the contrary, all the MLC plans were isocenter, the number of beams was equal to the number of nodes in each MLC plan. The non-isocenter beam character contributed the dose-engraving ability but made it harder to form more gathered high-dose distribution. However, it might be noted when the lesion was close to brain stem or optical chiasm with strict dose constraints in SRS/SRT, such as a pituitary adenoma case as shown in Figure 1 (c), Iris achieved higher HDRs of 130%PD and 120%PD, better high-dose distribution, and better optical chiasm sparing (Iris 0.03cc = 2377cGy, 0.2c = 1797cGy; MLC 0.03cc = 2458cGy, 0.2cc = 1932cGy). As Iris selected more nodes to meet the dose constraint with less loss of PTV coverage around the optical chiasm. MLC and Iris might be compared for tumor located close to the OARs to decide which achieved the better treatment plan.
As for the dose fallout outside PTV,results represented that MLC plan achieved shorter distance for DGD from 90%PD to 20%PD (p < .01), but no significant difference was found in DGD of 10% PD. Jang SY also found MLC achieved faster dose fallout in 50% (except for irregularly shaped targets or abutting with the OARs) and 10% isodose line of maximum dose, but both differences were not statistically significant. 13 Our study also found that DGD from 90% to 10% was positively correlated to rPTV with strong influences (MLC r ≥ 0.708; Iris r ≥ 0.772), which meant the larger size of tumor, the longer distance it needed for dose fallout. According to Table 2, the mean DGD difference between Iris and MLC plans increased with lower %PD, but the difference was smaller than 1 mm for 30%PD, and for 20%PD was 1.27 mm. All mean DGD differences were in the millimeter level. MLC plan also had a lower R2cm (p < .001), the R2cm difference were more than 10% in five patients. Among them, four patients had multiple lesions, and the rest one patient had a strict dose constraint to optical chiasm. R2cm of Iris plan needed to be noted for multiple targets. Reynolds 24 had stratified 495 CK plans into different size groups and recommended the ideal and minimally acceptable DGIs for each group, results in this study confirmed that all MLC and Iris plans satisfied Reynolds's recommended guidelines.
The receiving dose of OARs all satisfied the constraints suggested by the Timmerman Sheet. 20 No significant differences were found in OARs sparing between MLC and Iris plans. The volume-dose of brain stem was related to the beam path, if one beam just traveled through it, the D 0.03 cm3, D 0.5 cm3 would be directly affected. The beam paths were selected automatically by the inverse VOLO™ optimization algorithm in Precision version 3.3.0.0, the planner could only decide to set which region as ‘never’ or ‘exit only’, meant beams could not enter and exit through, beams could not enter but could exit through, respectively. Since the brain stem anatomically located at the center of the brain, even if only ‘exit only’ was set, it would affect the selection of beam paths for the target. Therefore, the dose interventions for brainstem were achieved by function. Results showed that the doses of brain stem in 23 cases were much lower than the constraints as the far distances from tumor to brain stem. As for the two cases of tumor close to brain stem, the two collimators also achieved similar brain stem D 0.03 cm3 and D 0.5 cm3 due to the same function setting. The anatomy of the brain stem was relatively regular, both collimators met the dose-engraving near the brain stem. But as for irregularly shaped optical chiasm, Iris provided better dose-engraving. The optical nerve L&R were not near the tumor in this study, so D 0.03 cm3 of them were also related to the beam paths. Since eye L&R were set as ‘never’, both lens L&R and eye L&R achieved quite low dose.
As for the treatment efficiency, the size of PTV was not significantly related to these efficiency parameters in both MLC and Iris plans. Compared to Iris, MLC plan decreased the average number of MUs per fraction, nodes, beams, and the treatment time by 67.14%, 41.5%, 74.06%, 51.64%, respectively. Such reductions were higher than results from Ianiro A's study (MU (33.3%), nodes (21.3%), beams (46.0%), treatment time (18.4%)) which also adopted the VOLO™ optimizer but was based on the CK-M6. 14 The treatment time estimated by TPS took into account the number of nodes, the robot arm traveling time from one node to another, the number of beam angles, the image time interval (estimated as 60 s), and the beam-on time. As for the MLC plan, the number of segments and the time to adjust the leaves were also within consideration. The patient setup time was not included in the treatment time. The robot arm moving time was closely related to the number of nodes while the beam-on time was closely related to the number of MUs due to the constant dose rate, it roughly explained the decrease ratio of the treatment time was between that of MUs and nodes. The time reduction by MLC might take an important advantage especially for the uncompliant and painful patients. In this study, the mean treatment time for MLC and Iris plans were 15 min, 32 min, respectively, which might provide a referenced time for the patient's treatment. But if the tumor was close to OARs with strict dose constraints, Iris plan might also be considered due to its better dose-engraving ability although it might take longer treatment time. It might be an attempt that using MLC generally but switching to Iris near critical OARs.
There were some limitations to the study. Firstly, this study was a single-center retrospective research and was constrained by the sample size. Although the statistical analysis of 25 patients might not fully represent all clinical scenarios, it suggested a trend that the further investigation might consider. Multi-center collaboration was needed to expand the samples and to refine the clinical scenarios for establishing quantitative guidelines. Another limitation of this study was that the clinical outcome data was absent at the present stage. Especially, it would be an important direction to assess patients’ outcomes from higher HDRs and more gathered high-dose distribution provided by MLC. The patients’ prognosis in the long-term would be recorded and analyzed continuously in the future.
Conclusion
This study compared multiple critical parameters between MLC and Iris plans for patients with intracranial tumors treated with CK-S7. MLC plan achieved higher nCI, higher HDRs from 110%PD to 135%PD, more centralized and gathered high-dose distribution inside PTV, shorter distance for DGD from 90%PD to 20%PD, lower R2cm, and higher treatment efficiency. There were no significantly differences of HI and OARs sparing between MLC and Iris plans. No significant correlation was found between rPTV and nCI, treatment time in both MLC and Iris plans. The positive correlations between rPTV and DGD from 90%PD to 10%PD were found in both MLC and Iris plans. MLC might take an important advantage especially for the uncompliant and painful patients. Iris showed a better dose-engraving ability, it might be taken into consideration especially when the tumor was close to OARs with strict dose constraints to decide which collimator was better for clinical treatment. The long-term clinical outcome would be concerned closely in the further study.
Footnotes
Acknowledgments
None.
Ethics and Patient Consent Statement
This was a retrospective study based on the treatment planning system. The study was approved by the Ethics Committee of Chinese Academy of Medical Sciences, Peking Union Medical College Hospital (approval number: I-25PJ1425). The requirement for patient informed consent was waived due to the retrospective nature of the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Key R&D Program of China, Ministry of Science and Technology of the People’s Republic of China (No. 2022YFC2404606).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
