Abstract
Introduction
Gastric cancer (GC) is a highly heterogeneous malignancy, necessitating novel therapeutic targets. B7-H3 and CD39, as immune checkpoints, are potential modulators of the tumor microenvironment and may influence the efficacy of immunotherapies.
Methods
B7-H3, CD39, and CD8 expression was assessed via immunohistochemistry (IHC) in 268 GC tissues and 80 gastric precancerous lesions. The correlation between B7-H3 and CD39 expression was analyzed using Spearman's correlation. Multiplex immunohistochemistry (m-IHC) was employed to determine the co-localization of B7-H3 and CD39 in GC tissues. Kaplan-Meier survival analysis and Cox regression models were utilized to evaluate clinical outcomes in different patient subgroups.
Results
Both B7-H3 and CD39 expression showed a stepwise increase during gastric carcinogenesis including chronic superficial gastritis (CSG), chronic atrophic gastritis (CAG), low-grade intraepithelial neoplasia (LGIN), high-grade intraepithelial neoplasia (HGIN) to GC, with significantly higher expression levels in GC tissues compared to all precancerous lesions (
Conclusion
The co-localized expression of B7-H3 and CD39 in GC patients is strongly associated with poor prognosis. This dual-target expression pattern provides novel insights and a theoretical foundation for the development of dual-target immune checkpoint inhibitors as potential therapeutic strategies.
Introduction
Gastric cancer (GC) ranks as the third leading cause of cancer-related mortality worldwide, with approximately 1.5 million new cases diagnosed annually. The disease predominantly affects middle-aged and older men, especially in East Asia.1,2 Advances in tumor microenvironment (TME) research have catalyzed progress in GC immunotherapy, particularly with the development of PD-1/PD-L1 inhibitors. While these therapies show promise in improving patient survival, they are hindered by low response rates and immune-related adverse events.3,4 Consequently, alternative immunotherapeutic targets, such as B7-H3 and CD39, are gaining increased attention.
B7-H3 (CD276) is a co-stimulatory molecule characterized by low expression in normal tissues but significantly elevated levels in tumor tissues.5–7 It facilitates immune evasion by impairing cytotoxic T cells and natural killer cells, 8 and contributes to dysregulated bio-signaling and altered energy metabolism in tumor cells.9,10 B7-H3 is a promising therapeutic target, with monoclonal antibodies and CAR-T cell therapies in clinical development showing encouraging results. Notably, DS-7300 and 131I-omburtamab have demonstrated safety, with DS-7300 progressing to Phase II trials following positive Phase I outcomes.11,12
CD39, first identified in Epstein-Barr virus-infected B-lymphocytes in 1982, is another critical immune molecule in the TME. As an ectonucleoside triphosphate diphosphohydrolase, CD39 regulates the conversion of extracellular ATP (eATP) into adenosine, balancing the pro-inflammatory immune response and the immunosuppressive effects of adenosine.13,14 CD39 is frequently overexpressed in various solid tumors, with elevated levels correlating with poor prognosis and disease progression.15–17 Targeting the adenosine signaling pathway—specifically through CD39 inhibition to prevent adenosine accumulation—represents an emerging therapeutic strategy.16,18 Combining CD39 inhibitors with anti-PD1/PD-L1 therapies has demonstrated superior efficacy compared to monotherapy, although resistance to PD1 inhibitors in some patients underscores the need for innovative treatment approaches.
Despite their distinct roles in cancer progression and immune evasion, the synergistic effects of B7-H3 and CD39 in GC remain poorly understood. Our previous research identified B7-H3 as a critical prognostic marker, with elevated expression in the TME strongly associated with poorer survival outcomes.19,20 Given the pivotal roles of B7-H3 and CD39 in shaping the TME and modulating immune responses, as well as the promising potential of dual-target therapies in oncology,21,22 further investigation into their interplay in GC is warranted. To clarify the terminology used in this study, we define “both high expression” as the concurrent high expression of both B7-H3 and CD39 within the same patient tissue sample. In contrast, “co-localization” refers to the specific spatial overlap where both molecules are expressed at the exact same physical location within the tissue architecture, as determined by techniques like multiplex immunohistochemistry. This study investigates both the phenomenon of concurrent high expression of B7-H3 and CD39 at the sample level and their precise spatial co-localization at the architectural level, as these distinct aspects provide complementary insights into the roles of these molecules in GC. In this study, we assessed B7-H3 and CD39 expression at distinct development stages of gastric carcinogenesis. We further analyze the relationship between B7-H3 and CD39 expression in GC. This study aims to address this gap by analyzing the co-expression of B7-H3 and CD39 in GC, evaluating their combined prognostic significance, and exploring their potential as biomarkers to inform immunotherapeutic strategies.
Materials and Methods
Patients and Specimens
This retrospective cohort study was conducted in accordance with the REMARK guidelines for tumor marker prognostic studies. 23 We collected specimens in different pathological stages of the precancerous gastric disease acquired from Pathology of the hospital from 2016 to 2021, obtaining 20 cases of chronic superficial gastritis (CSG), chronic atrophic gastritis (CAG), low-grade intraepithelial neoplasia (LGIN), high-grade intraepithelial neoplasia (HGIN) respectively. Additoonally, a total of 268 gastric cancer samples were collected from patients diagnosed in 2011 at a medical institution. Consecutive eligible patients meeting the inclusion criteria during the study period were enrolled.
Gastroscopic biopsy specimens were obtained from all cases except GC, which were collected through surgical resection.
All tissue samples, regardless of collection method, were harvested from the gastric antrum. All diagnoses were independently confirmed by two certified pathologists. Post-surgical resection specimens were fixed in neutral-buffered formalin and subsequently embedded in paraffin for histological analysis. This retrospective study incorporated a comprehensive dataset of clinicopathological characteristics, including age, gender, tumor volume, tumor differentiation, stage, and depth (as detailed in Table 1). Follow-up data for all 268 patients were collected from medical records and family contacts. Ethical approval for the study was granted by the Clinical Research Ethics Committee of the hospital. All GC patients underwent radical gastrectomy without neoadjuvant therapy.Based on prior GC biomarker studies anticipating Hazard Ratio (HR) ≥ 1.8 for co-expression markers (α = 0.05, power = 80%), 250 cases were required. Our cohort (n = 268) exceeded this threshold. All patient data were de-identified prior to analysis to ensure complete anonymity, with no possibility of tracing any individual's identity. The inclusion and exclusion criteria were defined as follows:
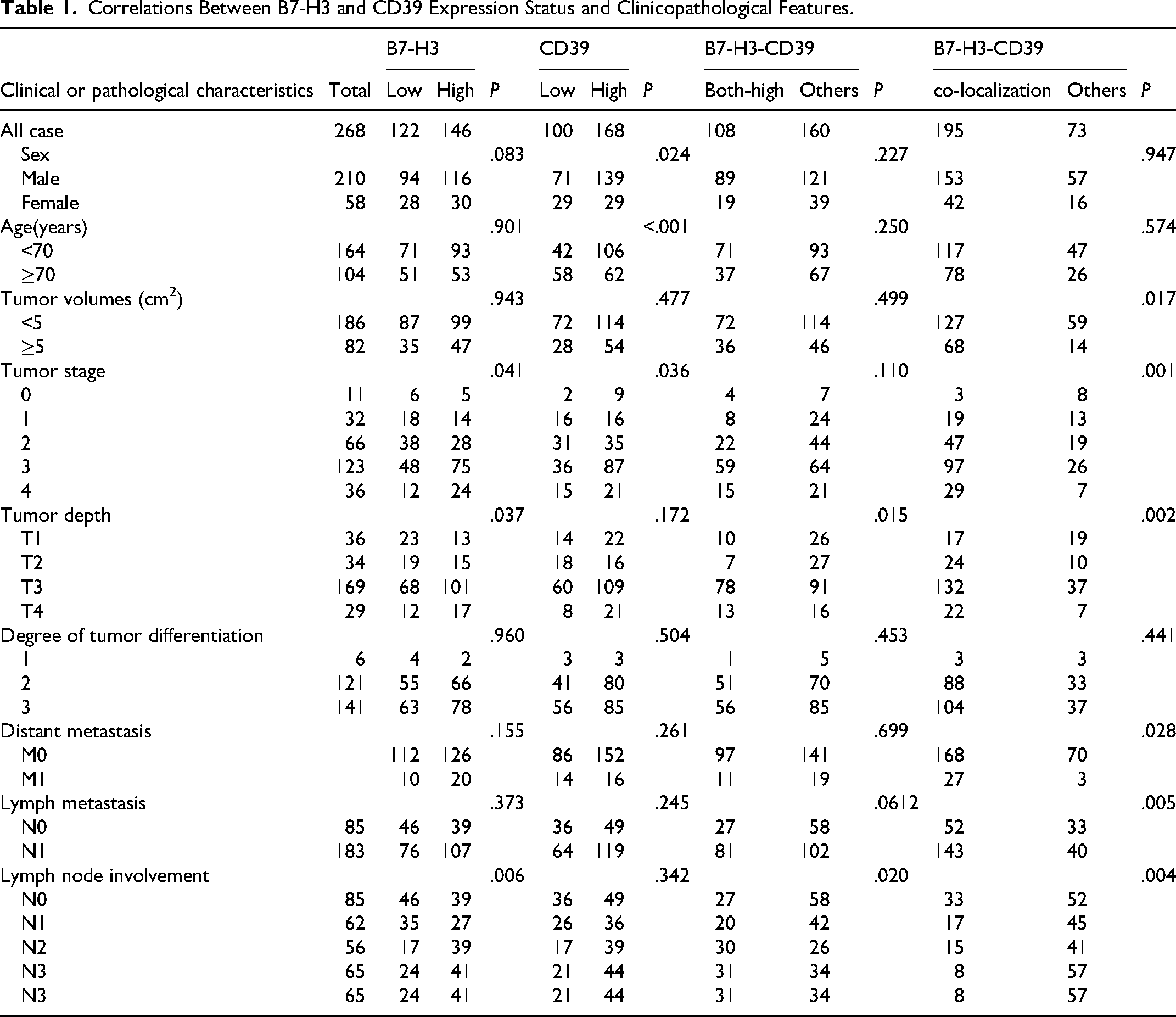
Correlations Between B7-H3 and CD39 Expression Status and Clinicopathological Features.
Inclusion Criteria
Samples meeting all of the following conditions were included:
Histopathologically confirmed gastric adenocarcinoma treated with radical gastrectomy; Tumor cellularity > 20% with preserved tissue integrity (no severe degradation or inadequate fixation) in archival FFPE blocks; Availability of complete clinicopathological data (including age, sex, tumor location, histological grade, and pTNM stage [AJCC eighth edition]); Absence of preoperative neoadjuvant therapy (chemotherapy or radiotherapy).
Exclusion Criteria
Samples were excluded if they met any of the following criteria:
Non-adenocarcinoma histology (eg, neuroendocrine carcinoma, lymphoma, or gastrointestinal stromal tumor [GIST]); Non-radical surgical procedures (palliative resection, exploratory laparotomy, or endoscopic submucosal dissection/mucosal resection [ESD/EMR] specimens); Inadequate tumor content or compromised sample quality (eg, excessive decalcification, autolysis); Concurrent active autoimmune disorders (eg, systemic lupus erythematosus, rheumatoid arthritis), HIV infection, or active syphilis; Missing essential clinical information (particularly pTNM staging data).

Tissue Microarray (TMA) Construction
Tissue Microarray (TMA) was constructed using an automated tissue array instrument. Each paraffin-embedded specimen was divided into two 1 mm cores, each containing tumor cell tissue and infiltrating immune cell tissue. Subsequently, 4-μm-thick TMA sections were created.
Immunohistochemistry and Scoring
4-μm-thick sections were cut from paraffin-embedded tissue blocks, mounted on polylysine-coated slides, and then deparaffinized and rehydrated using xylene and graded alcohol. The sections were treated by autoclaving in sodium citrate retrieval solution for 3 min. Endogenous peroxidase was blocked with 3% hydrogen peroxide for 15 min at room temperature. After washing twice in PBST, sections were blocked with 3% bovine serum albumin for 45 min, and then incubated overnight at 4 °C with primary antibodies. The following primary antibodies were used according to the manufacturer's instructions: mouse anti-human B7-H3 monoclonal antibody, 1/200 dilution (Cat No: 66 481-1-Ig, Proteintech, China), rabbit anti-human CD39 polyclonal antibody, 1/1000 dilution (Cat No: 14211-1-AP, Proteintech, China) and mouse anti-human CD8 monoclonal antibody, 1/10 000 dilution (Cat No: 66868-1-Ig, Proteintech, China). After primary antibody binding, slides were washed with PBST and then incubated with labeled polymer horseradish peroxidase rabbit/mouse antibody for 15 min (GP016129; Gene Technology, China). Immunoreactivity was detected using DAB reagent, after which the slides were counterstained with hematoxylin and sealed with neutral gum. Immunohistochemical sections were examined blindly and independently by two pathologists and scanned simultaneously by a Dmetrix imaging system (Dmetrix, Tucson, AZ, USA) with a K-Viewer imaging system (Jiang feng Bio, China). Sections were examined by staining at low magnification (×100). B7-H3 staining was scored by immunostaining intensity and ratio. Intensity was scored at high magnification (×200) and defined as follows: 0, 1+, 2+, and 3+. The percentage of B7-H3-positive cells in diseased glands or total tumor cells was estimated at four levels: 1 (<10%), 2 (10%-40%), 3 (40%-70%), and 4 (>70%). The immunoreactivity score was calculated as intensity multiplied by the percentage of stained cells. Scores of 0 were classified as no expression, 1-3 as weak, 4-7 as moderate, and 8 or more as strong. Patients with no and weak expression were categorized into the low expression group, and others into the high expression group. CD39 staining was scored by mean optical density intensity, defined as follows: 0, 1 (<10%), 2 (10%-50%), 3 (>50%). Scores of 0 were classified as no expression, 1 as weak expression, 2 as moderate expression, and 3 as high expression. Similarly, patients with no expression and weak expression were categorized into the low expression group, and others into the high expression group.
Multiple Immunohistochemistry and Scoring
Multiplex immunohistochemistry (mIHC) and antibodies specific for B7-H3, CD39, and CD8 were performed on gastric cancer TMA tissues as described above. Briefly, slides were dewaxed in xylene and then rehydrated with ethanol. Antigen retrieval was performed at boiling point in sodium citrate buffer (pH 6.0) for 15 min and endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 15 min at room temperature. Nonspecific antigen binding was blocked with goat serum solution for 30 min. Slides were incubated with the primary antibody B7-H3 (1:200, Cat No: 66481-1-Ig, Proteintech, China) overnight at 4 °C, and then horseradish peroxidase (HRP)-coupled secondary antibody was added for 30 min at room temperature. Next, slides were incubated with tyramide signal amplification (TSA) Fluorescein (Cat No: NEL701A001KT, Akoya) for 10 min at room temperature. Between subsequent staining runs, slides were microwaved to remove the Ab-TSA complex and blocked with goat serum solution, and the CD39 primary antibody was incubated between this cycle using TSA Cyanine 3 (Cat No: NEL704A001KT, Akoya) to correspond to the label. Similarly, in the last run, CD8 was visualized using TSA Cyanine 5 (Cat No: NEL705A001KT, Akoya), and finally, nuclei were visualized with DAPI (DAPI Fluoromount-G, Southern Biotech, USA). Representative images of each sample were captured and analyzed by Mantra and Inform software (Akoya Biosciences). Pathologic tissues from each patient were randomized in 4 multispectral regions (200x magnification field of view), the co-localization of B7-H3 and CD39 was quantified using a scoring system based on the percentage of double positive area, ranging from 0 to 12. A score of 0 indicates no co-localization expression, while scores of 1-4 represent low co-localization expression, 5-8 indicate moderate co-localization expression, and 8-12 denote strong co-localization expression. For the purpose of this study, moderate and strong co-localization expressions were considered as positive B7-H3 and CD39 co-localization. Additionally, CD8+ T cells, CD39+ CD8+ T cells were automatically quantified in each field of view region. The average distribution density of cell types was calculated for each patient's tissue locus. The median density of immune cells of interest in all tumors was used to classify tumors into high and low infiltration groups.
Statistical Analysis
Statistical analyses were performed using GraphPad prism 9.0 and SPSS software. Overall survival (OS) was defined as the time from the start of treatment to the date of death. It should be emphasized that OS was recorded in only 198 patients. The chi-square test was used to analyze the correlation between categorical variables. Correlations between variables were analyzed using Spearman rank correlation analysis. Survival analysis was performed using Kaplan-Meier curves, and Cox proportional risk models were used for univariate and multivariate analyses.
Results
B7-H3 and CD39 Expression at Different Pathological Stages of Precancerous Diseases of the Stomach
To investigate the expression and correlation of B7-H3 and CD39 in gastric cancer tissues, we first examined the expression levels of these two markers in gastric cancer tissues and non-neoplastic control specimens. During the progression of gastric precancerous lesions, B7-H3 and CD39 expression levels demonstrated significant dynamic changes. As shown in Figure 1A, immunohistochemical (IHC) results revealed representative expression patterns of both proteins across different pathological stages (CSG, CAG, LGIN, HGIN, and GC). Quantitative analysis indicated that the expression intensities of B7-H3 and CD39 in GC tissues were significantly higher than those in other precancerous stages (

Expression Patterns of B7-H3 and CD39 in Gastric Precancerous Lesions and Gastric Cancer Tissues. (A) Representative IHC Expression Patterns of B7-H3 and CD39 Across Different Pathological Stages (CSG, CAG, LGIN, HGIN, and GC). (B) Quantitative Analysis of Positive Expression Areas of CD39. (C) Quantitative Analysis of Positive Expression Areas of B7-H3. (D) Representative Multiplex IHC Expression Patterns of B7-H3 and CD39 Across Different Pathological Stages (CSG, CAG, LGIN, HGIN, and GC). (E) Quantitative Analysis of Multiplex IHC Expression of B7-H3 and CD39. (F) Representative Co-Localization Expression of B7-H3 and CD39 in GC. IHC Scale Bars: 50 µm; mIHC Scale Bars: 100 µm.
Co-Localized Expression of B7-H3 and CD39 in Gastric Cancer: Correlation with Clinicopathologic Factors and Prognosis
Furthermore, we analyzed B7-H3 and CD39 expression in 268 gastric cancer tissue samples using IHC (Figure 2A and B). High B7-H3 expression was observed in 146 samples, while high CD39 expression was found in 168. Spearman correlation analysis revealed a positive relationship between B7-H3 and CD39 expression in gastric cancer tissues (r = 0.2398,
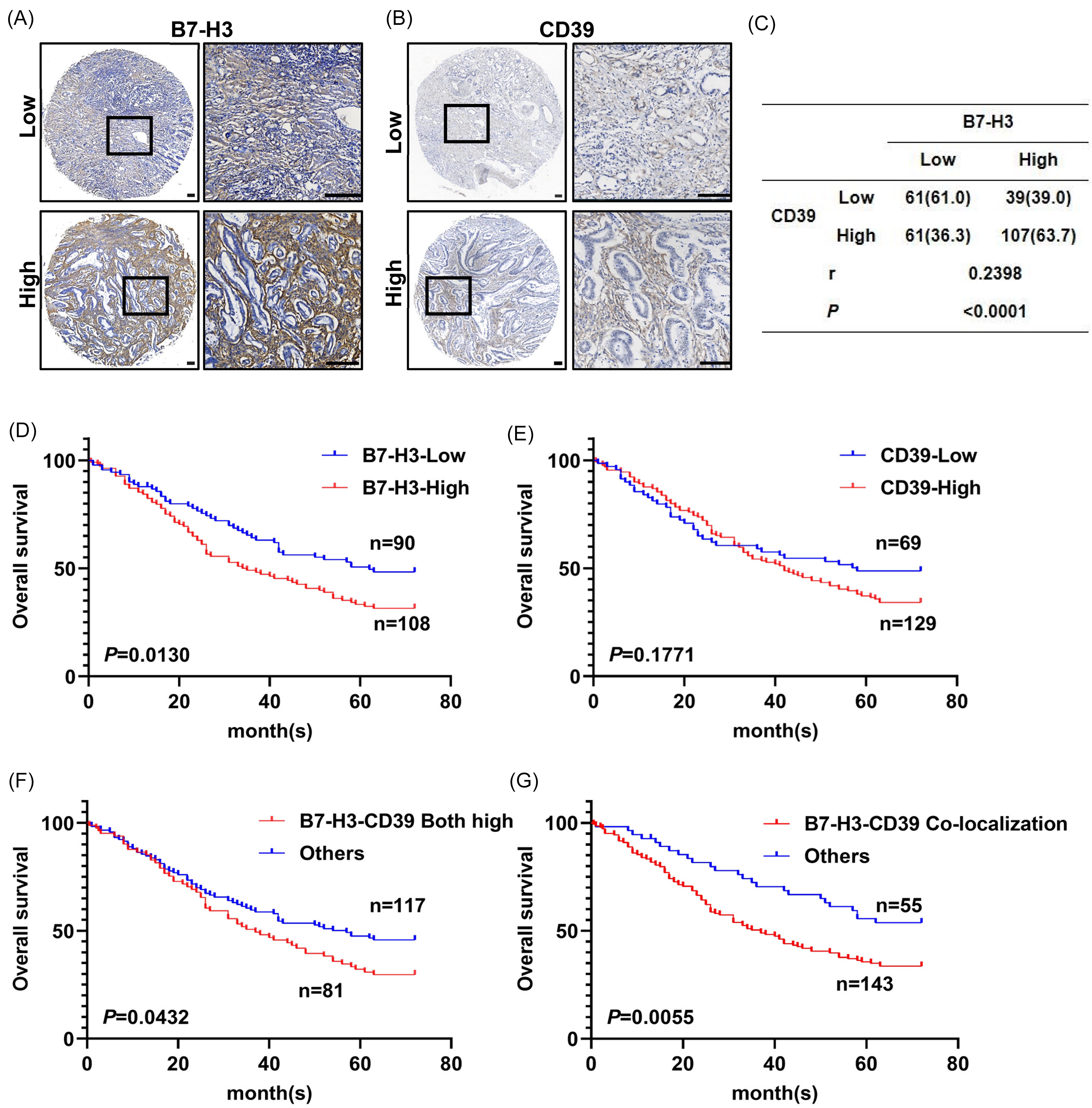
Co-Localization of B7-H3 and CD39 in Gastric Cancer Cells Indicates Poor Prognosis. (A, B) Representative Immunohistochemical Images Showing Low and High Expression of B7-H3 (A) and CD39 (B) in Gastric Cancer (GC) Specimens. (C) Correlation Analysis of B7-H3 and CD39 Expression. (F, G, H, I) Kaplan-Meier Survival Curves for Overall Survival of GC Patients Based on the Expression Status of B7-H3 (F), CD39 (G), Dual High Expression of B7-H3 and CD39 (H), and Co-Localized Expression Status of B7-H3-CD39 (I). Scale Bars: 100 µm.
Correlation analysis showed significant correlations between B7-H3 expression and tumor stage (
Prognostic analysis revealed that patients with high B7-H3 expression had significantly poorer OS compared to those with low expression (
Correlation of B7-H3 and CD39 Expression with CD8+ T Cell Infiltration in Gastric Cancer
Given the critical role of CD8+ T cells in antitumor immunity, we explored the correlation between B7-H3 and CD39 expression and CD8+ T cell infiltration. IHC revealed no significant correlation between B7-H3 expression and CD8 (r = 0.0756,

Absence of Correlation Between Co-localization of B7-H3 and CD39 Expression and CD8+ T Cell Infiltration in Gastric Cancer. (A, B) Representative Immunohistochemical Images Showing the Expression of B7-H3 with CD8 (A) or CD39 with CD8 (B) in the Same Patient. (C) Correlation Between B7-H3 Expression and CD8 Expression, and Between CD39 Expression and CD8 Expression. (D, E) Representative Multiplex Immunohistochemistry Images of B7-H3, CD39, and CD8 (D), and Correlation Analysis Between the Co-Localization Expression Score of B7-H3 and CD39 and the Extent of CD8 Infiltration (E). (F, G) Representative Multiplex Immunohistochemistry Images of CD39 and CD8, with Arrows Indicating CD39+ CD8+ T Cells (F), and the Proportion of CD39+ CD8+ T Cells within the CD8+ T Cell Population (G). Scale Bars: 100 µm.
Prognostic Significance of Co-Localized B7-H3 and CD39 Expression with CD8+ T Cell Infiltration
We analyzed the prognostic impact of B7-H3 and CD39 expression in relation to CD8+ T cells. Patients with high B7-H3 but low CD8 expression exhibited significantly reduced survival rates (

Kaplan-Meier Survival Curves for Gastric Cancer Patients Based on Specific Expression Statuses and Immune Cell Infiltration Levels. (A, B, C, D) Kaplan-Meier Survival Curves for GC Patients Stratified by B7-H3HighCD8Low (A) Expression Status 、 CD39HighCD8Low (B) Expression Status 、 B7-H3-CD39Both highCD8Low Expression Status (C) and B7-H3-CD39Co-localizationCD8low Expression Status (D). (E, F) Kaplan-Meier Survival Curves for GC Patients Based on CD8+ T Cell Infiltration Levels (E) and CD39+ CD8+ T Cell Infiltration Status (F).
Univariate Cox regression analysis identified tumor size, stage, depth, distant metastasis, high B7-H3 expression, and co-localization of B7-H3 and CD39 as significant predictors of survival (
Cox Regression Analysis of Risk Factors for Death.

Discussion
In this study, we identified a significant positive correlation between the expression of B7-H3 and CD39 in gastric cancer tissues, with notable co-localization in both tumor nests and peritumoral regions. While both molecules showed limited expression in gastric precancerous lesions, their coordinated upregulation peaked in GC tissues, suggesting a stage-specific synergy during malignant transformation. Multiplex immunohistochemistry confirmed that B7-H3 and CD39 colocalized in tumor and stromal compartments, yet exhibited distinct cellular tropism: B7-H3 was predominantly expressed in tumor and stromal cells, whereas CD39 additionally marked immune infiltrates. This tumor-enriched co-expression pattern implies a potential functional crosstalk between B7-H3 and CD39 in GC progression, challenging the conventional focus on their isolated roles in tumor biology. While previous studies have predominantly examined the individual roles of B7-H3 or CD39 in tumor biology, our findings based on data from 268 GC patients suggest that there may be a clinically relevant functional interplay between these two molecules.
CD39 primarily functions to hydrolyze Adenosine Triphosphate (ATP) into adenosine, which has dual effects: inhibiting the activity of T cells, Natural Killer (NK) cells, and dendritic cells (DCs) via A2A and A2B receptors, while simultaneously enhancing the function of regulatory T cells (Tregs), myeloid-derived suppressor cells (MDSCs), and M2 macrophages, thereby facilitating tumor immune evasion. 24 In contrast, B7-H3 suppresses T cell activity and enhances the secretion of immunosuppressive factors through interactions with unknown receptors on T or NK cells, promoting immune escape. 25 Given that patients with high co-localized expression of B7-H3 and CD39 exhibit the poorest prognosis, it is plausible that the synergistic immune evasion mechanisms mediated by these molecules play a critical role. Furthermore, multivariate analysis confirmed that co-localized high expression of B7-H3 and CD39 is an independent prognostic risk factor, supporting the potential of dual-targeted therapies against these molecules.
Beyond their immunosuppressive roles, B7-H3 and CD39 intersect in various oncogenic signaling pathways. B7-H3 activates the JAK/STAT signaling pathway, promotes epithelial-mesenchymal transition (EMT) through increased expression of matrix metalloproteinases (MMPs), and drives tumor migration, invasion, and metastasis.26,27 The activation of STAT3 further regulates cytokines such as IL-6 and TGF-β, enhancing immune evasion and malignant phenotypes.28–30 Additionally, B7-H3 contributes to metabolic reprograming by enhancing glycolytic capacity through increased Reactive Oxygen Species (ROS) production and HIF-1α stabilization. 9 CD39 expression, in turn, is regulated by stimuli including hypoxia, TGF-β, EMT, oxidative stress, and inflammatory cytokines—many of which are influenced by B7-H3.31,32 Hypoxia and TGF-β are particularly critical inducers of CD39 expression, suggesting a regulatory link between B7-H3 and CD39 signaling. Together, these molecules influence the metabolic landscape of the TME, with B7-H3 enhancing energy metabolism and ATP production during glycolysis,33,34 while CD39 directly modulates ATP depletion and adenosine accumulation.14,35 This metabolic synergy fosters a TME conducive to tumor proliferation and metastasis.
These findings underscore the therapeutic potential of targeting B7-H3 and CD39 through dual CAR-T cell therapies or bispecific antibodies. Such strategies could inhibit immune escape mechanisms, remodel the metabolic properties of the TME, and enhance the efficacy of antitumor immunity.
As key effectors in antitumor immunity, CD8+ T cells are critical targets of the immunosuppressive effects of B7-H3 and CD39. Our analysis revealed no significant correlation between B7-H3 or CD39 expression and CD8+ T cell infiltration, suggesting that these molecules may regulate T cells through mechanisms related to functional exhaustion rather than recruitment or proliferation.
Interestingly, we observed a substantial presence of CD39+ CD8+ T cells in gastric cancer tissues, particularly within the tumor stroma. While CD39+ CD8+ T cells are often considered bystanders associated with poor prognosis,36,37 our findings demonstrated that high infiltration of CD39+ CD8+ T cells correlated with improved overall survival. This aligns with studies in other cancers, such as head and neck tumors, where CD103+ CD39+ CD8+ tumor-infiltrating lymphocytes (TILs) were linked to better survival outcomes.38,39 These TILs exhibit a tissue-resident memory phenotype, enabling sustained immune surveillance and antitumor activity. This highlights the complexity of CD39's role in the TME, where its effects may vary depending on the cellular context. High expression of CD39 on CD8+ T cells may enhance immune surveillance and function rather than uniformly contributing to immune suppression. Further research is essential to delineate the specific regulatory roles of CD39 in different cell populations within the TME. This study also has several limitations. While we revealed the association between B7-H3/CD39 co-expression and poor prognosis and explored potential underlying mechanisms, direct experimental evidence elucidating the specific molecular interactions driving gastric cancer progression is lacking; moreover, the multiplex IHC approach employed cannot precisely resolve their subcellular co-localization or demonstrate direct molecular binding. As a single-center retrospective study, the generalizability of our findings requires validation in prospective multi-center cohorts. Finally, the therapeutic potential of dual targeting B7-H3 and CD39 remains to be supported by preclinical or clinical data.
Our study demonstrates the co-localized expression of B7-H3 and CD39 in gastric cancer tissues and establishes their association with poor prognosis. These findings provide a foundation for further exploration of their roles in tumor immunity and potential as therapeutic targets. Dual-targeting strategies that disrupt the interplay between B7-H3 and CD39 could reshape the TME, enhance immune recognition, and improve therapeutic outcomes in gastric cancer.
Conclusion
The present study observed that the expression of the immune-checkpoint molecules B7-H3 and CD39 increased progressively across the gastric carcinogenesis sequence, with the highest levels detected in established GC. Co-localization of these molecules was found to be associated with larger tumor volume, more advanced stage, deeper invasion, lymph-node involvement, distant metastasis, and reduced overall survival. These findings suggest this co-localization pattern may have potential as both a prognostic biomarker and a possible dual-target (B7-H3-CD39) for immunotherapy consideration. Multivariate analysis indicated this co-localization might serve as an independent prognostic biomarker. The observed lack of correlation with overall CD8+ T cell infiltration, combined with the prognostic associations, raises the possibility that this co-localization could contribute to an immunosuppressive microenvironment. Together, these results provide a basis for further investigation of dual B7-H3/CD39-targeted approaches as a potential therapeutic strategy in GC.
Footnotes
Abbreviations
Ethical Approval and Consent to Participate
This retrospective study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Soochow University (approval number: 2018121;2022030).
Informed Consent
Since this study was a retrospective study, the Ethics Committee of The First Affiliated Hospital of Soochow University has confirmed that no informed consent is required.
Author Contribution
All authors contributed to the study conception and design. Qiange Zhang and Ying Liu: Writing-original draft, Statistical analysis. Shenghua Zhan: Data curation, Resources. Cuiping Liu, Sisi Ding, Yu Shen, Lili Huang and Qi Ma: Resources, Funding acquisition. Siji Chen: Data collection. Hanqin Xuan and Ruipeng Wang: Material preparation. TingWang Jiang: Supervision. Lei Cao: Supervision, Data analysis, Writing-review & editing, Funding acquisition. All authors read and approved of the final manuscript.
Funding
This study was funded by grants from Science and Technology Project Plan of Suzhou (SKY2022129, SKY2022130), National Natural Science Foundation of China (81874163, 82270556), Suzhou Municipal Science and Technology Bureau (NO. SKJY2021086), the Project of State Key Laboratory of Radiation Medicine and Protection, Soochow University (NO. GZK1202218), Children's Hospital of Soochow University Key Research Program Incubation Project (2023ZDPY05) and Gusu Talent Project of Suzhou (GSWS2020106, GSWS2020011).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Material
All data used in this study are included in the article. Please contact the corresponding author for data requests.
