Abstract
Background
Spatially fractionated radiotherapy (SFRT) intentionally creates spatially-modulated peak-valley dose patterns for improving the tumor control or/and the sparing of organs at risk, compared to conventional radiotherapy (CONV). Evaluating non-uniform dose distributions using conventional physical dose does not fully account for the biological effectiveness of SFRT. In this study, we adopt the equivalent uniform dose (EUD) as a surrogate metric to evaluate non-uniform dose distributions.
Purpose
This work will develop a SFRT treatment planning method with EUD optimization, which is the first-of-its-kind to the best of our knowledge.
Methods
The SFRT scenario of proton GRID with uniform target dose is considered in this work for dose-only optimization (DO) and joint dose and peak-valley-dose-ratio (PVDR) optimization (JDPO) respectively. In addition to dose and PVDR optimization, SFRT treatment planning also optimizes the EUD. The EUD used in this work is based on cell survival as modeled by the linear-quadratic model. The EUD optimization problem is solved by: (1) iterative convex relaxation to decouple the nonconvex dose-volume-histogram constraint; (2) linearized alternating direction method of multiplier to efficiently handle the nonconvex minimum-monitor-unit constraint and nonlinear EUD objective.
Results
EUD optimization reduced the EUD and increased survival fraction. For example, for a head-and-neck patient, EUD optimization decreased the brainstem EUD from 5.52% to 3.86% for DO, and from 5.78% to 4.82% for JDPO, and increased the brainstem survival fraction from 72.9% to 81.6% for DO, and from 71.5% to 76.6% for JDPO. Furthermore, EUD optimization preserved PVDR from DO or JDPO.
Conclusions
A novel EUD optimization method is proposed for SFRT that can reduce the EUD and increase the survival fraction.
Keywords
Introduction
Compared to conventional radiotherapy (CONV), spatially fractionated radiation therapy (SFRT) intentionally delivers highly spatially modulated dose to the tumor target or/and the organs-at-risk (OAR), characterized by the peak-valley dose ratio (PVDR), for improving the therapeutic ratio. 1 Evaluating SFRT plan quality using physical dose, such as mean, peak or valley dose, may not fully account for its biological effectiveness.
Equivalent uniform dose (EUD) is a metric that quantifies the uniform dose distribution required to produce the same biological effect for a given nonuniform dose distribution. For example, it is shown in ref.2,3 that the EUD is a useful quantity to design corrections for various beam parameters and analyze the effects of relative biological effectiveness for different particle types and energies. In ref., 4 it is demonstrated that the EUD is a superior predictor of cell survival compared to the physical dose. This motivates the direct optimization of EUD during non-uniform dose distribution, which will be the primary objective of this work.
While the EUD optimization method developed in this work can be applicable to various SFRT scenarios, this work considers a specific SFRT scenario of proton GRID with uniform target dose (UTD) 5 to demonstrate the efficacy of EUD optimization for OAR sparing. The reasons to consider UTD are: (1) UTD is consistent with CONV and favored by preclinical evidences that the valley dose correlates the best with tumor control; 6 (2) the purpose of EUD optimization in this work is for OAR sparing, for which UTD conveniently allows the standard plan normalization to targets, so that the OAR sparing can be evaluated under the same plan normalization.
Methods
SFRT Treatment Planning with EUD Optimization
The SFRT treatment planning considered in this work is for UTD proton GRID with dose-only optimization (DO) and joint dose and PVDR optimization (JDPO) respectively. In addition to the optimization of dose and PVDR objectives, EUD is also optimized. The general form of SFRT treatment planning problem with EUD optimization is

In Equation (1),




The third term 
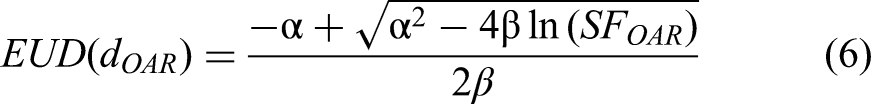
Optimization Algorithm
The optimization challenges of Equation (1) come from the nonconvexity of DVH and MMU constraints, the non-smoothness of function
We use quadratic functions for 
The active index 
With 
Using Equation (2) and (3), the function 
The optimization challenges for Equation (9) come from the L1 objectives with linear operator 
Due to the nonsmoothness of objective functions and multiple equality constraints, we use first-order optimization methods to solve Equation (11), eg, ADMM21–27 in this work.
Firstly, we define the augmented Lagrangian for Equation (11)
Then the ADMM algorithm solves Equation (11) by minimizing the augmented Lagrangian Equation (12) with respect to primal and dual variables alternatively as the follows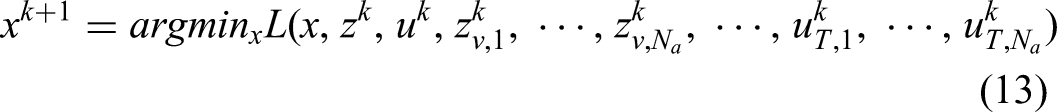

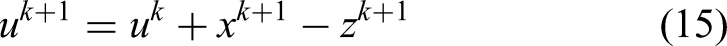


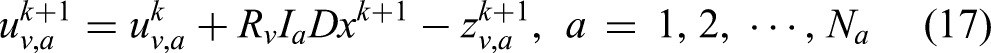



For the first subproblem,
Let
And
Using the definition of proximity operator and the first-order optimality condition, it is equivalent to the solution of the following linear system
Observing the positive definiteness of the system matrix, one can apply conjugate gradient method
29
to solve it. The detailed computation of the gradient of
The 
The 
The 
Similarly, the 
Material
The efficacy of EUD optimization is validated in comparison with CONV and SFRT (DO and JDPO respectively), by comparing the following five different scenarios: (1) conventional IMPT (CONV); (2) DO (
A generic proton pencil beam scanning (PBS) system was modeled, with energy layers ranging approximately from 70 MeV to 230 MeV, consistent with the specifications in matRad. Both treatment planning and optimization were implemented entirely within matRad, using its built-in dose calculation. The dose influence matrix
Clinically-used DVH constraints were adopted, and all plans were normalized to D95% = 100% in CTV. The conformity index (CI) was evaluated, defined as
In the following result section, the dose quantities are in percentage with respect to the prescription dose; the survival fractions are in percentage with respect to total volume of the structures under consideration; the values of PVDR and CI are unitless.
The proposed optimization algorithm is implemented in MATLAB R2023a using an in-house developed script. All experiments are performed on a desktop equipped with an Intel Core Ultra 9 processor, 64 GB RAM, and the Windows 11 (64-bit) operating system. The augmented Lagrangian parameters
Results
EUD and SF
EUD optimization reduced the EUD and increased SF for OAR. Specifically, EUD optimization decreased the brainstem EUD from 12.0% to 8.78% for DO and from 12.4% to 9.01% for JDPO in HN, reduced the esophagus EUD from 2.47% to 2.11% for DO and from 2.54% to 2.31% for JDPO in lung, reduced the rectum EUD from 4.45% to 4.16% for DO and from 4.29% to 4.09% for JDPO in prostate. EUD optimization also increased the brainstem SF from 72.9% to 82.6% for DO and from 71.5 to 81.4% for JDPO in HN, increased the esophagus SF from 60.1% to 65.2% for DO and from 59.0% to 62.3% for JDPO in lung, increased the rectum SF from 49.5% to 53.3% for DO and from 51.6% to 54.0% for JDPO in prostate.
OAR Sparing
In terms of OAR sparing, EUD optimization reduced the physical dose to OAR. For example, in HN, EUD optimization decreased the mean dose to the brainstem from 8.40% and 8.23% to 4.98% for CONV and DO respectively, and from 8.76% to 5.17% for JDPO. In lung, the mean dose to the esophagus was reduced from 6.83% and 8.27% to 4.29% for CONV and DO respectively, and from 7.73% to 5.96% for JDPO. For prostate, the mean dose to the rectum decreased from 20.5% to 18.7% for both CONV and DO, and from 20.7% to 19.2% for JDPO. These improvements are also demonstrated through the dose plots (a-e) and the DVH plot (f) and Figure 1(h) at 135°, Figure 2(h) at 240°, and Figure 3(h) at 90°.



PVDR
EUD optimization generally preserved PVDR for DO or JDPO, by comparing DO and DO-EUD or comparing JDPO and JDPO-EUD in Table 1. The PVDR was also compared by the BEV dose profiles (g) in Figures 1-3.
Plan Parameters.

The Dosimetric Quantities from top to Bottom: Max Dose for the Target Dmax, Conformity Index (CI), Mean Body Dose Dmean−Body, Mean OAR Dose Dmean−OAR, OAR Equivalent Uniform Dose EUDOAR, OAR Survival Fraction SFOAR, and PVDR per Field. Optimization Time (Time) is in Seconds. OAR Refers to Brainstem, Esophagus, and rectum for HN, Lung, and Prostate Cases, Respectively.
Discussion
In this work, we have developed a novel EUD optimization method for SFRT treatment planning, with feasibility demonstrated for UTD proton GRID. To address the unique optimization challenges from EUD optimization, we have developed an efficient optimization algorithm via linearized ADMM.
The results demonstrate that the proposed method effectively reduces the EUD of the OARs in both the DO and JPDO models, leading to a higher survival fraction. However, incorporating the EUD objective slightly compromises target coverage, as indicated by an increased maximum target dose and a decreased CI across all cases. Additional parameters presented in Table A1 (Appendix), including the EUD and tumor control probability (TCP) for the target, also show a modest degradation under EUD optimization. Nevertheless, the extent of this degradation is limited. However, EUD optimization results in lower predicted toxicities. As shown in Table A1, we report dosimetric metrics such as Vx−OAR (the percentage or volume of an OAR receiving at least x Gy), and D1cc−OAR (the minimum dose delivered to the hottest 1 cm³ of tissue) which serves as an important surrogate for toxicity risk in serial organs. From the results, we see that EUD optimization led to notable reductions in D1cc for critical structures, such as the brainstem and esophagus. These reductions may have meaningful biological implications, including a reduced risk of radiation-induced complications, such as necrosis or neuropathy, acute esophagitis,33,34 etc Regarding PVDR, EUD optimization generally yields lower values in the DO and JPDO models. However, this reduction is not substantial, and in certain instances, such as PVDR₁₃₅° for HN in the DO-EUD and JPDO-EUD models, the PVDR is even higher.
Note that the joint optimization of dose, PVDR, and EUD objectives is multi-criteria and may pose optimization tradeoffs.
35
In this study, we posed the uplimit in max target dose to be 120% of the prescription dose. However, while violating this uplimit, EUD and SF can be further improved by increasing the EUD regularization parameter
In this work, we use EUD as a metric and optimize it to achieve better OAR sparing. However, the traditional EUD model faces significant limitations when applied to SFRT. EUD assumes a homogeneous dose-response relationship across the tumor and normal tissues, which does not align with the highly heterogeneous dose distributions characteristic of SFRT. This model also neglects spatial dose modulation effects and fails to account for biological phenomena such as the bystander effect,36–38 and immune activation, 39 all of which are prominent in SFRT. Consequently, EUD may underestimate or misrepresent the therapeutic potential and toxicity risks in SFRT, limiting its reliability for outcome prediction and treatment optimization in this context. It is important to emphasize that this study serves as a preliminary effort toward establishing an optimization framework that is extensible to more sophisticated radiobiological objectives. For any specific biological model—whether described by partial or ordinary differential equations (PDE/ODE), or explicitly dependent on dose—our framework remains applicable, provided a mathematical relationship between the model and beam intensity can be established. In such cases, the proposed linearized ADMM algorithm can still be utilized. The primary technical concern lies in the convergence of the method. Although a rigorous convergence analysis is not provided in this work, a degree of smoothness with respect to the beam intensity is generally required for the biological model. When the objective function is nonsmooth or exhibits an unbounded Lipschitz continuous gradient, the algorithm may experience numerical instability. To mitigate this issue, advanced strategies such as Bregman iteration 40 or Moreau smoothing 41 can be employed to enhance stability and facilitate convergence. In summary, this work opens up a number of future research directions in biologically informed radiotherapy optimization.
In this work, we have only considered the EUD minimization for the purpose of OAR sparing. However, the same methodology can be adapted to maximize the EUD for targets with a heterogeneous dose distribution, such as LATTICE42,43 and modulated-target-dose GRID,44–47 for which the sign of the regularization parameter
The developed linearized ADMM can be generally applicable to other RT problems with nonlinear terms, such as energy layer optimization,48–52 normal tissue complication probability optimization, 53 and biological dose optimization. 54 The potential benefit of linearized ADMM is to improve both solution accuracy and computational efficiency. In contrast, proximal gradient descent (PGD) method28,55,56 can be easily trapped in local minimums.
All in all, the proposed EUD-based optimization framework demonstrates strong potential for future clinical translation in SFRT planning with proton GRID delivery. By incorporating EUD minimization, the method enables improved sparing of organs at risk while maintaining clinically acceptable target coverage. While EUD may not fully capture the spatial dose heterogeneity inherent in SFRT, it serves as a practical and computationally tractable surrogate for biological effect modulation. The observed reductions in dosimetric metrics indicate possible clinical advantages in mitigating normal tissue toxicity. Moreover, the computational efficiency of the linearized ADMM algorithm enhances the feasibility of integrating this approach into existing treatment planning workflows. With its flexibility to accommodate more advanced radiobiological models, the framework provides a promising foundation for the development of biologically informed radiotherapy planning strategies.
Conclusion
This work has developed a novel SFRT treatment planning method with EUD optimization and demonstrated the efficacy of EUD optimization for reducing EUD and increasing SF to improve OAR sparing.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251380609 - Supplemental material for Equivalent-Uniform-Dose Optimization for Spatially Fractionated Radiation Therapy
Supplemental material, sj-docx-1-tct-10.1177_15330338251380609 for Equivalent-Uniform-Dose Optimization for Spatially Fractionated Radiation Therapy by Ya-Nan Zhu, PhD, Weijie Zhang, PhD, Yuting Lin, PhD, and Hao Gao, PhD in Technology in Cancer Research & Treatment
Footnotes
Acknowledgment
The authors are very thankful to the valuable comments from reviewers. This research is partially supported by the NIH grants No. R37CA250921, R01CA261964.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the National Institutes of Health, (grant number R37CA250921, R01CA261964).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Appendix
Additional Plan Parameters.

| CONV | DO | DO-EUD | JDPO | JDPO-EUD | ||
|---|---|---|---|---|---|---|
| HN | EUD (Gy) | 2.03 | 2.04 | 1.96 | 2.06 | 1.97 |
| TCP(%) | 80.7 | 77.2 | 74.6 | 81.0 | 73.1 | |
| Dmax−OAR (%) | 80.7 | 77.2 | 74.6 | 81.0 | 73.1 | |
| V20−OAR (%) | 7.90 | 7.65 | 4.13 | 8.51 | 4.13 | |
| D1cc−OAR (%) | 48.3 | 44.7 | 31.0 | 45.6 | 32.6 | |
| Lung | EUD (Gy) | 2.05 | 2.08 | 2.04 | 2.09 | 2.10 |
| TCP(%) | 98.7 | 98.8 | 98.6 | 98.8 | 98.8 | |
| Dmax−OAR (%) | 44.6 | 48.5 | 35.7 | 39.6 | 37.1 | |
| V20−OAR (%) | 0.30 | 2.66 | 0.10 | 0.25 | 0.15 | |
| D1cc−OAR (%) | 24.7 | 35.6 | 21.4 | 28.4 | 23.7 | |
| Prostate | EUD (Gy) | 1.83 | 1.83 | 1.84 | 1.78 | 1.75 |
| TCP(%) | 72.7 | 72.6 | 73.2 | 69.3 | 66.9 | |
| Dmax−OAR (%) | 101.5 | 102.5 | 104.0 | 104.0 | 105.1 | |
| V43−OAR (%) | 3.29 | 3.35 | 2.75 | 2.86 | 2.81 | |
| D1cc−OAR (%) | 99.2 | 99.0 | 99.6 | 99.5 | 99.5 | |
The Dosimetric Quantities from top to Bottom: EUD and Tumor Control Probability (TCP) for the Target, max OAR Dose Dmax−OAR, the Percentage (or Volume) of a Specified Organ That Receives at Least x Gy of Radiation Dose Vx−OAR, minimum Dose Delivered to the Hottest 1 cm³ of Tissue (D1cc−OAR). OAR Refers to Brainstem, Esophagus, and rectum for HN, Lung, and Prostate Cases, Respectively.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
