Abstract
Traditionally, radiotherapy in metastatic renal cell carcinoma (RCC) was applied for the palliation of symptoms and the control of critical sites such as bone lesions and brain metastases. However, technological advances in radiotherapy (RT) planning and delivery made it possible to precisely deliver high dose radiotherapy at a confined target volume. One such approach is stereotactic body radiotherapy (SBRT). In patients presenting with limited metastatic burden, known as oligometastatic RCC (omRCC), SBRT can be of interest to maintain local control, in order to defer the onset of either a first or a subsequent line of systemic treatment. In this narrative review, we summarize the prospective and retrospective evidence on SBRT as a treatment option for omRCC, and give insight into the future perspectives.
Keywords
Introduction
Renal cell carcinoma (RCC) is the most common type of kidney cancer in adults, and the 14th most common type of cancer, representing around 2% of all cancer.1–3 Up to 30% of patients diagnosed with RCC present with metastatic disease.4,5 Approximately 15–25% of non-metastatic patients will eventually develop metastases.6,7 RCC most frequently metastasizes to lung, followed by bone, liver and brain. 8 Of all bone sites, the spine is most commonly involved. 9 Due to the availability of enhanced therapeutic options, the prognosis for these patients has improved over time. 10 Currently, the standard of care (SOC) treatment for metastatic RCC (mRCC) is based on systemic treatment (ST) more specifically: immune checkpoint inhibitors (ICI) and/or targeted therapy (TT).11–13 Furthermore patients with low-volume metastatic disease with a large primary tumor, or patients experiencing (near) complete response (CR) after upfront ST, might benefit from cytoreductive nephrectomy. 14 However, CR remains low (10%–15%) and currently this usually results in resistance to ST with only a minority of patients receiving long-term control with ICI.15,16 Radiotherapy can be used for the palliation of symptoms and disease control in critical sites such as bone lesions or brain metastases, and in combination with surgery for brain metastases, bone metastases or malignant spinal cord compression.17,18 Stereotactic body radiotherapy (SBRT), a method of external beam radiotherapy that precisely delivers a high dose of irradiation in one or few fractions to an extracranial target, might also be of use. 19 This in order to maximize local control (LC) of disease in the case of oligometastatic disease, as an addition to other treatment modalities, or to create a synergistic effect between them to increase response. SBRT works not only by directly damaging DNA, but also indirectly kills tumor cells by increasing hypoxia and causing vascular damage. 20 It can be delivered using a variety of advanced technologies, including volumetric modulated arc therapy, CyberKnife and tomotherapy. 21 Notably SBRT is already a well-established, safe, and effective treatment modality for primary RCC, as its radioresistance can be overcome by the high doses per fraction used in SBRT.22,23
The oligometastatic state of disease is defined as an intermediate stage between local and widespread metastatic disease. 24 In the absence of available biomarkers, it is typically defined in a rather practical manner, as the number of metastatic lesions visible on imaging limited up to 3–5 lesions. 25 The prospective evidence of SBRT as metastasis directed therapy (MDT) in this setting is limited. In standard practice, patients move from one ST to the next receiving on average 3–4 lines of therapy in total. 26 ST is frequently associated with significant toxicity resulting in worse quality of life. 26 Also by postponing the onset of ST or extending the duration of the current ST line and altering the course of the disease through elimination of resistant metastases, SBRT has the potential to improve survival outcomes. 27 The National Comprehensive Cancer Network guidelines endorse the use of SBRT for oligometastatic RCC (omRCC), while the European Association of Urology guidelines only recommend SBRT for clinically significant disease in order to achieve better LC or palliate symptoms.28,29 The European Society for Medical Oncology guidelines state one can consider SBRT for selected patients with low metastatic burden after multidisciplinary board review. 17 The purpose of this narrative review is to provide an overview of current evidence for SBRT in omRCC and provide an insight in future perspectives based on the ongoing clinical trials.
Defining Playground Rules
omRCC can present in a rather indolent manner, or can be a brief deceptive snapshot of rapidly progressive disease. The limited data on SBRT for omRCC together with the merging of different clinical situations into the same trial thereby blurring the outcome analyses, make it difficult to build up a sound base of evidence, and to clearly define which patients will benefit from a MDT approach. Besides different SBRT settings along with different treatment interests, study populations might be even more heterogeneous, as for example, ST is not always interrupted at the time SBRT is delivered, and treatment schedules of radiation may differ among physicians and for different organs treated. When interpreting data and considering SBRT for treating omRCC patients, a few basic rules need to be considered.
Various Disease Settings
When evaluating current evidence, a distinction should be made between different clinical scenarios. Most likely the different oligometastatic states of disease, specified by Guckenberger et al, implicate varying biological disease, and thus hold varying purposes for SBRT. 25 This results in different needs for the evaluation of clinical outcomes. Oligometastasis can occur in the absence of ongoing ST, whether at time of initial diagnosis of the primary tumor (“upfront” or “synchronous oligometastatic disease”), or after the initial primary diagnosis, called metachronous oligometastatic disease (Figure 1). Furthermore, oligometastatic lesions can present as residual tumor burden after a partial response to ST (persistent oligometastatic disease or “oligopersistence”), or as progression of a limited number of metastases on a background of otherwise stable disease during ST (also called “oligoprogression”). In other cases, oligometastatic disease and the need for SBRT may present when cessation of ST is prompted by side effects, such as persisting auto-immune disorders. SBRT can thus be interesting in various ways. It can thus be a part of a surveillance strategy in order to defer the start of ST, which may be ideally suited for patients expected to progress slow, with potential contraindications for ST, non-clear cell histology, as the efficacy of ST is generally lower compared to ccRCC, or patient preference.30,31 However it can also be applied in a synergistic combination with ST, to prolong the effective duration of the ST. 31 In case of progression under ST, SBRT is used to postpone the switch to subsequent line. This strategy has been applied in other urological cancers marking MDT as a promising strategy, capable of achieving sustained responses. 32
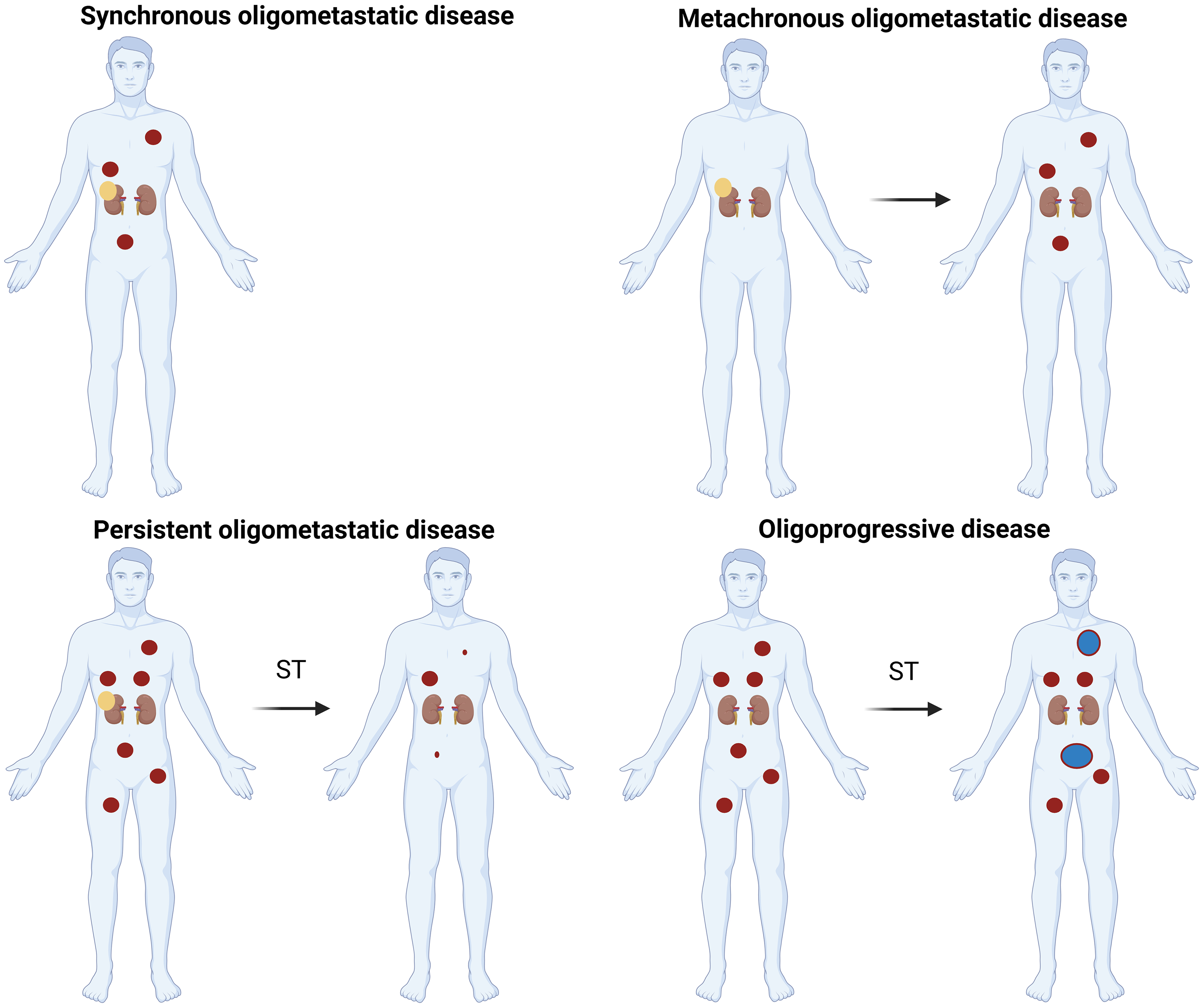
The Different Oligometastatic Settings Discussed. ST: Systemic Treatment, Yellow Dot: Primary Tumor, Red Dot: Metastasis, Blue Dot: Oligoprogressive Metastasis. Created in BioRender. Berghen, C. (2025) https://BioRender.com/wbyynx1.
Radiation Dose and Fractionation
SBRT can be delivered in different fraction schemes, ranging from single fraction to 10 fractions, with a variety in total dose, depending on the tolerance of the surrounding healthy tissues and preferences of the physicians. Although the optimal dose for maximizing tumor control (while minimizing toxicity) is not yet known, it appears to be important to deliver sufficient high dose to the target volume33–36 The comparison of those different doses is expressed by the biological equivalent dose (BED). The sensitivity of the renal cancer cells to the radiotherapy is measured by the alfa/beta value of the tissue. The alfa/beta value described in literature to calculate this BED however ranges vastly from 2.63 up to 10 with values around 3 being used most often.35,37–41 In the majority of the papers a BED > 100Gy is reported in order to overcome radioresistance and increase CR.35,42,43 A cumulative BED exceeding 100Gy correlates with a better two-year LC compared to a BED < 100Gy (96.2 vs 90.2%). 35 Some anatomical sites of metastasis are likely to benefit less from SBRT, particularly if close to critical surrounding normal tissues which limit the dose of radiotherapy. For example, the spinal site has been suggested to be associated with higher local failures. 44
Treating omRCC Lesions: All or Nothing?
The objective of local irradiation to create regression of metastatic cancer at distance from the irradiation site, is called the abscopal effect. It was firstly described in 1953 by Mole et al. 45 Multiple mechanisms have been proposed to explain this effect, such as the systemic secretion of specific cytokines and chemokines, a systemic immune response against the release of local tumor antigens, or local inflammation that can lead to a distant effect.46,47 With the advent of ICI in clinical practice for mRCC, new opportunities arose. 13 However, adding subablative SBRT to a limited number of metastatic lesions in patients treated with ICI monotherapy failed to demonstrate an improvement in PFS or overall survival (OS). 48 The phase II NIVES trial investigated the combination of ICI (nivolumab) and SBRT to a single metastatic lesion in polymetastatic progressive mRCC patients and failed to demonstrate additional benefit of SBRT, with an overall response rate (ORR) of only 17%. 13 Likewise the RADVAX RCC phase II trial assessed the outcomes of combined SBRT to one or two metastatic sites together with nivolumab plus ipilimumab. 49 The median PFS was 8.2 months (95% CI 4.6-18) with no significant difference compared to the median PFS of the CheckMate-214 trial. 50 Liu et al showed that radical SBRT to all lesions had a significant longer PFS compared to those with incomplete or no SBRT (26 vs 18.8 months, p = 0.043). 51 Even though ICs and SBRT may have synergistic local effects, data from these trials suggest that the abscopal effect may be relatively rare or non existing and targeting only few metastatic lesions instead of all oligometastatic and/or progressive lesions may not be the way to move forward. 31
Current Evidence for the Treatment of omRCC with SBRT
In a meta-analysis (‘SABR ORCA’) by Zaorsky et al in 2019, it was demonstrated that SBRT for omRCC was safe and resulted in a high LC of 89% at 1 year (95% CI 83.6-93.7). 43 In this landmark paper, 1 prospective trial and 27 retrospective studies were described, encompassing a total of 1602 patients and 3892 lesions (of which 1159 extracranial lesions). The 1-year survival rate was 86.8% (95% CI 62-99.8). Since this landmark paper, multiple retrospective and prospective studies on extracranial omRCC have been published. We will discuss these trials within the different settings (metachronous, synchronous omRCC and oligoprogressive mRCC) below. Regardless of the strong findings of these studies, one should take into account some general limitations: prospective trials mostly have a single-arm non-comparative design with short term follow-up. Both prospective and retrospective trials have considerably low patient numbers included, with only 1 retrospective trial evaluating more than 100 patients since 2019. Most of the trials had clear cell (cc) histology, favorable or intermediate IMDC scores, and one to three lesions treated. 52 The findings of the trials discussed below are summarized in Table 1.
Summary of Discussed Literature and Results of SBRT for omRCC.

Legend: P prospective cohort, R retrospective cohort, M metachronous, S synchronous, omRCC oligometastatic renal cell carcinoma, CI confidence interval, OPD oligoprogressive disease, NA not available, VMAT volumetric-modulated arc therapy, IMRT intensity modulated radiotherapy, CBCT conebeam computed tomography.
Metachronous and Synchronous Oligometastatic Disease
Multiple prospective trials and retrospective trials have been published since 2019 on metachronous and synchronous omRCC.33,35,37,38,51–53,56–64 Prospective trials were non-randomized, single arm trials and 1 prospective registry was a basket trial with different pathologies (10% RCC).33,57,59,64 No clear distinction has been made between synchronous and metachronous disease setting in all these prospective trials, however patients were treatment-naive or had received up to 2 previous lines of therapy which was stopped at least one month before trial eligibility in 1 trial.33,57,59
Hannan et al reported a one-year freedom from ST (FFST) probability of 91.3% (95% CI 69.5-97.8) in their phase II prospective trial, including 23 treatment-naïve omRCC patients with a maximum of 3 extracranial metastasis. 57 For a median follow-up of 21.7 months, they showed a 1-year PFS of 82.6% (95% CI 60.1-93.1) and 100% LC. Patients could receive repeated SBRT when feasible. 70% of their population presented with metachronous omRCC, with a median of 32.3 months between treatment of the primary tumor and SBRT for oligoprogression. The median time to start ST was 26.6 months (IQR 16.3-30.3). In another trial by Tang et al, a phase II single-arm feasibility study, 30 patients were included with up to 5 metastatic lesions. 33 Patients received SBRT (or hypofractionated doses if SBRT was not feasible) to all progressive lesions, while maintaining patients off ST. For a median follow-up of 17.5 months, they reported a median PFS of 22.7 months, and 1-year PFS of 64% (95% CI 69.5-97.8). 1-year ST free survival (STFS) was 82% (95% CI 70-98), while the 1-year adjusted STFS was 86% (95% CI 73-99). 33 Sequential radiotherapy may facilitate the deferral of initiation of ST and may allow for sustained ST breaks for select patients with omRCC. 33
In a prospective phase II trial, Siva et al (RAPPORT) reported on 30 patients, receiving SBRT for a maximum of 5 oligometastatic lesions per patient, in a population that had received up to a maximum of 2 ST lines, followed by eight cycles of pembrolizumab (200 mg, every 3 weeks). SBRT was delivered to all lesions suitable for SBRT. Whenever SBRT was not technically feasible, conventional radiotherapy was applied. 77% of patients were ST-naïve. For a median follow-up of 28 months, the ORR was 63% (95% CI 44-80), 1-year and 2-year PFS were 60% (95% CI 40-75) and 45% (95% CI 27-62) respectively and 1-year and 2-year OS was 90% (95% CI 72-97) and 74% (95% CI 53-87) respectively. 59 In a single-center retrospective study including 50 patients, Franzese et al evaluated SBRT in ST-naïve oligorecurrent RCC. 61 They found a median FFST of 63.8 months and a median distant metastasis-free survival of 17.9 months. Multiple metastasis were a negative predictive factor. 61 In clinical practice, when onset of ST can be postponed, a controlled imaging after a short period of 3–4 months can be performed to confirm the more indolent nature of the disease and real oligometastatic setting, although there is few evidence to support this practice. Besides metachronous disease, also presence of solitary lesions and non-bone locations had a positive impact on FFST. 56
Although not always clearly separated in trials, metachronous and synchronous metastatic populations are likely to present clinically with a different biology. Zhang et al reported in their retrospective study a clear difference between the metachronous and synchronous metastatic population with a median FFST of 25.5 months versus 5.6 months respectively (HR = 2.67). 53 In another large database including 63 patients with omRCC, the metachronous population was also associated with a longer FFST after SBRT, although it seems that also synchronous metastatic patients can benefit from SBRT in early setting. 63
Onal et al published a retrospective multicenter study exploring the need for next line ST (NEST) after SBRT. With the allowance of repeat SBRT, a median PFS of 18.3 months (95% CI 14.2-27.4), a one-year PFS of 64.9% and a median NEST-change of 15.2 months were reported. Two out of 17 switches in ST were due to toxicity on the previous line. A lower BED (<100Gy) was shown to have an impact on PFS but no impact yet on OS. Besides BED, also the number of fractions given, had a significant impact on OS favoring a low amount in a multivariate analysis by Onal et al. 35 The same author found, in a retrospective study with 42 patients, that the timing between start of TT (tyrosine kinase inhibitor, TKI) and SBRT in upfront oligometastatic patients, as well as the achievement of CR after SBRT was a prognostic factor for the median OS and PFS. After a median time of 18.2 months, 71.4% of the patients were still on the same line of TKI. 60 In a retrospective single-center study including 35 patients on ST, an univariate analysis showed a benefit in OS in patient receiving SBRT before TKI-failure (p = 0.006). Likewise a benefit in OS was found in patients with a short time interval (<6 months) between diagnosis of metastasis and start of radiotherapy (p = 0.046). 58
Oligoprogressive Disease (OPD)
Since 2019, 2 prospective trials have investigated the use of SBRT in the oligoprogressive setting for omRCC.27,55 None of the trials were comparative, being single-arm phase I/II trials encompassing 57 patients altogether.
Hannan et al delivered SBRT to a maximum of 3 oligoprogressive lesions in patients currently receiving firstto fourth line ST. 27 For a median follow-up of 10.4 months, SBRT extended the ongoing ST line with more than 6 months in 70% of patients (95%CI 49.9-91.1), with a median time to NEST of 11 months. The median duration of SBRT aided ST was 24.4 months. The duration of subsequent ST lines did not seem to differ from historical data, insinuating that SBRT does not undermine the benefit of subsequent lines, and may ultimately translate into a better OS. 27 Cheung et al reported on 37 patients with up to 5 oligoprogressive lesions, who had previous stability or response after at least three months of TKI therapy. 55 Cumulative incidence of changing ST was 47% (95% CI 32-68) at 1 year, the median time to change ST was 12.6 months. Their median PFS was 9.3 months, and they reported a 1-year OS of 92% (95% CI 82-100).
Multiple retrospective trials have been published since the landmark paper of Zaorsky, which are De et al, Gebbia et al, Schoenhals et al, Franzese et al, Franzese et al, Marvaso et al and Buti et al12,37,38,43,52,54,56,65 The latter of these trials reported on both oligorecurrent and oligoprogressive RCC. Patients on ICI experienced longer time to start NEST (9.2 months vs >28.4 months) in the series of Schoenhals. 12 The median time on ST before presenting oligoprogression seems predictive for the time to NEST after SBRT for OPD.54,55 In multivariate analysis, PFS was significantly correlated with both the number and size (diameter) of metastatic lesions.37,52 Sites of metastatic disease may inform about the aggressiveness of the cancer. Tumors that metastasize to the bone, liver, and brain have been associated with worse OS. 12 Oligoprogressive omRCC patients treated with SBRT while on an ICI-containing regimen appeared to have better PFS than those receiving either vascular endothelial growth factor or mammalian target of rapamycin (mTOR) inhibitors. Besides an improved response from ICI compared to older ST strategies, these patients may have received additional synergistic benefits from SBRT's antigen presenting properties and immune cell recruitment. 12
Toxicity and Combination with ST
The toxicity profile of SBRT is highly favorable. In the landmark paper of Zaorsky et al, the percentage of common terminology criteria for adverse events grade 3–4 caused by SBRT was 0.7% (95%CI 0-2.1). 43 The research that has been reported ever since, shows similar results. Tang et al delivered SBRT without ST, with 10% of patients showing grade 3–4 toxicity (back pain and muscle weakness, and hyperglycemia in one patient being treated for a pancreatic metastasis). 33 Moreover, SBRT does not seem to add toxicity when combined with ST in addition to TKI monotherapy or TKI combined with immunotherapy, with grade 3 to 4 events rarely being related to SBRT. 31 Indeed, in the study of Cheung et al, no grade 3 or higher toxicity was reported for SBRT combined with TKI. 55 Siva et al showed 4 out of 30 patients with grade 3 toxicity, though most likely related to systemic agents instead of SBRT. 59 In the trial of Hannan et al on oligoprogressive omRCC, 1 grade 3 toxicity was reported to be potentially related to SBRT: one patient experienced colitis with small bowel perforation 5 months after SBRT in the setting of concurrent mTOR and TKI therapy. 27 The phase I-II RAPPORT trial evaluated the safety and efficacy of complete SBRT combined with pembrolizumab in pretreated oligometastatic patients. 59 In the NIVES phase II trial (combination of nivolumab and SBRT to a single metastatic lesion in progressive mRCC patients), the combination of SBRT and nivolumab did not lead to any notable increase in toxicity, nor did it lead to an increase of therapy interruptions or delays. 13 The RADVAX RCC phase II trial, assessing the outcomes of combined SBRT to one or two metastatic sites together with nivolumab plus ipilimumab, showed no grade 3–4 toxicity and was deemed safe. 49 In the retrospective analysis of Schoenhals et al 1 grade 5 toxicity was described (hemoptysis, potentially related). 12 Tchelebi et al reported on a meta-analysis of 12 prospective trials with concomitant TKI and radiotherapy, which showed an increase in grade 3 toxicity. 66 Caution should be taken with SBRT administration to sites in proximity to the gastro-intestinal tract, in particular since patients may later receive ICI or TKI which may exacerbate the risk of perforation.
During SBRT, TTs, such as TKIs are either continued or paused up to the discretion of the treating physicians in most trials. Gan et al reported a stop of cabozantinib on average 7.2 days before start of radiation. 67 On the other hand, Cheung et al held TKI for a minimum of 1 week following the last fraction of SBRT. 55 A median pause of TTs of 14 days (2-21d) was disclosed by Kroeze et al. 40 Generally speaking, TT with concurrent SBRT is well tolerated. However caution is necessary when combining SBRT with TT, particularly when radiosensitive structures are close to the targeted lesion, especially when close to gastro-intestinal tract or respiratory tract. 54 In such instances, interrupting ST is reasonable. TKIs have a half-life ranging from 2.5 to 121 h, which should be kept in mind when choosing a timing to hold ST. 68
High-dose radiotherapy has been shown to induce an adaptive antitumor response, which may be enhanced by the combination of SBRT and ICIs. 13 RT might lead to an improved immune response achieved with the use of ICI, either through direct cytotoxic antitumor effect or by modifying the tumor microenvironment to promote an antitumor immune response. 69 Given the long half-life of antibodies, holding ICI-treatment during SBRT is unlikely to affect toxicity. Concurrent administration of SBRT and ICI is however considered safe. 12
The evidence regarding the concurrent use of SBRT with ICI or TT remains scarce or even absent for many combinations. This is particularly evident when examining site-specific analyses of SBRT delivery. In their systematic review and consensus recommendations, Kroeze et al highlight expert agreement on withholding certain systemic therapies, such as anti-VEGF antibodies, anti-EGFR antibodies, nivolumab plus ipilimumab, MEK inhibitors, and multikinase inhibitors, on the same day of SBRT administration. Additionally, a consensus was reached to interrupt MEK-inhibitors and multikinase inhibitors for up to two weeks, and to pause ipilimumab plus nivolumab and anti-EGFR antibodies for at least one week before or after SBRT. Importantly, there was a strong consensus across all ST that the SBRT dose should not be reduced and the fractionation scheme should not be altered. 70
Future Perspectives
What You See is What You Get? The Role of PSMA PET-CT and Whole Body MRI
MDT in omRCC is typically based on conventional imaging, with contrast enhanced computed tomography (CT) currently being the golden standard. 17 False negatives are however common in case of small metastatic locations. Improvement of imaging techniques might increase the detection rate and consequently the accuracy of the definition of omRCC.
Earlier studies with whole body magnetic resonance imaging (MRI) demonstrated an improved diagnostic accuracy regarding the detection of musculoskeletal metastasis compared to CT (97.7% vs 82%), with CT remaining superior in the detection of pulmonary metastases (88.5% vs 71.9%), while both methods had a similar performance regarding lymph node metastases (82.4% vs 83.4%). 71 Particularly lung nodules < 10 mm appeared difficult to interpret on MRI. 72
Importantly, technical improvements and optimization, including the addition of WB-diffusion-weighted MRI (WB-DWI/MRI) and T1-weighted gradient-echo volumetric interpolated breath hold sequence of the chest, with short echo time (< 2 ms) and thin slice thickness (< 3 mm) have substantially improved the performance of WB-MRI for detection of lymphadenopathies and metastases. 73 This is reflected in more recent trials. A study comparing WB-DWI/MRI with CT for staging renal cell cancer confirmed the significantly better performance of WB-DWI/MRI over CT for detection of bone metastases, with confirmed bone metastases detected in 55% of patients by WB-DWI/MRI versus only 43% by CT. 74 Additionally, meta-analyses have confirmed similar performance of WB-DWI/MRI to PET/CT for detection of primary and metastatic malignancies and for M-staging of non-small cell lung cancer, underscoring its ability for comprehensive cancer staging. 75 Regarding the detection of lung metastases, comprehensive data on the sensitivity and specificity of DWI and contrast-enhanced MRI are limited. A recent meta-analysis based on 8 studies showed pooled sensitivity of 91% and specificity of 76% for MRI, inferior to the reference standard technique, CT with pooled sensitivity of 100% and specificity of 99%. 76 While MRI shows acceptable accuracy for detecting lung metastases, it is unlikely to replace CT due to its lower sensitivity, specificity, positive and negative predictive value. 77
It should be noted however, that WB-DWI/MRI is not aimed for to replace CT-based staging of renal cell cancer. Rather, its very accuracy for staging metastases—particularly its higher detection rate for bone metastases compared to CT – irrespective of histological cancer subtype, make it a promising second-line imaging technique to assess total disease extent of patients suspected for oligometastastic disease and scheduled for SBRT. Prostate-specific membrane antigen (PSMA) is a cell surface protein expressed in prostatic tissue and also in neovasculature of other cancers, such as RCC. Multiple trials have evaluated PSMA positron emission tomography (PET)-CT in the staging of RCC compared to CT or MRI.78–80 PSMA PET-CT resulted in higher detection rates (87-88.9% vs 66.7%) compared to CT/MRI, and a significantly higher maximum standard uptake value when compared to fluorodeoxyglucose (FDG) PET-CT.81,82 This lead to an alteration of treatment management in 49% of the cases. 81 Especially when detecting bone lesions, PSMA PET-CT performed better than CT, however the detection of liver lesions was described worse compared to CT. 83 A pooled sensitivity and specificity for the detection of metastatic disease of 92% and 69.9% respectively, indicated that PSMA PET-CT can serve as non-invasive adjuvant tool to conventional imaging in the evaluation of staging RCC, particularly for ccRCC, with uncertain findings on conventional imaging.84,85 PSMA expression is reported to be lower in papillary RCC, and might not be appropriate for staging RCC subtypes other than ccRCC and chromofobe RCCs. 86
Improved imaging can also be used to guide radiotherapy. In the PEDESTAL study, a proof-of-concept study, PSMA-guided SBRT provided durable oncologic outcomes for omRCC, even at 5 years. 87 Others have investigated to role of PSMA PET-CT as a guidance for biology-guided radiotherapy (BgRT), a novel treatment modality that utilizes PET to guide radiation delivery. 88 In a cohort of 83 patients, BgRT seemed feasible in more than 60% of tumors. Suitable RCC targets often included moving targets located in lung or upper abdomen, and were characterized by PSMA PET as avid metastases surrounded by an area of non-avid normal tissue. 88
Other ligands for diagnostic targets, such as (18F)A1F-NYM005 that targets carbonic anhydrase IX, an anhydrase expressed in Von Hippel Lindau-mutated pathways and expressed on tumor cell surfaces in 95–100% of ccRCC, are currently under investigation. 89 In a prospective single center trial (18F)A1F-NYM005 appeared to be safe, and particularly useful for the metastatic setting (rather than primary), as there was notable activity in normal kidney tissue resulting in a lower tumor-to-kidney ratio. 89 However, these new ligands still have to make their entry in the clinic.
PSMA PET-CT may also play a future role in theranostics especially in ccRCC, with the association of neovascularisation of ccRCC and PSMA expression. Also the combination of PSMA TT and immunotherapy may be a promising approach. 90
Novel Strategies and Techniques in Radiotherapy
Further improvements of radiation techniques, such as microbeam radiotherapy, are currently being investigated in a pre-clinical setting. 91 Microbeam radiation therapy ensures a spatially fractionated radiation field instead of a homogeneous field 92 , with high-dose quasi-parallel microbeams (tens of micrometers wide) delivered spacing only few hundred micrometers apart, 91 allowing increased (very high) dose delivery with high precision. Besides an improved therapeutic ratio, it is thought to have the potential to modulate local immune response. 91 To the best of our knowledge, preclinical data on RCC are currently lacking, and most pre-clinical research has been done for brain tumors such as glioblastoma, and melanoma.
Already clinically available, proton therapy has been reported as a promising treatment option. 93 Emiting energy at a specific depth, proton therapy leads to a distinct energy peak known as the Bragg peak 93 , with reduced entrance dose en eliminated exit dose for each beam. Therefore, it could be of interest to reduce treatment toxicity for metastases with close relation to surrounding organs at risk. Only very limited data exist on treating omRCC with proton therapy 93 , with few reports in primary RCC setting 94
Topping up the Evidence: Ongoing Clinical Trials
The clinical behavior of mRCC varies from rapidly progressive to indolent. 95 The optimal patient selection and timing of SBRT in contemporary practice remains unclear. Multiple trials are currently ongoing and investigating SBRT monotherapy or in combination with ST for treatments of omRCC in both upfront, metachronous and oligoprogressive setting.
In the upfront/metachronous setting, the SOAR trial (NCT05863351) is currently ongoing. This is a phase III trial where patients are randomized between SOC (arm A) and (repeated) SBRT until progression, followed by SOC (arm B). Patients with two up to five oligometastatic lesions are included, primary endpoints of this trial are OS and toxicity incidence. In the same setting, the MDACC (ASTROs trial, NCT06004336) and the Consorzio Oncotech trial (NCT05578664) are two phase II trials currently investigating PFS. They randomize between SBRT versus SBRT followed by ST, and MDT (surgery or SBRT) versus Pembrolizumab plus MDT respectively. Another pilot trial is investigating SBRT for the treatment of synchronous or metachronous omRCC with the aim to report on toxicity (NCT02542202).
In the oligoprogressive setting an ongoing trial is NCT04974671, reporting on PFS after delivering SBRT to at least one lesion to patients on an ICI-regimen. The GETUG-STORM-1 is a phase II trial (NCT04299646) comparing ST versus SBRT plus ST (TKI or mTOR inhibitor or immunotherapies), with primary endpoint PFS, targeting a study population of 114 patients.
Additional answers might also come from the oligorare trial (NCT04498767). This is a randomized open-label multicentre phase III study, investigating the effect of adding SBRT to the SOC treatment on OS in patients with rare oligometastatic cancers (RCC included). Patients are randomized between SOC versus SOC and SBRT to all sites of known metastatic disease. Primary endpoint is OS. Other basket trials open for RCC inclusion are the SABR-COMET3 (NCT03862911) and SABR-COMET10 (NCT03721341). At last, the feasibility of combinations of SBRT and ICI/other ST are currently being investigated in several trials as well: NCT02781506, a phase II trial on nivolumab and SBRT, NCT03115801, a phase II randomized trial with immunotherapy plus radiotherapy in metastatic genitourinary cancers with ORR as primary endpoint, and NCT03693014, a phase II trial investigating the effect of hypofractionated radiotherapy in patients with limited progression on ICI.
Conclusion
SBRT results in excellent tumor control with a low toxicity rate. It can be an appropriate therapeutic option in patients presenting with synchronous or metachronous omRCC in order to postpone the onset of ST and its potential side effects. Furthermore, SBRT to oligoprogressive sites could achieve desirable disease control while extending the duration of the ongoing ST. Translational studies are needed to determine the underlying mechanisms of omRCC and how to optimize treatment in the future. A potentially synergistic association with immunotherapy also remains further to be investigated. Future randomized controlled studies are needed to ameliorate the detection of both inclusion and exclusion criteria that identify patients who are best suited for SBRT, and to learn more on the ideal timing of SBRT and ST combination, in order to fully establish the role of SBRT for mRCC patients as a solid SOC.
Footnotes
ORCID iDs
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
