Abstract
Long non-coding RNAs (lncRNAs) are known to play vital roles in human cancers. LncRNA TRPM2-AS has been found to be upregulated in various types of cancers. The elevated levels of TRPM2-AS are associated with important clinicopathological parameters such as tumor size, tumor stage, and lymph node metastasis, revealing that TRPM2-AS could be a potential target for cancer diagnosis, prognosis and treatment. Moreover, TRPM2-AS is involved in regulating the cell proliferation, migration, invasion, apoptosis, drug or radio resistance by serving as a competing endogenous RNA, directly bounding to proteins and regulating multiple signaling pathways. In this review, we comprehensively summarize the latest knowledge on the aberrant expression of TRPM2-AS, the relationship between TRPM2-AS and clinical features, and the detailed mechanisms of potential functions of TRPM2-AS in various cancer types. The current study highlights the potential of TRPM2-AS as a prognostic and therapeutic target in cancers.
Background
The malignant tumor is the main cause of human death. According to the latest Global Cancer Statistics, 1 there were approximate 20 million new cancer cases and about 9.7 million deaths in the year 2022. Although cancer mortality continues to decline, there are still 22.8% cancer deaths from noncommunicable diseases worldwide. The prognosis for cancer remains poor, despite advancements in therapy methods. Therefore, further research on the mechanisms of tumorigenesis and progression is necessary to identify effective biomarkers and therapeutic targets, which can provide new strategies for early diagnosis, prognosis evaluation, and treatment of malignant tumors.
Approximately 90% of the human genome is transcribed, but only 2% is made up of protein-coding genes. Long non-coding RNAs (lncRNAs) are a diverse class of RNA molecules that exceed 200 nucleotides in length and lack a protein-coding ability. 2 With the advancement of high throughout technologies and deeper scientific research, more and more lncRNAs have been identified as key players in a wide range of physiological and pathological processes, including chromatin modification, transcriptional regulation, and post-transcriptional regulation of proximal and distal genes.345–6 Due to the high efficiency, tissue specificity, and dynamic expression during development, 7 lncRNAs exhibit potential as diagnostic, prognostic indicators, and therapeutic targets for tumors.
The transient receptor potential melastatin 2 antisense RNA (TRPM2-AS), an antisense lncRNA transcribed from the TRPM2 gene locus mapped on chromosome 21, was first identified as melanoma-enriched antisense transcript in 2008. 8 The expression of TRPM2-AS was upregulated in melanoma and lung cancer, and its activation is consistent with the methylation status of CpG island. 8 Later, the increasing studies found that the aberrant expression of TRPM2-AS has been observed in various cancer types, including lung cancer (LC), 9 prostate cancer (PC), 10 colorectal cancer (CRC), 11 gastric cancer (GC), 12 osteosarcoma, 13 endometrial cancer (EC), 14 ovarian cancer (OV), 15 hepatocellular carcinoma (HCC), 16 laryngeal carcinoma, 17 retinoblastoma, 18 bladder cancer (BLCA), 19 esophageal cancer 20 and gallbladder cancer (GBC). 21 TRPM2-AS influences cancer cell proliferation, apoptosis, migration, and invasion through multiple mechanisms, such as epigenetic modification, interaction with microRNAs, and modulation of signaling pathways. This review aims to integrate current knowledge on TRPM2-AS, clarifying its role in diverse cancer types, mechanisms of action, and potential therapeutic implications.
Clinical Significance of TRPM2-AS in Cancers
TRPM2-AS acts as a tumor oncogene, is highly expressed in tumor tissues compared to normal tissues in many cancer types (Figure 1), indicating that TRPM2-AS overexpression could distinguish cancerous and normal tissues. Furthermore, altered expression of TRPM2-AS could serve as a diagnostic and prognostic marker in cancer. TRPM2-AS plays an important role in tumor biology, with the evidence that the up-regulation in TRPM2-AS expression is associated with adverse clinical outcomes, for instance tumor TNM stage, lymph node metastasis, distant metastasis, overall survival (Table 1, Figure 2).

The expression levels and clinical prognosis of TRPM2-AS in various cancers. The expression levels of TRPM2-AS in various cancer tissues were obtained based on the GEPIA online database analysis. * indicates p value < 0.05. Red indicates tumor tissue, blue indicates normal tissue. T: tumor number; N: normal number; BLCA: bladder cancer; BRCA: breast cancer; GBM: glioblastoma; OV: ovarian cancer; PC: prostate cancer; EC: endometrial cancer; SKCM: melanoma; CHOL: bile duct cancer; COAD: colon cancer; READ: rectal cancer; ESCA: Esophageal cancer; LIHC: liver cancer; GC: gastric cancer; LC: lung adenocarcinoma; LUSC: lung squamous cell carcinoma.

The clinical prognosis of TRPM2-AS in various cancers.
The Expression and Clinical Characteristics of TRPM2-AS in various Cancers.

Note: SKCM: melanoma; LC: lung adenocarcinoma; NSCLC: non-small cell lung cancer; GC: gastric cancer; CRC: colorectal cancer; EC: endometrial cancer; OV: ovarian cancer; GBM: glioblastoma; HCC: hepatocellular carcinoma; BLCA: bladder cancer; PC: prostate cancer; GBC:gallbladder cancer.
Role of TRPM2-AS in Respiratory System
TRPM2-AS in Lung Cancer
Lung cancer, accounting for 2.5 million new cases and the most frequently diagnosed cancer in 2022, presents a high death rate and poor prognosis. 39 After computational identification of TRPM2-AS, the over-expression level of TRPM2-AS is confirmed using qRT-PCR in matched tumorous/non-tumorous samples of lung tissues. 8 Furthermore, the expression level of TRPM2-AS is discovered to be associated with the methylation status of the CpG island. Later, the expression of TRPM2-AS is also found to be upregulated in non-small cell lung cancer (NSCLC) tissues compared to paired adjacent normal tissues.22,23 A higher expression level of TRPM2-AS is associated with higher TNM stages and larger tumor size. Kaplan-Meier curve shows that patients with high TRPM2-AS expression levels have poorer survival (hazard ratio (HR) = 1.239, p = 0.003) compared to those with low TRPM2-AS levels, revealing the potential of TRPM2-AS as a prognostic marker for NSCLC. Down-regulation of TRPM2-AS in NSCLC cells inhibits cell proliferation, migration and invasion, while promotes apoptosis in NSCLC cells. In another study, 24 a potential genome instability related competitive endogenous RNA (ceRNA) regulatory network using four lncRNAs including TRPM2-AS is constructed by multiomic bioinformatics analysis, indicating clinical references for immunotherapy and prognosis of lung cancer.
TRPM2-AS in Laryngeal Carcinoma
Laryngeal carcinoma is a common type of malignant tumor of the larynx, with 188,960 new diagnosis cases and 103,216 deaths in 2022. 1 Due to nonspecific symptoms, laryngeal carcinoma is often diagnosed at the advanced stages, causing delayed treatment and a worse prognosis. 40 LncRNAs are regarded as novel biomarkers for laryngeal carcinoma due to their aberrant expression and biological activities.41,42 TRPM2-AS is highly expressed in human laryngeal carcinoma tissues and cell lines, and elevated TRPM2-AS expression is found in patients with advanced clinical stage. 17 Knockdown of TRPM2-AS significantly inhibited cell proliferation, migration and invasion, offering new insights into TRPM2-AS targeted diagnostics and therapeutics in laryngeal carcinoma.
Role of TRPM2-AS in Digestive System
TRPM2-AS in Gastric Cancer
Gastric cancer (GC), the fifth most commonly diagnosed cancer worldwide (968,350 new cases, accounting for 4.9%) and the fifth leading cause of cancer death (659,853 deaths, accounting for 6.8%), 1 is a significant global health issue. Although current therapies have gained significant progress, the overall prognosis for GC patients is still poor with a high recurrence rate. LncRNAs have emerged as critical factors in cellular functions, influencing the development, growth, metastasis, and prognosis of GC. By analyzing RNA sequencing data from the GEO database (GSE70880, GSE224056) and TCGA database (TCGA-STAD), TRPM2-AS is identified due to its up-regulated expression level.12,28 Further functional enrichment analysis indicates that TRPM2-AS may be involved in key pathways, such as cell adhesion and the positive regulator in cell migration, underscoring its role in the development and progression of GC.
Several recent studies have found that TRPM2-AS is highly expressed in both GC tissues and cell lines.2526–27 Higher expressions of TRPM2-AS were significantly associated with TNM stage, invasion depth, and lymphatic metastasis. In a study by Xiao et al, 26 the expression level of TRPM2-AS was higher in T2-T4 stage than in T0-T1 (p < 0.001), higher TNM stages (p = 0.001), and patients with lymphatic metastasis (p = 0.001). Whereas, another study showed that the expression level of TRPM2-AS was higher in lower tumor grade (p < 0.001), suggesting that TRPM2-AS could be a biomarker for early-grade GC diagnosis. 28
Up-regulation of TRPM2-AS is associated with poor prognosis in GC. Kaplan-Meier plotter shows that the GC patients with higher TRPM2-AS expression levels have shorter overall survival (OS, HR = 1.37, p = 0.028) and recurrence free survival (RFS, HR = 1.46, p = 0.024). 26 Another study also showed that higher TRPM2-AS expression presented shorter OS (p = 0.0022), and over-expressed TRPM2-AS was a prognostic factor in GC patients by both univariate analysis (HR = 3.562, p = 0.006) and multivariate analysis (HR = 3.128, p = 0.013). 25
Over-expression of TRPM2-AS in GC cells contributes to cancer progression by promoting cell proliferation, migration, invasion and metastasis, while inhibiting apoptosis.25,28 Silencing TRPM2-AS inhibits cell proliferation (p < 0.001), enhances the apoptosis rate by 5-fold (p < 0.001), and reduced cell migration (p < 0.001). 27
TRPM2-AS in Colorectal Cancer
Colorectal cancer (CRC) accounts for 9.6% of newly diagnosed cancers and 9.3% of mortality, ranking as third most common malignancy and second cancer death worldwide. 1 LncRNAs as critical contributors are involved in the initiation, progression and metastasis of CRC.
Microarray analysis (GSE110715) reveals the up-regulation of TRPM2-AS in CRC, 11 which is validated in CRC tissues and colon cell lines. 29 TRPM2-AS down-regulation inhibits CRC cell proliferation, migration, and invasion. 30 Moreover, TRPM2-AS promotes methylation level with 90% sensitivity and 53% specificity (AUC = 0.71, p < 0.0004), 11 revealing that TRPM2-AS promoter methylation is negatively correlated with its expression. This suggests that the methylation status of the TRPM2-AS promoter could serve as a potential marker for CRC detection. However, the methylation status of the TRPM2-AS promoter is not associated with the clinicopathological characteristics.
TRPM2-AS in Esophageal Cancer
Esophageal cancer is a malignant digestive tract cancer with high mortality. It is reported an estimated 511,000 new cases and 445,000 deaths of esophageal cancer worldwide in 2022. 1 Increasing evidence showed that multiple lncRNAs were associated with tumorigenesis, metastasis, and prognosis of esophageal cancer.4344–45 Elevated TRPM2-AS expression is observed in esophageal cancer tissues and cell lines. 20 TRPM2-AS silencing inhibits cell proliferation, migration and invasion, while promotes cell apoptosis, uncovering that high expression of TRPM2-AS is related to poor prognosis of esophageal cancer.
TRPM2-AS in Hepatocellular Carcinoma
Liver cancer is the fifth most common cancer and the third leading cause of cancer death worldwide. 1 HCC is the most frequent subtype of liver cancer that originating from the primary liver cells. Abnormal lncRNA expression plays critical roles in the development and progression of HCC. 46 It is reported that the expression of TRPM2-AS is up-regulated in HCC tissues and cell lines. 16 TRPM2-AS is significantly correlated with tumor size (p = 0.005), AJCC stage (p = 0.014), and tumor differentiation (p = 0.021), but is not associated with age, sex, AFP, vascular invasion, or liver cirrhosis. Furthermore, HCC patients with high TRPM2-AS expression had a worse OS (p = 0.021), indicating that high expression levels of TRPM2-AS predict a poor prognosis in HCC. TRPM2-AS knockdown inhibits HCC cell proliferation, suggesting that TRPM2-AS might be a novel target for HCC therapy.
TRPM2-AS in Gallbladder Cancer
GBC, an aggressive carcinoma arising from the biliary tract, has a high mortality with a 5-year survival rate of less than 5%-15% in advanced GBC cases.47,48 LncRNAs can act as a tumor suppressors or oncogenic factors, lncRNAs aberrantly expressed in GBC patients are involved in malignant features that contribute to poor survival outcomes.4950–51 A recent study reported that exosomal TRPM2-AS was expressed significantly higher with high microvascular density in GBC tissues. 21 Besides, the authors found that the expression level of TRPM2-AS was positively associated with tumor size (p = 0.0098), lymph node metastasis (p = 0.0369), liver invasion (p = 0.0195), tumor differentiation (p = 0.0074), TNM stages (p = 0.0092), and CA19-9 levels (p = 0.0007). Furthermore, Kaplan-Meier analysis reveal that TRPM2-AS is associated with reduced OS (p = 0.0053) and RFS (p = 0.0089) in patients with GBC. Univariate (OS: HR = 2.401, p = 0.008; RFS: HR = 2.065, p = 0.016) and multivariate (OS: HR = 2.43, p = 0.016; RFS: HR = 2.124, p = 0.021) cox regression analysis demonstrate the independent prognostic role of TRPM2-AS in patients with GBC as well.
Role of TRPM2-AS in Urinary System
TRPM2-AS in Prostate Cancer
Globally, PC is the forth most common cancer in males and induces 396,792 deaths in 2022. 1 In addition to family history and genetic mutation, additional factors like smoking, diet, physical activity, specific medications, and occupation can increase the risk of PC. 52 Several lncRNAs have been reported to regulate cancer development and progression in PC.10,5354–55 TRPM2-AS, a newly identified lncRNA in PC, has been shown to be highly expressed and linked to poor clinical outcomes. 36 TRPM2-AS knockdown lead to PC cell apoptosis, revealing its role as a key regulator of cell survival. In addition, a large number of genes involved in unfolded protein response and the cell-cycle progression are found in TRPM2-AS knockout PC3 cells, 37 showing the role of TRPM2-AS as a novel therapeutic target.
TRPM2-AS in Bladder Cancer
BCLA is the ninth most common cancer worldwide and has been associated with incidence (613,791 new cases, 3.1%) and high mortality (220,349 deaths, 2.3%). 1 Due to the high occurrence rate, BCLA has become a growing challenge in clinical care. LncRNAs play a significant role in the development and may improve the prognosis of BCLA.56,57 A recent study found that TRPM2-AS was significantly elevated in BLCA tissues and cells. 19 Moreover, TRPM2-AS expression level is associated with tumor size (p = 0.049), histological grade (p = 0.003), TNM stage, pathological grade (p = 0.001), and lymphatic metastasis (p = 0.001). Additionally, TRPM2-AS enhances the viability and proliferation but impairs the apoptosis of BLCA cells.
Role of TRPM2-AS in Endocrine Reproductive System
TRPM2-AS in Endometrial Cancer
EC is an epithelial malignant tumor and one of the most common gynecological malignancies. EC patients are often diagnosed as clinically aggressive cancer, the incidence and mortality are increasing. 39 Many lncRNAs have been identified as potential targets for the diagnosis and treatment of EC.58,59 By using TCGA data, TRPM2-AS was found to be positively correlated with advanced histological grade, International Federation of Gynecology and Obstetrics stage, lymph node, distant metastasis, and poor prognosis. Lin et al identified TRPM2-AS as prognostic necroptosis related lncRNA using univariate (HR = 0.885, p = 0.043) and multivariate cox regression (HR = 0.885, p = 0.0015). 33 In addition, TRPM2-AS is identified as angiogenesis related and prognostic lncRNA using univariate (HR = 1.9, p = 0.0043) and multivariate cox regression (HR = 2.2, p = 0.00016) as well. 14 TRPM2-AS is proved to be overe-expressed in EC tumor tissues, and promotes proliferation, invasion, migration and angiogenesis.14,34
TRPM2-AS in Ovarian Cancer
OV is another highly lethal gynecological malignant cancer, posing a significant threat to women's health due to its high incidence, mortality rate and bleak prognosis. Based on recent discovery, lncRNAs are found to be involved in the proliferation, apoptosis, cell cycle, migration, invasion, metastasis and drug resistance of OV.6061–62 Ding et al detected the expression of TRPM2-AS increased in OV cell lines including EB0405, CAOV3, HEY, and SKOV3.15,63 Further cell assay demonstrates that TRPM2-AS contributes to cell proliferation, colony formation, cell migration and invasion as well.
Role of TRPM2-AS in Other Cancers
TRPM2-AS in Osteosarcoma
Osteosarcoma, a common bone malignancy with a poor prognosis, is slightly more common in males. 64 Many studies have focused on the roles of lncRNAs in osteosarcoma already, revealing that lncRNAs regulate various biological and pathological processes of osteosarcoma.65,66 TRPM2-AS, which is highly expressed in osteosarcoma tissues and cells, is associated with advanced enneking stage (p < 0.001), large tumor size (p < 0.001), and high histological grade (p < 0.001) in osteosarcoma. 13 High TRPM2-AS expression displayed obviously shorter OS. Loss function studies suggest that down-regulation of TRPM2-AS inhibits cell viability, proliferation and migration, and induces cell apoptosis. 32
TRPM2-AS in Gliomas
Gliomas is an aggressive cancer in the central nervous system with high mortality and morbidity. According to the histological characteristics and molecular changes, gliomas are divided into five categories. 67 Dysregulation of lncRNA is critical in the pathogenesis, progression, and malignancy of gliomas, revealing their important roles in gliomas.6869–70 Significant increase of TRPM2-AS is detected in glioma tissues, and higher expression of TRPM2-AS is positively connected with older age, male, advanced grade of cancer. 35 In addition, the upregulation of TRPM2-AS promotes the proliferation, migration, and invasion, while suppression of TRPM2-AS inhibits cell growth and metastasis. However, whether TRPM2-AS participates in cell apoptosis is still uncovered.
TRPM2-AS in Retinoblastoma
Retinoblastoma, the most frequent malignant intraocular tumor, occurs mainly in children and adolescents. The survival rates of retinoblastoma vary depending on the national income level. 71 Recent findings show that lncRNAs play crucial roles in retinoblastoma progression.72,73 Li et al found that higher TRPM2-AS expression was associated with advanced clinical stage (p = 0.007) and optic nerve invasion (p = 0.035) in patients with retinoblastoma. 18 Moreover, downregulation of TRPM2-AS significantly inhibits proliferation, migration and invasion, but elevates apoptosis in retinoblastoma cells.
Aberrant Expression Mechanism of TRPM2-AS in Cancers
An increasing number of lncRNAs have been discovered to be dysregulated in human cancers, and the underlying mechanisms that contribute to lncRNA dysregulation are gradually elucidated as well. It is reported that the activation of TRPM2-AS in melanoma was consistent with the methylation status of the CpG island, 8 but the underlying mechanism is poorly understood. Therefore, further investigations are needed to expand our knowledge on the interaction between CpG island and TRPM2-AS in cancer patients.
Transcriptional factors (TFs) are one of the largest families of transcriptional regulators that recognize specific DNA sequences to control gene transcription. 74 Evidences show that TFs may contribute to the aberrant expression of lncRNAs in cancers. 75 The overexpression of TRPM2-AS can be modulated by TFs ELK1 and SP1 in GC25,28 (Figure 3). TRPM2-AS expressing levels are notably decreased in cells after ELK1 knockdown, ChIP assays demonstrate that ELK1 could effectively bind to the sites from −1249 to −1240 in the promoter region of TRPM2-AS. 25 Dual-luciferase reporter assays confirm that ELK1 could remarkably elevate the activity of luciferase by using a mutant sequence. Based on these results, it is concluded that the overexpression of TRPM2-AS in GC was induced by ELK1. In another study, Zhang et al used online software to predict the binding potential of TFs for TRPM2-AS. Among three predicted TFs SP1, NFκB and p53, only SP1 was proven to activate TRPM2-AS transcription. 28

The aberrant expression of TRPM2-AS.
Posttranscriptional m6A modifications are involved in the pathogenesis of human cancers. A recent study found that the post-transcriptional m6A modification induced elevated TRPM2-AS in GBC, IGF2BP2 interact with TRPM2-AS and promote the stability of TRPM2-AS. 21 In detail, the 271 site of the m6A modification site is essential for the interaction of IGF2BP2 to TRPM2-AS. The authors suggest that the interplay between methyltransferases and binding proteins is very important for maintaining post-transcriptional m6A modification and is responsible for the stability and continuous overexpression of TRPM2-AS in GBC.
Regulatory Mechanisms of TRPM2-AS in Cancers
Till now, an increasing number of studies have suggested the oncogenic role of TRPM2-AS in tumor initiation, progression, metastasis, and radio or drug resistance. TRPM2-AS may exert its oncogenic function mainly by regulating various related gene expression profiles (Table 2, Figure 4).
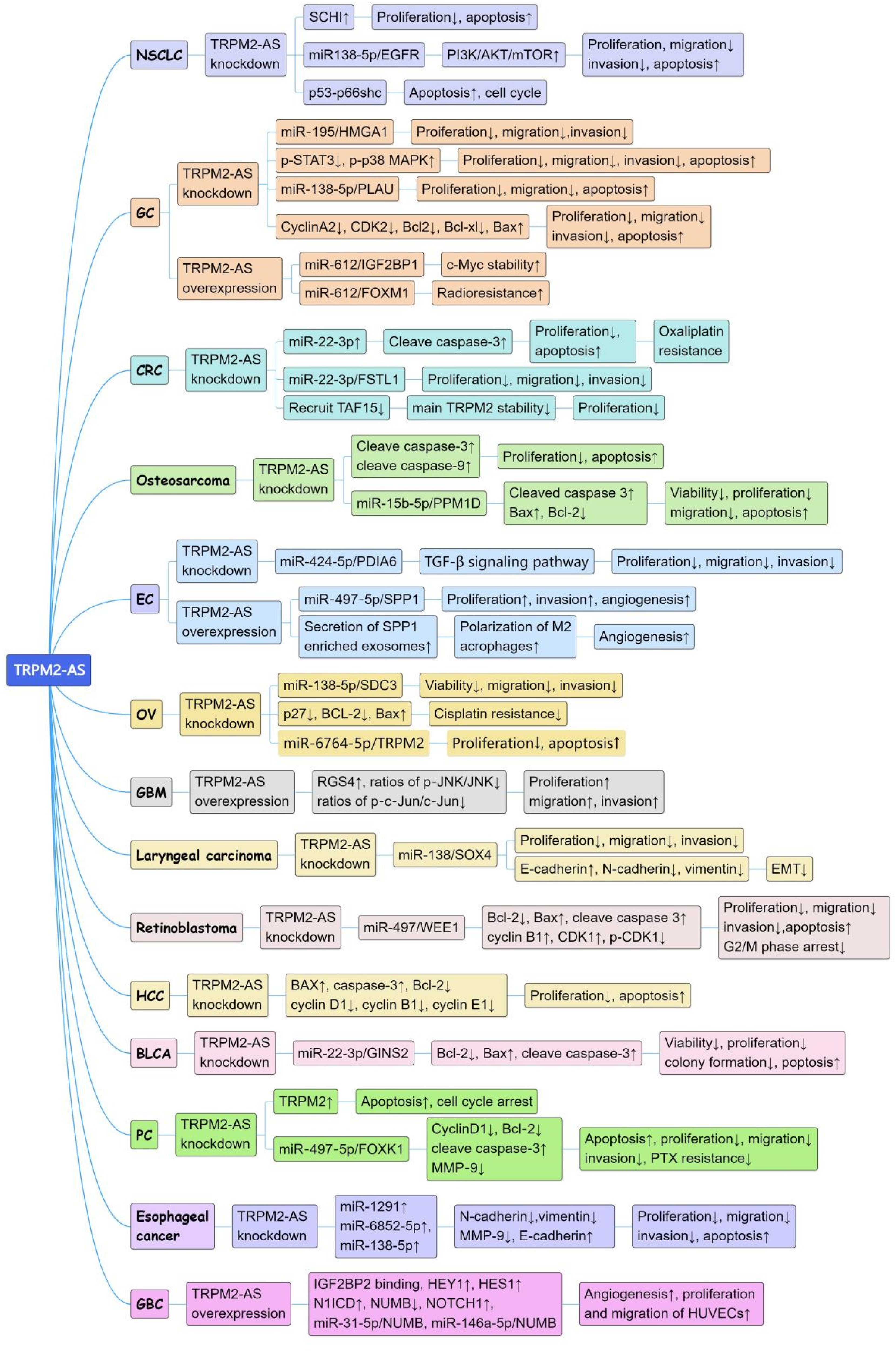
The regulatory mechanisms of TRPM2-AS in cancers. The arrow ↑ indicates the expression level increased, arrow ↓ indicates the expression level decreased. NSCLC: non-small cell lung cancer; GC: gastric cancer; CRC: colorectal cancer; EC: endometrial cancer; OV: ovarian cancer; GBM: glioblastoma; HCC: hepatocellular carcinoma; BLCA: bladder cancer; PC: prostate cancer; GBC:gallbladder cancer.
The Roles and Mechanisms of TRPM2-AS in Cancers.

Note: NSCLC: non-small cell lung cancer; GC: gastric cancer; CRC: colorectal cancer; EC: endometrial cancer; OV: ovarian cancer; GBM: glioblastoma; HCC: hepatocellular carcinoma; BLCA: bladder cancer; PC: prostate cancer; GBC:gallbladder cancer.
Involvement in Signaling Pathways
By affecting different genes expressions, lncRNAs are involved in a variety of cellular signaling pathways, for instance, JAK/STAT, 76 Wnt/β-catenin, 77 and Hippo signaling pathway. 78 TRPM2-AS is reported to participate in the pathological processes through p53, TGF-β, and NOTCH1 signaling pathways. It is reported that TRPM2-AS may regulate cell apoptosis through SHC1, 22 further study showed that TRPM2-AS regulated cell cycle, apoptosis, and resistance to cisplatin via suppressing p53-p66shc pathway in NSCLC. 9 In addition, the PI3 K/AKT/mTOR pathway is reported to be activated by TRPM2-AS/miR-138-5p, helping overcome both on-target and off-target resistance in therapy for advanced NSCLC. 23 Wang et al revealed that TRPM2-AS/PDIA6 axis could promote the proliferation and metastasis process by regulating the TGF-β signaling pathway through a classical SMAD-dependent pathway in EC. 34 While Zhang et al showed that TRPM2-AS inhibited GC cell apoptosis by promoting phosphorylated p38 MAPK and inhibiting phosphorylated STAT3. 28 In addition, TRPM2-AS up-regulation promotes glioma cell line U251 and U87 proliferation, migration, and invasion by inhibition of phosphorylated JNK and c-Jun and up-regulation of RGS4. 35 Recent findings showed that TRPM2-AS promoted angiogenesis by activating the NOTCH1 signaling pathway in GBC. 21 In detail, TRPM2-AS/PABPC1 can indirectly induce the activation of NOTCH1 signaling through miR-31-5p/miR-146a-5p-mediated NUMB mRNA degradation.
Involvement in ceRNA Network
Besides directly regulating target genes, lncRNAs can bind to miRNAs competitively acting as ceRNA, preventing the miRNAs degradation on their targets, ultimately participating in various biological processes. Luciferase assay, RNA pull-down assay, loss and gain function assay are commonly used to elucidate the ceRNA role of lncRNAs. Studies have shown that TRPM2-AS mediated ceRNA network (Figure 5) plays vital roles in the carcinogenesis of a variety of cancers, including GC, NSCLC, and OV.

TRPM2-AS ceRNA network involved in cancers. Red arrow indicates promotion, while green arrow indicates inhibition.
TRPM2-AS could play its oncogenic role as a miRNA sponge in GC. Huang et al revealed that miR-195 was a direct target of TRPM2-AS in SGC-7901 and MGC-803 cells. 25 Later, they demonstrated that TRPM2-AS could function as a ceRNA to modulate HMGA1 expression by sponging miR-195. In another study, Sun et al found that TRPM2-AS contributed to GC progression by sponging miR-138-5p to upregulate PLAU. 27 These results suggest a promising direction for GC therapy and diagnosis.
In addition to the TRPM2-AS/miR-138-5p axis in GC, it is also found TRPM2-AS/miR-138-5p axis participates in pathogenesis in other cancers. TRPM2-AS sponging miR-138-5p increase the levels of EGFR to promote cell proliferation, migration, and invasion of NSCLC and exacerbate xenograft tumors in vivo. 23 Besides, TRPM2-AS could sponge miR-138-5p to release SDC3, thus promoting the progression of OV and enhancing the cisplatin resistance in OV cells. 15 In laryngeal carcinoma, TRPM2-AS served as ceRNA binding to miR-138 directly, thus upregulating expression of SOX4 and promoting EMT. 17
miR-497 is another miRNA included in the TRPM2-AS ceRNA network. TRPM2-AS/miR-497 axis participate in G2/M arrest and cell apoptosis by inhibition of WEE1 in retinoblastoma. 18 In addition, TRPM2-AS could promote EC cell proliferation, migration and invasion, and angiogenesis of HUVECs through miR-497-5p/SPP1 axis. 14 Moreover, Shi et al reported that TRPM2-AS enhanced paclitaxel resistance in PC cells and suppressed cell apoptosis by reducing FOXK1 expression via sponging miR-497-5p. 38
Apart from miRNAs mentioned above, many other miRNAs are also reported to participate in the regulation of cancer progression. For instance, the TRPM2-AS/miR-424-5p axis directly targets PDIA6 to promote EC cell proliferation and metastasis through the TGF-β signaling pathway. 34 TRPM2-AS enhances the progression of BLCA by binding to miR-22-3p and thus increasing GINS2 expression, 19 and TRPM2-AS regulates CRC malignant behavior by inhibiting the miR-22-3p/FSTL1 axis. 30
Interaction with Proteins or mRNA
It has been reported that lncRNAs can connect to protein further affect cancer progress. TRPM2-AS interacted with fragment III of the protein PABPC1 via domain C. 21 The interaction could strengthen the interaction between PABPC1 and the NUMB mRNA, further promoting angiogenesis in GBC.
LncRNA also regulate tumor progression by affecting their nearby genes. TRPM2-AS, the an antisense and nearby gene of TRPM2, is positively correlated to TRPM2. 36 Moreover, TRPM2-AS maintained the mRNA stability of TRPM2 by directly interacting with RNA-binding protein TAF15, thus facilitating the proliferation of CRC cells. 29 A very recent published literature showed that TRPM2-AS could regulate TRPM2 by sponge miR-6764-5p, revealing both direct and indirect regulatory roles of TRPM2-AS. 63 TRPM2, a Ca2+ permeable non-selective cation channel, is widely expressed in multiple organs. TRPM2 has a vital role in cell growth and survival in different cancers 79 including, BC, 80 GC, 81 OV,63,82 oral squamous cell carcinoma. 83 TRPM2 exhibits characteristics similar to a plasma membrane ion channel that allows the entry of Ca2+ into GC cells, 84 which leads to a reduction in cellular bioenergetics, inhibition of cell invasion, and decreased cell survival. Using molecular and functional assays, silencing TRPM2 has demonstrated to inhibited GC cell growth and invasion though PTEN/Akt pathway. 85 Considering the relationship between TRPM2-AS and TRPM2, TRPM2-AS may make its role though sense RNA TRPM2 in cancers.
Involement in Drug or Radio Resistance
Due to no specific symptoms in the early stages, most cancer patients are diagnosed in advanced stages. Chemotherapy is still an effective method for cancer treatment till now. However, drug resistance is a crucial factor affecting the prognosis of cancer patients.
Paclitaxel, a well-known compound for chemotherapy, is commonly used for PC treatment. 86 In paclitaxel resistant PC cells, the expression level of TRPM2-AS was increased. 38 TRPM2-AS knockdown suppresses cell proliferation, migration and invasion, facilitated cell apoptosis in paclitaxel resistant PC cells. In addition, TRPM2-AS silencing enhanced the sensitivity of paclitaxel resistant PC cells to paclitaxel. Then, the evidence showed that TRPM2-AS could act as sponge of miR-497-5p, which may promote cell apoptosis and paclitaxel sensitivity in paclitaxel resistant PC cells. FOXK1, a target gene of miR-497-5p, abolished the influences of TRPM2-AS silencing on paclitaxel sensitivity in paclitaxel resistant PC cells. Thus, TRPM2-AS knockdown suppressed paclitaxel resistance in paclitaxel resistant PC cells by miR-497-5p/FOXK1 axis.
A549 cells treated with cisplatin exhibit higher expression of TRPM2-AS, and TRPM2-AS expression level was consistent with dose of cisplatin. 9 Overexpression of TRPM2-AS reduced the cisplatin induced apoptosis. Cisplatin resistance in A549/DDP cells might be inhibited by TRPM2-AS knockdown activating the p53-p66shc pathway, highlighting that TRPM2-AS may provide opportunities as a promising therapeutic target for NSCLC. In OV cells, knock-down of TRPM2-AS significantly increased apoptosis-promoting effect of the cisplatin. 15 TRPM2-AS could sponge miR-138-5p to release SDC3, thus enhancing the cisplatin resistance in OV cells.
Oxaliplatin, a third-generation platinum derivative, is approved for the treatment of patients with advanced CRC. TRPM2-AS is reported highly expressed in oxaliplatin resistant CRC cells. 31 TRPM2-AS knockdown inhibited the proliferation of oxaliplatin resistant CRC cells and promoted cell apoptosis by upregulating miR-22-3p, suggesting that TRPM2-AS might provide a potential target for reducing oxaliplatin resistance in CRC.
Radiotherapy is a common clinical treatment for GC. Resistance to radiotherapy is a major challenge in clinical and scientific research. It has been shown that lncRNAs are important regulators involved in radioresistance in various cancers.87,88 TRPM2-AS may play a role in regulating radioresistance in GC. 26 The expression of TRPM2-AS is significantly upregulated in GC cells when exposed to 2 Gy or 8 Gy irradiation compared to untreated GC cells. Reduced colony survival fractions and increased expression of the DNA damage marker γH2AX are detected in TRPM2-AS depleted GC cells when cells received 8 Gy irradiation. Mechanistically, TRPM2-AS may sponge miR-612, thus enhancing the expression of FOXM1 to promote GC radioresistance. Therefore, TRPM2-AS could be a useful target for clinically application in GC.
TRPM2-AS have been demonstrated to be associated with radioresistance 26 and chemoresistance when using paclitaxel, 38 cisplatin,9,15 oxaliplatin 31 in several cancer types. Therefore, TRPM2-AS may act as a novel target for cancer therapy especially when the patients are found to be resistant to drug or radio.
Regulating the Tumor Microenvironment
The tumor microenvironment is critical factor in cancer development and could be a potential target for cancer treatment. TRPM2-AS played an important role in tumor microenvironment of EC. 14 Using CIBERSORT analysis, the infltration levels of 12 immune cell subtypes showed statistical diference in EC tissues, and TRPM2-AS was positively related to regulatory T cell, NK cells, negatively related to M1 macrophage and plasma B cells. In addition, the expression of TRPM2-AS in M2 macrophages was increased. The expressions of M2 macrophage markers CD163 and CD206 were significantly increased when TRPM2-AS was over expressed, but was reversed by the knockdown of SPP1. Furthermore, knockdown of SPP1 also reversed the promoting effect of TRPM2-AS on angiogenesis of EC cells HUVECs. EC cells regulated the macrophages polarization by secreting SPP1 enriched exosomes. Taken together, TRPM2-AS could regulate the polarization of M2 macrophages and promote angiogenesis of HUVECs through SPP1.
Application Potential
As a Promising Biomarker for Cancer Prognosis
Kaplan-Meier curve showed that patients with high TRPM2-AS expression levels have poorer survival in cancers including NSCLC, 22 GC, 26 GBC, 21 PC, 36 HCC, 16 osteosarcoma 13 and EC. 14 Univariate and multivariate Cox regression analysis further confirmed TRPM2-AS as an independent prognostic factor in patients with GC, 25 GBC, 21 PC 36 and EC. 14 Thus these evidence revealed that TRPM2-AS might be a powerful prognostic biomarker for cancers.
As a Therapeutic Target for Cancer Treatment
Given that TRPM2-AS is highly expressed in over 14 types of cancer, it holds strong potential as a therapeutic target. The aberrant expression of TRPM2-AS in cancers could be partially attributed to the methylation status of the CpG island, m6A modifications, TFs SP1 and ELK1 interaction. The elevated level of TRPM2-AS is associated with key clinicopathological parameters such as tumor size, tumor stage, and lymph node metastasis. Moreover, TRPM2-AS silencing inhibits the biological behaviors of cancer cell growth, migration, metastasis and angiogenesis. The underlying mechanisms may associated with oncogenes such as NOTCH1, spp1, GINS2, and WEE1. Therefore, TRPM2-AS might be a potential target for cancer diagnosis and therapy.
Improvement of Radioresistance or Chemoresistance
TRPM2-AS have been demonstrated to be associated with radioresistance 26 and chemoresistance. TRPM2-AS knockdown attenuates radioresistance in GC, enhances the sensitivity to paclitaxel, 38 cisplatin,9,15 oxaliplatin 31 in several cancer types. Therefore, TRPM2-AS may act as a novel target for cancer therapy especially when the patients are found to be resistant to drug or radio.
Future Prospects and Conclusions
TRPM2-AS, acting as an oncogene, is highly expressed in human cancers. TRPM2-AS can promote tumor progression and play a crucial regulatory role in cell proliferation, migration, invasion, apoptosis, cell cycle, drug resistance, or radio resistance in various tumors. Most studies focus on its functions as a ceRNA through lncRNA-miRNA-mRNA signaling. Besides, TRPM2-AS may promote proliferation and metastasis process by regulating many signaling pathways, such as PI3 K/AKT/mTOR, NOTCH1 and TGF-β signaling pathway. Furthermore, TRPM2-AS could regulate the polarization of M2 macrophages and promote angiogenesis.
Although more and more function mechanisms of TRPM2-AS in cancers are continuously uncovered, most studies are phenotypic studies, or focused on ceRNA network. The depth mechanisms still need us to investigate. TRPM2-AS is an antisense and nearby gene of TRPM2. TRPM2-mediated Ca2+ signaling plays a critical role in regulating the growth and survival of various types of cancer cells. Considering the relationship between TRPM2-AS and TRPM2, there is huge potential for TRPM2-AS to make its role though sense RNA TRPM2 in cancers. Moreover, a recent study showed that TRPM2-AS played an important role in tumor microenvironment of EC, which give us a new direction to study the interaction between TRPM2-AS and tumor microenvironment including immune cells, immune evasion and tumor-immune dynamics etc. In addition, it is still important to use enlarged clinical samples to characterize the role of TRPM2-AS as a clinical indicator more precisely in cancers.
In conclusion, TRPM2-AS plays an oncogenic role in cancers and the underlying mechanisms are complex. The present understanding of TRPM2-AS functions is still limited. Thus, with increasing investigations on TRPM2-AS in cancers, more extensive mechanisms will be understood, leading to clinically targeted therapy for cancers in the future.
Footnotes
Acknowledgements
None.
Author Contributions
XZ contributed to the study conception and design. The first draft of the manuscript was written by SH and BL. The manuscript was revised and edited by XZ. HC, ZY and CR revised part of manuscript. All authors read and approved the final manuscript.
Data Availability
No data was used for the research described in the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics and Consent to Participate Declarations
Not applicable.
Funding
This study was supported by grants from the Science & Technology Department of Sichuan Province [grant number 2022NSFSC1538] and Development and Regeneration Key Laboratory of Sichuan Province [grant number 23LHNBZYB23].
