Abstract
Objective
This study assesses the physical properties of the silicone rubber (SR) bolus, compares its dosimetric characterization with those of gel and thermoset boluses, aiming to evaluate the feasibility and stability of utilizing SR bolus for head photon-beam radiotherapy.
Methods
Three types of boluses (gel, thermoset, and SR) were prepared with same dimensions. Firstly, the physical properties of SR bolus (density, tensile strength and hardness) were assessed pre-irradiation and post-irradiation. Secondly, the percentage of depth dose (PDD) curve was calculated using the treatment planning system (TPS) and measured with a plane-parallel. Thirdly, these three boluses were individually placed on the head phantom, followed by computed tomography (CT) scans to delineate the customized target area and organs at risk (OARs) for treatment planning. Over the subsequent three weeks, fan-beam CT (FBCT) images were obtained weekly and registered with the initial CT to modify the target area and OARs. Fourthly, the adhesion differences, dose distributions, and inter-fraction reproducibility of the three boluses were compared and evaluated.
Results
The results observed that post-irradiated SR bolus exhibited higher hardness values (p < .01) than the pre-irradiated one, with no significant differences in tensile strength and density. The PDD curves of all three boluses showed similarity within a difference range less than ±1%. The air gaps between the SR, thermoset, and gel boluses and skin surfaces were measured as 0.07 cm3, 2.96 cm3, and 4.82 cm3, respectively. Compared with gel bolus and thermoset bolus, the SR bolus demonstrated noticeable inter-fraction reproducibility.
Conclusion
The SR bolus can be fabricated at room temperature, adheres to irregular body surfaces, and retains its physical properties post-irradiation. Additionally, this bolus has comparable dosimetric characterization to other commercial boluses, and may provide notable protection for OARs. Therefore, the SR bolus is recommended as a feasible and stable material for head photon-beam radiotherapy.
Introduction
In the radiotherapy for superficial tumors, a bolus serves as an effective tool to ensure uniform dose distribution to the skin surface. 1 Over recent years, various specialized boluses have been reported and employed clinically. The conventional tissue-equivalent bolus, commonly referred to as the gel bolus, is a standardized product lacking customization and is extensively utilized in radiotherapy. 2 While the gel bolus is reusable and simple to manufacture, it does not accommodate every patient's needs nor all types of tissue. Previous research indicates that the gel bolus exhibits poor adhesion to the irregular body surfaces, resulting in air gaps between the bolus and skin, particularly in areas such as head and neck, or breast tumors.3–6 In contrast, the thermoset bolus demonstrates comparable build-up effects and superior skin conformity relative to the gel bolus.7,8 However, this material soften only at elevated temperatures and rapidly hardens, complicating its fabrication. Additionally, rigid thermoset bolus may compress the exposed wounds and sensitive skin. 9 Recently, the three-dimensional bolus (3D bolus) has emerged and been implemented clinically.10–12 The customized 3D bolus, crafted based on patient-specific body surface data obtained by CT scans, offers improved fit on irregular surfaces, minimizing air gaps. Alternatively, the creation of a 3D bolus is notably time-intensive, often spanning from several hours to days, and necessitates specialized printers and materials. 13
The silicone rubber (SR) has long been used as an impression material for dental repair and filling.14,15 Due to its structural similarities with human soft tissue and excellent biocompatibility, it has increasingly been employed in electron beam and brachytherapy in recent years.16,17 However, the feasibility of this material for conventional 6 MV photon beam radiotherapy remains unknown. Previous research explored the application of SR materials in postmastectomy and indicated that the SR' inherent gravity allows it to conform actively to the breast skin surface contour with high adhesion, which can be molded at room or body temperature after 3–5 min. 18 While these properties suggest that SR could potentially serve as a personalized bolus for individual patients, extending this material for areas with high curvature and unevenness on the nasal surface remains serious challenge. In this study, the SR' physical characteristics and dosimetry properties of three boluses (gel, thermoset, SR) were evaluated and compared, aiming to investigate the feasibility and stability of SR bolus for head photon beam radiotherapy.
Materials and Methods
Three Kinds of Bolus Manufacturing Methods
Gel bolus: Two pieces measuring 10 cm × 10 cm should be cut (Figure 1a) from a standard (25 cm × 25 cm) 5 mm thick gel bolus (Klarity, China). The bolus needs to be fixed on the surface of the nose of the anthropomorphic head phantom (RTsafe, Greece) (Figure 1b).
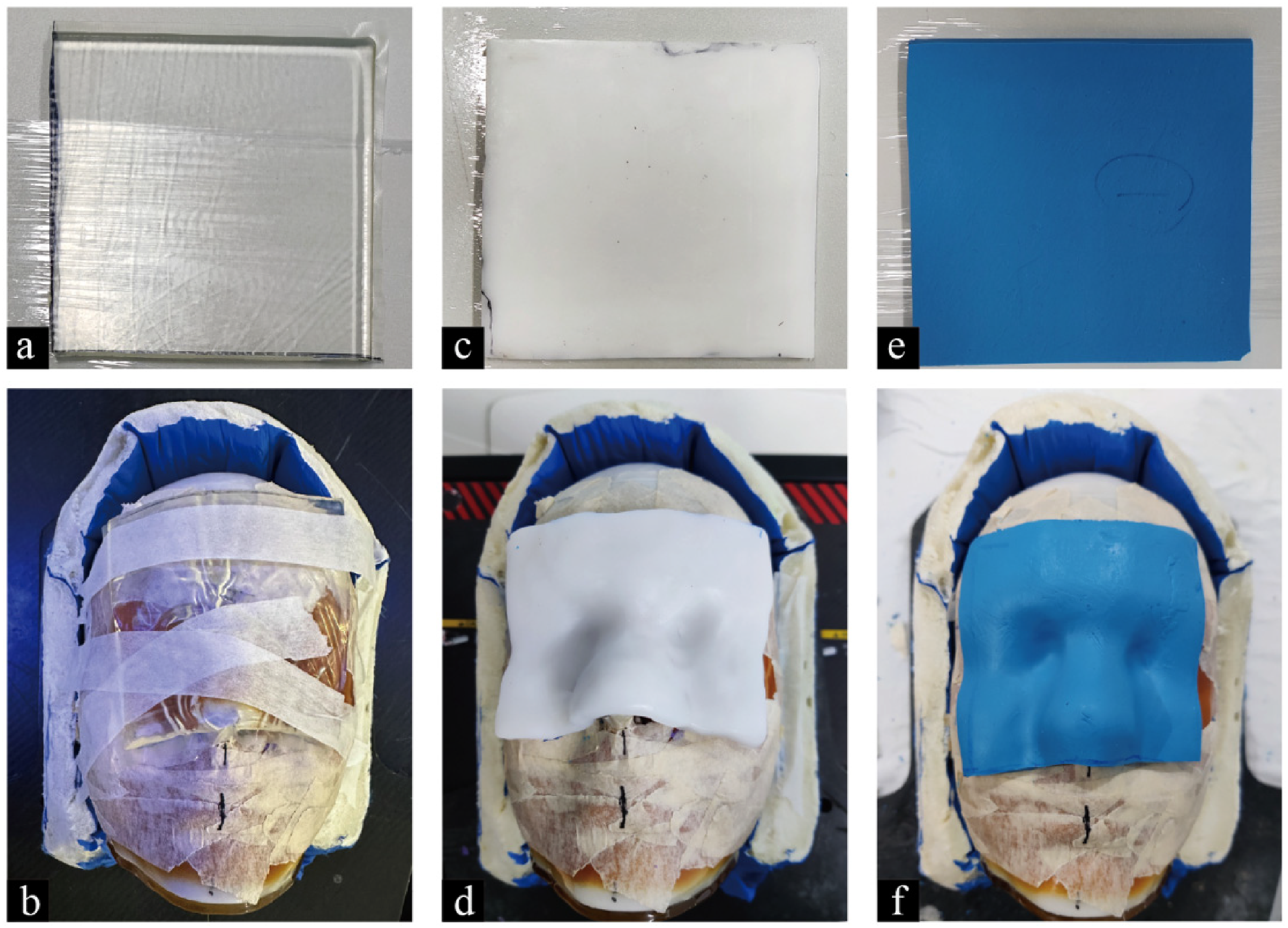
(a-b) the gel bolus with 5 mm thickness and covering the nasal cavity of the head phantom; (c-d) the thermoset bolus with 5 mm thickness and covering the nasal cavity of the head phantom; (e-f) the SR bolus with 5 mm thickness and covering the nasal cavity of the head phantom.
Thermoset bolus: Two pieces of 3 mm thermoplastic materials (Klarity, China) were placed in a 70°C constant temperature water tank. Upon softening, the two pieces were superimposed and cut into a 5 mm thickness of 10 cm × 10 cm size bolus (Figure 1c). The bolus covers the nose of the anthropomorphic head phantom, which is modeled according to the surface contour and cooled (Figure 1d).
SR bolus: The individualized SR bolus was made as previously described. 18 The same weight matrix and catalyst elastomeric initial impression (HUGE, China) are meticulously mixed at room temperature to avoid the formation of air bubbles. Subsequently, the mixture is placed in a mold with a thickness of 5 mm for flattening. After the mixed material cools down and hardens, it is removed from the mold and cut into two SR bolus with the size of 10 cm × 10 cm (Figure 1e). Flattening soft SR bolus with thickness of 5 mm is placed on the nose of an anthropomorphic head phantom, shaping it according to the contour of nose region with pressure of wet towel (Figure 1f).
Physical Characteristics Analysis
The material physical properties of the SR bolus with pre-irradiation and post-irradiation were compared under identical experimental conditions. The drainage method was applied for density analysis of the samples. Additionally, the shore durometer (LX-A, Aidebao, China) and tensile strength detector (WDW-1E, Pinsheng, China) were used to analyze the hardness and tear strength of the samples. After repeating each experiment three times, average value was taken as the final result. The experimental temperature was maintained at a constant 21 °C in the accelerator room. An independent sample test was used to analyze the difference of physical properties between pre-irradiated and post-irradiated SR bolus.
Dosimetric Evaluation
To evaluate the dose accuracy, the treatment plans were created for individualized SR bolus, gel bolus and thermoset bolus. Each bolus was placed on a water-equivalent phantom. The photon beam energy was set to 6 MV at a 0° gantry angle, with an open field size of 10 cm × 10 cm at a source-to-surface distance (SSD) of 100 cm, and the monitor units (MU) were set to 100. The percentage depth dose (PDD) at depths of 0 cm, 0.1 cm, 0.3 cm, 0.5 cm, 0.7 cm, 1 cm, and 2 cm below each bolus were measured using a plane-parallel chamber (PTW34001). The absolute doses at depths of 5 cm, 10 cm, 15 cm, and 20 cm in the phantom were measured using a farmer chamber (PTW30013). We took the average value of three measurements at each depth as the measurement value. For verifying the stability of SR bolus, two additional SR boluses were made with the same size and thickness and measured them at a depth of 0 cm. Furthermore, we calculated the PDD by the Monte Carlo dose calculation algorithm and absolute dose of each bolus using treatment plans system (uRT-TPOIS R001, China) with the same irradiation parameters as those used in the chamber measurements.
19
We compared the PDD of the SR bolus with that of the gel bolus and thermoset bolus, and the dose differences between the measured dose by chamber and the calculated dose by TPS were compared via equation (1) as follows,2,
9
The adhesion evaluation
A head phantom was used to evaluate the degree of adhesion between the bolus and skin surface. In our institution, the styrofoam combined with a thermoplastic mask were applied in head and neck neoplasm for ensuring the treatment accuracy. Each bolus was placed inside the mask as close to the skin as possible, covering the area from eyebrow arch to nose apex. The CT images were obtained with the following parameters: slice thickness = 2.5 mm, tube voltage = 140 kV, tube current = 250 mA, and field of view = 500 mm. The air gap volume was manually contoured in TPS to assess the degree of adhesion.
Comparison of Dose Distributions and inter-Fraction Reproducibility
To assess the clinical feasibility of dose distribution below the SR bolus, the treatment plan on the head phantom was compared with gel bolus and thermoset bolus. The placement method of each bolus and CT scanning parameters refer to the section 2.3. The upper and lower edge of CT scan corresponded to those of the bolus covering the nasal cavity region. The gross tumor volume (GTV) was delineated in the region of nose, with a 3 mm margin added as the plan target volume (PTV). Isotropic expansion of the PTV by 0.5, 1, 2, and 3 cm and intersecting the skin surface was defined as organs at risk (OAR-0.5 cm, OAR-1 cm, OAR-2 cm and OAR-3 cm, respectively), as shown in Figure 2. The planning parameters were set as follows: the x-ray energy = 6 MV; the gantry angle = 0 ~ 180° ; the field size = 10 cm × 10 cm; prescription dose = 200 cGy/fraction. The optimization parameters of the initial plans for the three boluses were consistent, with the dose of D98% normalized to 200 cGy. The D95%, D50%, D2%, homogeneity index (HI), and conformity index (CI) of the PTV, as well as Dmean and D1% of the OARs, were compared for each plan. The formulas for calculating HI and CI are as follows: HI = (D2% − D98%)/D50%, and CI = (VPTV95%)2/ (VPTV × V95%). 20

Visualization of GTV, PTV, OARs (OAR-0.5 cm, OAR-1 cm, OAR-2 cm, and OAR-3 cm) and air gap.
In addition, the fan-beam computed tomography (FBCT) image was scanned weekly in order to register with the initial plan, thereby facilitating the modification of the target and OARs. The initial plan's parameters were replicated without further optimization to calculate the dose of sequential FBCTs. The reproducibility of the bolus was evaluated by calculating the standard deviation of dose distribution between sequential FBCTs and the initial plan.
Results
Physical Characteristics
The physical properties of the SR bolus were analyzed and presented in Table 1. To ensure the stability of the samples, the interval between their formation and the measurement of physical properties is set at 50 days. This duration allows all types of cancer patients to complete the entire treatment process. The post-irradiation SR bolus exhibits higher hardness than its pre-irradiation one (p < .01). Additionally, the results show that there is no significant difference in tensile strength and density between pre-irradiated and post-irradiated SR bolus.
The Analysis of Physical Characteristics of SR Bolus.

Note: Each data represents the mean ± standard deviation.
Dosimetric Evaluation
Figure 3 illustrates the calculated PDD curve using the TPS, alongside the measured PDD values obtained from the chamber, as well as their absolute differences. The PDD curve for the SR bolus closely aligned with those of the gel bolus and thermoset bolus, both before and after the built-up region, with differences remaining within ±1%. For all three bolus types, the dose differences between measured and calculated PDD value were within ±3%, except for the thermoset bolus at a depth of 20 cm, which exhibited a difference of − 3.2%.

Comparison of the percentage depth dose curve of the calculated (line) and measured dose (white circle), and their difference (black circle) for SR bolus, thermoset bolus and gel bolus.
The Adhesion Evaluation of SR Bolus
The volume of air gap between bolus and body surface at initial plan (CT-simulation) and each FBCT is shown in Table 2. The air gaps between SR, thermoset, and gel bolus and skin surface were 0.07 cm3, 2.96 cm3, and 4.82 cm3, respectively. The SR bolus demonstrated superior adhesion and repeatability of bolus placement
The Volume of Air Gap Between Bolus and Skin (cm3) of Gel Bolus, Thermoset Bolus and SR Bolus.

Note: CT-sim = CT-simulation; FBCT = fan beam computed tomography; SD = standard deviation.
Evaluation of the Dose Distribution and Inter-Fraction Reproducibility
The DVH comparison of three boluses is shown in Figure 4. In general, the dose distribution of the SR bolus, gel bolus and thermoset bolus are similar. However, the dosimetric characteristics in the expanded OARs of the SR bolus are slightly lower than those of the thermoset bolus and gel bolus. The PTV dosimetry parameters are shown in Table 3 and Figure 5, and the OARs dosimetry parameters are shown in Table 4 (The form is placed at the end of the manuscript) and Figure 6. Compared to the gel bolus and thermoset bolus, the SR bolus exhibits significantly improved reproducibility of dose distribution

The DVH diagram of gel bolus, thermoset bolus and SR bolus.

Comparison of the PTV parameters of gel bolus, thermoset bolus and SR bolus.

Comparison of the OARs parameters of gel bolus, thermoset bolus and SR bolus.
The Evaluation of PTV Dosimetric Parameters.

Note: Each data represents the mean ± standard deviation. HI = homogeneity index; CI = conformity index.
The Evaluation of OARs Parameters.

Note: Each data represents the mean ± standard deviation. OARs = organs at risk.
Discussion
In this study, we investigated the physical and dosimetric properties of the SR bolus. We found that the physical characteristics of the SR bolus were not affected by head photon irradiation. Compared with gel bolus and thermoset bolus, which are commonly used in clinical practice, SR bolus had similar dosimetry characteristics to those boluses, and also provided better protection against OARs. Notably, the results of inter-fraction repeatability showed that the SR bolus not only had good adhesion, but also the dosimetric parameters of inter-fractions were stable.
The dosimetry property of the SR bolus was evaluated using a water-equivalent phantom, with depth measurements obtained from an ionization chamber. The calculated PDD curve of the SR bolus aligns well with those of the gel bolus and thermoset bolus, indicating that the SR bolus exhibits a similar build-up effect. Meanwhile, the dose difference between the calculated and measured PDD for all three boluses is within 3%
The adhesion of the SR bolus has improved compared to the thermoset bolus and gel bolus, with almost no air gap for irregular body surface. Both the SR bolus and thermoset bolus are custom-shaped to closely fit the body surface in real-time. However, the thermoset bolus requires soaking at 70°C for a few minutes to soften and harden quickly upon cooling, making the shaping process more difficult and posing a risk of burns. Furthermore, we noticed that the edges of the thermoplastic material tend to warp during the cooling and shaping process, which is the main reason for an increased air gap (3.08 cm³) with the skin surface. In contrast, the SR bolus is made at room temperature and has a suitable cooling time to shape carefully. It is worth mentioning that the adhesion of thermoplastic and SR materials is highly stable, with no significant difference in the air gap observed from FBCTs images (0.06 cm³ vs 0.07 cm³, 3.07 cm³ vs 2.98 cm³). The maximum air gap of the gel bolus is related to its own softness, and the cavity volume between FBCTs and initial CT didn't show a significant increase (4.71 cm³ vs 4.85 cm³).
Positioning repeatability is crucial for ensuring accuracy in radiotherapy across dozens of treatment fractions. Previous studies were confined to the initial plan's dose distribution, necessitating further investigation to determine if these dosimetric characteristics can be consistently replicated in subsequent treatments.9,21 In clinical application, the thermoset bolus and the SR bolus are typically integrated within the thermoplastic mask. However, in case of exposed wounds, the hardness and pressure of the former will increase skin sensitivity and pain. Conversely, the SR bolus is softer and more skin-friendly. Conventionally, poor adhesion of gel bolus is still a challenge for inter-fractional setup repeatability at each irradiation.10,21 In our study, the standard deviation of inter-fractions' dosimetry parameters was used as an evaluation metric. We observed that both the SR bolus and thermoset bolus performed with better reproducibility and stability in target dose distribution among inter-fractions compared to the gel bolus. Nevertheless, when applying the same prescription dose, the thermoset bolus achieved the lowest actual PTV dose (D98% 199.22 cGy), while the dose to the OARs was the maximum one. Considering all factors comprehensively, the SR bolus emerged as the most consistently reliable option, with its target dose distribution being equal to or surpassing other bolus types, and also reducing the dose absorbed by the OARs.
Several studies have employed SR material as a bolus. Chatchumnan et al confirmed that an SR-based bolus is suitable for electron beam treatment. 11 Canters et al initially filled the 3D-printed shell, generated from the electron beam treatment plan, with SR to fabricate the 3D-SR bolus. The subsequently performed a second CT scan to verify the geometric accuracy, a process that was both time-consuming and involved repetitive radiation exposure. 22 In our study, the SR bolus has well individuality in clinical practice, allowing for customized fitting according to each patients' tissue structure. A molding time of approximately 5 min is sufficient to mimic the tissue shape, making it clinically acceptable. Furthermore, following irradiation treatment, the SR bolus demonstrates a slight increase in hardness, while still maintaining the stability of its inter-fraction reproducibility and dose distribution. Hardness is characterized by its resistance to indentation and typically indicates the material's level of softness or rigidity, and the phenomenon of increased hardness potentially related to the material's intrinsic characteristics and tends to enhanced with an increase in radiation dose. 23
We also conducted a comparative analysis of the cost implications between the three bolus types. It is crucial to note that customizing commercial boluses to match the shape of a treatment field can lead to increased expenses. Both gel bolus and thermoset bolus necessitate shearing from a single piece of material and cost 2–4 times that of SR bolus, with gel bolus being the highest. In addition, there are two primary limitations to this study. Firstly, we solely examined the head site, which is one of the most common scenarios of surficial radiotherapy. Secondly, since the 0.5 cm is the most common bolus thickness used in clinical settings, we evaluated only this thickness. In future research, we plan to assess the performance of the SR bolus in other body regions and the influence of different thicknesses.
Conclusion
The SR bolus can be efficiently fabricated at room temperature. It actively adheres to the uneven body surface, forming a stable fixed connection with the thermoplastic mask. It maintains its physical properties after irradiation. In addition, this bolus has comparable dosimetric characterization and distribution to the gel bolus and thermoset bolus. It may provide notable protection for OARs. Therefore, the SR bolus is recommended for surficial radiotherapy procedures employing head photon beam technology.
Footnotes
Authors’ Contributions
Xue-mei Chen and Wei Wang carried out the data collection, statistical analysis and drafted the manuscript. Xiao-tong Huang and Wen-chao Diao participated in the data collection and statistical analysis. Xiao-tong Huang, Wen-chao Diao and Chen-di Xu reviewed the image registration. Le-cheng Jia and Hua Li exported the data and made technical guidance. Bin Li and Xiao-bo Jiang revised and finally approved the manuscript. All authors contributed to the article and approved the submitted version.
Data Sharing Statement
The data that support this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the crosswise project for Shanghai United Imaging Healthcare Co., Ltd. (grant number ZLDL-UIH-2022007), Guangdong Basic and Applied Basic Research Foundation (grant number 2020A1515110352), Special Scientific Research Fund for Medical Physics & Technology of Guangdong Biomedical Engineering Society (grant number 2022YXWL0101).

