Abstract
Keywords
Introduction
The incidence rate of lung cancer is the highest in the world. 1 The overall 5-year survival rate is only 16%–18%.2,3 The number of lung cancer cases and deaths in China has been ranked first among malignant tumors for 10 consecutive years. There were about 2,206,771 new lung cancer cases every year, of which non-small cell lung cancer (NSCLC) accounts for 85% and about 1,796,144 deaths due to lung cancer. 4 The basic treatment principle of lung cancer is to choose the appropriate treatment according to the tumor lymph node metastasis (TNM) stage. However, due to the heterogeneity of NSCLC, there are obvious individual differences in the treatment effect. At present, it is difficult to meet the needs of clinical treatment only based on TNM staging. Therefore, it is urgent to find more tumor markers closely related to NSCLC as therapeutic targets. 5
Tumor M2-pyruvate kinase (TuM2-PK) is a key enzyme in the glycolysis pathway and synthesis of nucleoside triphosphate. The expression level of TuM2-PK was increased in the serum of patients with various tumors, which was involved in the proliferation, migration, and metastasis of tumors.6-9 Carcinoembryonic antigen (CEA) is a glycoprotein with specific epitopes of human embryonic antigen, which is elevated in many tumors, such as lung cancer, digestive tract tumors, urogenital tumors, and breast cancer. 10 It has clinical application in monitoring the condition of lung cancer and judging the curative effect and prognosis. 11 CYFRA21-1 is highly expressed in various types of tumor epithelial cells, and gradually released into the blood circulation with tumor cell apoptosis. 12 In recent years, research projects have found that the concentration of TuM2-PK in the peripheral blood of tumor patients has significantly increased, which can be used for early diagnosis, prognosis assessment, and therapeutic monitoring of tumors. This study evaluated the diagnostic and prognostic value of TuM2-PK in NSCLC by detecting changes in serum levels in patients with NSCLC and benign lung diseases, and comparing it(them) with traditional CEA and CYFRA21-1 in NSCLC.
Materials and Methods
Patients
The subjects included 3 separate groups. The first group was 184 NSCLC patients with complete clinical data treated in our hospital from 2013 to 2015. The diagnostic methods of lung cancer include sputum cytology, computed tomography (CT) guided percutaneous lung biopsy, bronchoscopy, or surgical thoracoscopy. A pathologist interprets the cytology or histology of a biopsy. For patients diagnosed as primary NSCLC according to the 2015 World Health Organization Classification of Lung Tumors, staging was conducted according to the 8th edition of TNM classification. Staging examination of lung cancer includes clinical examination, CT of the chest, abdomen, and brain, abdominal ultrasound, bone scan, and positron emission CT. The second group included 60 sex- and age-matched consecutive cases with benign pulmonary tuberculosis, pneumonia, pulmonary mycosis, organized pneumonia, and pulmonary abscess. The third group included 90 healthy volunteers. The serum samples of the subjects were obtained from the Central Laboratory of Nanjing Brain Hospital. All 3 groups excluded the existence of other tumors. There was no significant difference in age and sex among the 3 groups. The study has been reported in accordance with the Standards for Reporting Diagnostic Accuracy Studies checklist. 13
The study was approved by the Ethics Committee of Affiliated Nanjing Brain Hospital, Nanjing Medical University (April 2010, approval number: NJXK202038), Nanjing, China and was carried out in accordance with national law and the current revised Declaration of Helsinki. All patients provided written informed consent before enrollment.
Measurement of serum TuM2-PK, CEA, and CYFRA21-1 Concentrations
After diagnosis, the patient took 3 mL of fasting venous blood before treatment. Centrifuge the sample at a speed of 2500 revolutions per minute for 15 min, and immediately store the supernatant at −80 °C until use. Electrochemiluminescence immunization (Roche Cobas e601) was applied to detect CEA and CYFRA21-1. For TuM2-PK testing, the serum sample was tested using a commercially available rapid test, ScheBo TuM2-PK Quick (ScheBo Biotech AG, Giessen, Germany) according to the manufacturer's instruction. Carry out the test on the automatic biochemical analyzer in strict accordance with the instructions of the kit. All tests were in duplicate and diluted appropriately, and the technicians were unaware of the clinical data. The positive results of the 3 markers were CEA >5.0 ng/mL, CYFRA21-1 >3.3 ng/mL, and TuM2-PK >15.0 units/mL.
Follow-up
All patients were followed up every 3 months after discharge. Chest CT, abdominal B ultrasound, head magnetic resonance imaging, and bone scanning were performed. Follow-up data were obtained through outpatient reexamination, telephone, and WeChat communication. All patients were followed up for 16 to 60 months, with an average of 37 months. The endpoint of follow-up was death or the deadline of follow-up.
Statistical Analysis
SPSS 20.0 statistical software was used for data processing. The measured data were expressed as (x ± s) according to the normal distribution. The one-way analysis of variance was used for comparison of multiple groups, and 2 independent samples t-test was used for comparison of 2 groups. The counting data were analyzed by using the χ2 test. The receiver operating characteristic (ROC) curve was drawn to evaluate the diagnostic value of TuM2-PK, CEA, and CYFRA21-1 in NSCLC, and the area under the curve (AUC) was calculated. Cox proportional hazard regression analysis was used to analyze survival-related variables. The Kaplan–Meier survival curve was drawn to evaluate the survival status of lung cancer patients with different levels of serum TuM2-PK, CEA, and CYFRA21-1. The logarithmic rank test was used to compare 1-year, 3-year, and 5-year survival rates. P < .05 was the difference and was statistically significant.
Results
Clinical Characteristics
One hundred male patients and 84 female patients with NSCLC were included in this study. Seventy (38.0%) were in stages I and II, 82 (44.6%) were in stage III, and 32 (17.4%) were in stage IV. Histopathological types were squamous cell carcinoma (SCC) in 69 patients (37.5%) and adenocarcinoma in 115 (62.5%). Sixty-seven (36.4%) cases were well differentiated, 59 (32.1%) cases were moderately differentiated and 58 (31.5%) cases were poorly differentiated.
Serum Levels of TuM2-PK, CEA, and CYFRA21-1 Were Elevated in NSCLC Patient
The serum levels of TuM2-PK were significantly higher in the NSCLC group compared with the benign lung disease (BLD) group (P < .01) and the healthy controls (HC) group (Figure 1A, P < .05). The serum levels of CEA and CYFRA21-1 were also higher in the NSCLC group compared with the BLD and the HC groups (Figure 1B and C, P < 0.05). There was no significant difference in serum levels of TuM2-PK, CEA, and CYFRA21-1 between the BLD group and the HC group (P > .01).
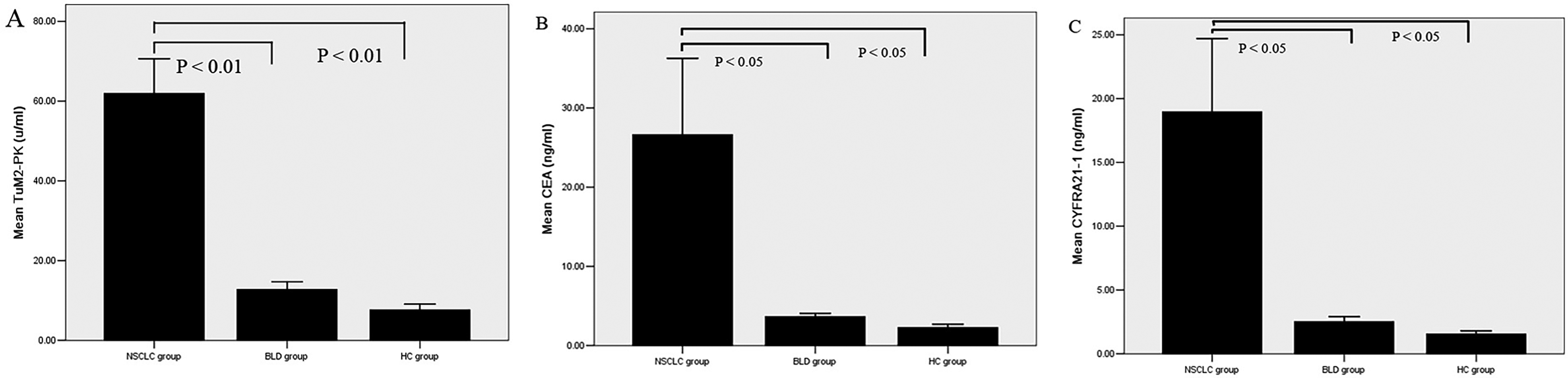
Serum levels of TuM2-PK (A), CEA (B), and CYFRA21-1 (C) in NSCLC patients, BLD patients, and HC.
Comparison of Serum TuM2-PK, CEA, and CYFRA21-1 Levels Depending on Clinical Characteristics in NSCLC Patients
The serum levels of TuM2-PK were higher in stage IV than those in stages I–III (P < .05). In addition, the serum levels of CEA and CYFRA21-1 increased with staging, and the serum CEA and CYFRA21-1 levels in stage IV NSCLC were higher than those of early NSCLC (P < .01; Table 1).
Comparison of Serum TuM2-PK, CEA, and CYFRA21-1 Levels Depending on Clinical Characteristics in NSCLC Patients.

Abbreviations: NSCLC, non-small cell lung cancer; TuM2-PK, tumor M2-pyruvate kinase; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin 19 fragment; ADC, adenocarcinoma; SCC, squamous cell carcinoma; TNM, tumor lymph node metastasis.
The levels of TuM2-PK, CEA, and CYFRA21-1 in patients with lymph node metastasis and distant metastasis were higher than those in patients without lymph node metastasis and distant metastasis (P < .05). The level of CEA in patients with adenocarcinoma was higher than that in patients with SCC, while the level of CYFRA21-1 was lower than that in patients with SCC (P < .05).
There was no significant correlation between TuM2-PK, CEA, and CYFRA21-1 expression and age, gender, and differentiation (Table 1).
The Diagnostic Value of TuM2-PK, CEA, and CYFRA21-1
ROC curves were plotted to determine the diagnostic efficiency of serum TuM2-PK, CEA, and CYFRA21-1 levels for NSCLC. The measurements of the different individual markers and their predictive value in the diagnosis of NSCLC were summarized in Table 2. Among the 3 biomarkers, TuM2-PK displayed the highest AUC (AUC = 0.814, 95% CI: 0.728-0.899; Figure 2A and Table 2) and satisfactorily discriminated NSCLC patients from the controls. CEA (AUC = 0.638, 95% CI: 0.529-0.747; Figure 2B) and CYFRA21-1 (AUC = 0.719, 95% CI: 0.614-0.824; Figure 2C) exhibited a better AUC in differentiating NSCLC patients from the controls (Table 2).

ROC curves for the serum levels of TuM2-PK (A), CEA (B), CYFRA21-1 (C), and TuM2-PK + CEA + CYFRA21-1 (D) in differentiating NSCLC patients and healthy controls.
Diagnostic Value of Serum Levels of TuM2-PK, CEA, and CYFRA21-1 and Their Combination in NSCLC Patients.

Abbreviations: TuM2-PK, tumor M2-pyruvate kinase; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin 19 fragment; NSCLC, non-small cell lung cancer; AUC, areas under the curves.
The combination of TuM2-PK, CEA, and CYFRA21-1 improved the classification capacity and yielded a better optimal diagnostic efficacy for NSCLC patients (AUC = 0.918, 95% CI: 0.856-0.980; Figure 2D and Table 2) than TuM2-PK, CEA, or CYFRA21-1 alone (P < .01).
Correlation of Serum TuM2-PK, CEA, and CYFRA21-1 Levels With Overall Survival
Next, we analyzed the association between serum levels of TuM2-PK, CEA, and CYFRA21-1 with the overall survival of patients with NSCLC by performing the Kaplan–Meier survival analysis. We found that the 1-, 3-, and 5-year survival rates of NSCLC patients with positive TuM2-PK, CEA, and CYFRA21-1 were lower than those of patients with negative TuM2-PK, CEA, and CYFRA21-1 (P < .05, Figure 3A–C and Table 3). The follow-up time of these 184 patients was from 16 to 60 months, with a medium time of 37 months. The Cox regression equation was employed to identify the potential factors that affect the overall survival of patients with NSCLC. Patients with positive TuM2-PK, CEA, and CYFRA21-1 were found to survive significantly shorter than patients with negative TuM2-PK, CEA, and CYFRA21-1 (P < .05).

Kaplan–Meier survival curve for NSCLC patients with different serum levels of TuM2-PK (A), CEA (B), and CYFRA21-1 (C).
Comparison of 1-, 3-, and 5-Year Survival Rates in NSCLC Patients With Different Serum Levels of TuM2-PK, CEA, and CYFRA21-1
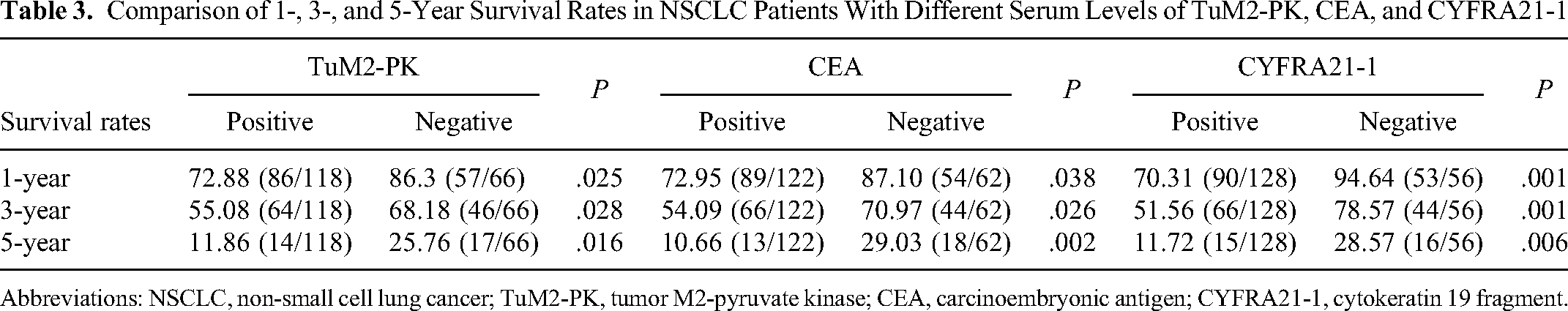
Abbreviations: NSCLC, non-small cell lung cancer; TuM2-PK, tumor M2-pyruvate kinase; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin 19 fragment.
Discussion
Lung cancer is often occult. Most patients have been diagnosed as advanced stage and have a poor prognosis. A large number of studies have confirmed that early diagnosis and treatment are the most effective ways to prevent and reduce mortality. Serological indicators in the diagnosis of lung cancer have the advantages of short diagnosis time and less trauma to patients, but their diagnostic accuracy and sensitivity are low, and their clinical application is limited.14,15
Pyruvate kinase is a key enzyme in the glycolytic pathway and plays a decisive role in the synthesis of triphosphate nucleotides. There are 4 isoenzymes of pyruvate kinase in mammals, namely L type, R type, M1 type, and M2 type, and the distribution of these 4 isoenzymes has relative tissue specificity. 16 In normal cells, M2-pyruvate kinase mainly exists in the form of the trimer, while in tumor cells, TuM2-PK is highly expressed and transforms into the mainly dimeric forms. Under the catalytic action of the oncogene-encoded kinase, both Ser and Wyr positions of TuM2-PK undergo phosphorylation. Due to the need for low-activity M2-pyruvate kinase in tumor cells to maintain their abnormal metabolic activity, low-activity M2-pyruvate kinase is overexpressed and appears in body fluids. 17
Previous studies have shown that the expression of TuM2-PK, CEA, and CYFRA21-1 is low in normal tissues, but they are highly expressed in tumor cells. 18 With the development of the disease, tumor cell apoptosis and autophagy increase, and cell components entering the blood circulation also increase. Therefore, the serum levels of TuM2-PK, CEA, and CYFRA21-1 increase.19,20 The results showed that the serum levels of TuM2-PK, CEA, and CYFRA21-1 in the NSCLC group were higher than those in the BLD and HC groups. The serum levels of TuM2-PK, CEA, and CYFRA21-1 in patients with TNM stages III and IV were higher than those in patients with TNM stages I and II, and the serum levels of TuM2-PK, CEA, and CYFRA21-1 in patients with TNM stage IV were higher than those in patients with TNM stage III, suggesting that the levels of serum TuM2-PK, CEA, and CYFRA21-1 increased with the increase of the TNM stage in patients with NSCLC. The reason may be that the higher the TNM stage, the higher the degree of NSCLC deterioration, the more likely tumor cells to metastasize, and because tumor cells often complete the metastasis process in the blood circulation system, the more tumor cells there are in the blood circulation, the higher the levels of TuM2-PK, CEA, and CYFRA21-1 in the blood. The results are consistent with those of some scholars at home and abroad.21,22 In different pathological types of lung cancer, the level of CEA was significantly higher in adenocarcinoma patients than in SCC, indicating that CEA can be used as an auxiliary diagnosis for adenocarcinoma, which was similar to previous studies.23,24 Previous studies have reported that the CYFRA21-1 levels in SCC were significantly higher than those in adenocarcinoma and small-cell carcinoma, which was consistent with the results of this study. 25
NSCLC is a multi-gene disease, and its occurrence and development involve a series of protein molecules or gene changes, so a single protein cannot fully explain the pathogenesis of NSCLC. 26 The results showed that the AUC of serum TuM2-PK, CEA, and CYFRA21-1 levels in the diagnosis of NSCLC were 0.814, 0.638, and 0.719, respectively, and the AUC of combined diagnosis of TuM2-PK, CEA, and CYFRA21-1 was 0.918, suggesting that serum TuM2-PK, CEA, and CYFRA21-1 and their combination have certain diagnostic value for NSCLC. The results also showed that the 1-, 3-, and 5-year survival rates of TuM2-PK, CEA, and CYFRA21-1 positive NSCLC patients were lower than those of TuM2-PK, CEA, and CYFRA21-1 negative NSCLC patients, respectively. The reasons may be that the mitogen-activated protein kinase (MAPK) signaling pathway is one of the signal transduction pathways closely related to the pathogenesis of NSCLC, while TuM2-PK, CEA, and CYFRA21-1 can activate the intracellular MAPK signaling pathway, then promote the development of NSCLC.27,28 Therefore, NSCLC patients with serum TuM2-PK >15 units/mL, CEA >5.0 ng/mL, and CYFRA21-1 >3.3 ng/mL should be paid more attention to, and effective interventions should be made to improve the prognosis of patients.
Several limitations of our study warrant discussion. First, we performed the study at a single center with a relatively small sample size. Second, the expression of TuM2-PK in the serum of lung cancer patients was detected, but the expression of TuM2-PK in lung cancer tissues was not detected. Third, the specific mechanism of the relationship between TuM2-PK expression and NSCLC was lacking. Further perspective trials should be performed.
Conclusion
In conclusion, the serum levels of TuM2-PK, CEA, and CYFRA21-1 increased with the increase of the TNM stage in NSCLC patients. Serum TuM2-PK, CEA, CYFRA21-1, and their combination have certain diagnostic values for NSCLC. The prognosis of NSCLC patients with serum TuM2-PK >15 units/mL, CEA >5.0 ng/mL, and CYFRA21-1 >3.3 ng/mL is poor. Clinicians should pay more attention to NSCLC patients with elevated serum levels of TuM2-PK, CEA, and CYFRA21-1. Our results showed that serum TuM2-PK, CEA, and CYFRA21-1 have clinical value in the diagnosis and prognosis of NSCLC. However, the mechanism of the relationship between serum TuM2-PK, CEA, and CYFRA21-1 levels and NSCLC is still unclear and needs further study.
Footnotes
Abbreviations
Author Contributions
Y.W. and C.H.X. carried out most of the experiment and wrote this manuscript; Q.Z. and Y.C.W. did the enzyme-linked immunosorbent assay; Y.H.X. collected data; L.L. and C.H.X. helped the design and all through the research. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a grant from the Medical Scientific Research Project of Jiangsu Provincial Commission Health and Family Planning (H2017048).
