Abstract
Introduction
Pelvic malignancies are common cancers in human beings. According to the latest statistics in the United States, the incidence of pelvic malignancies is about 28.2%, including cancers of the reproductive system, rectal, anus, anal canal, anorectum, and urinary bladder. 1 Radiotherapy is one of the most important treatments for pelvic malignancies, and hematological toxicity (HT) is a common side effect. Severe HT may lead to treatment interruption, infection, and affect the dose and cycle of chemotherapy, potentially endangering the prognosis of patients. 2
Bone marrow is an important hematopoietic organ and can be divided into red marrow, also known as active bone marrow (ABM), which has hematopoietic function, and yellow marrow, which does not have hematopoietic function. In adults, about 50% of the ABM is located in adjacent vertebrae, pelvis, and femoral neck. 2 Cytotoxic drugs and radiation can induce the differentiation of ABM into adipocytes and increase the fat content of bone marrow, thus inhibiting hematopoiesis. 3
Some studies have explored the relationship between dose-volume characteristics of anal 4 and cervical cancer4–9 patients’ ABM using positron emission tomography (PET)/computed tomography (CT) due to rising concern for ABM conservation during pelvic radiotherapy. Although PET/CT has some advantages in identifying ABM, there is no uniform standard for the optimal uptake value of ABM, and it is difficult to apply PET/CT to every patient considering the actual situation. In contrast, magnetic resonance imaging (MRI) technology is relatively mature, and ABM can be well distinguished on MRI because of the difference in composition.10,11 Similar to skeletal muscle on the MRI T1 sequence, the ABM comprises 20% protein, 40% water, and 40% fat. On the other hand, yellow bone marrow has a high signal on the MRI T1 sequence, with 80% fat, 15% water, and 5% protein.12,13
Several studies have established a correlation between ABM dose-volume parameters and HT.2,5–7 However, the optimal technique for identifying pelvic ABM remains unclear. Moreover, no study has examined the relationship between ABM dose-volume parameters and HT in patients with pelvic malignancies undergoing radiotherapy, irrespective of tumor type. To address this gap, we conducted a study aimed at evaluating the clinical value of MRI-based ABM delineation in patients with pelvic malignancies. Specifically, we analyzed the association between HT and dose-volume parameters of both ABM and total bone marrow TBM.
Materials and Methods
We conducted a retrospective analysis of data from 116 patients with pelvic tumors who underwent radiotherapy at the First Affiliated Hospital of Guangxi Medical University between January and July 2022. All patient details have been de-identified. The inclusion criteria included: (1) age ≥18 years; (2) all patients were diagnosed with pelvic malignancies by pathology; (3) the blood routine and liver and kidney function tests were normal before radiotherapy; (4) the pelvic MRI was completed before radiotherapy; (5) perform a complete blood count every week at our hospital during treatment; and (6) complete and searchable medical records were available. The exclusion criteria included: (1) previous history of chemotherapy or pelvic radiotherapy; (2) previous history of other cancer; and (3) combined with hematopoietic or infectious diseases. This study was approved by the Ethics Committee of the hospital (IRB: 2022-E468-01). The reporting of this study conforms to STROBE guidelines. 14
All patients received pelvic intensity-modulated radiotherapy (IMRT). IMRT plans were generated with 7-9 coplanar fields using a 6-MV photon beam. The prescribed dose for the PTV of the IMRT plan was 45-60 Gy/25-30 F. If patients had positive lymph nodes, a concomitant boost dose was given to these areas, and the prescribed dose for PGTVnd was 56-66 Gy/25-30 F. CT simulation and pelvic MRI scans were completed before radiotherapy, and then the data sets of CT images were imported into the Varian treatment planning system. The pelvic contour generated by the system was used as a surrogate for TBM, defined as the contoured volume of the sacrum, coccyx, hip bones (ilium, pubis, and ischium), L4/L5 vertebral bodies, and proximal femur to lesser trochanter, as illustrated in Figure 1A.

Example of pelvic ABM and TBM delineation. (A) The TBM on the CT image (the green line). (B) MRI T1 sequence image. (C) The ABM on MRI T1 sequence (the red line). (D) The ABM and TBM on CT-MRI fusion.
Fusion of pelvic MRI with localization CT was performed to outline ABM (Figure 1B-D). MRI T1 sequence allows for accurate determination of bone marrow cell content.11,15 Adipose tissue can be identified by its short T1 relaxation time and high signal intensity in T1 sequences, whereas yellow bone marrow can be differentiated from ABM based on its high-fat content.11,15 ABM shows reduced signal intensity, that is, lower than subcutaneous fat, but higher than intervertebral disc or muscle tissue. The ABM was delineated based on T1 sequences, where regions with signal intensities similar to or slightly higher than that of muscle were identified. To reduce subjectivity, one radiation oncologist outlined the ABM, which was then reviewed and modified by a senior physician. If there was any apparent inconsistency between two radiation oncologists, a chief radiation oncologist will re-evaluate it and make the final decision.
Our dose constraints for organs at risk (OARs) are as follows, TBM V10 ≤ 90%, V20 ≤ 75%, small intestine V40 ≤ 30%, V50 ≤ 10%, rectum V40 ≤ 60% (except for rectal and anal canal cancer), bladder V50 ≤ 50%, femoral head V30 ≤ 15%, Dmax ≤ 50Gy, spinal cord Dmax ≤ 42 Gy. Priority was given to balancing the target coverage and dose limitation requirements of important OARs such as the small intestine, rectum, bladder, and femoral head. Finally, the dose constraints of TBM were pursued as far as possible and were not mandatory to meet all requirements.
All patients were required to undergo blood routine examination every week during the treatment period and increase the frequency of examination if necessary. The lowest values of white blood cell (WBC), absolute neutrophil count (ANC), hemoglobin (Hb), platelet (PLT), and lymphocyte (Lym) were recorded from the beginning to 3 months after the end of radiotherapy and graded according to the Common Terminology for Adverse Events (CTCAE). Grade 2 or higher HT (HT2+) was defined as any grade 2+ toxicity for WBC, ANC, Hb, and PLT, but did not include Lym.
For the calculation of sample size, we set β and α to 0.1 and 0.025, respectively. According to the reductive effect on HT in cervical cancer patients, we hypothesized ABM group was able to reduce Grade ≥ 2 HT from 62.1% to 33.3%. 16 The sample size of two groups was estimated to be 116 by two-sided test.
Statistical Analyses
SPSS26.0 software was used for data analysis. In the descriptive statistical analysis, continuous variables were expressed by mean ± SD, and categorical variables were expressed by frequency and percentage. For paired sample analysis, we first tested the normality of each group of data. For those with normal distribution, we used the paired sample t-test, and for those who do not conform to the normal distribution, we used Wilcoxon signed rank test. Linear regression models were used to analyze the correlation between dose-volume parameters and the lowest values of blood cell count parameters (WBC, ANC, Hb, PLT, and Lym). Univariate and multivariate logistic regressions were used to analyze the correlation between HT2+ and dosimetric parameters, and then the receiver operating characteristic (ROC) curve was used to evaluate the value of dosimetric parameters in predicting HT.
Results
Patient Characteristics
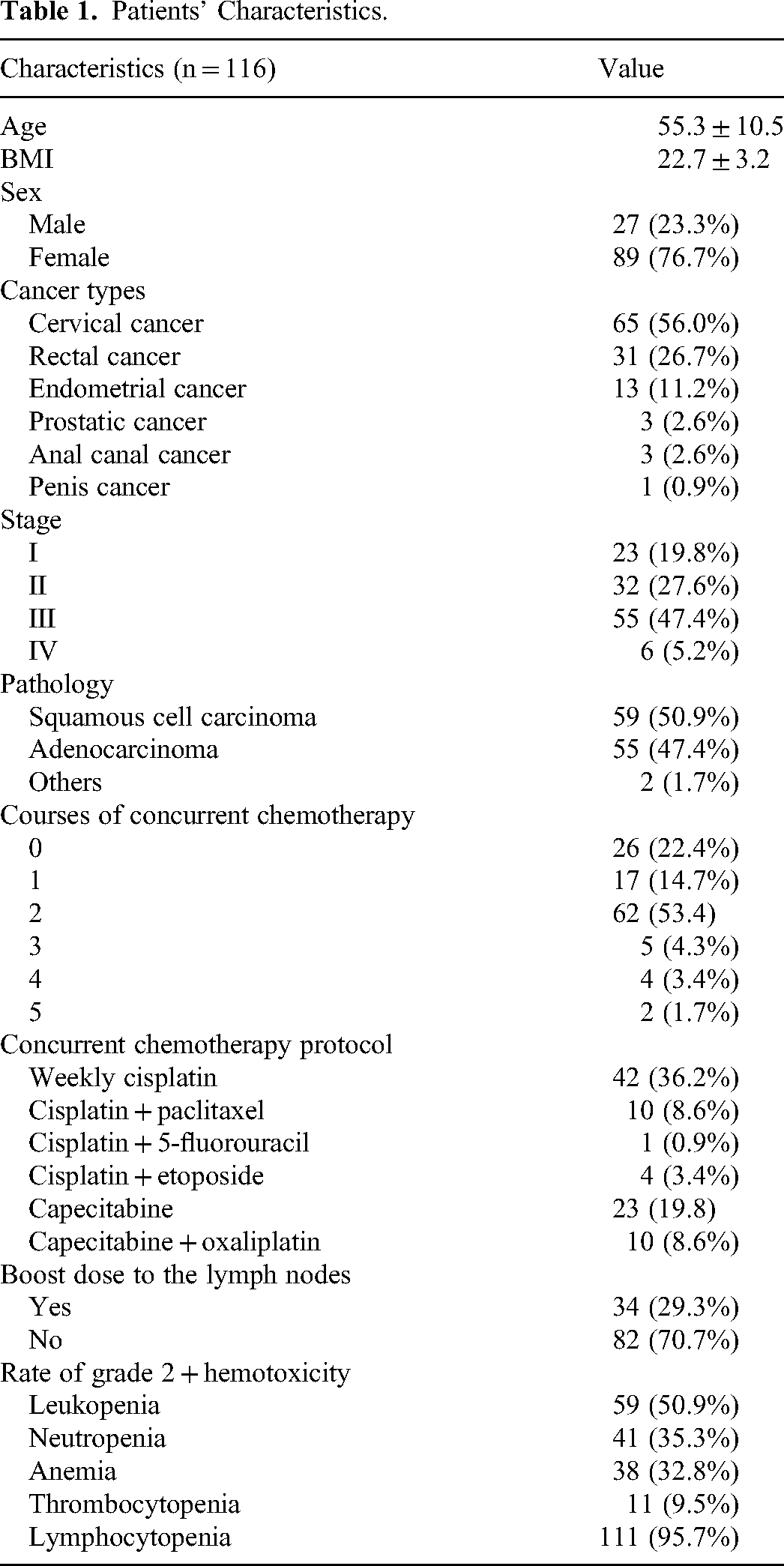
The patient characteristics were shown in Table 1. The average age of the patients was 55.3 ± 10.5 years and the mean BMI was 22.7 ± 3.2. There were 27 men and 89 women. Among all patients, cervical cancer accounted for 56.0%, rectal cancer for 26.7%, and endometrial, prostate, anal canal, and penile cancers together accounted for 17.3%. There were 23 patients with stage I, 32 patients with stage II, 55 patients with stage III, and 6 patients with stage IV. The pathological classification of patients was mainly squamous carcinoma accounting for 50.9%, adenocarcinoma accounting for 47.4%, and other types of carcinoma accounting for 1.7%. Ninety patients (77.6%) received concurrent chemotherapy during radiotherapy, of which 2 courses accounted for the largest proportion. Weekly cisplatin (36.2%) accounts for the largest proportion of concurrent chemotherapy protocols, followed by capecitabine (19.8%). Thirty-four patients (29.3%) had a simultaneous integrated boost dose to their positive lymph nodes. Of the 116 patients, 68 (58.6%) had HT2+. And of these patients, 59 had grade 2 or higher leukopenia, 41 had grade 2 or higher neutropenia, 38 had grade 2 or higher hemoglobin decrease, 11 had grade 2 or higher thrombocytopenia, and 111 had grade 2 or higher lymphocytopenia. The changing trend of WBC, ANC, Hb, PLT, and Lym over time is shown in Figure 2.

Weekly blood cell count ratio variation during the treatment course.
Patients’ Characteristics.
Dose-Volume Parameter
The radiation dose-volume indicators were shown in Figure 3. ABM-V5 (98.16%), ABM-V10 (94.04%), ABM-V15 (90.64%), ABM-V20 (84.58%), ABM-V25 (72.07%), ABM-V30 (62.24%), ABM-V35 (51.45%), and ABM-V40 (38.99% ± 6.44%) were significantly lower than TBM-V5 (98.99%), TBMV10 (95.38%), TBM-V15 (92.83%), TBM-V20 (87.34%), TBM-V25 (77.52%), TBM-V30 (65.97%), TBM-V35 (55.12%), and TBM-V40 (41.33%±8.35%) (

Averaged dose-volume histogram comparison showing ABM and TBM dosimetric parameters in pelvic cancer patients. ***
Logistic Regression Analysis
Univariate and multivariate logistic regression analysis was used to establish the correlation factors of HT2+ in pelvic malignancies (Table 2). The single-factor analysis suggested that sex, BMI, courses of concurrent chemotherapy, boost dose to the lymph nodes, ABM V5, ABM V10, ABM V15, ABM V20, and TBM V5 were related to HT2+ (
Univariate and Multivariate Logistic Regression Analysis of Factors Associated with the Development of Grade 2+ Hematologic Toxicity.

Linear Regression Analysis
Linear regression analysis was performed between dosimetric parameters and blood cell nadir. The WBC nadir was significantly correlated with ABM V10 and ABM V15 (
Linear Regression Model Parameters.

ROC Curve
We performed ROC curve analysis on ABM parameters. ABM V5 (>99.34%, AUC = 0.64,
Receiver Operating Characteristic Curves Analysis for Grade 2+ Hematologic Toxicity.

Discussion
Radiotherapy plays an important role in the treatment of pelvic malignancies such as cervical and rectal cancer. Study has shown that the low lumbar spine, sacrum, iliac bone, and proximal femur contain about half of the total ABM in the body. 17 The pelvic bone is the main hematopoietic site of body, and this part of bone marrow is affected during pelvic radiation therapy. Myelosuppression is a common adverse reaction in patients with pelvic malignancies during treatment. It has been reported that 69.5% of patients with cervical cancer receiving chemoradiotherapy have HT2+. 18 HT has been reported to be associated with ABM related parameters in cervical,19,20 rectal,21,22 and anal cancer.4,23 This report is the first, to our knowledge, to investigate the correlation between MRI-based ABM radiation dose-volume parameters and HT occurrence in patients with pelvic malignancies, irrespective of cancer type.
At present, 99mTc sulfur colloid SPET, 5 atlas,9,24 18F-FDG-PET, 6 MRI,21,25 and other imaging methods have been used to distinguish ABM. Wang et al 5 found that ABM volume, ABM-V30, and ABM-V40 were significantly associated with grade 3+ HT in 39 patients with cervical cancer who defined ABM based on 99mTc sulfur colloid SPET. Meng et al 6 baseline 18FDG PET/CT pelvic ABM volume percentage helps predict the severity of HT in patients with locally advanced cervical cancer undergoing chemoradiotherapy. Although PET/CT has some value in identifying ABM, it is not acceptable to every patient due to its high price. Kuncman et al 21 found that ABM based on MRI delineation was predictive of absolute lymphocyte count nadir and platelet nadir (PLTnadir%) in rectal cancer. MRI examination is relatively common, and red and yellow bone marrow is easy to be identified on the T1 sequence. However, it remains unclear how MRI-defined ABM radiation dosimetry parameters are related to HT in patients with pelvic malignancies, irrespective of cancer type. Therefore, we performed image fusion of the T1 sequence and localization CT to aid in the delineation of ABM.
There was a significant difference between ABM and TBM dosimetric parameters, effectively reducing V5-40 of ABM. Wang et al 22 found that the increase of ABM V5 was significantly correlated with the decrease of the WBC, ANC, and PLT nadirs. This is similar to our study. We found that ABM V5-V20 was related to HT2 + in univariate regression analysis. Chen et al 26 analyzed 97 cases of uterine cervical/endometrial cancer patients who received radiotherapy with or without chemotherapy, and multivariate analysis showed that the V10 of functional bone marrow 1 was, respectively, significantly associated with grade ≥ 2 anemia. This is similar to our results. In the multivariate regression analysis in this study, we found that ABM V10 and ABM V15 were significantly associated with HT2+. Studies have shown that bone marrow hematopoietic stem cells are sensitive to low-dose radiation, and damage to these cells is a principal cause of HT. 27 Furthermore, chronic myelosuppression and irreversible morphologic bone marrow changes can occur at doses above 30 Gy. 22 Yet, it is known that hematopoietic ABM is not distributed uniformly throughout the bones. A considerable portion of the medullary canal is composed of yellow marrow, which is composed primarily of fat. Contouring the entire medullary canals on simulation CT thus overestimates the volume of ABM, unnecessarily constraining the IMRT plan. Therefore, this study showed that the volume of ABM receiving low-dose radiation is important in the development of HT.
It is also well known that concurrent chemotherapy is an important factor that causes HT. This study likewise found that the courses of concurrent chemotherapy were significantly associated with HT2+. In addition, this study found that the male sex, low BIM, and boost doses to the lymph nodes were high-risk factors for the occurrence of HT2+. However, multivariate analyses of our study did not find the relationship between HT2+ and low BIM and boost doses to the lymph nodes. In the linear regression model, we found that ABM V5 was predictive of ANC and Lym nadir. In addition, we also found that ABM V10 and ABM V15 were predictive of WBC, ANC, Hb, and Lym nadir. It is important to note that almost all of the patients included in the current study had grade 2 lymphopenia. Lym is extremely sensitive to ionizing radiation.
Rose et al 28 suggested ABM V10 < 95% and V20 < 80% reduce the risk of grade 3 leukopenia. Huang et al 19 discovered that patients with the lumbosacral spine (LSS) V10 < 87% and LSS mean < 39 Gy were more likely to avoid HT2+. And study has shown that the absolute volume of ABM 10 Gy <179 cubic centimeters (cc) and 20 Gy <186 cc all predicted HT3+. 7 In this study, we found that ABM V5 > 99.34%, ABM V10 > 95.3%, ABM V15 > 92.48% and ABM V30 > 59.82% can predict the occurrence of HT2 + . Some of our cutoff values are slightly larger than the conclusions of previous studies, and we consider it is because we have included a variety of pelvic tumors, among which there are differences in radiation methods and doses. Retrospective studies have shown that the volume of ABM in the pelvis is an index for predicting HT. For example, Wang et al 5 found that ABM volume < 387.5 cc based on 99mTc-sulfur colloid tomography can predict HT, with a sensitivity and specificity of 84.2% and 85%, respectively. In 31 patients with cervical cancer, Zhou et al 7 found that pelvic ABM volume < 738 cc can predict HT with high specificity. However, the sample size of these studies is very small, and the conclusions are different. In summary, pelvic ABM is important for the development of HT. This study supports the notion that bone marrow is a parallel organ, depending on the threshold of a functional subunit. Therefore it is clinically significant to reduce the ABM irradiation dose volume, especially V5-V20, during radiotherapy, thus reducing the incidence of acute myelosuppression.
Our study has some limitations, first of all, it is a retrospective study and the data come from a single center, which may lead to regional bias. Secondly, the delineation of ABM is somewhat subjective, and MRI does not provide a quantitative assessment of ABM. Nevertheless, we still believe that the subjectivity in ABM will be smaller than the volume difference between TBM and MRI-based ABM.
Then, we use data from cervical cancer to calculate the sample size of pelvic malignancies, and the inclusion of other pathologies might have diluted the results. Further prospective studies are needed to explore the qualitative indicators of ABM based on MRI and to confirm that the protection of pelvic ABM based on MRI could reduce the incidence of HT2+.
Conclusion
Dosimetric parameters of ABM V5-V20 were significantly associated with HT2+ in pelvic malignancies. ABM V5, ABM V10, and ABM V15 were predictive of WBC, ANC, Hb, and Lym nadir. We conclude that MRI-based outlined ABM better predicts the occurrence of HT in pelvic radiotherapy patients compared to CT-based outlined TBM.
Footnotes
Abbreviations
Authors’ Note
All authors contributed to the study conception and design. Ting Gao was major contributor in writing the manuscript. Ting Gao and Liqun Wei collected and processed data. Li Jiang, Shanshan Ma, and Wei Zhang were responsible for the statistical data. Fang Wu and Yong Zhang contributed to the manuscript's critical assessment and revised the final draft of the document. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the First Affiliated Hospital of Guangxi Medical University, Approval Number: 2022-E468-01. Informed consent for patient information to be published in this article was not obtained because this is a retrospective study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Development and Application of Appropriate Medical and Health Technologies in Guangxi [Nos. S2018025], and Scientific Research projects of Health Commission in Guangxi [Nos. Z20211395].
