Abstract
Colorectal cancer (CRC) is the third most frequently found cancer in the world, and it is frequently discovered when it is already far along in its development. About 20% of cases of CRC are metastatic and incurable. There is more and more evidence that colorectal cancer stem cells (CCSCs), which are in charge of tumor growth, recurrence, and resistance to treatment, are what make CRC so different. Because we know more about stem cell biology, we quickly learned about the molecular processes and possible cross-talk between signaling pathways that affect the balance of cells in the gut and cancer. Wnt, Notch, TGF-β, and Hedgehog are examples of signaling pathway members whose genes may change to produce CCSCs. These genes control self-renewal and pluripotency in SCs and then decide the function and phenotype of CCSCs. However, in terms of their ability to create tumors and susceptibility to chemotherapeutic drugs, CSCs differ from normal stem cells and the bulk of tumor cells. This may be the reason for the higher rate of cancer recurrence in patients who underwent both surgery and chemotherapy treatment. Scientists have found that a group of uncontrolled miRNAs related to CCSCs affect stemness properties. These miRNAs control CCSC functions like changing the expression of cell cycle genes, metastasis, and drug resistance mechanisms. CCSC-related miRNAs mostly control signal pathways that are known to be important for CCSC biology. The biomarkers (CD markers and miRNA) for CCSCs and their diagnostic roles are the main topics of this review study.
Keywords
Overview of Cancer Stem Cells in Colorectal Cancer
Colorectal cancer (CRC), the second most lethal cancer globally, accounted for 9.4% of all cancer-related fatalities in 2020. Its global incidence is predicted to quadruple by 2035, with the greatest increase in less developed countries due to older population growth. New cases and deaths are more noticeable in higher-income areas. There are three main types of colon cancer: sporadic, inherited, and colitis-related. In the colon or rectum, abnormal glandular epithelial cell proliferation is what causes it. The number of CRC cases is increasing globally due to genetic and environmental factors. Patients with chronic ulcerative colitis and Crohn's disease have a higher risk. Factors include dietary and lifestyle choices, family history, and chronic inflammation. CRC develops through four phases: metastasis, progression, metastasis, and promotion. The initiation stage involves losing the APC suppressor gene, the promotion stage activates the KRAS oncogene, and the metastatic stage involves the deletion of the P53 suppressor gene 1 (Figure 1).

The four phases of the development of CRC. Created by Biorender.com.
Carcinogenesis is the process of developing cancer through genetic, epigenetic, biochemical, and histological changes. Despite advancements in cancer research, there is still a lack of clarity on the true mechanism of cancer growth. New methodologies have contributed to understanding cancer's pathogenesis. Cancer is a diverse disease with various differentiated and proliferating cells with different markers, leading to tumor formation, recurrence, metastasis, and therapeutic resistance. Cancer stem cells (CSCs) contribute to this heterogeneity. 2
Normal and Cancer Stem Cells
Adult stem cells, induced pluripotent stem cells, and embryonic stem cells are three types of undifferentiated cells. Adult stem cells originate from tissue, while pluripotent stem cells originate from early morula phase embryos or the blastocyst's inner cell mass. Induced pluripotent stem cells are derived through somatic cell reprogramming and share characteristics with stem cells. Stem cells create adult cells through differentiation 3
Stem cells, which specialize in tissue during embryonic development, play a crucial role in replenishing and repairing mature tissues. Their self-renewal and pluripotency make them promising for clinical treatments. Stem cell transplantation has been shown to heal damaged organs. A unique subset of tumor-initiating cells, also known as CSCs, is implicated in cancer development. 4
Dick and colleagues discovered stem cells in leukemia in 1994, advancing the idea that cancer tumors develop hierarchically. CSCs are now being identified and isolated from various human malignancies, influencing cancer pathogenesis and therapeutic development. CSCs divide, proliferate, clonally expand, and differentiate rapidly to increase stem cell population and develop resistance to cancer treatment interventions. 5
CSCs are key agents in cancer growth, capable of self-renewal, sphere formation, migration, invasion, and resistance to various treatments. Initially found in leukemias, CSCs can also develop solid tumors. Dedifferentiated somatic cells, cancer cells undergoing EMT, or mutations in healthy stem cells can all cause them. MicroRNAs play a role in regulating CSCs, though the biological mechanisms behind their expansion remain unclear 6 (Figure 2).

Unique characters and mechanisms of cancer stem cells. Created by Biorender.com.
CSCs, which are cancer-specific, are crucial in the emergence of drug resistance and tumor heterogeneity. Dysregulation in signaling pathways related to stemness properties explains their tumorigenic potential. Despite exhibiting similarities to normal stem cells, CSCs exhibit oncogenic potential for cancer pathogenesis. Various theories have been proposed regarding their creation and operation, while normal stem cell formation and tissue development functions remain well-understood. 7
The stem cell division theory suggests that cancer develops from genetic mutations in normal stem cells, leading to the production of CSCs, which in turn cause tumor growth and metastasis. The hypothesis posits that tumor cells differ, explaining the heterogeneity of tumors. It argues that only a small fraction of tumor cells can split and become tumorigenic. 8
Moreover, CSC-induced tumors can be transplanted into different generations and contain a mixed population of the original tumor's tumorigenic and non-tumorigenic cells. The general idea is that CSCs have the capacity for self-renewal and heterogenic differentiation, which leads to the development of tumors (Figure 3).

(A) An overview of the origin of cancer stem cells (CSCs). (B) Cells of origin in cancer. Created by Biorender.com.
Normal differentiated cells experience forced dedifferentiation and epigenetic reprogramming, which results in the formation of CSCs. CSCs have distinct stem cell niche compositions, with tumor-suppressing niches consisting of signaling pathways. In contrast, tumor microenvironmental cells, like cancer-associated fibroblasts, are present in CSC habitats, promoting CSC differentiation and proliferation through growth factors or cell–cell interactions. 9
Biomarkers and Signaling Pathways of Colorectal Cancer Stem Cells
The exact origin of CRC cells and the mechanisms behind carcinogenesis are still largely unknown. Tumors are now understood to be hierarchically organized structures of several cell types rather than just being “bags stuffed with homogenous malignant cells.” These cell types can influence metabolic, physiological, and morphological alterations that eventually promote tumor growth, the likelihood of management failure, and tumor recurrence. They can also contribute to the tumor's heterogeneity. 10
Cancers are physiologically heterogeneous and not structured into systems, and every cell within a tumor has the capacity to grow and metastasize into other organs. Either intrinsic (like fluctuations in transcription factors or proteins) or extrinsic (like environmental signals, and immunological reactions), which alter typical cell activity, are used to explain any functional errors in the model. The hierarchy of the cell class developing at the top, which is distinct from the rest of the hierarchy and contains a variety of capacities for self-renewal, explains cellular heterogeneity. 11 CSCs differ from typical stem cells in terms of their morphology, physiology, and metabolism, as well as how susceptible they are to certain chemotherapeutic treatments. 5
The existence of CSC may help to explain, in part, the high likelihood of cancer recurrence in patients who have undergone chemotherapy and surgery. Despite this, a lot of work has been made in locating potential colorectal cancer stem cell (CRCSC) biomarkers that could identify the disease's stage of development, patient risk groups, the likelihood of recurrence and spread, as well as individuals who are most likely to not respond to specific treatments. 12 So, this review is mostly about the current state of CRCSC biomarkers and the pathways linked to the growth of CRC from the point of view of the CSC model. This could be important because there is a lack of knowledge about particular CRCSC biomarkers.
Signaling Pathways of Cancer Stem Cells in Colorectal Cancer
Targeting these signaling pathways may offer a promising approach to the treatment of CRC by disrupting CSC maintenance and overcoming resistance to chemotherapy. However, the complexity of these pathways and their crosstalk with other signaling pathways in the tumor microenvironment (TME) pose challenges for the development of effective CSC-targeted therapies13–22 (Table 1).
Signaling Pathways of Cancer Stem Cells in Colorectal Cancer.
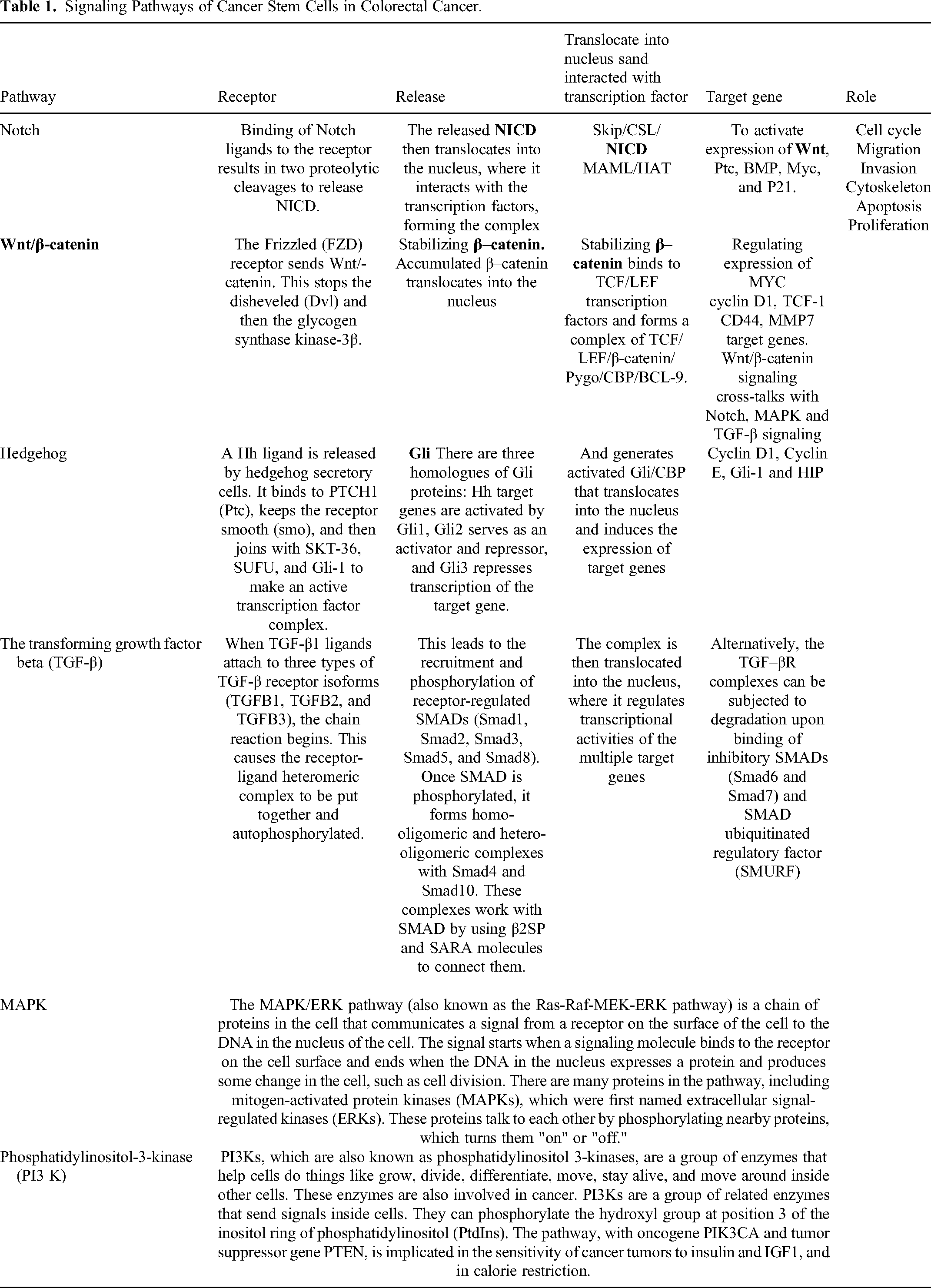
Wnt Signaling Pathway
The Wnt signaling system is responsible for various intracellular functions, including stem cell fate determination, cell polarity, proliferation, and differentiation. Dysregulation of this cascade can lead to epithelial dysfunctions, such as colon cancer. It is possible that the non-canonical pathway, which can stop catenin-dependent overactivation of the Wnt pathway, can help fight cancer. The amount of unbound catenin in the cytoplasm controls the Wnt cascade's activity. When Wnt binds to the LRP/Frizzled complex, its downstream targets are phosphorylated. This stops GSK-3 from working and causes more nonphosphorylated-catenin to build up. 13
When p300 and CREB bind to lymphoid enhancer-binding factors and T cell transcription factors, they work with catenin that has not been phosphorylated. This causes genes that control cell fate, proliferation, stem cell maintenance, and embryogenesis to be expressed. When other Wnt cascade targets, such as CD44, LGR5, and ASCL2, are harmed, they cause cells to grow too much and lose their stem cell compartments. This is similar to how Wnt signaling networks are connected to EpCAM signaling processes. 14
EpICD is an EpCAM intracellular domain that is released when presenilin-2 and TACE break down EpCAM. This controls the transcription of Wnt target genes. CD133 can enhance β-catenin signaling, inhibiting cancer cell development. Bmi signaling regulates Wnt pathway activities reciprocally. 15 Bmi1 can stop DKK1 from working, which raises the Wnt signaling pathway and starts the transcription of genes like c-Myc, Lgr5, CD44, and Bmi1. Hyperactivation of DKK1 can modify ALDH-1 activity, affecting cancer cell survival strategies. Mutations that activate, stabilize, or inactivate the APC gene activate the Wnt transcriptional pathway. Using siRNA to block catenin lowers ionosphere development, which is an important part of cancer development. On the other hand, turning up TCF/LEF and c-Myc makes it easier for ionospheres to form. 16
NOTCH Signaling Pathway
Notch signaling cascade components are expressed in intestine bottom crypts, crucial for controlling progenitor and goblet cell differentiation. However, cancer cells exhibit higher levels of aberrant hyperactivation of these components than healthy colonic mucosa cells. Overexpression of Notch pathways in cancers like colon cancer is linked to poor prognosis and therapeutic response. Notch signaling, which causes ALDH1A1 deacetylation, is associated with aggressive metastatic cancer. MSI-1 signaling can control Notch activity, promoting cell survival and proliferation. Blocking the Notch pathway eliminates CD133-positive cells, preventing tumor cell proliferation and growth. Not every member of the Notch cascade is connected to colon cancer formation.17,18
TGF-β Signaling Pathway
The TGF superfamily, comprising over 30 proteins, plays a crucial role in tissue homeostasis and cell cycle inhibition. It is expressed in healthy immune, nodal, and epithelial cells and is one of the most frequently changed cell signaling cascades observed in many cancer types. Hyper-activation of this pathway encourages angiogenesis and immunosuppression, which may be essential for cancer development, tumor invasion, and metastasis. The extracellular matrix's composition, cell adhesion, differentiation, and apoptosis are all linked to TGF-signaling. 19
Some proteins, including Smad6 and Smad7 and Smurf, can break down TGF-R complexes, which can hurt cells in the digestive tract. Mutations in stem cell populations may increase carcinogenesis and increase the likelihood of additional mutations. Targeting these cell types may be more effective in treating colon cancer patients. 20
The TGF-R complex is responsible for controlling the transcription of target genes in the nucleus. Inhibitory SMADs and SMAD ubiquitinated regulatory factors can destroy these complexes. Gastrointestinal epithelial cells are known for constant proliferation and differentiation, making mutations in stem cell populations more likely to increase the likelihood of carcinogenesis. Targeting these cell types may be more effective in treating colon cancer patients. The tbr2 gene is vulnerable to replication errors, frameshift mutations, and microsatellite instability, accounting for 40% to 80% of all colon cancer cases. In vivo tests show that it is less likely to cause tumors, and mice that have both SMAD and 2SP genes deleted have a higher chance of getting aggressive colonic adenomas. 21
Hedgehog Pathway
The hedgehog signaling system is crucial for stem cell maintenance, polarity, migration, and differentiation in embryonic development. Its three homologues are Desert Hedgehog (Dhh), Indian Hedgehog (Ihh), and Sonic Hedgehog (Shh). Dysregulation of this pathway increases tumor growth in various tissues, including CRC. Hyper-activation of Hh may control CSC maintenance. RASP acylation of the H-N-terminus is what starts the cascade. 22
When Hh is released, it binds to the Drosophila patched gene (Ptch1), which keeps the smoothened receptor stable and stops it from going to the primary cilium. For Hh signaling to happen, the Cubitus interruptus (Gli) gene family of zinc finger transcription factors, which has three homologues, needs to be turned on. The Hh-Gli pathway is overactive in CRC stem cells and primary human colon carcinoma cells, which changes how cells divide, how tumors grow, how long they live, and how they spread. 23 Hh signaling is very important for keeping the CD44+/CD24/low phenotype in CSCs, which helps cancer spread. Cyclopamine's ability to stop Hh cascades has been shown to stop EMT and metastasis. This shows that the Hh pathway may be useful for medication therapy. 24
Crosstalk Between Signaling Pathways
Abnormal stem cell pathway activation in CSCs rarely occurs in isolation, potentially impacting their response to external signals. Studies show that Notch may be able to block Wnt signaling without Su (H) being involved. This could lead to cancers that are linked to high levels of catenin in the nucleus. When the Wnt/-catenin pathway was blocked in mice, the expression of Jagged1 in ovarian cancer cells went down. This suggests that the Notch and Wnt pathways may be linked in a backward loop. 25
The Wnt cascade, which can alter Gli3 expression, influences the Shh/Gli patterning of the Hh pathway. It is possible for Hh signaling to stop the nuclear-catenin/TCF4 complex. This stops Wnt signaling in the colonic epithelial cell cascade. mTOR/PI3 K/Akt, Raf/Ras/Erk/Mek, and other pathways are also connected to Notch signaling and how cells form and divide. 26 A study found that aberrant Wnt or Hh activities in APC mutant mice increase the risk of colon cancer. Hes1 and NICD are more abundant in cells where mTOR is always active. This is most likely because of STAT3/Jagged/p63 cascades. Treatment with mTOR inhibitors reduced Notch and mTOR signaling in cells. PI3 K/Akt-based methods can also control TGF-/ALK5 activity, which could lower the TGF-mediated cell death or cycle arrest caused by insulin, interleukins, and other stimuli. 27
Wnt signaling and TGF-b/BMP pathways are also mutually linked. Lef/-catenin/Smad, the pathway's downstream components, work together to synergistically regulate the activities of the common genes. The Hh route and p53 molecules have been linked in several studies 28 ; Figure 4
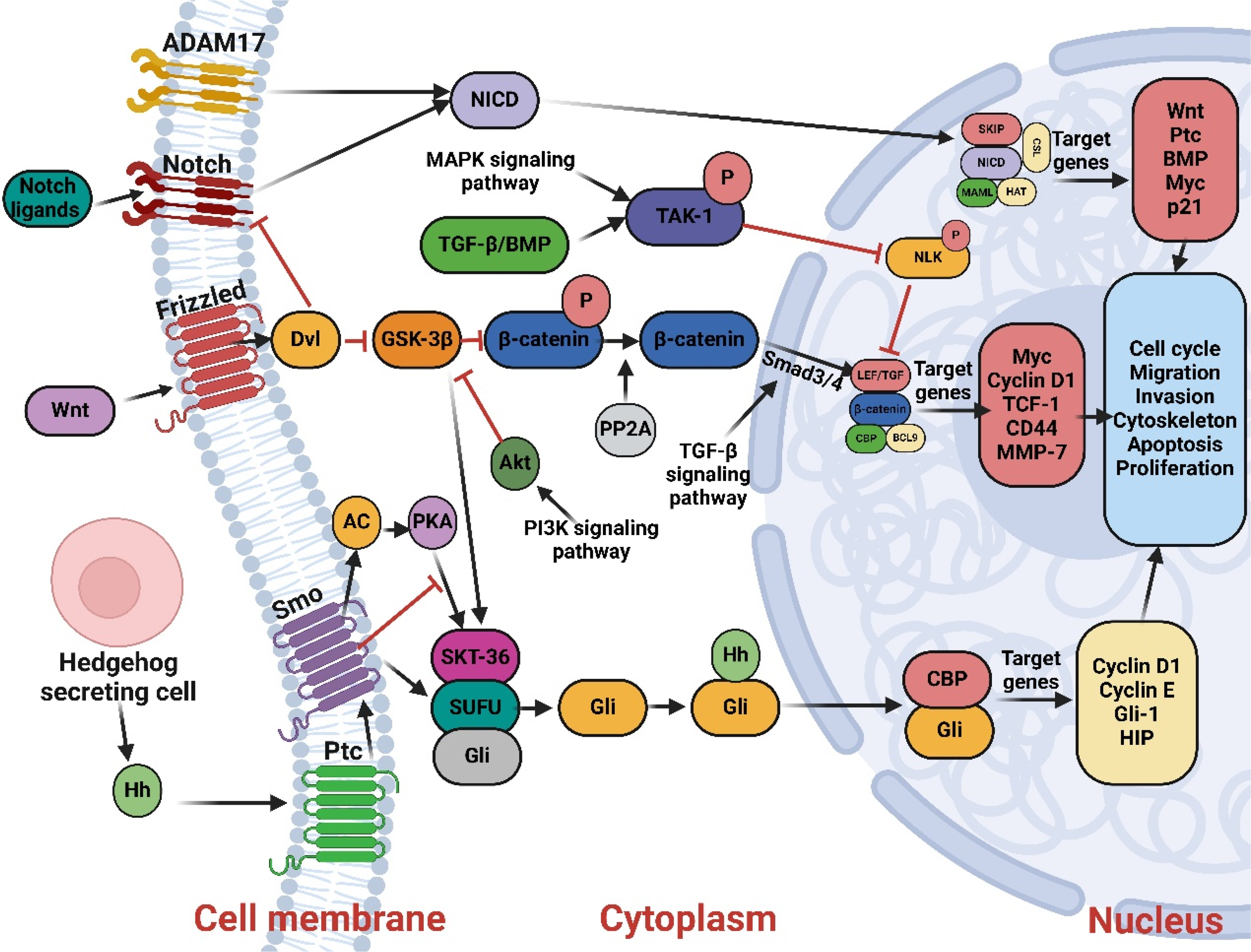
Crosstalk between signaling pathways of colon cancer. Created by Biorender.com.
General Criteria of Colorectal Cancer Stem Cells (CRCSC)
To research CRCSC biology, it is crucial to identify and validate their nature using molecular markers like fluorescent microscopy and fluorescence-activated cell sorting (FACS). Two research teams led by John Dick and Ruggero De Maria conducted the first tests to confirm the existence of CSC-like cells in CRC. To be classified as a CSC, CRCSC and other organs and tissues must meet specific requirements, including self-renewal, multilineage differentiation, and tumorigenic potential. Self-renewal refers to a cell's ability to divide into two cells, one undifferentiated stem cell and the other a differentiated lineage. 29
CSCs have multilineage differentiation potential, which means they can change into different types of tissue cells, like enterocytes, goblet cells, paneth cells, and enteroendocrine cells. After in vitro differentiation, CRCSCs should create cell types resembling these types. Two stemness characteristics of CSCs are self-renewal and multilineage differentiation, while tumorigenic potential is a cancer cell's characteristic. The capacity of a cell to enter tissue and develop a tumor after transplantation serves as a measure of its tumorigenic potential. The tumorigenic potential of CRCSCs has been consistently assessed by injecting them into immune-deficient mice. 30
The CRC tissue digest is grown in a medium without serum that contains growth factors and hormones. This lets colon CSCs be chosen and unique spheres be made. The expression of CD133 or other markers can be used to recognize and isolate colon CSCs. Two methods, MACS and FACS technologies, can purify CD133 cells. Isolating and growing CSCs in vitro has significant therapeutic implications, as the acquired cells can be molecularly defined, which can be applied to create new therapeutic techniques. These techniques rely on anticancer agents, which can only kill cancer cells that are actively dividing by inducing the intrinsic apoptotic pathway. Colon CSCs become resistant when they make too much IL-4 on their own, but this can be stopped to make them more vulnerable to death signals. 31
Further research into the population of CSCs’ selectively activated signaling transduction pathways will be possible thanks to the possibility of isolating and growing CSCs in vitro. This will help us better understand the mechanism underlying their survival strategy and define novel anti-tumor therapy approaches.
Isolation of Colon Cancer Stem Cells
It has been found that colon cancer has a population of colon stem cells because the cells' surfaces show signs of not being fully grown yet and the Wnt signaling pathway is very active. Efforts have been made to develop treatments to eliminate this population. Techniques preserving tumor stem cell characteristics in tissue culture using human and transgenic mouse material are crucial. Tumor fragments are digested and cultivated in a serum-free, modified neurobasal medium with growth factors. This method preserves self-renewal and differentiation into all lineages in the initial cancer. Identifying variables that sustain or produce a CSC phenotype and molecular mechanisms driving the CSC population remain active areas of inquiry. Even though tumor cells are very different from each other, stromal cells in the intestine can give them colon CSC traits. However, human colon CSCs cannot be transplanted into an immune-competent host, limiting their use in studying the interplay between cancer and the immune system (Figure 5).

Isolation of colon cancer stem cells. Created by Biorender.com.
The following four techniques—transplantation into recipient animals, cell isolation by FACS, cell culture-based selection systems, and lineage tracing—can be used to identify cCSCs (Figure 6) 31 ; FACS; in vitro cell culture; in vivo transplantation; lineage tracing. Arrows spanning sectors indicate the complementary use of various techniques. For instance, in vitro cell culture and/or in vivo transplantation assays can validate the identification of cCSCs by cell sorting (pink arrows); Xenografting is usually done with cCSCs that have been cultured and/or genetically labeled (light green arrow); and human cCSC lineage tracing is mostly done by transplanting cCSCs that have been genetically changed in culture (dark green arrow). 29 Created by Biorender.com.

Main methodologies contributing to the definition of CCSCs.
Biomarkers of Cancer Stem Cells in Colorectal Cancer
Tumor subpopulations express diverse stem cell markers and have varied roles. CD133, CD144, CD24, CD166, CD44, CD29, ALDH1, LGR5, and CXCR4 are colorectal adenocarcinoma stem cell markers. However, research on CSCs’ prognostic and predictive roles in CRC progression is sparse. This tumor cell subpopulation's phenotypic, molecular, and functional features in CRC's original location and metastases are important for creating new diagnostic and therapeutic techniques (Figure 7).
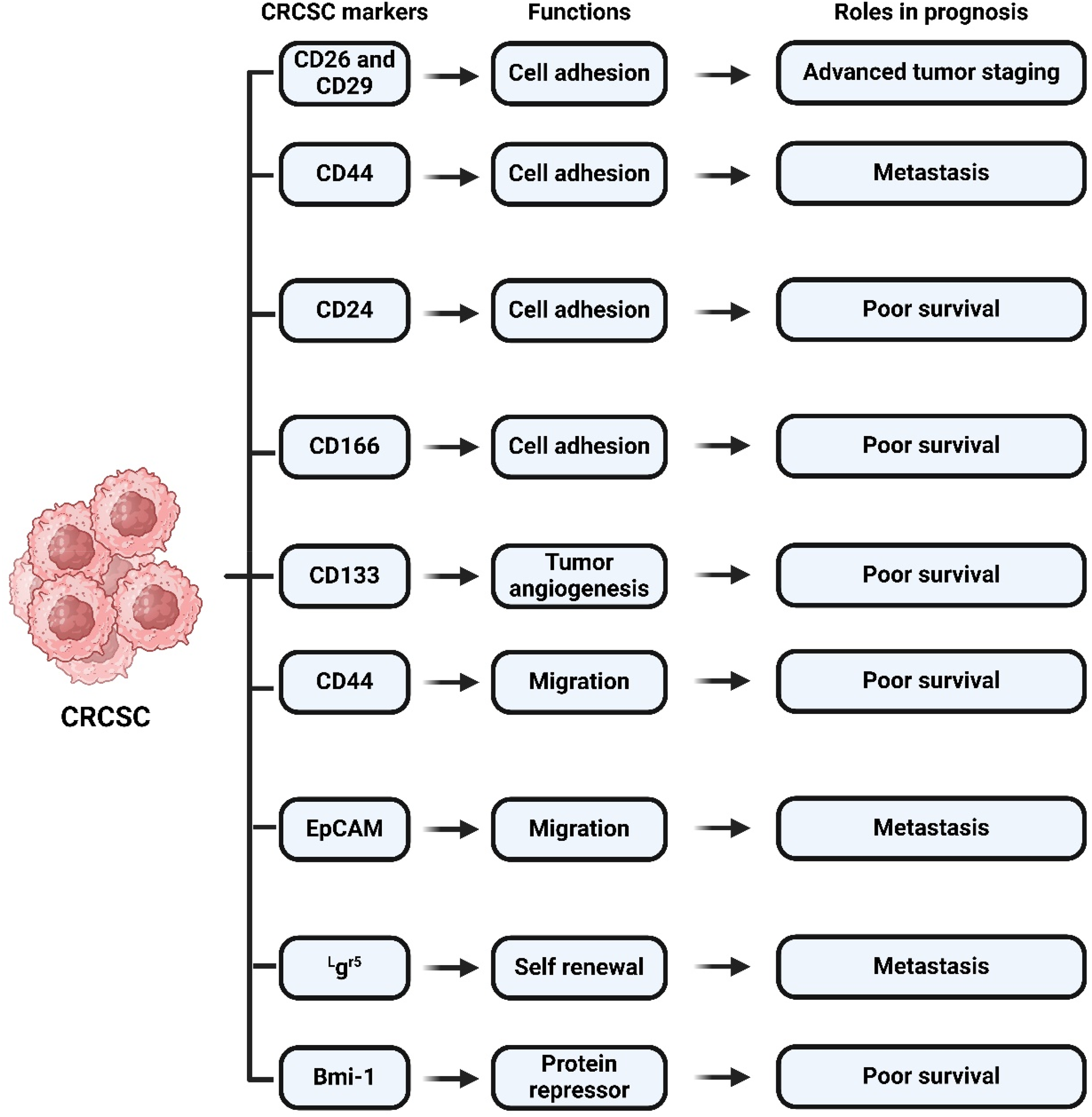
Biomarkers of cancer stem cells in colorectal cancer. Created by Biorender.com.
Circulating Tumor Cell
Liquid biopsies detect two main biomarkers, circulating tumor cells (CTCs) and circulating tumor DNA (ctDNA), which provide complementary information. However, ctDNA analysis is limited due to its fragmentation and small concentrations, and its role is only at the genomic level. CTCs can be analyzed at genomic, transcriptomic, and proteasomic levels using in vivo and in vitro functional assays. Isolation of CTCs allows for morphology and phenotyping studies, enabling comparison with the primary tumor. CTCs also offer an accessible, non-invasive source of tumor material for cancer patients.
The primary tumor sheds tumor cells into the bloodstream that can infiltrate as single cells, clusters, strands, or single files. They were first described in 1869 and have become increasingly important in preventing metastasis and relapse. Millions of CTCs are released in circulation, but only some can reach their destination and induce metastasis at distant sites. CTCs can resist shearing forces and collisions with blood cells, but those that have undergone epithelial-mesenchymal transition (EMT) can resist these forces. EMT allows CTCs to change from the epithelial phenotype to the mesenchymal phenotype, enabling invasion and invasiveness. When colorectal cancers start to spread, they use EMT and signaling pathways like transforming growth factor, wingless/integrated WNT, platelet-derived growth factor, and IL-6 to do it. This turns on transcription factors that create the mesenchymal phenotype.
CD24
A phosphatidylinositol anchor keeps CD24, a glycoprotein that sticks to the outside of cells and acts like mucin, in place. B lymphocytes express CD24, play a role in cell proliferation. CD24 favorably promotes the maturation of activated T cells and granulocytes, as well as other immune system cell types. According to reports, the protein may also play a role in the emergence of several cancer types. It was discovered that about 50–68% of CRCSC express CD24. There is a positive relationship between CD24 expression and the degree of differentiation, lymph node metastasis, and different stages of adenocarcinoma development. However, there was no discernible link between the presence of the marker and a shorter patient survival time. The relevance of the CD 24 protein as a CRCSC marker is less persuasive and has not been thoroughly proved. 32
CD26
There is a 110-kDa cell surface glycoprotein receptor called CD26. It is found on activated T, B, NK, macrophages, epithelial, and endothelial cells. It may have functions in T cell activation via the collagen receptor molecule and thymic ontogeny. CD26 targets Rantes, a critical regulator of monocyte chemotaxis and interferon-inducible chemokines. Studies suggest CD26 may be involved in the mobilization and migration of hematopoietic stem cells (HSC). The expression of the biomarker may be altered in various malignancies, but only one study suggests it may play a role in the pathogenesis of CRCSC. 33
CD29 1-Integrin
CD29 is a protein with a broad extracellular domain, transmembrane stretch, and small intracellular region that activates cell growth, migration, survival, and proliferation activities. It contains progenitor and stem cells in normal intestinal mucosal cells. A small group of primary colorectal cancers have both CD24 and CD29 markers. These markers are thought to be a tumor-initiating factor that helps stem cells multiply and spread. CD29 expression is elevated in colon tissues after adjusting for disease stage, despite being lowered during the late stages of CRC progression and linked to worse disease outcomes. However, there are still questions about its relationship with stem cell populations and its role in malignancy prognosis. 34
CD44
CD44 is a cell adhesion and transmembrane hyaluronic acid receptor glycoprotein that sends signals outside of cells. It changes the Wnt and beta-catenin pathways that follow. Angiogenesis, homing and activation of lymphocytes, hematopoiesis, cell adhesion, and migratory processes are all impacted by CD44. A number of CSCs have been identified to express CD44. The interaction of CD44 and CD54 can be particularly useful in identifying rectal cancer cells. It is yet unclear how CD44 affects the emergence of cancer and metastases, though. This explains why various cancer types frequently express various CD44 splice variants. More research is needed to find out if the CD44 molecule is a good way to identify CRCSCs because it has mixed and controversial prognostic implications for patient survival, lymph node size, and tumor grade progression. 35
CD133
This is a cell surface marker called the prominin-1 protein, which is also known as the Penta span-transmembrane cholesterol-interacting CD133 protein. It is found in many cancerous tissues, such as intestinal bottom crypt cells and bone marrow-derived endothelial progenitor cells. Studies have shown that the glycoprotein may act as a marker for CRC stem cells. CD133+-enriched cell populations are better able to engraft and start solid tumor formation in immunodeficient mice than unsorted CRC cell populations. 35
With the help of CSC, CD133+ cells can go from not-dysplastic parts of adenoma-polyp-lesions to not-dysplastic serrated hyperplastic polyps and then to dysplasia, adenomas, and cancers. This suggests that CD133 expression is upregulated more frequently during the initial stages of CRC progression, aiding in tumor growth. Epigenetic mechanisms through hypermethylation of the CD133 gene promoter may result in CD133+ expression in CSC. There is a strong link between higher levels of CD133 and worse prognostic outcomes and resistance to 5-FU-based chemotherapy in the development of cancer. 32
Moreover, CD133+ cell populations are more resistant to conventional radiation therapy, which explains the higher risk of CRC relapse and other hazards associated with radiotherapy. However, CD133's value is still up for debate because CD133 cell populations can also cause tumor formation in animals with compromised immune systems. Colon cancer cells can switch between the CD133+SW620 and CD133SW620 subpopulations when they are under a lot of stress or when high levels of hypoxia are present. When human embryonic colon and kidney cancer cells bind to one of the two ETS sites, it controls the P5 promoter of the CD133 gene. HIF-1 and HIF-2 are responsible for doing this. All of these data point to CD133's crucial role in the development and spread of cancer, suggesting the possibility that it could serve as a predictive biomarker for CRCSC. 5
CD166
A transmembrane protein called CD166, also known as ALCAM or activated leukocyte cell adhesion molecule, has five extracellular immunoglobulin-like domains. T and B lymphocytes all have CD166 on their outer surfaces. In the base of the crypt and progenitor niche cells, or CRCSC, in colonic epithelial cells, CD166 was seen. A shorter survival time is correlated with CD166 expression with a CRC diagnosis. Colorectal polyp count, tumor size, lymph node metastasis, and a worse clinical prognosis are all directly associated with each other. Contrary to our findings, there is a significant association between CD166 markers and CRC outcomes. Our findings suggest that colon polyps to carcinoma transition events must involve upregulation of CD44 and CD166; however, the precise role is still understood. 36
Aldehyde Dehydrogenase
Aldehyde dehydrogenase (ALDH1) has been identified as a CSC marker for several malignancies. Catalysis and the irreversible oxidation of aldehydes to their corresponding carboxylic acids are two of ALDH1's known functions. When cancer is poorly differentiated, in normal tissues, or in patients with metastatic colon disease, there is an elevated expression of ALDH1. Malignant colonic stem cells were also found to have ALDH1. This gene was expressed higher up in APC animals, near the top of the crypt, where normal epithelial cells changed into adenomas. 37,38
Bmi1
Bmi1, a stem cell biomarker, plays a crucial role in the maintenance and self-renewal of stem cells across various tissue types. It also acts as an epigenetic chromatin modulator of target genes and is part of the polycomb repressive complex (PRC1). Bmi1-expressing cells are often found in the small intestine. Diphtheria toxin therapy selectively eliminates Bmi1-expressing cells, disrupting the normal crypt composition. The question remains whether Lgr5-positive cells that express Bmi1 compensate for their loss. Interestingly, when Lgr5-expressing cells were removed, there were three times as many Bmi1-expressing cells as controls. Many CSC types respond to Bmi-1. 39
The oncoprotein Bmi1 has been linked to the growth, spread, and metastasis of CRC, which typically has poor clinical outcomes and survival rates. However, the biomarker has only been documented to play a role in CRCSC. The conventional Bmi1 signaling pathway appears to be necessary for the functioning of CRCSC and the advancement of CRC. 40
Epithelial Cell Adhesion Molecule (EpCAM)
A transmembrane glycoprotein known as EpCAM mediates intracellular adhesion, cell migration, proliferation, differentiation, and signaling that is particular to epithelial cells. EpCAM expression has been noted in epithelium-derived cell populations as well as a variety of human cancer tissues. FACs that focused on the tumorigenic potential of EpCAMhigh/CD44+ and EpCAMlow/CD44 were the ones who first identified EpCAM. It should come as no surprise that EpCAMhigh/CD44+ were able to start a tumor's progression while EpCAMlow/CD44 failed to establish any colonies. When xenografted on NOD/SCID mice, the active EpCAM caused a type of cancer heterogeneity that is usually seen in the original tumors. 41
EpCAM+/CD44+ cell surface indicators may contribute to CRCSC chemoresistance. Combining EpCAMhigh/CD44+/CD166+ cell lines seems to be better at forming cell colonies, being resistant to drugs, and having lower rates of spontaneous apoptosis than EpCAM populations. Also, it has been discovered that EpCAM expression is negatively connected with the primary tumor grade and that higher expression levels are seen for early stages of cancer progression. It has also been linked to less EpCAM expression when a metastatic cell can get into the tumor, when the invasive tumor margin gets ingested, and when lymph node metastases are present. The lymph node stage correlates with lower EpCAM expression rates. These results suggest that EpCAM may play a part in cellular signaling systems. More research needs to be done on the protein to fully understand its clinical, prognostic, and therapeutic implications. 42
Leucine-Rich Repeat-Containing G Protein-Coupled Receptor 5 (Lgr5)
Lgr5 belongs to the group of orphans GPCR receptor proteins. The protein has seven trans membranes and an extracellular stretch with a lot of leucine. Lgr5 was first found in colon crypt base columnar progenitor cells that had changes that made the Wnt/-catenin pathway stronger. Patients with simultaneous expression of several stem cell genes, including Lgr5, may have 10-fold higher odds of developing colon cancer than those with low levels. Furthermore, there is a correlation between increased lymph node metastases levels and lower patient survival rates when Lgr5 is overexpressed. These discoveries outlined Lgr5's critical function in CR carcinogenesis and suggested that it might be a useful CSC marker. 43
Musashi-1 (Msi-1)
It was discovered that a protein called Msi-1, which binds to RNA, can compete with the elF4G translation initiation factor of two messenger RNAs (mRNAs): p21/Waf1 and neural stem cells. These findings imply that Musashi-1 may be crucial for CSC carcinogenesis and tumor development. Msi-1 participates in the regulation of Bstemness, which controls Wnt/Notch pathways, in CRCS C. On the other hand, Msi-1 is only found on CRCSC as a stem cell progenitor. The protein's function has only been thoroughly studied in relation to the nervous system, so no conclusions have been drawn about Msi-1's impact yet. Together, these results show the significance of Msi-1 in the emergence of CRCSC and demand additional research. 44
Molecular Markers of Cancer Stem Cells of Colorectal Cancer
Non-Coding RNAs (ncRNAs)
Epigenetics has been shown to play a crucial role in controlling the behavior of CRCSCs and CRC cells. Long-non-coding RNAs (lncRNAs) and microRNAs are examples of ncRNAs. They control gene expression and a number of cellular processes by working with their target messenger RNAs to either break down or stop translation. Several studies have examined the connection between the gene expression profile of miRNAs and the clinical outcome of CRC. MiRNAs control many cellular processes, such as cell division and self-renewal. This makes SCs resistant to environmental factors that would normally make most cells go through the G1/S checkpoint. It has been suggested that tumor cells should use the same techniques SCs used to get past this checkpoint. 45
Role of miRNA and Oncogenic Signaling Pathways in Human Cancer Stem Cells
miRNAs, small noncoding RNAs, regulate gene expression through feedback mechanisms, leading to mRNA degradation. Deregulation of miRNA expression can affect multiple transcripts and cancer development and spread, making them closely linked to cancer development. 46
MiRNAs, epigenetic factors in cancer, regulate gene expression and may serve as therapeutic targets and diagnostic biomarkers. miRNA expression levels vary between colorectal neoplastic and normal tissues. Anti-tumor miRNA mimics have been shown to reduce cell proliferation, and migration, and induce apoptosis in CRC in vitro and animal models. miRNAs may play a crucial role in the clinical diagnosis, management, and prognosis of CRC patients. 47
miRNAs play a crucial role in the development of chondrocyte-derived stem cells (CCSCs), with different expression profiles in differentiated cells and tissues. By messing up key signaling pathways, changes in miRNA expression in CCSCs can cause angiogenesis, metastasis, and drug resistance. MiRNAs regulate the efficiency of CCSCs’ interaction with these pathways to preserve SC pluripotency. 17
miRNAs are essential in the development, progression, and prognosis of human cancer. Oncogenic miRNAs, which regulate tumor-repressor genes, can overexpress and silence these genes. Oncogenes, miRNAs targeting oncogenes, are often found in vulnerable regions, where mutations or deletions can lead to overexpression. Around 50% of these miRNAs contribute to cancer development, with low- and high-expression miRNAs serving as tumor suppressors and oncogenes. 9
Changes in the way miRNAs are expressed can have a big effect on how cancer spreads by making it more resistant to treatment, able to invade new tissues, and stopping cells from dying. MiRNAs regulate cell-derived stem cell (CSC) traits like self-renewal, drug resistance, and tumorigenicity, and can modulate or degrade genes and signaling pathways essential for CSCs’ maintenance and function. 48
miRNAs are crucial for maintaining, reprogramming, pluripotency, and other regulatory mechanisms of stem cells, highlighting their role in the development of mammals. They focus on different differentiation pathways, demonstrating their importance in the growth of CSCs, and highlighting their essential role in stem cell development9,49; Figure 8. deregulate various signaling pathways involved in the regulation of CSCs as well as cell cycle, self-renewal, and differentiation, EMT, and drug resistance when acting as tumor suppressors or oncogenes30,48,49;

Molecular markers of cancer stem cells of colorectal cancer. Created by Biorender.com.
Potential Therapeutic Strategies Targeting Cancer Stem Cells in Colorectal Cancer:
Targeting CSCs is a promising approach for the treatment of CRC, and several potential therapeutic strategies have been investigated. The following are some examples of potential therapeutic strategies targeting CSCs in CRC:
Small molecule inhibitors: target specific signaling pathways involved in CSC maintenance and have been developed. For example, inhibitors of the Wnt/β-catenin pathway, such as PRI-724 and LGK974, have shown promise in preclinical studies.50–52 Monoclonal antibodies: They target CSC-specific surface markers, such as CD133 and CD44, and have been developed. For example, a monoclonal antibody against CD133 (AC133) has been shown to reduce tumor growth and metastasis in preclinical models.53–56 Immunotherapy: Immunotherapy approaches that target CSCs have also been investigated. For example, chimeric antigen receptor (CAR) T-cell therapy targeting CD133 has shown promise in preclinical studies.57,58 Natural compounds: Natural compounds that have been shown to target CSCs in CRC include curcumin, resveratrol, and sulforaphane. These compounds have been shown to inhibit CSC growth and reduce tumor formation in preclinical models.59,60 Repurposed drugs: Repurposed drugs that have been approved for other indications have also been investigated for their potential to target CSCs in CRC. For example, metformin, a drug used to treat diabetes, has been shown to inhibit CSC growth and reduce tumor formation in preclinical models.61,62 Combination therapy: Combining CSC-targeted therapies with conventional chemotherapy or radiation therapy may offer a more effective treatment strategy. For example, the combination of a Wnt/β-catenin pathway inhibitor and 5-fluorouracil (5-FU) has been shown to reduce tumor growth and increase survival in preclinical models.63,64
Moreover, it is important to note that while these approaches show promise in preclinical studies, their efficacy and safety in humans need to be evaluated in clinical trials. Additionally, identifying the appropriate patient population and developing biomarkers that can accurately predict response to CSC-targeted therapies are important areas of future research.
Future Directions and Challenges in Targeting Cancer Stem Cells in Colorectal Cancer
The targeting of CSCs is a potential strategy in the area of CRC therapy.65,66 Nevertheless, other challenges must be overcome to establish efficacious medicines specifically targeting CSCs. There are several prospective avenues and obstacles in the pursuit of targeting CSCs.
In order to find CRCSCs in CRC, scientists need to create reliable biomarkers that can find and separate CSCs within the TME. Understanding the molecular mechanisms behind CSC resistance is crucial for developing effective therapeutic strategies. The TME is crucial for CSC survival and multiplication, and future therapeutic approaches should focus on targeting the TME to disrupt the relationships between CSCs and surrounding cells and tissues. Combination therapy, which integrates CSC-specific therapeutic approaches with conventional chemotherapy or radiation therapy, has the potential to be more effective, but the ideal dosage and combination of these interventions must be determined. Preclinical models are needed to accurately replicate the heterogeneity of CRC and the TME to assess the effectiveness of CSC-targeted medicines. Existing models, such as cell lines and xenografts, have limitations that hinder their ability to capture the intricate nature of actual malignancies. Clinical trials are essential for assessing the safety and effectiveness of CSC-targeted treatments in CRC patients. Careful planning of trials, considering factors like patient selection and endpoints, is essential to ensure the outcomes are meaningful and can be effectively applied in a therapeutic context.
Conclusion Remarks
In conclusion, the identification of molecular biomarkers and signaling pathways in CSCs has greatly enhanced our understanding of CRC. These advancements have paved the way for the development of personalized therapies that target specific molecular abnormalities in CSCs, potentially improving patient outcomes. However, further research is still needed to fully elucidate the complex mechanisms involved in CSC biology and identify additional molecular targets for therapy. Ultimately, the continued investigation of CSCs in CRC holds great promise for the development of more effective and personalized treatments for this disease.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
