Abstract
Objective
1q21 gain/Amp is one of the most common cytogenetic abnormalities. There are controversies about its effects on prognosis and may be associated with inferior outcomes in patients with newly diagnosed multiple myeloma (NDMM). To explore the optimal induction treatment, we analyzed and compared the efficacy of combinations of bortezomib-lenalidomide-dexamethasone (VRD) and only bortezomib-based triplet regimens without lenalidomide (only bortezomib-based) as induction therapy in patients with NDMM with 1q21 gain/Amp.
Methods
Seventy-six NDMM patients with 1q21 gain/Amp who were admitted to our center from 2016 to 2022 were retrospectively analyzed in this study. The progression and efficacy of the patients were observed.
Results
Within our study group, the overall survival rate stood at 75.0%, and the progression-free survival (PFS) rate reached 40.8% in NDMM patients with 1q21 gain/Amp. The best outcome assessment was that 17.1% achieved complete response (CR) and 44.7% achieved very good partial response (VGPR). Patients in the VRD group had a deeper response (VGPR: 63.6% vs 37.0%, P = 0.034), lower disease progression rate (31.8% vs 70.3%, P = 0.002), longer sustained remission (median 49.7 months vs 18.3 months, P = 0.030), and longer PFS (median 61.9 months vs 22.9 months, P = 0.032) than those treated with only bortezomib-based induction therapy. No significant differences were found among patients with partial response or better (86.4% vs 77.8%, P = 0.532) or CR (27.3% vs 13.0%, P = 0.180). Multivariate analysis showed that only bortezomib-based induction therapy (P = 0.003, HR 0.246, 95% CI 0.097-0.620), International Staging System stage III (P = 0.003, HR 3.844, 95% CI 1.588-9.308) and LMR <3.6 (P = 0.032, HR 0.491, 95% CI 0.257-0.940) were significantly associated with adverse PFS.
Conclusions
When compared with the sequential administration of bortezomib and lenalidomide or only bortezomib-based protocols, NDMM patients with 1q21 gain/Amp may benefit more from VRD as initial treatments.
Keywords
Introduction
Multiple myeloma (MM) ranks among the most prevalent hematological malignancies, characterized by significant heterogeneity. 1 Cytogenetics is an important factor leading to its heterogeneity and variable prognosis. 2 1q21 gain/Amp is a common genetic abnormality and may be associated with poor prognosis in newly diagnosed multiple myeloma (NDMM). 1q21 gain/Amp occurs in approximately 40% of NDMM patients,2,3 and its effect on prognosis is controversial. NDMM patients with 1q21 gain/Amp are at an increased risk for drug resistance, disease progression, and death. 2 Even in the era of novel agents and combination treatment strategies, the majority of studies found that 1q21 gain/Amp was still a poor prognostic predictor. 4 Based on the different pharmacological mechanisms and synergies of proteasome inhibitors (PIs) such as bortezomib and immunomodulators such as lenalidomide, the bortezomib-lenalidomide-dexamethasone (VRD) regimen improved the prognosis of all MM patients. 5 Therefore, both NCCN and Chinese guidelines have recommended VRD as the preferred first-line treatment for NDMM patients.6,7 However, it is still unclear whether NDMM patients with 1q21 gain/Amp can benefit from the combination of bortezomib and lenalidomide or one agent-based treatment. Therefore, this paper aims to investigate the clinical efficacy, prognostic effect, and survival of NDMM patients with 1q21 Gain/Amp treated with a VRD regimen as frontline treatment.
Methods
Patients
In this retrospective research, clinical data were gathered from 76 patients diagnosed with NDMM exhibiting 1q21 gain/Amp. The patients received treatment with either VRD or only bortezomib-based triplet regimens at our hospital between 2016 and 2022. The patients were selected consecutively in this study. All patient details have been de-identified. The diagnosis of MM was established based on the criteria set forth by the International Myeloma Working Group (IMWG). 6 The reporting of this study conforms to STROBE guidelines. 8
Study Treatment
Patients were initially assigned to groups with the following treatments: bortezomib plus cyclophosphamide and dexamethasone (VCD), bortezomib plus thalidomide and dexamethasone (VTD), bortezomib plus doxorubicin and dexamethasone (PAD), and VRD. The VCD regimen involved 35-day cycles including subcutaneous bortezomib 1.3 mg/m² administered on Days 1, 8, 15, and 22; intravenous cyclophosphamide 300 mg/m² on Days 1, 8, and 15; and intravenous dexamethasone 20 mg on Days 1, 2, 8, 9, 15, 16, 22, and 23. The VTD regimen consisted of 35-day cycles comprising subcutaneous bortezomib 1.3 mg/m² on Days 1, 8, 15, and 22; intravenous dexamethasone 20 mg on Days 1, 2, 8, 9, 15, 16, 22, and 23; and oral thalidomide 100 mg daily. The PAD regimen consisted of 35-day cycles involving subcutaneous bortezomib 1.3 mg/m² on Days 1, 8, 15, and 22; intravenous dexamethasone 20 mg on Days 1, 2, 8, 9, 15, 16, 22, and 23; and intravenous doxorubicin 25 mg/m² on Day 1. The VRD protocol involved 35-day cycles including subcutaneous administration of bortezomib at a dosage of 1.3 mg/m² on Days 1, 8, 15, and 22, and intravenous administration of dexamethasone at 20 mg on Days 1, 2, 8, 9, 15, 16, 22, and 23; and oral lenalidomide at either 10 mg or 25 mg once daily for 21 days.
Maintenance therapy included bortezomib and lenalidomide (Vd), lenalidomide and dexamethasone (Rd), bortezomib plus lenalidomide and dexamethasone (VRd), and ixazomib and dexamethasone (Id). Vd regimen consisted of 2-week cycles with subcutaneous administration of bortezomib at a dose of 1.6 mg/m2 and intravenous infusion of dexamethasone at a dosage of 20 mg. Rd regimen involved either 10 mg or 25 mg of oral lenalidomide once daily for 21 days, combined with 20 mg of oral dexamethasone once daily on Days 1, 8, 15, and 22 within each 28-day cycle. The VRd regimen involved 2-week cycles with subcutaneous administration of bortezomib at a dose of 1.6 mg/m² and intravenous infusion of dexamethasone at a dosage of 20 mg, along with oral lenalidomide administered once daily at either 10 mg or 25 mg for 14 days. Id comprised 4-week cycles of oral Ixazomib 3 mg or 4 mg once a day on Days 1, 8, 15, and oral dexamethasone 20 mg once a day on Days 1, 8, 15 and 22.
Patients treated with bortezomib or ixazomib were administered acyclovir at a dose of 400 mg twice a day to prevent herpes zoster. Thrombosis prophylaxis, in the form of aspirin, was provided to patients receiving thalidomide or lenalidomide, given the absence of treatment-related contraindications. The determination of all treatment protocols was made through agreement reached by both clinicians and patients. All treatment regimens obtained written informed consent from patients or their families, agreeing to the use of data for research analysis.
Assessments
Assessments of response were conducted based on the response criteria outlined by the IMWG. 9 The primary objective of this analysis was to assess progression-free survival (PFS), with additional objectives focused on determining the best treatment response, including complete response (CR), very good partial response (VGPR), and PR. Other outcomes were disease progression rate and influencing factors of PFS.
Follow-up
All patients were monitored for disease progression or mortality. The surviving patients were last followed up on December 20, 2022. Excluded from the analysis were cases lost to follow-up. PFS was defined as the time from the initial diagnosis to disease progression or death unrelated to progression and associated with the disease. Overall survival (OS) was defined as the time from the initial diagnosis to the last follow-up date or death.
Statistical Methods
All data obtained in this study were subjected to analysis using SPSS 23.0 and GraphPad Prism 7.0. Patient demographics, disease characteristics, and treatment details were evaluated using descriptive statistics. Measurement data were presented as mean ± standard deviation or median. Fisher's exact test and the Wilcoxon run-sum test were employed for analyzing categorical and continuous variables, respectively. The log-rank test and Kaplan-Meier method were utilized for estimating time-to-event endpoints and conducting between-group comparisons for PFS. Univariate survival analysis was conducted using the Kaplan-Meier (log-rank) method, while Cox regression was employed for multivariate survival analysis. All P-values were two-sided; a value of P < 0.05 was considered statistically significant.
Results
Patient Background
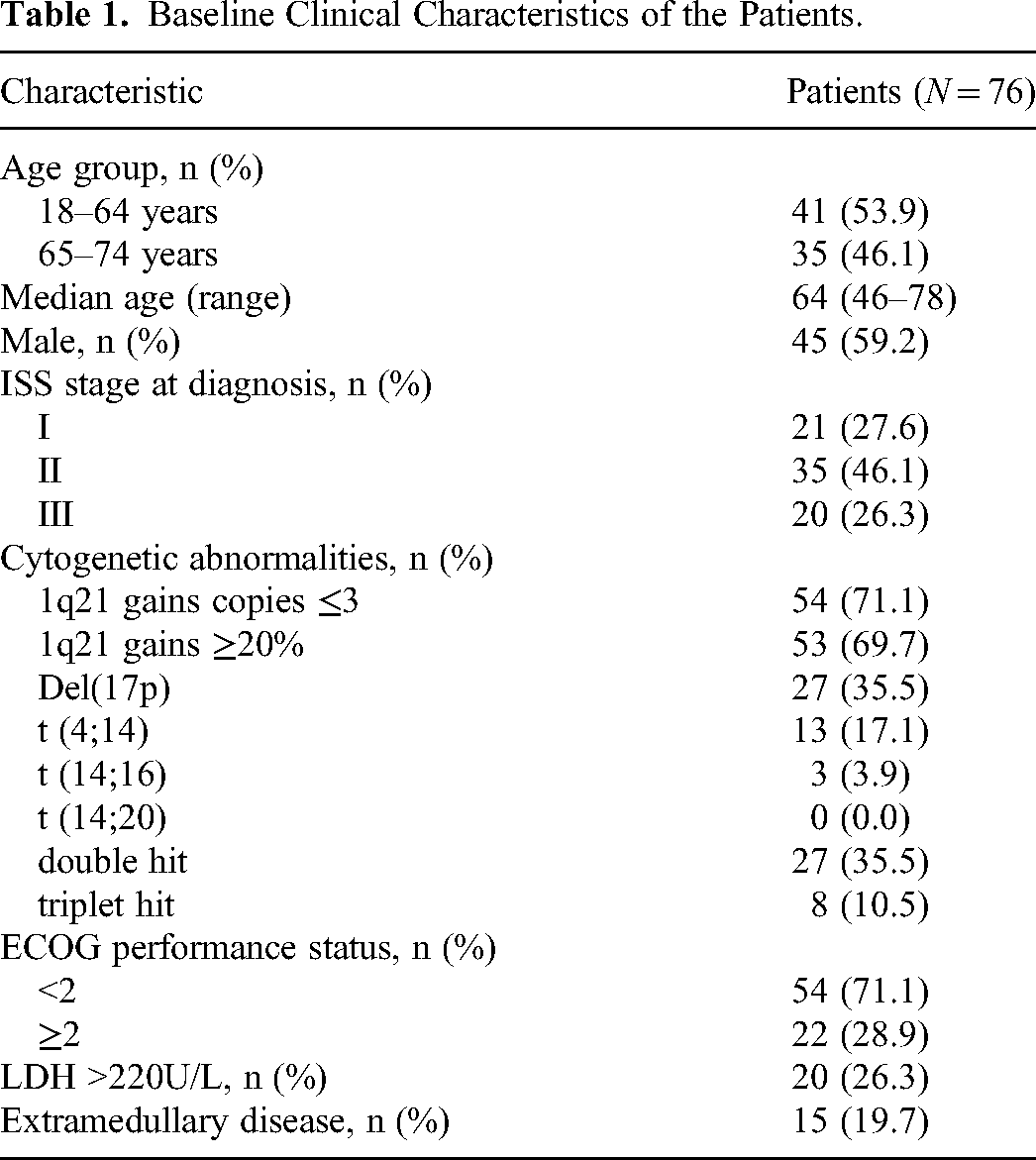
Seventy-six NDMM patients with 1q21 gain/Amp were included in this study, and baseline demographic and a summary of disease characteristics are provided in Table 1. The median age was 64 (range: 46-78) years. A total of 59.2% of patients were male. According to the International Staging System (ISS), 46.1% of patients had ISS stage II disease, and 26.3% had ISS stage III disease. A total of 28.9% of the patients had ECOG scores ≥2. The 1q21 acquisition clone size was defined as ≥5%. Among the 76 patients with 1q21 gain/Amp, 54 (71.1%) patients had 1q21 gain, and 22 (28.9%) had 1q21 amplification. Cytogenetic and FISH analyses showed that 46.1% of the patients were at high cytogenetic risk, including 35.5% of patients with del 17p, 3.9% with t (14;16) and 17.1% with t (4;14), and no patients were concurrent with t (14;20). In addition, 35.5% of patients could be classified as double hit, and 10.5% were triple hit according to the mSMART 3.0 risk stratification criterion. 9 The percentage of patients with extramedullary lesions was 19.7%.
Baseline Clinical Characteristics of the Patients.
Treatment Regimen
Fifty-four patients received only bortezomib-based regimen induction, including VCD (n = 27; 50.0%), VTD (n = 20; 37.0%), and PAD (n = 7; 13.0%), and 22 received VRD regimen induction. The median cycle of the best response after treatment was 4 (range, 3-13) in the only bortezomib-based treatment group, and the median cycle was 3 (range, 3-9) in the VRD group. At the data cutoff, the median follow-up time for all patients was 25.0 months (range, 5.7-73.7), with 30.9 and 25.0 months for only the bortezomib-based regimen and VRD, respectively. Among the 76 patients, 59 patients entered the maintenance therapy stage, the median cycle of maintenance therapy in the only bortezomib-based treatment group was 14 (range, 1-64), and the median cycle of maintenance therapy in the VRD group was 8 (range, 1-23). The treatment regimen of the cohort is shown in Figure 1. In the bortezomib-only group, 3.7% of patients received autologous hematopoietic stem cell transplantation (ASCT), and 13.6% of patients received ASCT in the VRD group. Other patients were ineligible for or unwilling to receive ASCT.
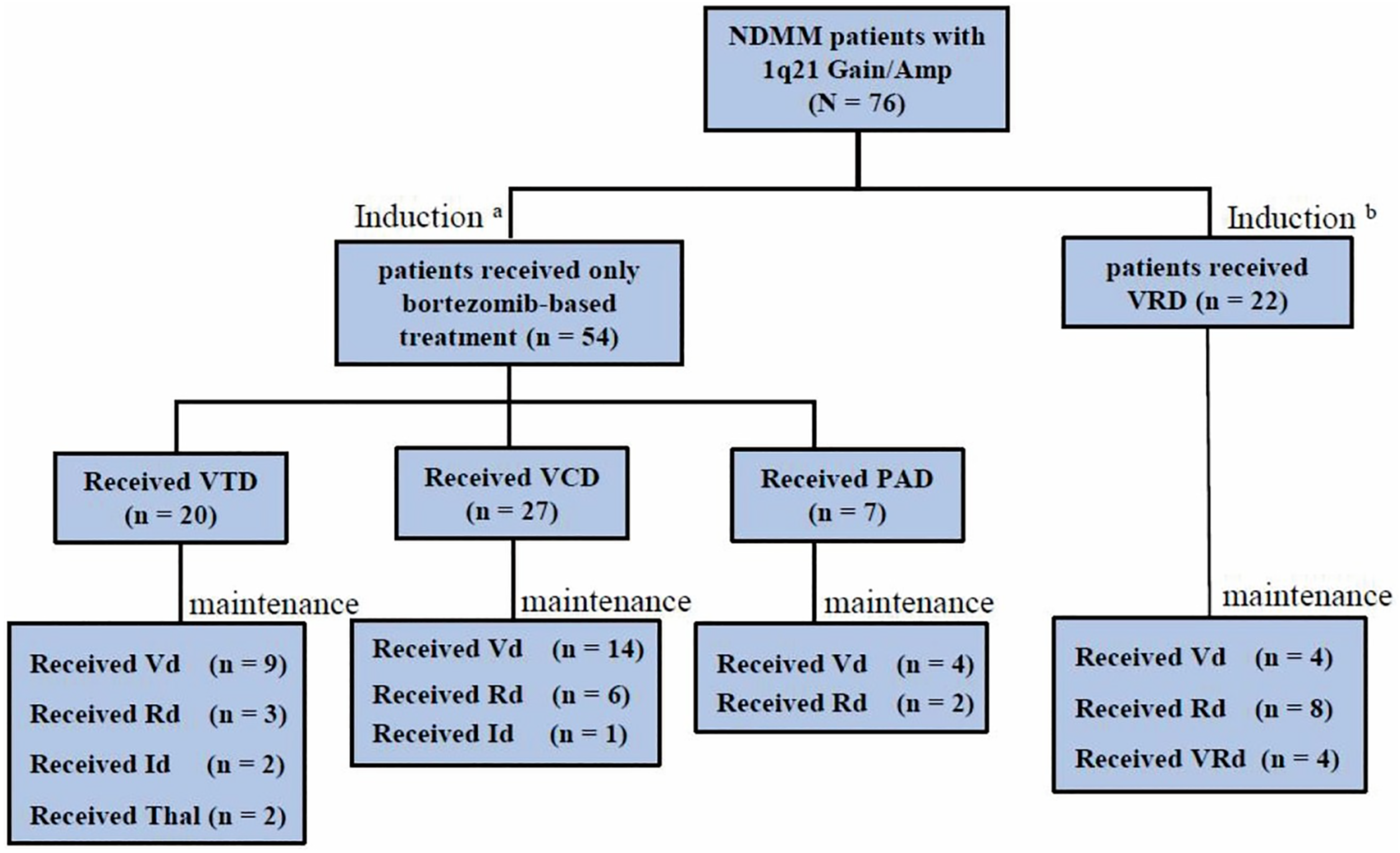
The treatment regimen of the cohort. VTD, bortezomib with thalidomide and dexamethasone. VCD, bortezomib with cyclophosphamide and dexamethasone. PAD, bortezomib with doxorubicin and dexamethasone. VRD/VRd, bortezomib with lenalidomide and dexamethasone. Vd, bortezomib and dexamethasone. Rd, lenalidomide and dexamethasone. Id, ixazomide and dexamethasone. Thal, thalidomide. a3.7% patients received ASCT. b13.6% patients received ASCT. ASCT, autologous hematopoietic stem cell transplantation.
Response Rate and Survival Outcome
The best outcome assessment was that 17.1% achieved CR and 44.7% achieved VGPR in this study. Sustained remission is defined as the best remission to relapse. The median time of sustained remission was 20.7 months (95% confidence interval [95% CI] 15.9-25.5 months) in all cases. Figure 2a displays the PFS, and Figure 2b illustrates the OS for the entire cohort. Furthermore, Figures 2c-f present the PFS according to risk stratification by induction regimens, VGPR, ISS and LMR. Among the 76 patients, the median PFS was 28.1 months (95% CI 21.3-34.9 months), and the median OS was 60.9 months (95% CI 47.9-73.9 months). At the data cutoff, a total of 45 (59.2%) patients experienced disease progression.

Kaplan-Meier survival curves. (a) Progression-free survival (PFS) of the whole cohort, (b) overall survival (OS) of the whole survival; PFS according to risk stratification by induction regimens (c), VGPR (d), ISS stage (e) and LMR (f).
The results of univariate analysis showed that NDMM patients with ISS stage III and only bortezomib-based induction therapy were identified as risk factors associated with inferior PFS (Figure 2c and e). Multivariate analysis was conducted on a cohort of 76 NDMM patients, incorporating findings from univariate analysis, which included clinical and cytological characteristics such as ISS stage, LMR, therapeutic regimen, and treatment response. Both multivariate analysis and univariate analysis showed that obtaining a VGPR or better response had no impact on PFS (P > 0.05). Only bortezomib-based induction therapy (P = 0.003, HR 0.246, 95% CI 0.097-0.620), ISS stage III (P = 0.003, HR 3.844, 95% CI 1.588-9.308) and LMR <3.6 (P = 0.032, HR 0.491, 95% CI 0.257-0.940) were significantly associated with adverse PFS in multivariate analysis.
Impact of VRD on 1q21 Gain/Amp
As mentioned above, 54 patients received only the bortezomib-based regimen, and 22 patients received the VRD regimen. Baseline characteristics were nearly equivalent between the VRD and only bortezomib-based groups (Table 2). VRD as induction therapy resulted in superior outcomes over only PIs-based induction therapy. We further explored the effects of induction therapy on patient outcomes, and the response to treatment is shown in Figure 3. VRD was associated with deeper response (VGPR: 63.6% vs 37.0%, P = 0.034), lower disease progression rate (31.8% vs 70.3%, P = 0.002), longer sustained remission (median 49.7 months vs 18.3 months, P = 0.030), and longer PFS (median 61.9 months vs 22.9 months, P = 0.032) than those who received only bortezomib-based treatment. The median OS was 60.9 months in the bortezomib-based treatment only group, and the median OS was not reached in the VRD group, but there was no statistical significance between the two (P = 0.200).
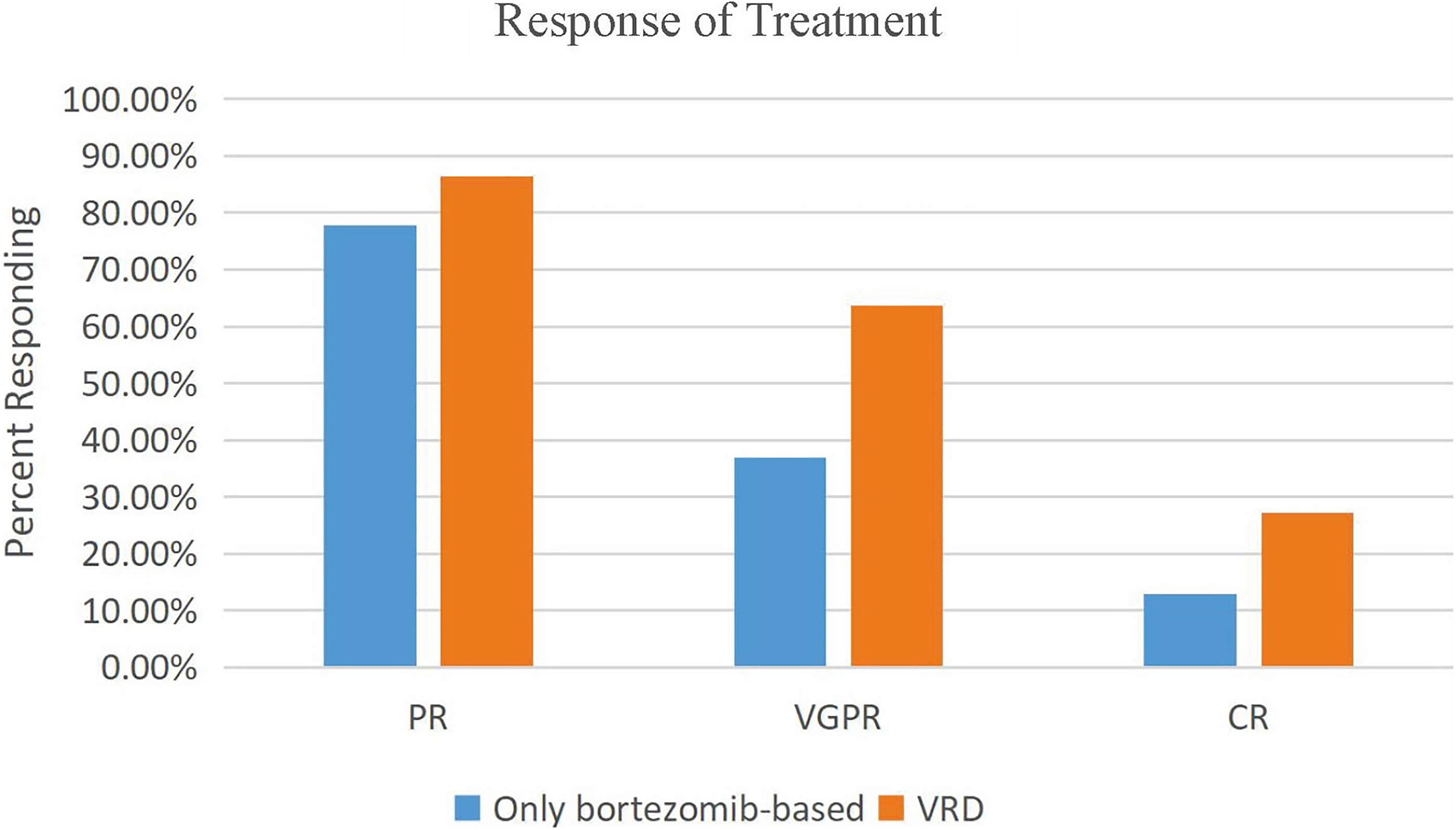
Best response to only bortezomib-based treatment and VRD. Percentage of patients achieving at lease a complete response (≥CR), very good partial response (≥VGPR), and partial response (≥PR) to PXD and VRD induction is demonstrated. CR, complete response; VGPR, very good partial response; PR, partial response; VRD, bortezomib-lenalidomide-dexamethasone.
Comparison of Clinical Features and Outcomes Data Between the Only Bortezomib-based Treatment Group and VRD Group

High-risk cytogenetic abnormalities were detected by fluorescence in situ hybridization (FISH) or metaphase cytogenetics, including del 17, t (4;14), t (14;16).
CR, complete response; VGPR, very good partial response; PR, partial response; VRD, bortezomib-lenalidomide-dexamethasone.
Discussion
This retrospective single-center study shows that chromosome 1q21 abnormalities are associated with poor outcomes in NDMM patients in the real world. MM patients with 1q21 gain/Amp may have a shorter median PFS and a higher disease progression rate. We found that the deep response rate, relapse rate and survival outcome of patients with 1q21 gain/Amp could be appropriately improved by bortezomib plus lenalidomide and dexamethasone (VRD) therapy.
MM is a genetically intricate disorder marked by diverse clinical heterogeneity in response rates and survival outcomes. 2 Compared to patients without 1q21, patients with 1q21 gain/Amp may have a much deeper response and were more likely to obtain a VGPR or better.4,10,11 Although there are controversies, most researchers considered that patients with 1q21 gain/Amp had shorter durations of PFS and OS than patients without this cytogenic abnormality.12–14 This means that a deeper response may not indicate a survival benefit. Some studies have discussed the relationship between response dynamics and survival.11,15 It seemed that patients who responded to chemotherapy and achieved CR rapidly were prone to suffer clonal evolution and early relapse.11,15 Another potential rationale was that the response depth of CR or VGPR was not enough for survival benefit, and most patients relapsed despite achieving CR in patients with 1q21 gain/Amp. Therefore, minimal residual disease (MRD) negativity may be a better indicator. The phase 3 MAIA and ALCYONE studies not only provided evidence that MRD-negative status was associated with improved PFS but also confirmed that sustained MRD negativity lasting ≥6 and ≥12 months was important for survival outcome. The data supported utilizing MRD persistence as a predictive and prognostic instrument in NDMM. 16 Mohan M et al also concluded from a 568 consecutive MM cohort that obtaining MRD slowly and sustained MRD negativity were much better than obtaining MRD rapidly but MRD conversion. 17 Patients who lose MRD negativity may have similar poor outcomes regardless of the initial risk stratification. 17 Our study had similar results to that of this 1q21 gain/Amp MM cohort where patients had significantly poor outcomes in terms of PFS and OS, and the median PFS and OS were 28.1 and 60.9 months, respectively. The ≥VGPR group did not have a better PFS in patients with 1q21 gain/Amp. However, it is worth mentioning that these patients treated with VRD had a higher ≥VGPR rate and had a superior PFS. Further exploration showed that compared to the patients treated with only bortezomib-based protocols initially, patients with 1q21 gain/Amp had longer sustained remission in the VRD group. This result suggested that the duration of remission was more important for long-term survival than deep remission.
The debate continues as to whether NDMM with 1q21 gain/Amp could benefit from PIs or IMiDs.10,18 Tang HKK et al reported that +1q was an adverse factor for OS in MM uniformly treated with bortezomib-based induction, which was partially alleviated by ASCT. 18 Similarly, in another study, the authors concluded that the application of bortezomib-containing induction chemotherapy in patients with 1q2l amplification can efficiently improve the remission rate. The study reported by Xiao Hu et al showed that the administration of lenalidomide-based treatment could prolong OS in NDMM patients harboring 1q+. However, in the study, the author also speculated that the crude protective effects of lenalidomide could be partly attributed to the better health status of patients, given that lenalidomide is used more frequently in patients with better renal function. 10 The use of upfront ASCT, daratumumab-based treatment, and bortezomib-based treatment did not demonstrate a significant association with differing OS or PFS compared to patients who did not receive these treatments. 10 It seemed that either bortezomib-based therapy alone or lenalidomide-based therapy alone was inadequate for NDMM patients with 1q21 gain/Amp. Although NCCN and Chinese guidelines regard the VRD regimen as a preferred first-line therapy for NDMM, it is unclear whether VRD could sufficiently benefit patients with 1q21 gain/Amp. The data from Timothy M. Schmidt et al suggested that when treated with RVD induction, patients with +1q should be considered at very high risk for early progression in MM when ≥four copies are detected or in the context of other high-risk cytogenetic abnormalities. Patients with 1q21 gain/Amp do not benefit significantly from RVD regimens. 4 The study of Efstathios Kastritis et al reported the opposite result where the use of the PI and IMiD combination was associated with somewhat better outcomes but only partly mitigated the effects of +1q21. 12 However, in the era of novel agents, a head-to-head comparison of all these drugs is still lacking, particularly for individuals with 1q21 gain/Amp. We tried to compare the efficacy of only bortezomib-based regimens and VRD as initial treatments for patients with 1q21 gain/Amp. As mentioned above, patients had longer sustained remission in the VRD group. Both univariate and multivariate analyses indicated that induction therapy was an independent factor for PFS in NDMM with 1q21 gain/Amp. Patients initially treated with VRD seemed to be liable to acquire VGPR or better (63.6% vs 37.0%, P = 0.034) and had a lower disease progression rate (36.4% vs 70.3%, P = 0.006) and superior PFS (median 61.9 months vs 22.9 months, P = 0.032) than those who received only bortezomib-based treatment. In our previous study, sequential administration of only bortezomib-based treatment as an initial therapy followed by Rd as a continuous treatment may not be inferior to VRD for first-line treatment in the whole NDMM cohort regardless of cytogenetics. 19 In this study, it was shown that only the bortezomib-based regimen had a low VGPR rate and a high disease progression rate in 1q21 patients compared with VRD. This result indicated that synergistic mechanisms of bortezomib and lenalidomide may account for overcoming some adverse effects of 1q21 gain/Amp rather than one agent-based chemotherapy or sequential strategies.
Combination treatments with different new agents could increase OS and decrease drug resistance in myeloma patients.20,21 The mechanisms of how PIs and immunomodulatory drugs act synergistically and overcome the resistance of 1q21 gain/Amp remain unclear. MM tumors bearing the amp 1q21 abnormality are associated with decreased TRAF3 expression, potentially resulting in increased noncanonical nuclear factor kappa B (NF-κB) pathway activity. 22 As reported, bortezomib could increase the inhibitory kappa B (IκB) protein level by inhibiting its ubiquitination degradation and then stabilizing the NF-κB complex, inducing the apoptosis of MM cells.21,22 Lenalidomide prevents the proliferation of MM cells by binding to cereblon and promoting the ubiquitination degradation of the transcription factors Ikaros (IKZF1) and Aiolos (IKZF3).21,23 Furthermore, lenalidomide promotes the activation of diverse immune cells such as CD8, CD4, and NK T cells, while suppressing regulatory T cells. 23 Bortezomib preferentially blocks the ubiquitination degradation of cereblon but not IKZF1/3 at the clinical dose, which enhances the anti-MM effect of lenalidomide. 23 Based on the different pharmaceutical mechanisms and interactions of the two agents, we hypothesized that VRD combination treatments could decrease drug resistance and improve the efficacy of MM patients. This is also true for MM patients with 1q21 + .
Conclusion
In conclusion, 1q21 gain/Amp may indicate poor prognosis for NDMM patients. Compared with the sequential administration of bortezomib and lenalidomide or only bortezomib-based protocols, NDMM patients with 1q21 gain/Amp may benefit more from VRD as initial treatments. Although there was no difference in the final OS benefit, more patients could attain continuous deep remission and were more likely to have a superior survival rate. Pharmacokinetic coordination may be the main reason that 1q21 patients can benefit from VRD. Although the guidelines recommend that only bortezomib-based protocols as initial treatment for NDMM patients are applicable, VRD should be emphasized as the preferred choice for patients with 1q21 gain/Amp. The primary limitations of this study are the single-center retrospective study and the relatively small sample size. Therefore, a larger sample size and prospective clinical trials are warranted to confirm the results, and further studies are needed to explore how the VRD regimen improves the outcomes of NDMM patients with 1q21 gain/Amp.
Footnotes
Acknowledgments
Not applicable.
Authors’ Contributions
Study design: QZ, JW, and FX; data collection: QZ and JW; data analysis: QZ, JY, and FX; data interpretation: QZ, JW, and FX; preparation of manuscript: QZ and FX; literature search/analysis: QZ, JY, YZ, JS, and YL; funding collection: FX, QZ, and JW.
Code Availability
Not applicable.
All authors have read and approved the final manuscript.
Consent for Publication
Written informed consent to publish was obtained from study participants.
Data and Materials Availability
The datasets analyzed in the present study can be obtained from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study adheres to the tenets of the Declaration of Helsinki. Ethical permission was obtained from the Ethics Committee of Mianyang Central Hospital on April 20, 2021 (No. S2021029), Mianyang, China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the No. 2021YJ013 research project from Mianyang Central Hospital, by the No. S20074 research project from the Medicine Association of Sichuan Province and by the No. 2022NSFSC1603 research project from the Sichuan Science and Technology Program.
