Abstract
Globally, hepatic cancer ranks fourth in terms of cancer-related mortality and is the sixth most frequent kind of cancer. Around 80% of liver cancers are hepatocellular carcinomas (HCC), which are the leading cause of cancer death. It is well known that HCC may develop resistance to the available chemotherapy treatments very fast. One of the biggest obstacles in providing cancer patients with appropriate care is drug resistance. According to reports, more than 90% of cancer-specific fatalities are caused by treatment resistance. By binding to the 3'-untranslated region of target messenger RNAs (mRNAs), microRNAs (miRNAs), a group of noncoding RNAs which are around 17 to 25 nucleotides long, regulate target gene expression. Moreover, they play role in the control of signaling pathways, cell proliferation, and cell death. As a result, miRNAs play an important role in the microenvironment of HCC by changing immune phenotypes, hypoxic conditions, and acidification, as well as angiogenesis and extracellular matrix components. Moreover, changes in miRNA levels in HCC can effectively resist cancer cells to chemotherapy by affecting various cellular processes such as autophagy, apoptosis, and membrane transporter activity. In the current work, we narratively reviewed the role of miRNAs in HCC, with a special focus on tumor microenvironment and drug resistance.
Introduction
Globally, hepatic cancer ranks fourth in terms of cancer-related mortality and is the sixth most frequent kind of cancer. 1 Approximately 80% of liver cancers are classified as hepatocellular carcinomas (HCC), which are the primary cause of cancer-related fatalities. 2 Despite the advancements in therapeutic approaches, advanced-stage HCC continues to have a dismal prognosis, with a life expectancy of approximately 6 months following diagnosis. 3
Several factors contribute to an increased risk of HCC. These factors include age, cirrhosis, genetic conditions, exposure to toxins, excessive alcohol consumption, and smoking. Additionally, infection with hepatitis B or C viruses is recognized as a significant risk factor for HCC development. In recent years, nonalcoholic fatty liver disease (NAFLD) and its more severe form, nonalcoholic steatohepatitis (NASH), have emerged as important causes of HCC in Western countries. NAFLD and NASH have also become leading indications for liver transplantation following hepatitis C virus (HCV) infection.4,5
When it comes to treatment, radiofrequency ablation (RFA) is currently considered the primary therapy for small HCC tumors (≤2 cm) due to its comparable effectiveness to surgical excision. RFA presents advantages such as causing less disruption to surrounding tissues and allowing for quicker recovery. In individuals with intermediate-stage HCC, transcatheter arterial chemoembolization is often considered as an alternative initial therapy. However, its effectiveness is limited due to the counteractive effects of vascular growth triggered by oxygen deprivation. 5 For late-stage unresectable HCC, where the options are limited, the multitargeted kinase inhibitor sorafenib is considered the most effective treatment. 6 While it may provide only a marginal improvement in patient survival, sorafenib is currently the only systemic treatment available for advanced HCC.6,7 Furthermore, some natural compounds, such as sanguinarine, have the potential to regulate microRNA-16 (miR-16) expression and its impact on the treatment of HCC. 8 However, these findings need further investigation.
Through attachment to the 3'-untranslated region (3'-UTR) of target messenger RNAs (mRNAs), microRNAs (miRNAs), a cluster of RNA molecules that do not encode proteins which are around 17 to 25 nucleotides long, regulate target gene expression. Moreover, they play an essential role in the regulation of signaling pathways, cell growth and division, and cell death. 9 Many mRNAs can be subjected to the influence of a single miRNA, and several miRNAs can work together to suppress a single mRNA. 10 Therefore, miRNA-guided posttranscriptional control of the gene expression is achieved. P53, RAS/MAPK, PI3 K/AKT/mTOR, WNT/β-catenin, MET, MYC, and transforming growth factor beta (TGF-β) are only a few of the molecular pathways that are deregulated during the development of HCC. Several important pathways that are connected to cancer can be impacted by genetic, epigenetic, and aberrant miRNA expression changes. 11 Consistent dysregulation of miR-122, miR-199, miR-221, and miR-21 appears to be particularly significant in HCC. Notably, miR-199a and miR-122 are 2 of the miRNAs that are most frequently present in healthy liver tissue. 12
It is generally recognized that HCC can quickly acquire inherent resistance to chemotherapy medications that are now accessible.13,14 Drug resistance in the treatment of cancer patients is a primary obstacle in achieving successful care. According to reports, over 90% of cancer-specific fatalities are caused by treatment resistance. 15 Resistance to drugs used for treating cancer may be categorized into 2 groups determined by the traits of resistance exhibited by cancer cells: primary drug resistance (PDR) and multidrug resistance (MDR). Cancerous cells have the potential to grow unresponsive to a single kind of anticancer medication in PDR, but they may retain their susceptibility to anticancer medications from other categories. Cells afflicted with cancer develop new structures and processes in the MDR that make them resistant to anticancer medications. The main factor for HCC chemotherapy failure is MDR. Compared to cancers without MDR, those with it have a number of distinguishing characteristics. For instance, tumors with MDR manifest high levels of the point of apoptosis initiation, cellular glucose metabolism in the presence of oxygen, deficient oxygen levels, and drug-efflux transporter activity. 16 Many studies have repeatedly showed that the variable involvement of miRNA expression an important role in the incidence and development of HCC, and a significant quantity of miRNAs are directly involved in the treatment resistance of cancer cells.15,17,18 miRNAs have the ability to either increase or decrease hepatoma cell drug resistance based on several regulatory mechanisms. A thorough grasp of the fundamental mechanism behind drug resistance is provided by all the studies conducted on the compounds used in chemotherapy. Knowing the processes by which miRNAs contribute to the emergence of drug resistance might aid in the creation of fresh methods for combating it in cancer therapy.19–23
The tumor microenvironment (TME), which includes the TME of HCC, is intricate and involves interactions among tumor cells like HCC cells, the blood arteries around them, stromal cells, and immune cells. These interactions are crucial for carcinogenesis, angiogenesis, and metastasis. These interactions also involve metabolites, growth factors, cytokines, chemokines, noncoding RNAs, and immunological checkpoint molecules.24–26 A model for the relationship that exists between the TME and tumor growth is liver cancer. An complicated microenvironment is created by the chronic inflammation of liver damage resulting from infection with a hepatitis virus as well as the generation of cytokines and growth factors inside the parenchyma. 27 TME affects tumor development, metastasis, and eventually prognosis. Hence, the primary function of TME is to engage dynamically with cancerous cells. 28 In the current work, we narratively reviewed the function of miRNAs in HCC, with an especial emphasis on the surroundings of the tumor and its connection to resistance against drugs.
Hepatocellular Cancer: Mechanism of Initiation
Over 90% of HCC cases are associated with chronic liver disease, with cirrhosis being a major risk factor. Cirrhosis can be caused by various conditions such as chronic alcohol consumption, α1-antitrypsin deficiency, haemochromatosis, and primary biliary cholangitis, all of which are significant risk factors for HCC. In cirrhotic patients, HCC is the leading cause of death.29,30
NAFLD is another chronic liver disease that has lately raised concerns due to its increasing occurrence and association with metabolic diseases such as diabetes and obesity. 31 Another risk factor is viral infections with hepatitis B virus (HBV), HCV, and HDV that are also risk factors for HCC, with HBV infection and include around 50% of cases. 32 However, the chances of HCV leading to cancer have significantly decreased due to the use of antiviral drugs that help patients achieve sustained virological response (SVR). 30
Nonalcoholic Fatty Liver Disease
NAFLD affects around 25% of people on the planet. Nevertheless, only a small percentage of them progress to more severe stages such as end-stage liver disease (ESLD), which is observed in fewer than 13% of individuals diagnosed NAFLD. 31 NAFLD has various stages that correspond to distinct signs in liver tissue, beginning with nonalcoholic fatty liver and advancing to NASH, fibrosis, and ultimately cirrhosis. After cirrhosis, patients may develop HCC and ESLD. 33 Changing from NAFLD to HCC involves many factors, such as insulin resistance, which disrupts the metabolism of glucose, ketones, and lipids, resulting in extra free fatty acids (FFAs) that affect the normal mitochondrial function in liver cells, lipotoxicity occurs when glucose metabolism pathway interfered with high levels of free fatty acids and leads to increase the production of toxic byproducts. 34 These factors make measuring the effect more difficult. HCC cells exhibit higher expression levels of genes that promote de novo lipogenesis, a characteristic unique to cancer cells compared to normal cells. These results in an increased accumulation of lipids in HCC cells due to elevated production of reactive oxygen species (ROS) and steatosis, which further contributes to oxidative stress in patients with NASH. Consequently, HCC cells adapt and thrive in the lipotoxic environment created by excessive lipid accumulation and oxidative stress. In patients with NAFLD, there is an abundance of FFAs being processed by the liver mitochondria. This excessive influx of FFAs leads to incomplete beta-oxidation, reduced ketogenesis (a process involving the production of ketone bodies from fatty acids), and an excess production of ROS, causing oxidative stress.35,36 Cancer outcomes is more linked to overload of ROS with inflammation which not only seen in patients with NAFLD but also in patients with hepatitis.
Hepatitis B Virus
Almost 296 million people, including more than 6 million children under 5 years old, have hepatitis B, and 25% of chronic hepatitis B infections turn into liver cancer. 37 HBV is a DNA virus and by inserting itself into the host genome could cause insertional mutagenesis which in turn will activate cancer-causing genes. HBV can raise HCC chance even in patients who don't have cirrhosis, although the majority of patients with HBV that result in HCC have cirrhosis at diagnosis.
The risk of HCC has increased dramatically due to East Asia's high frequency of endemic HBV infection. That is beyond what is considered cost-effective for surveillance in both men (at 40 years old) and women (at 50 years old). Consequently, implementing surveillance programs has become necessary in this region. 30
Patients in their early 30s or 40s in Africa show symptoms of HCC, most likely as a result of being exposed to aflatoxin B1, a carcinogen frequently present in contaminated food. The combined effect of aflatoxin B1 and HBV infection increases the likelihood of developing HCC in these individuals. Fortunately, the implementation of HBV vaccination programs in certain parts of Asia has led to a decrease in HCC cases. The success of these vaccination efforts highlights the effectiveness of preventive measures in reducing the incidence of HCC. However, it is important to note that many regions are still in the process of introducing general vaccination programs and have yet to experience the full benefits of widespread HBV vaccination. 30
Hepatitis C Virus
Chronic HCV infection is the primary underlying liver disease in patients with HCC in Japan, North America, and Europe. 38 Unlike HBV, which can integrate itself into the genome, HCV cannot do so. Therefore, the risk of HCC development in HCV-infected individuals is mainly associated with the presence of cirrhosis or chronic liver damage characterized by bridging fibrosis. However, with the advent of direct-acting antiviral (DAA) therapy, an increasing number of patients with HCV infection have achieved a SVR, indicating successful elimination of the virus from their body. As a result, the risk of HCC in these patients has significantly decreased. Studies have shown that achieving an SVR through DAA treatment has led to a reduction in HCC risk by 50% to 80%. Nevertheless, a considerable number of patients, particularly those belonging to ethnic or racial minorities or individuals residing in low socioeconomic areas, have not undergone testing for HCV and are unaware of their infection status. This lack of awareness contributes to delayed diagnosis and treatment. 30 Furthermore, patients who have developed cirrhosis due to HCV infection continue to face a persistent risk of developing HCC, even after achieving an SVR through successful treatment. The risk of HCC remains high, with an annual incidence rate of more than 2%. Consequently, these individuals require regular and vigilant surveillance to detect any potential signs of HCC at an early stage.
Tumor Microenvironment in HCC
The TME can change in such a way that can be one of the major tumorigenesis factors. Here are some of the components that enter TME and some of the processes that occur there.
Carcinogenic Factors
Chronic inflammation contributes to tumor development. 39 Inflammatory processes followed by alcoholic abuse, NASH, and chronic infection with HBV or HCV is associated with HCC. 40 Moreover, obesity, smoking, diabetes mellitus, and NAFLD are the other risk factors, 41 and molecularly, TERT, TP53, and CTNNB1 are the most frequent mutant genes.42–48
Carcinogenic Processes
Angiogenesis
To provide tumor growth, generating new blood vessels from the former ones, called angiogenesis, is vital for oxygen and nutrient supply, damaging substances disposal, and metastasis. 49 Liver tumors exhibit abnormalities in the vascular system, consequently, vessels are irregular and extremely leaky. Hence the region develops hypoxia and/or necrosis which have high tumorigenesis potential. Atypical microvasculature is generally seen in 2 forms: (1) arterialized: tight vessels engulfed by smooth muscle cells and (2) capillarized: nonfenestrate capillaries with laminin support in the basement.50–52
Solid tumors’ angiogenesis (including HCC) relies on 3 fundamental steps in endothelial cells: activation, proliferation, and migration. Angiogenic factors activate endothelial cells. Due to protease secretion, they weaken cell interactions and consume the basement membrane and extracellular matrix. Matrix proteins release several angiogenic factors (described in detail below) after decomposition which secondarily stimulate endothelial cells. Then, owing to proliferation and migration, endothelial cells form a tubular structure which ultimately develops new blood vessels. 53
Angiogenic Factors
Vascular endothelial growth factor (VEGF): liver cells and endothelial cells express VEGF and VEGF receptor (VEGFR)-1 and 2, respectively.54–57 Whereas communicating with VEGFR-1 results in hepatocyte proliferation, VEGFR-2 signaling stimulates sinusoidal endothelial cells proliferation. 58
Fibroblast growth factor-2 (FGF-2): El-Assal et al 59 found a relation between FGF-2 and vessel density in HCC.
Angiogenin: similarly, in a study with 41 patients with HCC, Hisai et al 60 support the idea angiogenin expression correlated with microvasculature mass.
Hypoxia
Insulin-like growth factor-2 (IGF-2) and hexokinase II secreted due to hypoxia trigger the growth of HCC.61,62 Hypoxia, also stimulate Rac and angiogenic factors including IGF-2, VEGF, and hypoxia-inducible factor 1 (HIF-1).62,63 Id-1 and Rac (differentiation/DNA synthesis inhibitor) result in the elevation of HIF-1 lifetime, leading to VEGF upregulation. Moreover, hypoxia can lead to the downregulation of some tumor suppressors like p53 and von Hippel-Lindau.64–67
Inflammation
Inflammatory processes caused by alcoholic/NASH and viral hepatitis play important roles in the pathogenesis of HCC. 68 Free radicals secreted by immune cells in inflammation can harm DNA, thus, neoplastic transformation may develop. 40 Tumor-associated macrophages (TAMs) originating from Kupffer cells (KCs) or monocytes arrive in tumor tissue by various factors including chemokines secreted from tumor cells and inflammatory cells.40,69 TAMs’ cytokines and growth factors may trigger HCC formation processes. 40 One of the most potent released driver molecules is IL-6. 40 According to Naugler et al, IL-6 mice have a lower prevalence of HCC than wild-type mice. 70 furthermore, inflammation positively impresses metastasis progress. 71
Fibrosis
Fibrosis process, happens at the end stages of chronic inflammatory conditions like cirrhosis, is a complex mechanism that augments the risk of HCC development. Unbalances in the extracellular matrix (ECM), accumulation of ECM contents, and disorganization of the liver structure are characteristics of hepatic fibrosis. 72 Hepatic stellate cells (HSCs) owing to modifying the TME and affecting the fibrosis process, have a crucial role in HCC development. HSCs transform into myofibroblasts in chronic hepatic injury conditions, afterward, they secrete compounds that promote fibrosis. These factors include cytokines like TGF-β, hepatocyte growth factor (HGF), VEGF, bFGF, PDGF, and other factors like matrix metalloproteinases (MMPs), the tissue inhibitors of metalloproteinases, and ECM proteins. 73 The more fibrosis advances, the more HSCs are stimulated and with a vicious cycle, more fibrosis is developed again. 74
Cellular Components of TME
The liver is an immune organ that always exposes to small intestine pathogens and this status requires a rich structure of immune cells. Commonly responses and reactions to the pathogens, do not cause inflammation; however, imbalances and liver-involved diseases may lead to chronic inflammation and this inflammatory process has been proposed as a critical risk factor inducing HCC.75,76 Since responses are modulated by cells, the most crucial cells involved in HCC development are discussed in the following sections.
Nonimmune Cells
Natural Killer Cells
Natural killer (NK) pioneers in defensive mechanisms against viral infections and HCC carcinogenesis in the liver. Due to its key role in balancing immune responses, abounding investigations aimed at NK cells. Hypoxia, altering NK receptors (NKRs) function, and immune modulatory molecules influences lead to NK cell dysfunction and consequently cause poor prognosis and impairment of antitumor immunity.77,78
Kupffer Cells
A significant part of liver immune homeostasis is due to KC activities. 79 However, influenced KCs by pro-inflammatory molecules and repressor cells, trigger KC-expressed receptors on myeloid cell-1 (TREM-1) that are a key factor in the initiation of HCC.80,81
Dendritic Cells
Dendritic cells (DCs) as dedicated antigen-presenting agents are capable to evoke T cells against tumor-associated antigens which cause the progression of the malignancy. Unique vaccines based on DCs have effective results in the treatment of many cancers, most significantly HCC and prostate cancer, melanoma, and renal cancer.79,82
Hepatic Stellate Cells
HSCs are stockpiles of lipids and vitamin A, and they are mainly found in the Disse's space. By a successive process, KCs release multiple signaling proteins such as TGF-β1 which trigger the collagen and extracellular matrix substances production in HSCs. Moreover, Activated HSCs secrete leptin. Therefore, the mentioned process boosts fibrosis.83–85
Liver Sinusoidal Endothelial Cells
Liver sinusoidal endothelial cells (LSECs), which is characterize by the most significant basement membrane and wide fenestrations, transport loads from sinusoidal space to the parenchyma or Disse.86,87 Physiologically, fenestrations are crucial to liver function. Through the circulation system, they serve as the primary means of communication between the liver and the rest of the body. 88 Also, in the liver, spleen, and bone marrow, sinusoids are irregularly shaped tubular spaces through which blood passes. 89 LSECs recruit leucocytes into the liver tissue through a long process of adhesion and pass within a transcellular way. Owing to exorbitant immunosuppressive leukocyte arrival in pathological conditions, pathogens may promote tumorigenesis.90–92
Immune Cells
Myeloid-Derived Suppressor Cells
Myeloid-derived suppressor cells (MDSCs) will spread in pathological status and synthesize immunosuppressive enzymes including arginase and inducible nitric oxide synthase. 93 Cytokines like IL-1β, MCP-1, VEGF, GM-CSF, and G-CSF are able to activate MDSCs to infiltrate. 94
Tumor-Associated Macrophages
Macrophages (Ms) are derived from circulating monocytes in the bloodstream, undergoing a process of differentiation and maturation. Once differentiated, these macrophages then migrate from the bloodstream into various tissues throughout the body, where they play crucial roles in immune responses, tissue homeostasis, and the clearance of cellular debris. This migration of macrophages helps to establish a localized immune response within tissues, allowing them to fulfill their diverse functions in tissue health and immunity. 79 Between 2 phenotypes, M1 and M2, the M2 phenotype promotes tumor initiation, advancement, and metastasis process.95,96
Tumor-Associated Neutrophils
Similar to TAMs, tumor-associated neutrophils (TANs) have 2 phenotypes, N1 which suppress tumor and N2 which promote tumor progression.97,98 Evidence shows that multiple factors can modulate the activity of TANs such as interferon type 1, TGF-β, and CD8+ T cells.99,100 This facilitatory role of TANs is a hot topic of interest nowadays.
Tumor-Infiltrating Lymphocytes
The population of tumor-infiltrating lymphocytes, not singularly, but coupled with CD8 + cytotoxic T lymphocytes (CTLs) count is a crucial factor to evaluate HCC progression.101,102
Regulatory T Cell
Research indicates that regulatory T cell (Tregs) in special conditions may exhibit a compromising role in immune responses. 103
CD8+ Cytotoxic T Lymphocytes
CTLs’ immune functions are associated with patient survival, nevertheless, although the competence of their immune responses is limited.79,104
Signaling Molecules and Pathways
There is substantial evidence that, at different stages of HCC, a variety of molecules communicate with each other and lead to inflammation and HCC. Cytokines, IL-6, VEGF, and FGF-2 are the ones that promote carcinogenic processes, by contrast, TGF-β has an antitumor role. 105 The most significant chemokines are CXCR3 (inflammatory cells chemotaxis and cancer cells advancement),106,107 X-C motif chemokine receptor1 (XCR1) (antigen presentation and cancer progression),108,109 C-C motif chemokine ligand 2 (CCL2) (promotor of HCC invasion), 110 CXC (hepatocyte repairing and alteration of TME in HCC favor).111,112 Sex hormones are also related to the incidence and prognosis of HCC. Evidence suggests that the difference between male and female sex hormones leads to poor prognosis and more mortality in male patients. 113 Interestingly, for female subjects, after menopause, this protective property is attenuated. 114 Exosomes as a particular signaling pathway may promote angiogenesis, and tumor invasion. 115
Effect of miRNAs in Cancer and Cancer Therapy
MiRNAs are tiny RNA molecules, about 22 nucleotides long, that do not make proteins but do regulate gene expression. They are created through a multistep process involving the Drosha complex, which produces pre-miRNA from primary miRNA transcript in the nucleus, followed by exportin 5-mediated nuclear export of pre-miRNA. 116
miRNAs possess the ability to modulate the expression of a wide range of genes, thereby exerting specific effects on various biological processes. When it comes to cancer, miRNAs are generally classified into 2 categories based on their role in gene regulation: tumor-suppressor (TS) miRNAs and onco-miRNAs. 117
Onco-miRNAs that are firmly established include the miR-17-92 cluster, which is situated in intron 3 of the C13orf25 gene at 13q31.3 and is overexpressed in lung cancer. 118 Overexpression of miR-21 has been observed in liver, 119 stomach, 120 breast, 121 and ovarian cancer. 122 While miR-181 is upregulated in oral squamous cell carcinoma, breast cancer, prostate cancer, and gastric cancer. 123
TS miRNAs that are well-established include let-7 family members, which have been linked to several cancers such as breast CSCs, pancreatic cancer, epithelial-mesenchymal transition, and high grade serous ovarian cancer. 124 Additionally, miR-29c is significantly downregulated in pancreatic cancer, which correlates with hyperactive Wingless-related integration site (Wnt) signaling pathways. 125
TS miRNAs prevent the onset of cancer by regulating the expression of oncoprotein-coding genes. They achieve this by promoting apoptosis, modulating the TME, inhibiting oncogene expression, stopping endothelial to mesenchymal transition, and suppressing cell proliferation. 126
In experiments conducted with PANC-1 cells, the introduction of miRNA-877-3p mimics resulted in a decrease in the growth rate of these cells. Conversely, when miRNA-877-3p inhibitors were applied, it led to an enhancement of PANC-1 cell proliferation. 127 Furthermore, miR-877-3p was found to exert suppressive effects on the growth of bladder cancer cells. This was achieved through its ability to increase the expression of p16, which is a tumor suppressor gene. The upregulation of p16 contributed to the inhibition of bladder cancer cell growth. 128
The tumor suppressor miRNA, known as miR-34a, plays a crucial role in impeding the growth of melanoma tumors by targeting the FLOT2 gene. 129 Increased expression of miR-34a leads to a reduction in both the mRNA and protein levels of ZEB1, whereas the suppression of miR-34a results in elevated ZEB1 levels. 130 ZEB1 is a transcription factor that exhibits high expression in various human cancers, including breast, liver, and lung cancers. By suppressing ZEB1, studies have shown a decrease in cancer cell proliferation and metastasis, suggesting that inhibiting ZEB1 holds promise as an effective approach for cancer treatment. 131 Similarly, in another context, miRNA-144-5p inhibits the expression of ITGA3 in thyroid cancer (TC). When miRNA-144-5p is overexpressed, it counters the tumor-promoting effects caused by the overexpression of ITGA3 on various biological functions associated with TC 132 (Figure 1).

The tumor suppressor microRNA (miRNA) mechanism in cells. (1) When damaged DNA is created after a problem with DNA is identified, tumor suppressor genes are activated. (2) These genes produce different RNAs and proteins by expression. (3) The proteins produced by these processes can act as transcription factors and produce other proteins. In this stage, these proteins can work in cooperation with other RNAs to process mature RNA. (4) The produced proteins can destroy the cell by using one of the 3 mechanisms of cell arrest (ie, inhibition of CDK and Cyclins), cell repair and cell apoptosis. Any change in the above process causes malignant cells. At the top of the page, a number of miRNAs that can be activated or inactive in the regulation of malignant cells in various studies with the presented mechanisms are also given.
Reduced expression of miR-30a leads to a decrease in its ability to target ZEB2, which is associated with increased cell plasticity, migration, and in vivo dissemination. 133 In medulloblastoma cells, there is a link between Myc-miR-29 and B7-H3, where Myc upregulates B7-H3 to promote tumor angiogenesis, which can be inhibited by miR-29 overexpression-mediated B7-H3 downregulation. 134 Tumor-suppressor miRNAs like miR-322 can boost immune responses, as seen in its ability to suppress galectin-3 expression. 135
Research has revealed diverse characteristics of onco-miRNAs. For instance, in esophageal squamous cell carcinoma, elevated miR-508-3p levels are linked to reduced disease-free and overall survival rates. 136 Additionally, miR-508-3p is highly connected with mesenchymal features in ovarian cancer, and it indirectly regulates the expression of several epithelial-to-mesenchymal transition (EMT)-associated genes. 137
Elevated miR-513a-5p levels indicate an unfavorable prognosis for patients with breast cancer. 138 In nonsmall-cell lung cancer, miR-510-3p drives cancer growth and hinders apoptosis by targeting phosphatase and tensin homolog (PTEN). 139 Overexpression of miR-135 in gastrointestinal cancers is linked to reduced overall survival in affected individuals. 140
Studies suggest that the upregulation of miR-135b in gastric cancer cells can increase their ability to proliferate, migrate, and invade by targeting the tumor suppressor gene CAMK2D, which belongs to the Ca2+/calmodulin-dependent protein kinase subfamily. Interestingly, the administration of miR-135b antagonist in vivo has been found to reduce tumor growth and metastatic potential in xenograft models 141 (Figure 2). Another oncogenic mechanism involves the inhibition of miR-9 activity by miR-424 and miR-503, which leads to a loss of its ability to maintain cells in an undifferentiated state and promotes cell lineage commitment and growth. 142

Onco-miRNAs mechanism in cells. (1) RAS and MYC genes are effective oncogenes in cell growth mechanisms. (2) These genes produce desired proteins and RNA. A number of these RNAs (miRNAs) and the cells associated with them are mentioned. (3) Growth hormone binds to some proteins, causing the transcription factor to activate and produce the desired protein. (4) Other proteins also contribute to cell growth, survival, and cell activity. Mutations in the shown genes 1 can disturb the mentioned mechanism and cause excessive production of cells, causing them to become malignant.
Effect of miRNAs in HCC Microenvironment
The TME contains cellular and noncellular components. The primary cellular components of the stroma of HCC, which produces extracellular components of the TME like ECM, different proteins, growth factors, cytokines, and proteolytic enzymes, are cancer-associated fibroblast (CAF), immune cells, HSC, and endothelial cells. 143 HCC is among the most hypoxic tumor kinds, which is another contributing factor. Poor arterial perfusion, rapid O2 uptake by tumor cells, and specific metabolism frequently result in hypoxic conditions within TME. 144
Changes in immunological phenotype caused by HCC tumor cells affect the number of immune cells in TME. 145 A recent study shows that reduced miR-539 promotes the synthesis of MMP-8 and increases TGF-β1 signaling, which results in M2 macrophage differentiation in TME. 146 Additionally, miR-15a, miR-16, and miR-107 promote M2 macrophage differentiation by paracrinely activating Wnt/β-catenin signaling in macrophages. 147 Higher levels of the chemokine CCL22, which draws Treg cells, are produced as a result of increased TGF-β inhibiting miR-34a expression. 148 Attracted Treg may help HCC immune system escape by lowering the proportion of CD8+ T cells in TME. 149 Evidence indicates that patients with HCC having an increase of miR-561-5p had lower CX3CL1 levels. Moreover, the absence of NK cells at the progression of HCC results from the loss of CX3CL1, which causes immune escape and metastasis via suppressing the production of interferon-γ and TNF-α.150,151 The other study demonstrates that TGF-β2 and bone morphogenetic protein 2 (BMP2) released into the TME by TANs cause the development of miR-301a-3p in HCC. Overexpression of miR-301a-3p induces the production and secretion of CXCL5 in HCC, which attracts more TANs to TME. 152 Lastly, the altered immune phenotype in the TME manifests as an increase in M2 macrophage, Treg, and TAN and a decrease in CD8+ T cells and NK cells, which can promote the growth and proliferation of HCC as its metastasis.
In HCC, aerobic glycolysis and hypoxia cause acidification, resulting in a pH differential between TME inner and outer layers.153,154 Hypoxia promotes the miR-377-5p and miR-576-3p expression, which can lead to the stimulation of HIF-1α production in HCC.155,156 Additionally, by the downregulation of miR-154-5p, HIF-1 increases the PLAGL2 level. Moreover, PLAGL2 activates EGFR/AKT signaling, promoting HIF-1α synthesis and creating a feedback loop that worsens TME hypoxia. 157 MiR-100-5p is also negatively related to HIF-1α, which inhibits the synthesis of lactate dehydrogenase A (LDHA). An enhanced level of LDHA leads to increased glycolysis and TME acidification. 158 According to findings, miR-10b and miR-21 generated by the acidity of TME in HCC increase cancer cell proliferation and metastasis and may work as therapeutic targets in addition to prognostic molecular markers. 159 Additionally, acidic TME can trigger the secretion of some extracellular vesicles with particular RNAs. One of them is miR-21, which not only promotes the production of cytokines for angiogenesis but also triggers HSC shift to CAF. 160 Patients with metastases in their lungs had increased levels of tumor-derived exosomal miR-1247-3p, which is produced in acidic TME and causes normal tissue fibroblast to shift to the CAF. IL-6 and IL-8 are released by HCC CAFs and are known to facilitate sorafenib resistance, migration, and EMT. 161
HCC has a hypervascular TME with an abnormal vascular network and hyperangiogenesis that aids in HCC invasion and metastasis. 162 Several studies demonstrate the part miRNAs play in the angiogenesis of HCC. Reducing miR-140, miR-26a, miR-195, and miR-503 levels leads to increasing VEGF expression, promoting angiogenesis.163–166 Additionally, the downregulation of miR-214 activates HDGF paracrine pathway, which in turn aids in the hypervascularity of HCC. 167 Another study has shown that in HCC cells, miR-125b overexpression reduced PIGF expression and changed the angiogenesis index. 168 Plus, it has been reported that miR-125b could lead to resistance against chemotherapeutics (paclitaxel, in this case) by suppressing the expression of pro-apoptotic Bcl-2 antagonist killer 1 (Bak1). 169 The rise of some miRNAs expression also can induce angiogenesis in TME. miR-146a is critical in promoting the endothelial cells’ angiogenic activity in HCC by boosting PDGFRA production and reducing BRCA1. 170 Recent research indicates that miR-424-5p increased HCC angiogenesis and cell proliferation by increasing E2F7 and activating VEGFR-2 signaling 171 (Figure 3).

The role of miRNAs in the microenvironment of HCC. Liver cancer cells can grow and cause HCC through overexpression or inhibition of miRNAs through 3 pathways: (1) affect the number of immune cells in TME, (2) acidification due to aerobic glycolysis and hypoxia, and (3) hyperangiogenesis. Abbreviations: CAF, cancer-associated fibroblast; ECM, extracellular matrix; HCC, hepatocellular carcinoma; HSC, hepatic satellite cells; LDHA, lactate dehydrogenase A; miRNA, microRNA; MMP-8, matrix metalloproteinase-8; TANs, tumor-associated neutrophils; TGF-β1, transforming growth factor-β1; TME, tumor microenvironment; Treg cells, T regulatory cells.
The ECM in HCC solid tumors contains extracellular fluid and multiple components like fibrin and glycoproteins. Changes in ECM can affect tumors by increases in proliferation and migration. HSCs and mesenchymal stem cells (MSCs) expression miR-1246 and miR-34a result in Wnt/β-catenin signaling activation, which inhibits E-cadherin, debilitates intercellular junctions and induces EMT.172,173
Moreover, by the sponge miR-26a/b-5p and activation of the IL-6/JAK2/STAT3 pathway and Wnt/β-catenin signaling, carcinogenesis and EMT rise in HCC. 174 In tumor tissues, lysyl oxidase-like 2 (LOXL2) is markedly overexpressed in patients with HCC. LOXL2 changes ECM components and HCC cell adherence in the TME and the development of metastatic niches. MiR-26 and miR-29 in HCC can downregulate LOXL2 transcription 175 (Table 1).
Summary of the Effect of miRNAs in HCC Microenvironment.

Abbreviations: CAF, cancer-associated fibroblast; ECM, extracellular matrix; EMT, epithelial-to-mesenchymal transition; HCC, hepatocellular carcinomas; HSC, hepatic stellate cell; miRNA, microRNA; TME, tumor microenvironment.
Effect of miRNAs in HCC Chemoresistance to Current Treatments
Primary liver cancers, such as HCC, are known to exhibit high resistance to chemotherapy. In these malignancies, up to 100 genes have been found to be linked to treatment resistance pathways. 176 However, recent studies have shed light on the role miRNAs play in drug resistance in HCC through various mechanisms, including autophagy, membrane transporters, and disruption of apoptosis 177 (Table 2).
Summary of the Effect of miRNAs in HCC Chemoresistance to the Current Treatments.

Abbreviations: EMT, epithelial-to-mesenchymal transition; HCC, hepatocellular carcinomas; KRAS, kirsten rat sarcoma; LOXL2, lysyl oxidase-like 2; MDR, multidrug resistance; miRNA, microRNA; PTEN, phosphatase and tensin homolog;
Recent research has shown that Huh-7, a particular HCC cell line, expresses miR-27b, miR-146b-5p, miR-181a/d, and miR-146a and that leads to MDR against drugs such as doxorubicin (dox), carboplatin, cisplatin, mitomycin C, and vincristine by targeting proteins such as PTEN, Kirsten rat sarcoma (KRAS), and P53. 178 Another study has shown that angiogenesis and EMT in HCC can result from upregulating miR-32-5p by triggering the PI3K/Akt pathway and suppressing the expression of PTEN, which ultimately results in MDR. 179 Additionally, increasing miR-181a while decreasing RASSF1 expression in Hep3B cell lines results in changes to MAPK signaling, which reduces apoptosis in cancer cells and ultimately leads to resistance to sorafenib. 180
MiRNAs can induce drug resistance through various mechanisms, including the effect on membrane transporters. Overactivity of these transporters can lead to resistance to chemotherapy by altering drug flow into tumor cells. 181 Findings have shown that reducing miR-491-3p, miR-122, and miR-223 levels can increase ABCB1 transporter activity, leading to resistance to doxorubicin.182–184 Furthermore, resistance to doxorubicin is shown by an increase in the membrane transporter activity of ABCC1 caused by a decrease in miR-133a and miR-326. 185
One of the ways of chemotherapy resistance is increased autophagy induced by cellular stress. In autophagy, cells degrade their nonfunctional organelles and proteins to maintain metabolic homeostasis. 186 Autophagy-related genes (ATGs) are a group of genes that can induce autophagy by different stressors. 187 Sorafenib sensitivity can be raised and sorafenib resistance can be avoided by increasing the amount of miR-142-3p by inhibiting ATG5 and ATG16L1 and reducing autophagy. 188 Another research revealed that reducing the level of miR-26a/b can decrease the effect on ATG1 and increase autophagy, leading to resistance to doxorubicin. 189 Furthermore, the level of miR-520b is important in response to doxorubicin in patients with HCC, and its reduction through the effect on ATG7 leads to resistance to doxorubicin. 190 Also, reducing the level of miR-199a-5p via a similar mechanism leads to the chemoresistance of cisplatin. 191 miR-101, by inhibiting autophagy through the effect on ATG4D and increasing apoptosis in the HepG2 cell line, leads to greater efficacy of chemotherapy with cisplatin, and the findings suggest that reducing miR-101 can also lead to chemoresistance of cisplatin. 192
One of the features of cancer cells is the lack of apoptosis, and chemotherapy drugs used in cancer treatment attempt to induce apoptosis in these cells. Factors that prevent apoptotic cell death may lead to chemoresistance in cancer treatment. 193 PTEN is one of the genes that can induce apoptosis by inhibiting Akt signaling. Increased levels of miR-21 can lead to resistance to sorafenib by reducing PTEN expression. 194 Additional research has demonstrated that elevated levels of miR-216a/217, miR-19a-3p, miR-222, and miR-494 can result in sorafenib resistance via the PTEN/Akt pathway.195–198 Additionally, miR-205-5p can lead to resistance to 5-fluorouracil by reducing PTEN expression. 199 Increased levels of miR-760 can increase PTEN expression and inhibit resistance to doxorubicin by inhibiting Akt signaling. 200 The B-cell leukemia-2 (Bcl-2) family is another group of genes that can cause drug resistance in cancer treatment by affecting apoptosis. 201 According to a study, raising the expression of miR-34a by suppressing Bcl-2 expression will improve HCC's susceptibility to sorafenib and avoid sorafenib resistance. 202 In the Bcl-2 family, Mcl-1 is a crucial member that is inhibited by miR-363. Patients with HCC who have low levels of miR-363 have higher expression of Mcl-1 and its anti-apoptotic activity, which makes them resistant to cisplatin. 203 Patients with HCC develop resistance to cisplatin as a result of decreased levels of miR-363, which increase the expression of Mcl-1 and its anti-apoptotic action. 204 In addition, HBV infection can induce resistance to sorafenib by downregulating miR-193b expression and increasing Mcl-1 expression. 205 Another gene that contributes to HCC's poor prognosis and its cells’ anti-apoptotic activity is KRAS. In HCC, the level of miR-622, which directly reduces KRAS expression, is decreased, leading to resistance to sorafenib 206 (Figure 4).
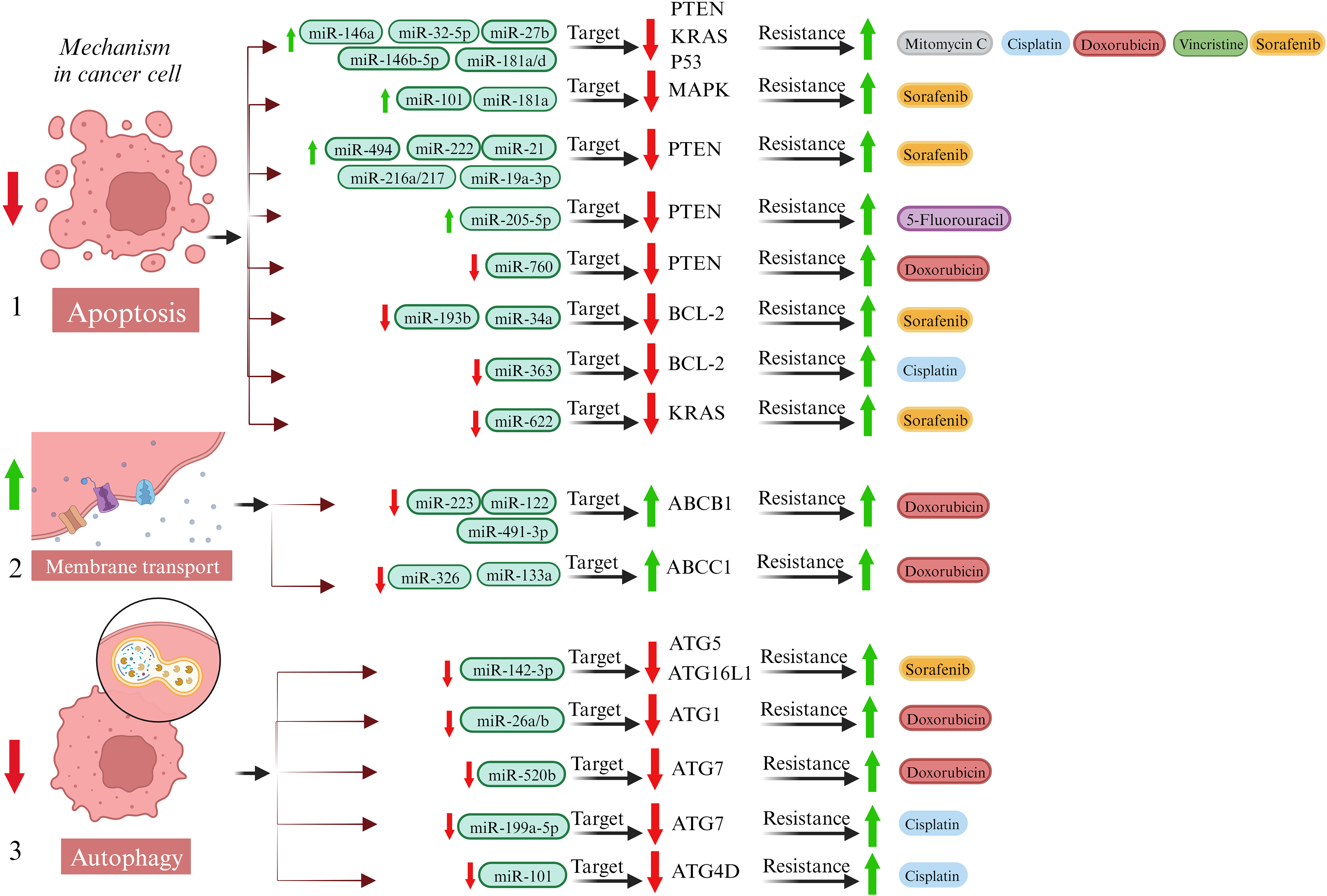
Mechanism of drug resistance in HCC cells. (1) One of the ways cancer cells resist is by reducing apoptosis. This reduction results from the overexpression or inhibition of miRNAs and reducing the expression of the indicated genes and proteins. As a result, increasing cancer drug resistance. (2) Increasing membrane transporters activity is also considered as a way of resistance in cancer cells. Following miRNA inhibition, the activity of the mentioned transporters increases and they become resistant to doxorubicin. (3) Autophagy is another mechanism cancer cells use to enhance drug resistance. Following inhibition of miRNAs, ATGs are also inhibited. As a result, cancer drugs are resistant through this mechanism. Abbreviations: ATGs, autophagy-related genes; HCC, hepatocellular carcinoma; KRAS, Kirsten rat sarcoma; miRNA, microRNA; PTEN, phosphatase and tensin homolog.
It is promising that miRNAs can be used to treat HCC. A miRNA-targeted therapy and miRNA replacement therapy are 2 miRNA therapeutic strategies used to treat HCC. An exogenous pathway is used to import miRNA or miRNA mimics that have therapeutic effects on HCC into hepatocytes in miRNA replacement therapy. For example, in studies using adeno-associated viral vectors that deliver miR-3-3p to mice's livers, the mouse liver tumor progression was significantly slowed and survival was significantly improved. 207 However, miRNAs produced in the absence of exogenous factors can result in immunoreactive and toxic responses, and their method of delivery may also limit their potential applications. In contrast, miRNA-targeted therapy involves the use of miRNA inhibitors to increase the levels of certain miRNAs in the expression level and influence the role that miRNAs play in cancer cells by regulating the expression levels of certain miRNAs. Using peptidyl-prolyl cis-trans isomerase NIMA, Pu et al have found that API-1 interacts with the enzyme by targeting one of its subunits, Pin1, so that it regulates miRNA biogenesis and inhibits HCC development, which could be used as a therapy for the disease. 208 MiRNA has been investigated for its potential as a therapeutic approach for HCC, however, there are still many challenges that need to be overcome before such approaches can be used in the clinic. miRNA therapy will, however, become an essential strategy for treating HCC in the future as a result of the continuous development of related technologies and research in this area.
There is still no clear understanding of how HCC develops at the molecular level. HCC medical management has recently incorporated genetic predisposition and immune-targeted treatment. Prognosis assessment, treatment response prediction, and new molecular targets will substantially benefit from this approach. HCC risk can be reduced with vaccines and antiviral therapies. There is a need for awareness and a strategic approach to reduce the risk of NASH and HCC. It is expected that in the future, serum biomarkers will replace imaging and screening modalities or diagnoses modalities as the primary diagnostic method. A more critical role will be played in the future by early diagnosis and medical management of HCC. In order to better understand the molecular basis of HCC, treatment methods are developing and being made more specific. Efforts need to be made to understand the mechanisms of immunotherapy resistance as well as to discover more predictive biomarkers to assist in the therapeutic decision-making process. 209 The identification of a large number of miRNAs as anticancer regulators has led to an understanding of their role in carcinogenesis, invasion, and metastasis of HCCs. Moreover, miRNAs derived from circulating extracellular vesicles and secreted by cancer cells have been shown to be potential biomarkers and potential therapeutic targets in cancer, and there are many miRNA-based treatments available that are being developed to become safer and more effective, as well as miRNA-based biomarkers. For new miRNA-based anticancer therapies to be successful in treating HCC, further research is needed to determine the possibilities. In order for circulating miRNAs to be used as diagnostics in clinical routine, it is still necessary to resolve several issues before their use is accepted. There is no doubt that normalization is a controversial topic. There can be a significant impact on the biological interpretation of data based on the choice of a reference gene, as the choice of a reference gene can have a major effect on the measurement of transcript levels. Furthermore, standardization should also be applied to miRNA processing, including the collection and storage of samples, as well as the isolation of RNA and reverse transcription. 210 Additionally, commercial samples collection kits are extremely expensive. As a result, it is essential to establish an inexpensive and time-saving method for collecting samples. There may be discrepancies in the conclusions that can be drawn from different studies on the same exosomal miRNA when it comes to diagnosing HCC and predicting its outcome. There is also a reason for the different conclusion that is primarily due to the fact that the study actually included only a small number of research subjects. 211 A large disparity between the groups of research subjects contributes to inconsistent results in the tests, because there are large individual differences between them. In the future, miRNAs are likely to play a very important role in diagnostic and therapeutic algorithms if the current limitations can be overcome.
Conclusion
Overall, we have significant cross-talk between TME and HCC cells. The miRNAs play an important role in this process by impacting immune phenotype, hypoxia, acidification, angiogenesis, and ECM composition. Moreover, changes in miRNA levels in HCC can effectively resist cancer cells to chemotherapy by affecting various cellular processes such as autophagy, apoptosis, and membrane transporter activity. Further studies on miRNAs and their use with chemotherapy drugs can significantly reduce tumor growth and improve patient outcomes.
Footnotes
Abbreviations
Acknowledgment
We would like to thank Dr Ali Akbar Moghadamnia. All figures are created using BioRender.com.
Data Availability
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
