Abstract
Introduction
Compared to other conventional medical imaging modalities, such as computed tomography (CT), cone beam CT, and ultrasound images,1–3 magnetic resonance imaging (MRI) offers superior soft tissue contrast, providing a clearer visualization of both tumors and organs at risk (OARs). MRI encompasses various parameters that reflect diverse tumor characteristics, including tissue oxygenation, cell density, and cell function features. Notably, MRI employs non-ionizing radiation, enabling repeated imaging without increasing patients’ radiation exposure. These distinctive attributes make MRI highly suitable and widely adopted in image-guided radiotherapy. 4
Current commercial magnetic resonance linac (MRL) systems, such as Elekta Unity by Elekta AB in Stockholm, Sweden, have made MR-guided radiotherapy a reality.5–7 However, the presence of a magnetic field inevitably affects the distribution of radiation dose. Numerous studies have also reported the electron return effect (ERE), which occurs when secondary electrons return back into tissue-air interfaces, leading to significant dose changes,8–10 particularly in air cavities and tissue-air interfaces. In high-field designs, the dose distribution closely corresponds to that of the tissue-air interface, resulting in the appearance of hot and cold areas within the air cavity of the target area. This effect is particularly concerning when the target area is a hollow organ with adjacent air. 11
External beam radiotherapy holds a crucial role in treating esophageal malignancies, serving as a significant modality in the preoperative setting for patients with operable tumors, and as a definitive treatment option for those ineligible for resection due to medical or technical reasons.12,13 Additionally, the esophageal site presents challenges for MR-guided radiotherapy due to its complex motion characteristics. Moreover, the close proximity of the esophagus to critical OARs, such as the lung, heart, and spinal cord, further complicates the treatment process.
With MRL, achieving sufficient visualization of soft tissue tumors allows for accurate real-time online location verification and dose adjustment without overdosing critical OARs. This high-quality MRI technique enables noninvasive real-time visualization and tracking of tumors. While there has been an increasing interest in the impact of the magnetic field on dose distribution with the development of clinical applications of MRL, related studies remain limited. Chuter et al quantified the interaction of anatomical changes with ERE in head and neck cancer patients, revealing no significant clinically relevant difference in doses between the planning target volume (PTV) and OARs associated with weight loss in 0 or 1.5 T fields. 14 Uilkema et al assessed the impact of a 1.5 T transverse magnetic field on dose delivery in rectal cancer patients and found that changes in air content within a few percentage points had an impact on the dose distribution. 11 Chen et al investigated the dose effects of ERE at air-tissue and lung-tissue interfaces during intensity-modulated radiation therapy (IMRT) planning. 8 They found that doses on tissue interfaces were significantly affected by the presence of a transverse magnetic field during MR-guided RT when the magnetic field was not included in plan optimization. 15 However, for tumors located in the esophagus, where air cavities are irradiated, no comprehensive statistical comparison of IMRT plans with and without the inclusion of a magnetic field has been reported. Hence, it is necessary to investigate the significant differences between 0 and 1.5 T regarding the diametric parameters of IMRT plans and the doses delivered to tissues containing air cavities.
The esophagus is known to exhibit both inter- and intra- fraction motion, and motion can be a particular concern in radiation treatment, especially for MR-guided radiotherapy. Therefore, another focus of this study was to assess the dosimetric impact of esophageal motion on the esophagus and motion during intra-fraction radiotherapy.
Materials and Methods
Patient Selection
We conducted a prospective review of data from 22 patients with esophageal cancer who received MR linac radiotherapy. The median age of the patients was 65 years (range: 51-77 years). Additionally, we included 78 patients with newly diagnosed esophageal cancer who were treated with conventional linac between May 2021 and April 2023. The mean age at the time of diagnosis was 61.3 ± 9.3 years (median, 63.5 years; range, 43-85 years), and 58.9% of the patients were male. Squamous cell carcinoma accounted for 98.4% of the tumors. Among them, 35 patients underwent 4DCT scan simulation, with exclusion of patients having poor pulmonary function or preexisting respiratory problems. Before the treatment initiation, written informed consent was obtained from all patients. Fifty-seven patients are included in the study in our institution (Shandong cancer hospital). The patient characteristics are detailed in Table 1.
Patient Characteristics.

Patient Characteristics.

Each patient underwent a 4DCT scan using a 16-slice CT scanner (Philips Brilliance Bores CT, Netherlands). Patients were positioned in the supine position with arms stretched over their heads using a vacuum bag, followed by laser alignment. Laser cross-marked points were applied to the bilateral axial midline and the anterior midline as metal markers. Axial CT mode was used to acquire images from the neck to the mid-abdomen. Scans were conducted during free breathing without any breathing control. The Real-Time Position Management (RPM) Respiratory Gating System (Varian Medical Systems, Palo Alto, CA) monitored each patient's respiration by tracking infrared markers on the abdomen. The system signaled the scanner to label each CT image with a time tag. Philips Advantage 4D software sorted the reconstructed 4DCT images into 10 respiratory phases labeled 0% to 90% based on these time tags, where 0% corresponded to end inspiration (EI) and 50% corresponded to end expiration (EE). The slice thickness of the 4D scan was 3 mm and no slice gap. All patients do not create contrast media during the scan.
Structure Delineation
All 4DCT datasets of each patient were registered to the reference 4DCT scan corresponding to the EE phase (respiratory phase 50%, GTV50) using tools in the treatment planning system (TPS) based on bony landmarks for comparison. The same senior physician determined the primary tumor (considered the gross tumor volume, GTV), the clinical target volume (CTV), and the PTV for each image dataset, maintaining a consistent window level and window width throughout. The GTV centroid positions were obtained using the Monaco (v5.40.04, Elekta AB, Stockholm, Sweden) TPS. Volumes were constructed using 3 approaches: GTVmean, which was the average of all 10 phases of each GTV; GTVmip, which was the contour delineated from the maximum intensity projection; and GTVall, which combined all 10 GTVs. The displacement in each direction between the center of the GTV50 contour on the simulated scan and the center on the subsequent scan was used to evaluate the interscore displacement between the scores. The displacement was calculated as the GTV50 centroid coordinate on the subsequent dataset minus the reference dataset. The coordinates of the center of mass on the GTV50 were obtained.
An additional 5-mm margin was added to the heart to form the heart wall. To quantitatively evaluate the ERE-induced dose increment in the tissue-air interfaces, the skin was contoured to include the first 1 mm under the outline of the body, and the interfaces of tissues containing GTV air cavities were contoured to include the first 1 mm inside the air cavities. OARs, including the sternum, lungall, heart, spinal cord, and chest wall, were also contoured in all phases.
Optimization Strategies in IMRT Planning
In the Monaco TPS, treatment plans were formulated based on CT images obtained from patients in a free-breathing state. The Monaco TPS employed the GPU Monte Carlo dose (GPUMCD) algorithm, enabling consideration of a 1.5 T magnetic field. For each patient, the TPS employed specific parameters: a minimum segment area of 4 cm², a minimum segment width of 0.6 cm, medium fluence smoothing, a minimum of 4 monitor units (MU) per segment, a maximum of 120 segments, and a statistical uncertainty per calculation set at 1%. The prescribed dose was 60 Gy in 30 fractions, with a requirement that 95% of the PTV be encompassed.
For practical considerations, 4D-CT scans representing the 2 extreme phases of the breathing cycle, EE and EI, were acquired for each patient to account for uncertainties arising from respiratory motion across the entire respiratory cycle. Five types of step-and-shoot IMRT plans were generated for each patient:
The first plan (plan0T) was developed without the influence of a magnetic field (0 T). The second plan (plan1.5T) was directly recalculated under a 1.5 T magnetic field, maintaining the same segment shapes and MUs as the first plan. The third plan (plan1.5T_reOpt) was reoptimized under a 1.5 T magnetic field, using the same cost function as the first plan. The optimization process for plan1.5T_reOpt involved resetting the calculation engine settings after completing and saving plan0 T, changing the beam type, and optimizing the plan using the same cost function as plan0 T. “PlanEI” and 5. “PlanEE” were generated by recalculating the dose distribution of plan1.5T_reOpt (using the same segment aperture shapes and MUs as plan1.5T_reOpt) without undergoing plan optimization for the EI and EE phases.
To minimize the calculation uncertainty of the Monte Carlo (MC) calculation, all plan optimizations were executed 3 times for each plan. Consequently, dosimetric evaluation for each tumor target and OARs was based on averaged values.
Data Analysis Endpoints
To compare these plans, dose-volume histograms of all structures and corresponding evaluation parameters were calculated. A series of dose-volume parameters were extracted from the dose distributions for the tumor and OARs that could be affected by the prescribed dose. The series was V100%, D95%, and the dose heterogeneity index (HI) and conformity index (CI) for targets. The HI was defined as (D20%–D80%)/Dprescription, Dprescription: prescribed dose. The closer the HI value was to 0, the better the homogeneity was. The CI was defined as CI = TV1*TV1/TV*VR1, where TV is the volume of the target, and TV1 is the portion of the PTV within the prescribed isodose line. VR1 is the treated volume of the prescribed isodose line. The closer the CI value was to 1, the better the conformity was.
Several parameters were compared for the OARs: (1) the maximum dose (Dmax) and the maximum dose covering 1 cm3 volume (D1cc) to the spinal cord; (2) the Dmax and D1cc to the sternum; (3) the V5, V20, and V30 to the lung wall; (4) the V30 and V40 to the heart; (5) and the maximum and mean doses for the skin, lung wall, heart wall, chest wall, and the interfaces of tissues containing air cavities.
Model Phantom
An optically stimulated luminescence dosimeter (OSLD) system (RadPro International GmbH, Germany) was used to measure the real dose for the water phantom, as described in our previous research. 16 The water phantom was designed in-house to assist in MRI data acquisition and the simulation of the real environment of the position of the esophagus. The phantom is a thorax-shaped container filled with a solution of 0.4 mL Mncl2 to replicate the relaxation time (T2) of muscle tissue surrounding the thorax tumor (Figure 1). To assess the difference in the dose delivered in the magnetic field, OSLDs were used to measure the dose in the MRL and conventional linac conditions. First, the phantom was adjusted on the couch and irradiated with OSLDs lined up at different measurement points on the surface of the phantom (Figure 1a). Second, the OSLDs were placed on the surface of the emulational OARs (eg, esophagus and heart) (Figure 1b and c). The OSLDs were taped to the phantom while still ensuring that the OSLDs were in firm contact with the surface of the phantom. For all measurements, each irradiation was repeated 5 times, and the data from the OSLD measurements were averaged.

The phantom is a thorax-shaped container filled with a solution of 0.4 mL Mncl2 to replicate the relaxation time (T2) of muscle tissue surrounding the thoracic tumor.
Calibrate the absolute dose of the accelerator output according to the IAEA TRS277 report. Calibration strip: under the SAD = 143.5 cm, refer to the underwater 9.7 cm, the gantry angle is 0°, the field size is 10 cm × 10 cm, and the number of MUs is 100 MU.
The dose was calibrated at 100 MU = 100cGy at the reference depth. The dose rate was kept at 400 to 425 MU/min during the experiment. In order to ensure the accuracy of the output dose measurement, ensure that the ionization chamber is static in water ≥ 15 min before the measurement, and the dosimeter is fully preheated at the same time. The absolute dose was measured when the gantry angle was 0°, 90°, 180°, and 270° respectively.
Statistical Analysis
The statistical significance of differences between all dosimetric parameters was tested by analysis in SPSS v24.0 (IBM Corp.). Values of P < .05 were considered to represent statistically significant results.
Results
The differences in dose-volume metrics between the treatment plans designed for 1.5 and 0 T are depicted in Figure 2. Table 2 summarizes various dosimetric parameters for the PTV, heart, lung (whole lung), spinal cord, skin, heart wall, chest wall, sternum, and air cavities for all patients (Figure 3).

The differences in dose-volume metrics between the treatment plans designed for 1.5 and 0 T are shown.

Comparison between TPS calculation and OSLD measurement results for VitalBeam and Unity.
Changes in Various Dose-Volume Parameters for PTV, OARs, and Skin in Patients.

There was a significant difference in PTV metrics (V100%, D95%, homogeneity index (HI), and CI) between plan0T and plan1.5T, and also between plan0T and plan1.5T_reOpt (P < .05). All plan1.5 T metrics were significantly worse in the presence of a 1.5 T magnetic field than those acquired without a magnetic field. Specifically, V100%, D95%, and CI for the PTV of plan0T were higher than those of plan1.5T, but HI values were all less than 3%. For the upper and middle sites, the changes in V100%, D95%, and CI for the PTV were all less than 4%, and HI for the PTV was higher than 4%. Reoptimization of the plan under the magnetic field (plan1.5T_reOpt) slightly mitigated this effect. However, for the lower site, the changes in V100%, D95%, and HI for the PTV were higher than those for plan0T, resulting in worse conformability. Nevertheless, the reoptimized plan improved these effects to some extent. The details are shown in Table 3.
Comparison of Dose-Volume Data Between the Original Plan and PlanEE and Between the Original Plan and PlanEI for Magnetic Field and No Magnetic Field.
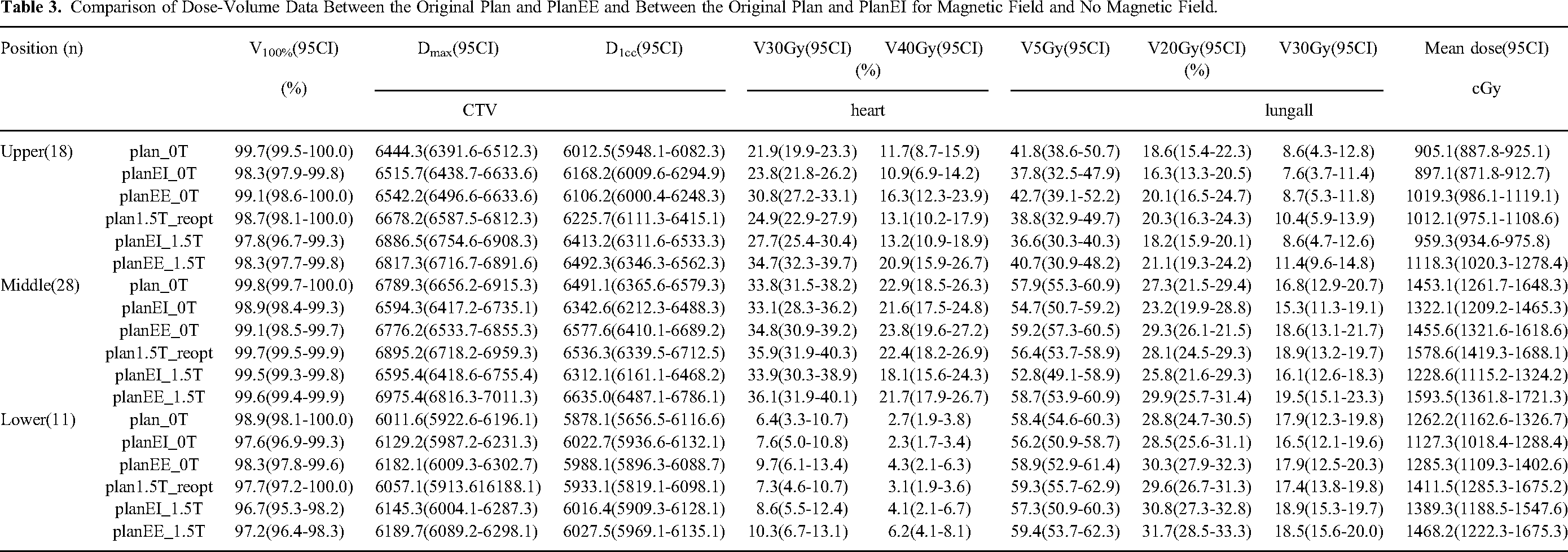
Compared to non-magnetic field conditions, the doses at air-tissue interfaces (chest wall and heart wall) were slightly higher in some cases (with an average increase of 3.0 Gy for 4.3% of the tissue). However, with plan reoptimization, the aforementioned effects were mitigated. The doses for the skin, spinal cord, heart wall, chest wall, sternum, and air cavities were all significantly increased compared to plan1.5T for Dmax and D1cc. Notably, for skin dose, instances of middle and lower esophageal cancer showed an increase of more than 20%. The doses of other OARs (spinal cord, heart wall, chest wall, sternum, and air cavities) were also increased by 1% to 9%. The optimized plan considering the magnetic field significantly reduced the changes in Dmax and D1cc for the skin. There was a significant increase in V20 and V30 for the whole lung (lungall) in plans considering a magnetic field. However, for upper and middle esophageal cancer, V5 for lungall decreased significantly. Overall, there were no significant differences in most dose metrics of the OARs between the recalculated and reoptimized plans. The reporting of this study conforms to STROBE guidelines. 17
Discussion
The study focused on 3 main aspects: the variation in plan quality under a magnetic field, the dose differences at skin and tissue-air interfaces, and the investigation of intrafraction displacement and volume variation in primary thoracic esophageal carcinoma.
The esophagus has a unique anatomical structure, starting from the cricoid cartilage at the lower edge of the sixth cervical vertebra to the gastric cardia at the level of the 11th thoracic vertebra, with a length of approximately 25 to 30 cm in adults. During radiotherapy, the esophageal position is affected by factors like cardiovascular pulsation and respiratory movement, leading to uncertainties and changes in lesion motion in different parts. Target areas for lower thoracic esophageal cancer are located beyond the head and foot direction, with a larger placement distance compared to the upper and middle parts of the chest. The target zone's movement within each patient's fraction is also highly variable.
The study showed that for upper and middle locations, V100% was slightly worse at 1.5 T compared to 0 T, with a maximum difference of 4%. However, there was no significant difference for the lower location, possibly due to changes in air cavities in the absence of a 1.5 T magnetic field. Including the magnetic field in plan optimization effectively reduced its impact on dose distribution, consistent with previous studies by Chen et al. 8
Dose metrics for targets were significantly worse in plan1.5T compared to plan0T, especially for upper and middle targets. After reoptimizing the plan under the magnetic field (plan1.5T_reOpt), target metrics were close to those in plan0T. Increases in homogeneity index (HI) were observed for recalculated plans in the presence of a 1.5 T magnetic field, but they were eliminated with the inclusion of the magnetic field in plan optimization. OARs were less affected by the magnetic field, indicating that the target volumes were less robust to its presence. This effect is mainly caused by changes in dose deposition due to the magnetic field's influence on the trajectory of secondary electrons. The impact on esophageal dose deposition is significantly influenced in a magnetic field environment due to both tumor movement and the presence of cavities. Tumor movement alters the scattering environment, while the presence of a cavity amplifies the magnetic field's effect on the motion of charged particles.
Regarding intrafractional displacement, the study observed larger displacements in the SI direction than in the LR and AP directions throughout the treatment, particularly for lower esophageal cancer. The details are shown in Table 4. The study suggests expanding the incisal margin reasonably in the SI direction for patients with lower esophageal cancer.
Magnitude (cm) of Interfraction GTV Centroid Motion in 4DCT Scans.

The study also investigated the effects of intrafractional anatomical changes in doses to tumors treated with MRL. Esophageal motion did not significantly affect the dose distribution in this regard. However, EE values for total lung V5, V20, and V30 and heart V30 and V40 in 4D-CT plans were larger than those in EE plans, indicating a slight increase in dose to OARs and the target area due to the magnetic field's presence (Table 3). Anthropomorphic phantom measurements showed higher point doses when using the VitalBeam system compared to Unity. Despite the differences between MRL and conventional linac methods, plan reoptimization under a 1.5 T magnetic field improved the feasibility of achieving acceptable treatment plans for esophageal cancer patients. The skin dose increased significantly under the 1.5 T magnetic field, and there were some differences in dose distribution at tissue-air interfaces.
Conclusion
The study highlights the significant impact of a 1.5 T magnetic field on plan quality and the importance of plan reoptimization to achieve clinically acceptable treatment plans for esophageal cancer patients.18,19 The dosimetric effects on skin and tissue-air interfaces warrant consideration, especially for cardiac tissue, in radiotherapy using a 1.5 T linac. Based on the experience of our other studies, 20 it may be possible to reduce the effect of magnetic field on uneven dose distribution by increasing the number of fields and penetrating fields. Following studies should increase the follow-up of clinical results, confirm the complications of patients, and further analyze the clinical value of MRI-guided radiotherapy. The findings emphasize the need to address motion-related uncertainties and consider the effects of magnetic fields in the treatment planning process.21–23
Footnotes
Acknowledgments
We thank all the patients for allowing us to analyze their data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study was approved by the Human Research Ethics Committee of Shandong Cancer Hospital, Jinan, China (SDTHEC2021003157). Written informed consent was obtained from each participant.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by National Key Laboratory of Advanced Drug Delivery and Release Systems, the National Natural Science Foundation of China under grants 82102173, U23A20461, 82172072; Academic promotion program of Shandong First Medical University (2019LJ004), and by Taishan Scholars Program of Shandong Province, China (grant no. ts20120505).
