Abstract
Conforming to the current replace–reduce–refine 3Rs’ guidelines in animal experiments, a series of explorative efforts have been made to set up operable biomedical imaging-guided platforms for qualitative and quantitative evaluations on pharmacological effects of tumor vascular-disrupting agents (VDAs), based on the chick embryos (CEs) with its chorioallantoic membrane (CAM), in this overview. The techniques and platforms have been hierarchically elaborated, from macroscopic to microscopic and from overall to specific aspects. A protocol of LED lamplight associated with a new deep-learning algorithm was consolidated to screen out weak CEs by using the CAM vasculature imaging. 3D magnetic resonance imaging (MRI) and laser speckle contrast imaging (LSCI) to monitor the evolution of CE and vascular changes in CAM are introduced. A LSCI–CAM platform for studying the effects of VDAs on normal and cancerous vasculature of CAM and possible molecular mechanisms has been demonstrated. Finally, practical challenges and future perspectives are highlighted. The aim of this article is to help peers in biomedical research to familiarize with the CAM platform and to optimize imaging protocols for the evaluation of vasoactive pharmaceuticals, especially anticancer vascular targeted therapy.
Introduction
It has become a global trend in biomedical research to more strictly respect animal welfare from the viewpoints of science, legislation, and public awareness. Consequently, the European Parliament has issued European Directive 2010/63/EU on the protection of animals used for scientific purposes. 1 This directive announced a replace–reduce–refine 3Rs’ guideline: when designing scientific research, these three elements are encouraged to be implemented by the institutional hierarchy to protect live animals.
Accordingly, the chick embryo (CE) and chorioallantoic membrane (CAM) models could be excellent alternative platforms well complied with the current 3Rs’ principle. The allantois of the CE emerges at about 3.5 embryonic days (ED 3.5), and afterwards rapidly increases its size and forms an extra-embryonic vesicle. During this continuous enlargement, the allantois fuses with the chorion, and consequently the CAM is formed. Between the two (chorionic and allantoic) layers of membranes, an extremely rich vascular network is generated in the stroma.2,3 Such unique enriched vasculature provides a biological microenvironment that not only enables CE's growth, but also becomes exploitable for biomedical research with characteristics of being easily visible, accessible, and affordable. 4
Consequently, the CE and particularly its CAM have become useful scientific platforms for research on vasculature and angiogenesis,5,6 cancer,7,8 bioengineering materials,9,10 drug screening and development,11,12 cellular mechanism,13,14 stem cells, 15 etc. Various types of tumor cell lines, tumor cell suspensions, and patient-derived tumors (xenografts, PDX) have been tested in the CAM models.16,17 Meanwhile various innovative anti-cancer therapies have also been tested on CAM models.4,7,18–21
Among the most effective tools, precise medical imaging techniques have been proved as excellent noninvasive manners to advance the understanding of chick allantois and CAM. The intuitive 3D images have formed concrete bases for setting up CE platforms to facilitate anticancer pharmaceutical evaluations.
Small-molecule vascular-disrupting agents (VDAs) act on the existing tumoral vasculature, particularly by making endothelial cells round up to bleb, which physically obstructs the cavity of tumoral capillaries. Further, increased vascular leakage and resistance, and exposed basement membrane induce vascular occlusion or blood supply shutdown, and thus resulting in tumor ischemic or hemorrhagic necrosis.22,23 The underlying molecular mechanism is that VDAs bind to one of the cytoskeleton components, tubulin, at its colchicine-binding site, which triggers the depolymerization of tubulin. This kind of disruption selectively acts on immature endothelial cells that are already retarded with another cytoskeleton component, actin, together causing both the shape and function of such endothelium unsustainable. 22 Therefore, VDAs could be ideal candidates for demonstration of the feasibility and efficiency of the CAM platform in preclinical safety, efficacy, and toxicology testing of vasoactive anticancer pharmaceuticals (Figure 1). We have developed a dual targeting pan-anticancer theragnostic strategy called OncoCiDia, 24 in which VDA is the first drug to selectively destroy tumoral vessels for inducing necrosis. Such a therapeutic strategy has been supported by both animal and human studies.22,25

Schematic mechanisms of tubulin-binding tumor vascular-disrupting agents (VDAs). (A) vasoconstriction due to VDAs effects; (B) blood flow leakage due to the exposure of basement membrane; (C) VDAs cause endothelial shape alteration and disorganization, and then basement membrane exposure; (D) endothelial round up and bleb, and thus resistance increase in vessel, flow stasis; (E) blood flow.
However, a number of safety issues have been reported from clinical trials with VDAs. 26 Besides other adverse effects including dyspnea, tumor pain, abdominal pain, neurotoxicity, and headache,26–28 cardiovascular toxicities are prominent with hypertension as the most common one. 28 These systemic side effects or off-target toxicities are rather difficult to explain by VDAs’ specificity on tumoral vasculature. Such uncertainties call for further research to gain more insights into the nonspecific effects of VDAs on normal vasculature and to elucidate underlying molecular mechanisms.
The most crucial requirements to monitor vascular alterations are the measurement methods with steady, visible, and quantifiable features. Emerging noninvasive techniques can continually measure in vivo vascular status and changes, in real time and at high resolutions, and facilitate the assessment of vascular-targeting drugs in laboratory or preclinical assays. 29 Nondestructive techniques have been applied to image the CAM vasculature, including X-ray, magnetic resonance imaging (MRI), gamma-rays, ultrasound, optical approaches, etc., which are supplemented most recently by artificial intelligence (AI). Laser speckle contrast imaging (LSCI) 30 is emerging as a novel noninvasive approach with versatile characteristics for in vivo comprehensive exploration of vasculature.31,32
This overview article aimed to summarize the recent attempts for studying the pharmaceutical effects of VDAs on both normal and tumoral vasculatures, as examples for anti-cancer vascular-targeting therapy. Based on the CE and CAM platforms,3,33 relevant technical tools including modern imaging modalities such as MRI, MicroCT, digital radiography (DR), and quantitative LSCI34–36 have been applied, in combination with AI technologies, 37 histomorphology, and immunohistopathology 38 at cellular and molecular levels.
This serial experimental research was exempted from institutional ethics committee approval since CMs are not considered as animals according to the European Directive 2010/63/EU.
CAM Vasculature Imaging Techniques and Limitations
The CAM is one of the classic models for research related with blood vasculature. Various imaging modalities have been set up for visualizing, monitoring, and evaluating the changes of CAM vasculature. The vascular changes appear increasingly interesting, especially when the interactions occurred after introducing the tested pharmaceuticals and biological compounds. Here the techniques for measuring and monitoring discernable CAM vasculatures are reviewed systematically from macroscopic to microscopic and from overall to specific aspects.
The most conventional approaches are destructive ones in which the vasculature of the dissected CAM is observed by a camera, a stereoscope, 39 or a microscope. With such approaches, the CE is not alive and the CAM is not intact, which is impossible for in vivo studies.
Nondestructive imaging instruments have been developed to image the CAM vasculature, which can be classified into lamplight, X-ray, MRI, gamma-rays, acoustic, and optical techniques, most recently assisted by deep learning algorithms or AI. Those modalities can measure structural changes in the vasculature including the number and diameter of blood vessels and certain functional parameters, for example, hemodynamics and blood oxygen levels.29,40 Clinical CT, MRI, PET, and ultrasonography (US) are unable to achieve enough resolution for visualizing microcirculation and monitoring the vascular alteration in real-time in the CAM. In addition, the contrast agent (or radiotracer) infusion may make embryos fatal. 41 However, optical techniques have special advantages for imaging CAM vessels in vivo with high resolution. Blood possesses a very typical feature to react to the spectrum of light, by which optical imaging can capture various important parameters, including vascular structure, blood perfusion, flow volume, and flow velocity in the CAM. These innovative optical methods mainly include optical coherence tomography, laser Doppler technique, and laser speckle technique. However, coherence tomography is not eligible for blood flow imaging because of the interference with internal tissue background. 42 Although laser Doppler is a standard clinical method to measure blood dynamics, it is restricted by low resolution and long-duration measurement and not real-time monitoring. Therefore, a LSCI technique LSCI–CAM platform was developed, which can create prompt, high-resolution perfusion images. 43 This new platform allows to visualize the vascular structure at the microscopic level, to continuously and dynamically record the changes in the vasculature, and to carry out quantitative analysis and assessment, especially to evaluate the pharmacological effects. LSCI is still not optimal to detect and monitor functional and structural changes in deep tissues with a limitation of a few millimeters. 29 Yet the platform has the potential to monitor blood vessels in tumors transplanted superficially on the CAM, in order to assess the efficacy of anticancer pharmaceuticals. The specifications of two laser imaging techniques are compared in Table 1.
Comparison of Techniques Laser Doppler Versus Laser Speckle.

Source: Perimed AB, www.perimed-instruments.com and www.tcpo2.com.
Vascular Illumination with LED Light Assisted by Deep Learning
The CAM vasculature can be imaged by LED light and the weak CEs can be screened out by a novel AI-assisted protocol for imaging detection. This technology has been performed in the industry, especially in vaccine production. 44
The characteristics of blood vessels and other features in the CE become detectable after illumination by an LED source of white light in a dark space. Afterwards, an AI deep learning protocol is conducted to classify the images, so the live and weak CEs can be distinguished by their activity and viability. Such new deep learning frameworks helped develop a new set of convolutional neural networks (CNNs). Compared with traditional image processing methods, CNNs have better feature extraction capabilities and can accurately extract vascular features from CEs. A residual multiscale fire block (RMFB) was first designed to extract multiscale features from CE images and perform feature fusion at different scales, effectively by using the overall color information and blood vessel information of the image. Weak embryo detection network (WEDNet) is constructed through the cascade of RMFB modules, which deliver the signals from every scale for the comprehensive analyses to detect the weak CEs (Figure 2). According to the detection information, judgment instruction is transferred to a machine that picks weak embryos out. The experimental results showed that the detection of WEDNet can reach 99.35% of accuracy. 44

(A) Original image; (B) egg embryos dataset. The samples shown in (a) and (b) are living samples and the samples shown in (c) and (d) are weak.
Morphological Development of CE with Allantois Monitored by a Clinical MRI Scanner
The CAM is formed by fusion of allantois with chorion during the development of CEs. Prior to the research of CAM vasculature, the precise measurement on dynamic morphology of allantois urges noninvasive 3D holographic imaging on the CE and its organs. Especially the holographic 3D images during a full-length embryonic period deemed imperative for the poultry industry and biomedical science. Among excellent techniques of biomedical imaging modalities, MRI outstands for its balanced features of spatiotemporal resolutions, soft tissue contrast, free of ionizing radiation, and 3D morphological and functional capabilities, which are well feasible for in ovo studies on CE evolution. With bio-safety assurance, 3.0T clinical MRI scanner (MAGNETOM Prisma; Siemens, Erlangen, Germany) was employed to observe dynamically anatomical growth from embryonic day 1 (ED1) until ED20, capable of 3D reconstruction of in ovo allantois for quantitative assessment. 45
After the viability checking of CMs by deep learning method described in the previous section, CE eggs were taken randomly for MRI scanning on each ED. A Cooling regime of 0°C ice bath for 60 min was applied immediately prior to MRI scanning in order to prevent motion artifacts induced by CE movement. Series of MRI T2- and T1-weighted (T2WI, T1WI) images at axial, coronal, and sagittal orientation were acquired to generate 3D images. Allantois and entire CE regions were semiautomatically segmented by the software of intensity-based thresholding and region-growing algorithms. Afterwards, the system generated 3D allantois morphology with its volume automatically calculated. Scanned CE eggs were returned to the incubator until hatching to test their hatchability. 45
As shown in Figure 3, MRI images from ED2 to ED4 could illustrate the structural changes of latebra, a minor channel linking to the center of the yolk. It is a transitional structure for the CEs to absorb nutrients from yolk core into circulation of yolk sac. According to MRI theory, the yolk on both T1- and T2-weighted images should appear hyperintense with bright signals, because of its lipid-rich contents. Controversially, an interesting discovery is that the yolk appeared hypointense with a dark signal on T2 images. This phenomenon implies that the hyperintensity from the lipid was offset by another mechanism. Iron as a superparamagnetic metal is also rich in egg yolk, which can shorten both T1 and T2 relaxation times, and thus lead to such phenomenon. This method provides a new clue for noninvasive quantification of trace elements in the egg. 45

3D morphology of CE and its allantois depicted by using a 3.0 T MRI scanner. (A) a clinical 3.0 T MRI system; (B) T2-W images; (C) 3D morphology of allantois as segmented in red from ED2, ED6, ED12 and ED19 on T2-W MRI images. AL, albumen; Y, yolk; AS, air space; Red cycle, blastoderm; White arrow, latebra body; Yellow arrow, neck of the latebra; Green arrow, the nucleus of Pander.
The evolution of the CE in vivo was followed by 3D MRI across a full-length period (from ED1 to ED20). The noninvasive precise 3D imaging database of the CE and allantois has been acquired, with the progressive morphology and data measurement as new references for poultry industry and biomedical sciences. 45 The process of CAM formation has been demonstrated vividly. Allantoic fluid underwent a paraboloid-shaped profile, which raised sharply from ED8, with a peak on ED12, and then decreased until ED20. After cooling regime and 3.0 T MRI scanning treatment, CEs can reach 100% hatchability, which proves biosafe of this protocol. 45
LSCI–CAM Platform for Studying Vasculature Evolution of the CAM
After imaging clarification of CAM development, it comes the time to characterize CAM vasculature and to establish a LSCI–CAM platform for baseline and anticancer pharmaceutical studies. 5
A full-field laser perfusion imager (FLPI) from Moor Instruments (www.moorclinical.com), was assembled in the center of LSCI–CAM platform, as illustrated in Figure 4. A CE was placed inside an egg holder. The shell of the CE was opened along the edge of air cell as an observation window and an injection window of 1.0 cm × 1.0 cm was opened on the lateral side of the CE. Importantly, the CAM must be entirely intact and a major vessel must be positioned underneath the injection window. The CAM was scanned by FLPI in real-time through the observation window and the acquired imaging data were transmitted to the computer for analysis. 5
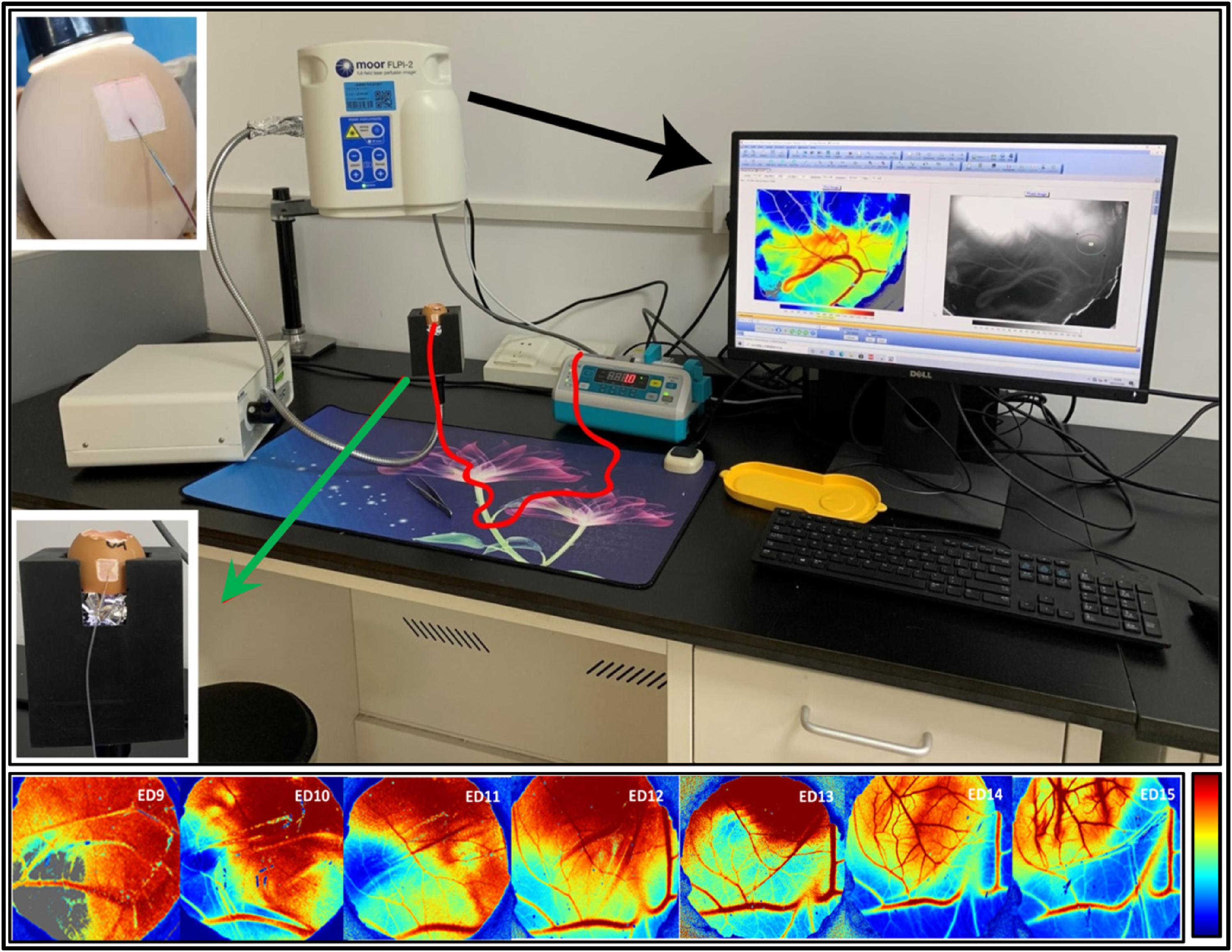
A LSCI-CAM platform for baseline and VDA studies (upper part). Color-coded real-time LSCI perfusion images of CAM vessels on specific EDs from ED9 to ED15 for the baseline study (lower row). The color bar on the right is a perfusion value (PV) indicator, showing the degree of the blood perfusion.
For intravascular infusion tests, a custom-made catheter needle was inserted into a major vessel through the injection window, and the other end of the catheter was connected with a microinjection pump enabling slow infusion of a solution at a designed rate. This platform was successfully established to acquire, quantify, and demonstrate vascular hemodynamic information from the CAM. 5
To collect baseline parameters and determine the best ED for anticancer pharmaceutical studies, six CEs were employed for monitoring vascular evolution in the CAM. On each ED from ED9 to ED15, CEs were taken out one by one for 30 min FLPI measurement. The blood perfusion value (PV) and the diameter of the same vascular section of individual eggs were measured quantitatively. Average PV during 30 min and vessel diameter on each ED were analyzed. All CEs with open air cells remained alive from ED9 to ED15 (Figure 4 lower row). Through comparing vascular diameter and PV, ED12 was determined as the best time for upcoming anticancer pharmaceutical studies with statistical significance (p < 0.001). A reverse correlation between vessel diameter and PV was found (p < 0.002). Intravascular infusion and simultaneous LSCI acquisition in ovo proved feasible. 5
In order to rehearse for upcoming anticancer pharmaceutical studies, 20 CEs were infused by physiological saline through the major vessel in the injection window. The duration of both intravascular infusion and simultaneous LSCI acquisition was set for 30 min. At the end of infusion, 1% Evans blue solution was injected via the same catheter as a verification step. Blue vessels could be viewed from the observation window, which ensured that the needle still remained within the vessel.
To verify the connectivity between CAM vessels and the embryonic circulations in the CE, postmortem procedures were executed. Barium sulfate suspension of 0.5 ml was administrated into the major vessel in injection window. The CE was placed in a µCT scanner (NANOTOM, GE Phoenix, USA). The vasculature in the whole circulation of different parts in a CE was captured with a high resolution at 25 µm and reconstructed for 3D rendering. Immediately proceeding, the whole CE was cracked out into a plastic dish and imaged by a clinical digital mammographic unit (Prime, Siemens, Germany) for DR. One piece of a major vessel was excised from observation window for HE staining. Histomorphology and contrast-enhanced angiography delineated the affirmative connection of CAM vasculature with embryonic circulation. 5 3D µCT images in superficial, in-depth, and vessel-subtracted views of whole CE were reconstructed into videos (refer Supplementary Video file of Chen et al. 5 ).
LSCI–CAM Platform for Studying VDAs Effects on Normal Vasculature of CAM
The new LSCI-CAM platform was applied to study VDAs on normal CAM vasculature and molecular mechanisms of drugs, on ED12 as optimally predetermined in the previous section.
According to the maximum tolerated doses (MTDs) derived from animal studies and clinical trials, C118P (a new VDA) or CA4P (a hallmark VDA) were dissolved with 0.9% saline into three concentrations: 2, 5, and 10 mg/ml. Six treatment groups received intravascular infusions of the three concentrations of VDAs against a vehicle control group with 0.9% saline, respectively (n = 6, each). Through the injection window, 0.5 ml solution was constantly infused into a major CAM blood vessel for 30 min, via a microinjection pump at a speed of 1 ml/h. Multiparametric measurements were performed simultaneously on the LSCI–CAM platform for real-time monitoring of vascular and hemodynamic alterations. Conventional monitoring parameters include blood perfusion (Flux) and vascular diameter; in addition, IKOSA deep learning platform (www.ikosa.ai) produced by KML Vision GmbH (www.kmlvision.com, Graz, Austria) contributed two new parameters of vascular density and vascular total length. In total, four parameters have been collected and analyzed at an analytical interval of every 5 min. Relative values were adopted for quantifications and comparisons of the multiple parameters, due to the variations among individual eggs. 5 A relative value is the value at each measurement point against the initial value on time point zero.
These four parameters were successfully processed with two examples shown in Figure 5. Each pair of four parameters was positively correlated, verifying that the four parameters have the same trend corresponding to the effects of VDAs. Furthermore, quantitative analyses demonstrated that the normal vasculature of the CAM was impacted negatively by VDAs with statistical significance (p < 0.0001), providing evidence for concerned side-effects of VDAs among clinical patients (Figure 6A). Furthermore, such effects appear to be dose-dependent.
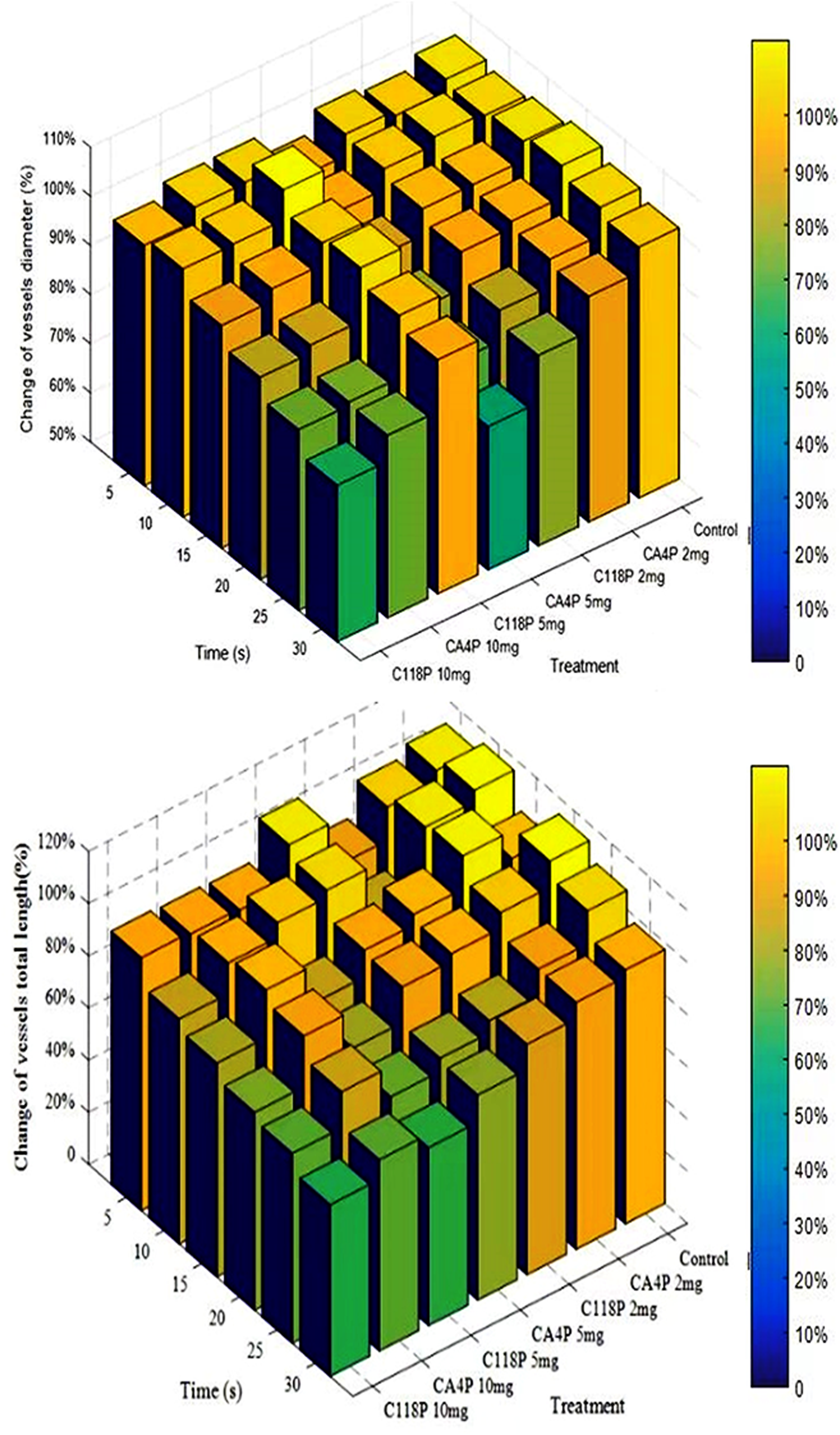
Qualitative trend in 3D plots of relative values from multiple parameters comparing saline control group with two VDAs in 3 concentrations each. Upper: vessel diameter; Lower: vessel total length. The color bars on the right indicate the degree of the relative parameter changes in function of the time, drugs, and dosages.
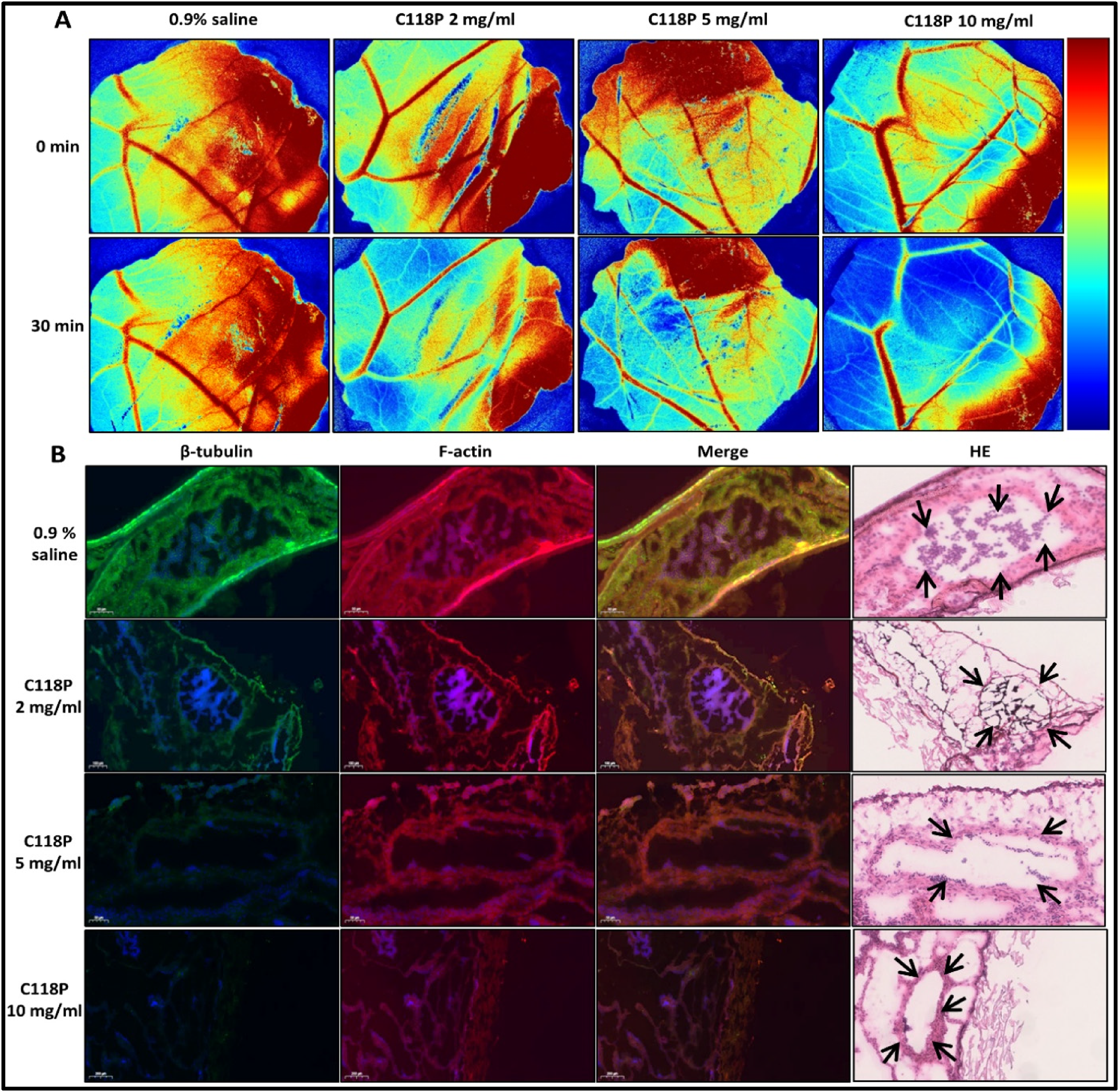
VDA on normal vasculature of CAM and related molecular mechanism. (A) FLPI captured FLUX images of blood perfusion in the CAM sequentially generated by the LSCI-CAM platform during VDA intravascular infusion. Horizontal line lists 0.9% saline control, 2, 5, 10 mg/ml of C118P; vertical line lists time points for imaging captures; (B) FIHC microscopies on vascular endothelial cytoskeleton in CAM. Pictures of β-tubulin (green fluorescence) and F-actin (red fluorescence) and merged images are displayed against the treatment with 0.9% saline (control) and C118P (2, 5, 10 mg/ml), to demonstrate cytoskeletal changes. Nuclei are counterstained with DAPI (blue). On HE stained slide, arrows indicate the focused blood vessels with endothelial cells.
To explore the molecular mechanisms underlying VDAs’ pharmacological effects, conventional histopathology and fluorescent immunohistochemistry (FIHC) for endothelial cytoskeleton of the CAM were assayed. FIHC analyses include two major proteins of cytoskeleton, β-tubulin and F-actin, which were qualitatively demonstrated and quantitatively analyzed. FIHC verified the results of the four evolving parameters by correlation analyses and exposed molecular mechanisms 5 (Figure 6B). This novel LSCI–CAM platform could be useful for in vivo evaluations of anticancer pharmaceuticals. As a good example, for the first time, VDAs have been comprehensively evaluated in vivo in the CAM for multiparametric hemodynamic assessment with the verifications at the molecular level. It paves a new way for preclinical screening of the drugs interfering with tubulin and actin as the two major proteins in the cytoskeleton.
The Effects of VDAs on Tumor Vasculature as Studied on the LSCI–CAM Platform
Next, we have utilized LSCI–CAM platform to explore the pharmacologic effects of VDAs on tumors grafted or inoculated in the CAM with histopathology proofs.
In total, four approaches of transplantation of two tumor cell lines, that is, Lewis lung carcinoma (LLC) and hepatocellular carcinoma (HepG2) cell line obtained from the cell bank of Chinese Academy of Sciences (Shanghai, China), were implemented in the CAM of CE air cell on ED8. Each approach of different tumor cell-line included 12 CEs for the transplantation (n = 12). Approach 1: silicon ring in 5 mm diameter was directly placed and adhered to the surface of vascular zone on the CAM. Cell suspension of 50 μL at 5 × 105/ml concentration was dropped in the center of silicon ring. Approach 2: cell suspension of 50 μL at 5 × 105/ml concentration was directly injected or implanted onto the vascular zone of CAM after mixing with 100 μL of Matrigel. Approach 3: a glass rod was used to gently abrade a piece of CAM surface in order to expose the capillaries, while preventing bleeding, with the rest implantation steps similar to Approach 2. Approach 4: cancer cell suspension of 50 µl at 5 × 105/ml concentration was infused into the mesoderm of the CAM under the observation window by an insulin syringe. CE eggs were placed back to the incubator afterward.
On ED16, tumor formation was evident. However, in the recent pilot experiments, tumor take rate can be achieved only in 30% of CEs, which just allows qualitative assessment and verification. Histopathological results affirmed successful tumor formation with the presence of typical cancerous morphology, as shown in an example of approach 4 by injecting cell suspension directly into the mesoderm of the CAM (Figure 7C).

Tumor transplantation resulted from using approach 4. (A) green arrows indicate tumor tissue in the CAM; (B) tumor biopsy after the dissection; (C) cancer cells with heterogenous dedifferentiation in both nuclei and cytoplasm by histopathological verification of HE staining.
On ED 16, two VDAs (C118P or CA4P) were prepared in two concentrations, 2 and 10 mg/ml. CEs were randomly divided into four groups for the treatment by two concentrations of two VDAs, respectively (n = 6, each). Another vehicle control group was treated by 0.9% saline. The blood perfusion and vascular diameter of major tumoral vessel were qualitatively observed and quantitatively analyzed for 30 min under intravascular infusion on the LSCI–CAM platform. The vasculature of tumor in CAM was negatively affected by VDAs, appearing dose–dependent manner. Such effects with tumor vessels were much more prominent than normal vasculature.
The conventional histopathology, CD34, and FIHC assays of tumors in the CAM were qualitatively analyzed and compared with normal vessels in CAM adjacent with tumor. The established LSCI–CAM platform proves a feasible and easy-to-handle technique for in vivo studies on anticancer pharmaceutical evaluations. This proof-of-concept study has demonstrated various approaches of tumor transplantation based on CAM model, which can be expanded to studies with more widely accessible tumor transplantation.
Conclusions
By utilizing different in vivo imaging modalities, vascular morphology and changes in CE and CAM can be portrayed. A novel imaging-guided LSCI–CAM platform has been established for anticancer pharmaceutical evaluations. By taking VDAs as examples, we demonstrated promising applications of LSCI–CAM platform for qualitative and quantitative evaluations of particular pharmaceutics.
Research Outlook and Practical Challenges
Various factors might impact the take rate of transplanted tumors on the CAM model. The four approaches of transplantation mentioned above did not yield high enough take rates. Nevertheless, on the basis of the above protocols and desired outcomes for the quantitative analyses and for the studies on molecular mechanisms, the take rate of tumor transplantation is major direction for improvement.
As recently discussed in the literature, the take rates for transplantation of various cancers rely more on unique intrinsic properties than on differences in experimental techniques. For instance, there were reportedly 80%–100% take rates for sarcoma, glioblastoma, renal cell cancer, and nasopharyngeal carcinoma 46 ; but merely 50% for giant cell cancers and squamous cell carcinoma in the head and neck; and 0% for LLC. 46 In contrast, in our recent study, a 30% take rate was achieved with LLC (work in progress).
LSCI–CAM platform was properly implemented on CE tumor models with typical vasculature, instead of tumor nodules and micro-tumors commonly seen in other reports. 46 Hence, more cancer cell lines should be tested with LSCI–CAM platform, for example, renal cell cancer, sarcoma, glioblastoma, and nasopharyngeal carcinoma, in order to verify intrinsic properties of different tumor types and to increase the take rate.
In addition, experimental techniques can be improved for the take rate increment. Patient-derived xenografts (PDX) model can be directly transplanted inside the mesoderm of CAM, which is an efficient approach to simulate the tumor microenvironment. As reported, the transplantation with PDX derived from glioblastoma 47 and sarcoma 48 can achieve more than 80% of take rate. Tumor transplantation with more kinds of PDX should be evaluated.
Another proposed protocol is to mix cancer cell suspension with Matrigel, which can be directly injected into the mesoderm of the CAM, facilitating the accumulation of tumor cells in the CAM.
Using green fluorescent protein (GFP) is an excellent method to label tumor cells.20,49,50 The tumor cells labeled with GFP are constitutively expressed in the tumors that are visually discernable, indicating the growth and metastasis of tumors by fluorescent signals in the CAM and CE.
Improved cell culture could be a potentially useful method to drive tumor cells more adaptable in CAM microenvironment.
Short life range of the CEs is one of the main drawbacks of CAM model, which limits the tumor growth and its vascularization. Ideally, first-generation tumor can be transferred on to a fresh CAM for continuous growth. Progress has been made in such exploitation. 51
Grown vessels in tumor xenografts can be visualized by DR imaging. Contrast agents (eg barium sulfite suspension) and Evans blue can be mixed and injected into the CAM vasculature, and DR imaging may then capture and identify vessels in the tumor, which can assist the quantitative hemodynamic measurement of the tumor on LSCI–CAM platform. 52 Recently we noticed that hypericin, the second small molecule compound used in Oncocidia, has also been studied on the CE-CAM platform, although its unique property of necrosis-avidity was not realized by Blascakova et al.
Footnotes
Acknowledgments
VEPYMO Poultry Breeding NV (Belgium) and Shanghai Poultry Breeding Co., Ltd are acknowledged for their generous supply of the check embryonic eggs in this study. Mr. Peng Cheng from Gene&I Scientific Ltd. is acknowledged for his technical support on full-field laser perfusion imager (FLPI). Mr. Siegfried Schwarz from KML Vision GmbH (Graz, Austria) is acknowledged for sincerely providing IKOSA deep learning platform and technical assistance. Nanjing Sanhome Pharmaceutical Co. Ltd (Nanjing, China) is acknowledged for their generous supply of C118P.
Author Contributions
Conception and design by L.C., Y.N., and Y.L.; literature search, all; writing original draft preparation, L.C., Y.N., and Y.L.; review and editing, all; supervision, M.Y., Y.N., and Y.L.; administration, M.Y., Y.N., and Y.L.; conducting the experiments, and analyzing and interpreting the data, L.C., Y.F., X.Z., YS.L., Y.X., J.X., and YZ.L.; drafting the manuscript, L.C.; revision, Y.N. and Y.L. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
Not applicable, because this serial experimental research was exempted from institutional ethics committee approval.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
