Abstract
Introduction
Cervical cancer (CC) is the most common gynecological cancer in developing countries. 1 Persistent infection with high-risk human papilloma virus (HPV) types is an important determinant of CC development, progressing from cervical intraepithelial neoplasia (CIN), carcinoma in situ of the cervix (CIS) to CC. 2 The survival time of patients with metastases and relapses has been significantly prolonged with the application of immunotherapy, such as immune checkpoint inhibitors; however, the final curative effect has not been achieved. Thus, new diagnostic biomarkers and therapeutic targets need to be further studied. 3
Recently, the tumor microenvironment and immunosuppression have gradually become the focus of research. T helper (Th) cells are crucial immunomodulatory cells in the human body. Th1 cells play a tumor-killing role, and Th2 cells are involved in immunosuppression. When the Th1/Th2 equilibrium of tumor cells is disrupted, Th1 cells drift into Th2 cells. Th2 cells were found to be dominant, the Th1/Th2 ratio was reduced, and the patient showed immunosuppression, tumor immune escape, and tumor cell proliferation and metastasis. 4 Immune checkpoints are inhibitory signals in the immune system that maintain autoimmune tolerance and immune homeostasis. Immune cells in the tumor microenvironment or tumor cells overexpress inhibitory immune checkpoints, such as cytotoxic T lymphocyte-associated antigen-4 (CTLA-4), programmed death 1 (PD-1), and programmed death ligand 1 (PD-L1). 5 The application of anti-immune checkpoint CTLA-4, PD-1, and PD-L1 monoclonal antibodies in various tumor treatments has attracted unprecedented attention for blocking immune checkpoint strategies in tumor immunotherapy. Patients with melanoma receiving anti-PD-1 and PD-L1 therapies show less immunotoxicity; however, a considerable number of patients are not sensitive to PD-1.6,7 In addition, the efficacy of immunotherapy in CC can only be determined from phase I or II clinical trials. The involvement of other important immune checkpoint molecules in the tumor immune escape of CC has become a hot topic of current research.
LAG-3 is a recently discovered immunosuppressive protein coded by LAG-3 gene located on chromosome 12 (12p13.3) and is one of the most promising immune checkpoint along with PD-1 and CTLA-4. 8 It is a member of the type I immunoglobulin superfamily, and it is composed of 498 amino acids and contains 4 extracellular immunoglobulins. 9 LAG-3 is mainly expressed in activated CD4+ T cells, CD8+ T cells, and natural killer cells. 10 LAG-3 is abnormally expressed in a variety of tumor microenvironments and plays an important role in the tumor immune response. 11
Soluble LAG-3 (sLAG-3) has been proposed to be derived from the proteolytic cleavage of surface LAG-3, which is found in serum and plasma. The function may be similar to that of synthetic LAG-3 fusion protein (sLAG-3-Ig), which can bind to major histocompatibility complex II, thus inhibiting the binding of LAG-3 and its inhibitory function. 12 In addition, dendritic cells (DCs) are induced to activate, mature, and migrate to secondary lymphoid organs to facilitate the upregulation of surface molecules and reduction of the antigen-capturing ability, and the production of interleukin 12 (IL-12), tumor necrosis factor-alpha, and other cytokines can stimulate T-cell proliferation and promote the Th1 response. 13 sLAG-3 was first discovered in patients with inflammatory diseases, and its expression level increased when the treatment was effective. 14 Studies have reported the changes in the expression level of sLAG-3 in patients with breast 15 and gastric cancer. 16 However, the significance of the expression in CC remains unknown.
We developed an enzyme-linked immunosorbent assay (ELISA) kit for human sLAG-3 based on an improved double-antibody sandwich ELISA method and successfully performed quantitative detection of human sLAG-3 using this kit. Plasma sLAG-3 expression levels were examined in 139 patients with CC-related diseases and 116 healthy individuals. Importantly, we identified an association between sLAG-3 expression and CC progression.
The aim of this study was to investigate sLAG-3 expression in patients with CIN, CIS, and CC and to determine the association between this protein and the pathological features and T cells of the immune microenvironment. This study suggests that sLAG-3 can be developed into a more sensitive target for CC diagnosis and related immunotherapies in the future.
Materials and Methods
Antibodies, Reagents, and Cell Lines
This study follows the Equator guidelines and is consistent with the ARRIVE 2.0 guidelines.
17
Commercial recombinant LAG-3 Ig protein was obtained from R&D Systems (Minneapolis, MN, USA). Anti-LAG-3 antibodies 4F4 and 4E12 were produced in our laboratory. Plasma levels of sLAG-3 were determined by an ELISA developed in our laboratory using 4F4 as a capture antibody and biotinylated 4E12 as the detection antibody. Chinese hamster ovary (CHO) cells derived from Baidu Cell Bank were stably transfected with empty vector (CHO/mock) or human LAG-3 vector (CHO/LAG-3). Transfection efficiency was confirmed using flow cytometry (Supplemental document 1)
Generation of a Novel Anti-Human LAG-3 Monoclonal Antibody
The protocols adhered to the principles of adequate animal care and complied with the Guidelines for the Care and Use of Laboratory Animals. 18 This study was approved by the Institutional Animal Care and Use Committee (Supplemental document 2). The recombinant LAG-3 Ig protein was used as an immunogen. Freund's adjuvant was used to immunize the mice with the fusion protein. In this study, the purchase order number of mice was 201911A407. The dose of antigen for the first immunization was 10 to 50 μg/mouse, followed by full emulsification with Freund's complete adjuvant and multiple subcutaneous injections, usually 0.8 to 1 mL/mouse, 0.2 to 0.3 mL/point. The second immunization was performed 4 weeks after the first immunization; the dosage route was the same as above, and the mice were fully emulsified with Freund's incomplete adjuvant. The third immunization was performed 3 weeks after the second immunization; the dose was the same as above without adjuvant, and the antigen was dissolved in normal saline and injected intraperitoneally. Three weeks after the third immunization, the fourth immunization was administered at the same dose as above, without an adjuvant, and the antigen was dissolved in saline and injected into the intraperitoneal cavity. After 7 to 10 days, mouse orbital blood was collected to measure the titers. If the titer reaches 1:104, immunization was boosted 3 to 5 days before fusion. The antibodies were purified from mouse ascites using a Protein G-Sepharose CL4B affinity column (Pharmacia, Uppsala, Sweden).
Characterization of LAG-3 mAb
Western blotting was used to analyze the binding ability of the monoclonal antibodies (mAbs). Recombinant LAG-3 Ig was separated on a 10% nonreducing gels. Afterwards, the proteins were transferred onto a membrane, which was then washed with phosphate-buffered saline (PBS) containing 0.1% Tween 20 (PBST) and blocked with 5% nonfat dried milk. The target bands were stained with 2 monoclonal antibodies, 4F4 and 4E12 (1:250 dilution), followed by horseradish peroxidase (HRP)-labeled goat anti-mouse IgG mAb (Affinity Biosciences, OH, USA). The membranes were visualized using enhanced chemiluminescence reagent (Applygen Technologies Inc., Beijing, China). A commercial LAG-3 antibody (ab209236; Abcam, Cambridge, UK) was used as the positive control (1:500 dilution). The experiment was repeated thrice.
The binding ability of the antibodies was verified using immunohistochemistry and immunofluorescence. Cells were digested, counted, and spun into cell slivers. The samples were fixed with 4% paraformaldehyde for 15 min, washed with PBS, and blocked with 5% bovine serum albumin (BSA) for 30 min at 37°C. The anti-LAG-3 mAbs 4F4 and 4E12 were added and incubated overnight at 4°C. After washing, the sections were incubated with HRP-labeled goat anti-mouse IgG antibody (Affinity Biosciences) at 37°C for 30 min, after which they were treated with diaminobenzidine solution for color development and hematoxylin for counterstaining. A commercial LAG-3 antibody (ab209236; Abcam) was used as a positive control.
For the immunofluorescence assay, a total of 2 × 104 cells were plated onto the chamber of a Lab-Tek 8 chambers slide (Thermo Fisher Scientific, CA, USA) overnight. The cells were then fixed with 4% paraformaldehyde (Thermo Fisher Scientific) and permeabilized in 0.1% Triton X-100 (Sigma-Aldrich, St. Louis, MO, USA) for 30 min at 4°C. After washing thrice and incubating with a blocking solution (5% BSA in PBS), the cells were incubated with the LAG-3 mAb 4F4 at 4°C overnight. The following day, the cells were washed thrice and incubated with goat anti-mouse Alexa Fluor 594 (Thermo Fisher Scientific) in the dark for 1 h. Finally, the cells were washed and incubated with 4′,6-diamidino-2-phenylindole for 20 min at room temperature in the dark. All the matched samples were photographed under a microscope at identical exposure times.
The binding capacity of the antibody to recombinant human LAG-3 protein was determined using indirect ELISA (R&D Systems). Plates were coated with a carbonated buffer solution (CBS) containing LAG-3 Ig at 4°C. The cells were incubated with 3% BSA (Beyotime, Shanghai) for 2 h, after which biotin-labeled LAG-3 mAb was added and cultured at 37°C. Subsequently, the cells were stained with HRP-streptavidin (Sigma-Aldrich) at 1:10000 for 1 h at 37°C. After suspension in 2.0 mol/L sulfuric acid, absorbance was measured at 450 nm (Bio-Rad Laboratories, Hercules, CA, USA).
Establishment of sLAG-3 ELISA
Mouse anti-LAG-3 mAb (clone 4F4, 5 μg/mL) was coated with sodium phosphate buffer in a 96-well plate (Corning, NY, USA) overnight at 4°C. The plates were blocked with 2% BSA in PBS at 37°C for 2 h. Samples or purified human LAG-3 fusion proteins were added and incubated at 37°C for 2 h in duplicate. The plate was washed with PBST thrice and incubated with 0.5 μg/mL biotinylated mouse anti-LAG-3 mAb (clone 4E12) for 1 h at 37°C. All antibodies were titrated to optimal working concentrations. The plate was washed with PBST thrice followed by incubation with HRP-streptavidin (1:7000; Sigma-Aldrich) for 1 h at 37°C. After washing with TBST 10 times, the plate was incubated with tetramethylbenzidine (TMB; Sigma-Aldrich), and the reaction was stopped using 2.0 mol/L concentrated sulfuric acid. Absorbance was measured at 450 nm using a microplate reader (Bio-Rad Laboratories). The minimal detectable concentration was 0.1214 pg/mL for sLAG-3.
For the ELISA competition test, 96-well plates were incubated overnight at 4°C with unlabeled anti-LAG-3 mAb 4F4 or 4E12 in CBS. After blocking, recombinant human LAG-3 protein was added to the plates and incubated at 37°C for 2 h. The plates were then stained with biotin-labeled 4F4 or 4E12 for 1 h at 37°C and treated with TMB and sulfuric acid solution, respectively. Finally, absorbance was measured at 450 nm using a microplate reader.
Stability, Precision, and Specificity of sLAG-3 by ELISA
Three samples of known concentrations (5, 2.5, and 1.25 ng/mL) were tested 20 times on the ELISA plates to analyze the precision of the ELISA system. In addition, the stability of the ELISA was evaluated. After the precoated plates were preserved for 0, 10, 20, or 30 days at 4°C, 3 sLAG-3 concentrations (5, 2.5, and 1.25 ng/mL) were detected to assess variation in the ELISA system.
Determination of sLAG-3 in Plasma of Patients and Normal Controls
To determine plasma sLAG-3 in patients and normal controls, 48 CIN, 13 CIS, and 78 CC cases with detailed medical records, diagnosed and treated between 2019 and 2022, were selected as subjects. For all patients, records included age, HPV infection, tumor staging, and metastasis. The study was subject to strict ethical approval (Supplemental document 3). All the participants signed an informed consent form. The staging was based on the 2018 Federation International of Gynecology and Obstetrics staging guidelines. 19 The exclusion criteria were as follows: presence of other types of cancer or receipt of antitumor therapy before admission; or hepatitis, syphilis, and other infectious or autoimmune diseases. Peripheral blood was collected in purple anticoagulant tubes (Bao-kang Organism, China) with EDTAK2 anticoagulant lining on an empty stomach the morning after admission. The blood was centrifuged, and the plasma was collected and stored at −80°C. The expression of sLAG-3 in plasma was determined using an ELISA kit.
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 8.0 (GraphPad Software Inc., La Jolla, CA, USA) and SPSS 25.0. sLAG-3 has a non-normal distribution and is represented as the median and interquartile range. The Mann-Whitney U test was used to analyze differences between groups. The Kruskal-Wallis test was used for comparisons among multiple groups. The predictive value of sLAG-3 for disease diagnosis was determined via multivariate logistic regression analysis. The receiver operating characteristic (ROC) curve and the area under the ROC curve (AUC) were calculated. Statistical significance was set at P < .05.
Results
Identification of 2 Novel Anti-Human sLAG-3 mAbs (Clones 4F4 and 4E12)
Two specific anti-human sLAG-3 mAbs (clones 4F4 and 4E12) were characterized and used in subsequent experiments. The heavy and light chains of 4F4 and 4E12 were IgG1 and κ, respectively. First, the specificity of the mAb for human LAG-3 was determined by western blotting. Recombinant LAG-3 Ig protein and a commercial LAG-3 antibody were used as the antigen and positive control, respectively. Western blotting showed that 4F4 and 4E12 recognized the target protein bands (Figure 1A). Immunohistochemistry showed that 4F and 4E12 reacted with LAG-3-transfected CHO cells (Figure 1B). Immunofluorescence assays were performed using 4F4 and 4E12 mAbs as primary antibodies and showed that 4F4 specifically recognized LAG-3-transfected CHO cells, but not mock-transfected CHO cells (Figure 1C). Indirect ELISA was used to identify LAG-3 monoclonal antibodies. The results indicated that 4F4 and 4E12 could bind to LAG-3 Ig protein (Figure 1D).

Identification of 2 novel anti-human LAG-3 mAbs (clones 4F4 and 4E12). (A) Western blot results showing that 4F4 and 4E12 could recognize the target protein bands of LAG-3. (B) Immunohistochemistry assay images showing that 4F4 and 4E12 could react with CHO/LAG-3. (C) Immunofluorescence assay images showing that 4F and 4E12 could react with CHO/LAG-3. (D) The binding ability of the antibody to immobilized LAG-3-Fc was detected via indirect ELISA. The results re-present the average absorbance at 450 nm. Abbreviations: CHO, Chinese hamster ovary; ELISA, enzyme-linked immunosorbent assay; LAG-3, lymphocyte activation gene-3; mAb: monoclonal antibody.
Establishment of a Novel Sandwich sLAG-3 ELISA
An ELISA competition assay showed that unlabeled and biotin-labeled monoclonal antibodies could bind to different sites of the LAG-3 protein, indicating that 4F4 and 4E12 recognize different epitopes of LAG-3 (Figure 2).

Competitive assay via ELISA. The data are expressed as mean absorbance at 450 nm. Abbreviation: ELISA, enzyme-linked immunosorbent assay.
After optimizing the working concentrations of the coating and detection mAbs, 5 μg/mL 4E4 mAb and 0.5 μg/mL biotin-labeled 4F12 mAb were used as the capture and detection antibodies, respectively, and a sensitive ELISA kit was developed. The detection limit of LAG-3-Fc was 0.1214 to 500 pg/mL, and the R2 value was 0.9922 (Figure 3A). At the same time, the kit did not cross-react with other soluble molecules, such as sPD-L1, sCD80, or sB7-H3 recombinant proteins (Figure 3B-D). To further study the potential applications of ELISA kits, it is necessary to understand the accuracy and stability of the system. First, the intra-assay and inter-assay precision was evaluated. The results showed that the difference between the ELISA wells and plates was small (coefficient of variation [CV]% < 5%). Stability determination showed no significant loss of signal intensity at the different time points (CV% < 10.5%). Therefore, long-term storage of precoated ELISA plates did not affect the quality of sLAG-3. The results showed that a highly stable and highly sensitive sLAG-3 sandwich ELISA system was established (Tables 1 and 2).

Standard curves of the developed ELISA and assessment of accuracy and stability. (A) The linear detection line shows a working range from 0.1214 to 500 pg/mL. R2 is the correlation coefficient. (B)-(D) The protein levels of PD-L1, B7-H3, and CD80 were determined via ELISA. Abbreviations: ELISA, enzyme-linked immunosorbent assay; PD-L1, programmed death ligand 1.
Stability of the sLAG-3 ELISA System

Abbreviations: CV, coefficient of variation; ELISA, enzyme-linked immunosorbent assay; SD, standard deviation; sLAG-3, soluble lymphocyte activation gene-3.
Sensitivity of the sLAG-3 ELISA System

Abbreviations: CV, coefficient of variation; ELISA, enzyme-linked immunosorbent assay; SD, standard deviation; sLAG-3, soluble lymphocyte activation gene-3.
LAG-3 Expression in Different Patient Groups Based on Clinical Parameters
After rigorous screening, a total of 139 patients and 116 healthy subjects were enrolled in the study cohort . Table 3 shows the patient data (Supplemental document 4). No significant difference was observed in patient ages, and because most patients had cancer at an early stage (stage I), the volume of the cancer tissue may have been small (diameter < 4 cm). As shown in Figure 4A, we found that the sLAG-3 level in patients with CC, including CIS and invasive carcinoma of the cervix, was significantly lower than that in normal controls (M = 3.76 ng/mL vs M = 8.36 ng/mL, P < .0001, Figure 4A). sLAG-3 ELISA showed that compared to precancerous lesions and carcinoma in situ, the average sLAG-3 level in patients with CC (including squamous carcinoma and adenocarcinoma) was lower. In addition, a significant difference in the degree of decline between cancer and precancerous lesions was observed (M = 3.44 ng/mL vs M = 3.89 ng/mL vs M = 6.01 ng/mL vs M = 8.52 ng/mL, P < .0001, Figure 4B). Based on the staging criteria for CC, we divided the patients into stages I, II, and III. The expression level of sLAG-3 significantly decreased with the increase in clinical stage (M = 1.97 ng/mL vs M = 3.38 ng/mL vs M = 3.89 ng/mL, P < .01, Figure 4C). As shown in Table 3, differences in tumor diameter and metastasis were observed at different stages of CC. Finally, we divided patients with CC into 2 groups according to the metastatic status of the lesions and found that patients in the metastatic group had a significantly lower level of sLAG-3 (M = 2.94 ng/mL and M = 4.27 ng/mL for the metastatic and nonmetastatic lesions, respectively, P < .0001, Figure 4D).

(A) sLAG-3 expression levels in the normal (HC) and cancer groups. (B) Expression of sLAG-3 based on tumor grade (CIN, CIS, CC, and HC). (C) Expression of sLAG-3 based on tumor stage (Phase I, II, or III). With tumor progression, sLAG-3 expression significantly decreased. (D) Expression of sLAG-3 based on metastasis stage. P < .05, P < .01, P < .001, and P < .0001. Abbreviations: CC, adenocarcinoma and squamous cell carcinoma; CIN, cervical intraepithelial neoplasia; CIS, carcinoma in situ; HC, health control; NS, Non-statistical significance; sLAG-3, soluble lymphocyte activation gene-3.
Patient Demographics and Clinical Profiles (N = 139).

Abbreviations: CC, cervical cancer; CIN, cervical intraepithelial neoplasia; CIS, carcinoma in situ; HPV, human papilloma virus; N, number; SCCA, squamous cell carcinoma antigen.
Potential Diagnostic Value of sLAG-3
A multiple logistic regression model was used to analyze predictors of CC progression. As shown in Table 4, low-level sLAG-3 was an important independent predictor of CIN (B = −0.185, 95% CI: 0.731-0.946, P < .05), CIS (B = −0.653, 95% CI: 0.385-0.704, P < .05), CC stage I (B = −0.701, 95% CI: 0.409-0.602, P < .05), CC stage II (B = −1.154, 95% CI: 0.193-0.517, P < .05), and CC stage III (B = −1.916, 95% CI: 0.066-0.329, P < .05). Finally, ROC analysis was performed to determine the sensitivity and specificity of sLAG-3 levels for the diagnosis of CC metastasis. The AUC of sLAG-3 levels was 0.7816 (95% CI: 0.6657-0.8976; P < .05, Figure 5). The optimal threshold for predicting CC metastasis was found to be 3.41 ng/mL, with sensitivity and specificity values of 79.17% and 68.52%, respectively. This indicates that sLAG-3 is a potential diagnostic marker for CC.
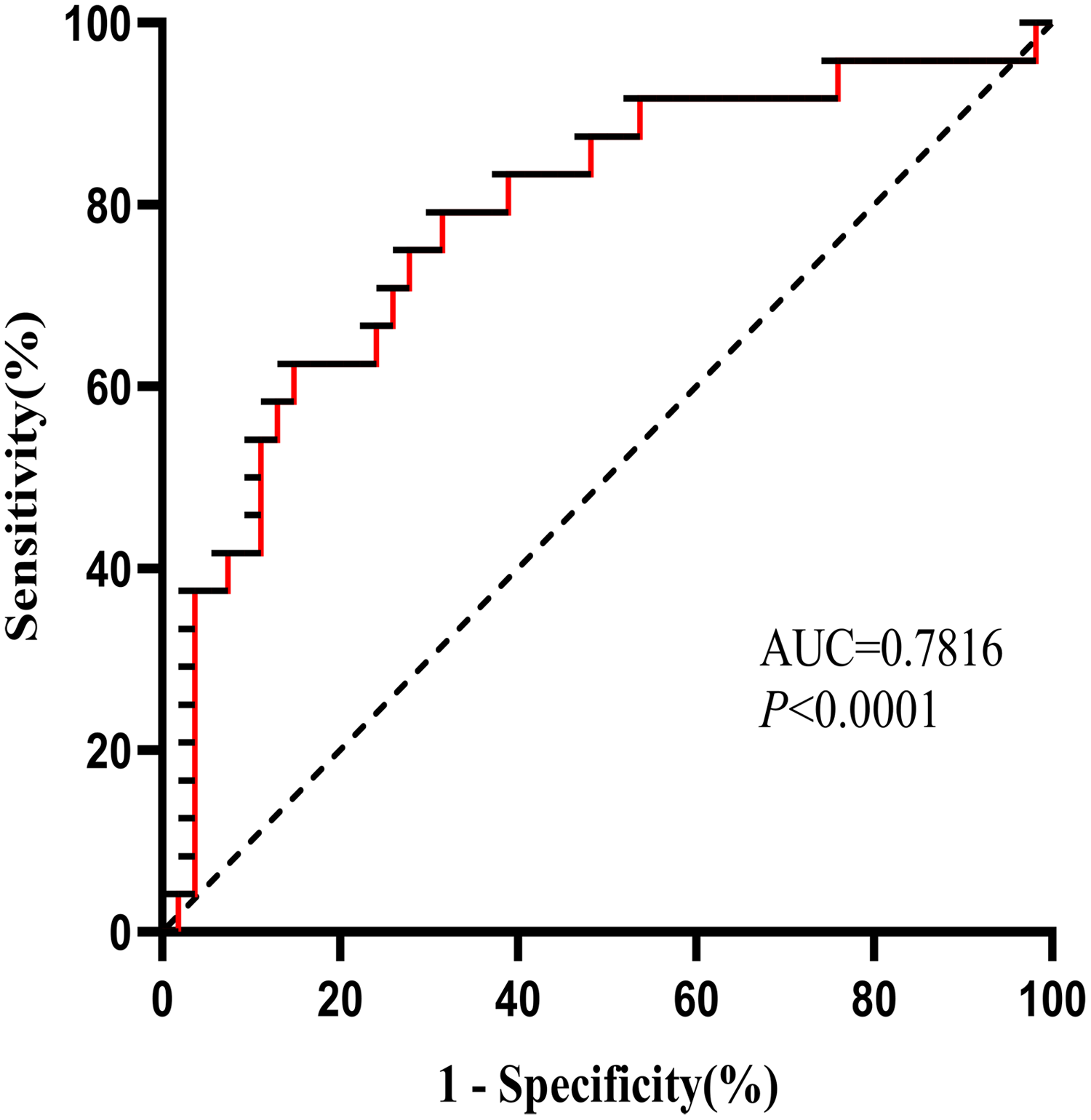
ROC curve of sLAG-3 in patients with metastatic cervical cancer. The ROC curve is used to show the diagnostic and predictive ability. Abbreviations: ROC, receiver operating characteristic; sLAG-3, soluble lymphocyte activation gene-3.
Multivariate Logistic Regression Analysis of sLAG-3 Level Associated With Cervical Cancer Disease Development.

Abbreviations: B, coefficient of beta; CC, cervical cancer; CI, confidence interval; CIN, cervical intraepithelial neoplasia; CIS, carcinoma in situ; SE, standard error; sLAG-3, soluble lymphocyte activation gene-3.
Discussion
In recent years, immune checkpoints and their application in therapy have become popular topics in the field of cancer research, especially LAG-3, which has attracted increasing attention. In the present study, we screened and characterized 2 novel mAbs against human LAG-3, namely 4F4 and 4E12. Notably, 4F4 and 4E12 bind to different antigenic epitopes. 4F4 was used for immunofluorescence assay. Western blotting and immunohistochemistry showed that both 4F4 and 4E12 could bind to recombinant LAG-3-Ig protein.
Currently, several well-characterized assays for LAG-3 detection are available for humans and mouse samples. For example, the human LAG-3 kit of Abcam (ab193707) has a detection range of 45 to 12,000 pg/mL. In this study, we established a sandwich ELISA kit to detect sLAG-3 using 2 anti-human LAG-3 mAbs, with a wide working range of 0.1214 to 500 pg/mL. Double-antibody sandwich ELISA has high sensitivity and specificity and can accurately quantify antigens with easy experimental operation. Therefore, the establishment of this kit lays a solid foundation for further research on sLAG-3. Serum levels of sLAG-3 in patients with CIN, CIS, and CC, and normal controls were detected using this kit.
Current studies have shown that the tumor microenvironment plays an important role in promoting immune tolerance and tumor metastasis, among which one of the most prominent features is the expression of various negative co-stimulatory molecules. 20 Importantly, LAG-3 has been identified as a promising target because it is one of the important inhibitory receptors leading to T-cell exhaustion.21–23 LAG-3 was found to be highly expressed in CD8 + T cells in a preclinical mouse model of ovarian cancer, and blocking its expression induced an increased antitumor response. 24 A previous study reported that increased LAG-3 expression in leukemic cells correlated with shorter time to treatment and poor outcome in chronic lymphocytic leukemia. 25 Many other studies, such as the one mentioned above, have demonstrated the important role of LAG-3 in mediating tumor immune escape. We speculated that LAG-3 expression plays an important role in the progression of CC. With the extension of HPV infection time, the viral replication load gradually increases and LAG-3 constantly plays a negative regulatory role, which can reduce the proliferation and activity of tumor killer cells. This results in immune escape, which is conducive to the growth and metastasis of cervical tumors and causes the progressive trend of the disease from CIN to CC. However, this study found that sLAG-3 negatively correlated with disease progression; the expression level of sLAG-3 in patients with CC was lower than that in patients with CIN. In the progression from CIN to CC, sLAG-3 can play an important role in shunting.
In the present study, we investigated the expression of sLAG-3 in CC cell lines. Both membrane-bound and soluble forms of negative co-stimulatory molecules play important roles in immune regulation.26–28 sLAG-3 plays an important role in the generation and proliferation of antigen-specific CD8 + T cells and promotes the transformation of CD8 + memory cells into initial T cells. Importantly, it induces DC activation and maturation, stimulates T-cell proliferation, and promotes Th1 cell response; thus, it may be an indicator of Th1 cell activation. 29 Relevant studies have confirmed that the administration of recombinant sLAG-3 as a vaccine adjuvant with antigen or tumor cells can result in the immune response of Th1 cells, thus better inhibiting tumor growth and increasing the survival rate and time of tumor-bearing animals. 30 Recombinant sLAG-3 fusion protein IMP321 has been used as a single therapy or in combination with chemotherapy for tumor treatment, and several randomized clinical trials have shown its potential application. 31 Studies have shown that sLAG-3 is involved in positive or negative immune regulation, and changes in its plasma levels are involved in cancer development, prognosis, and treatment. For example, Triebel et al 32 detected sLAG-3 in patients with breast cancer positive for estrogen receptor and/or progesterone receptor, and its expression level was positively correlated with the prognosis but was not significantly correlated with the clinicopathological characteristics of the patient. Of note, the plasma sLAG-3 levels in patients with CC were significantly lower than those in normal subjects. Moreover, with an increase in tumor stage and disease progression, that is, from CIN to CC and from CC stages I to III, serum levels of sLAG-3 decreased gradually. Further research revealed that plasma sLAG-3 levels were lower in patients with CC without metastasis than in those with metastasis. Currently, an important reason for the high incidence of CC is that patients with CIN are not accurately diagnosed or treated promptly at the early screening stage. Therefore, it is important to improve the detection rate of CIN as the existing diagnostic molecular markers for cervical lesions are inadequate. Based on these findings, LAG-3 may be used as an indicator of cancer progression risk.
Researchers have found that sLAG-3 has positive implications in the treatment of melanoma and advanced head and neck cancer.33,34 In a cooperative study for melanoma, an increase in soluble IL-2 and sLAG-3 after only one cycle of ipilimumab was observed. 35 This study also found that sLAG-3 had diagnostic value for CC and related diseases. Multivariate logistic regression analysis showed that sLAG-3 had diagnostic and predictive value for CIN, CIS, and CC at stages I, II, and III. Similarly, it has a predictive value for CC metastasis. Our findings have several important implications for future research. Patients with CIN and CIS with low sLAG-3 expression levels may be at risk of cancer progression, which requires active intervention. For patients with confirmed CC, if sLAG-3 expression is low, the possibility of advanced stage or metastasis should be considered, and a careful selection of treatment options is required. In addition, when patients enter the stage of invasive cancer, ensuring that patients with different stages receive better treatment is another important problem. Surgical treatment is preferred in the early stages (stages IA-IB1), while nonsurgical treatment is preferred in the late stages (stages IIB-IV). There is no uniform standard for patients with intermediate stages (stages IB2-IIA). 36 If surgical treatment is selected and high-risk factors are confirmed after surgery, additional radiotherapy will be required postsurgery. Several clinical studies have confirmed37–39 that more complications will occur if additional radiotherapy is administered after surgery. Therefore, if an indicator can be used to predict high-risk factors (such as lymph node metastasis), stratified management of patients will have great clinical significance. For patients with CC with predicted high-risk factors, direct selection of nonsurgical treatment to avoid unnecessary surgical trauma will greatly improve the prognosis. Given the predictive value of sLAG-3 identified in this study, it is expected to guide clinical treatment regimens.
Despite these findings, we acknowledge that this study has certain limitations as the expression levels of sLAG-3 in only some samples were examined. We will expand the samples to support our findings in the future.
Conclusion
In summary, the following conclusions are drawn from this study. Firstly, we have developed 2 new mAbs targeting human LAG-3. These antibodies are valuable for studying the biological functions of LAG-3, such as in western blotting, immunohistochemistry, immunofluorescence, and ELISA. Furthermore, we established a sandwich ELISA kit, sLAG-3, that can sensitively and specifically detect in plasma samples, providing technical support for studies on the role of LAG-3 in related diseases. Finally and most importantly, we found that the plasma sLAG-3 expression level is negatively correlated with the progression of CC, which demonstrates its diagnostic and predictive value. From this perspective, the above findings have application value for sLAG-3 antibodies in the clinically assisted diagnosis of patients with tumors.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231202650 - Supplemental material for Clinical Significance of Soluble LAG-3 (sLAG-3) in Patients With Cervical Cancer Determined via Enzyme-Linked Immunosorbent Assay With Monoclonal Antibodies
Supplemental material, sj-docx-1-tct-10.1177_15330338231202650 for Clinical Significance of Soluble LAG-3 (sLAG-3) in Patients With Cervical Cancer Determined via Enzyme-Linked Immunosorbent Assay With Monoclonal Antibodies by Yang Li, Wenwen Wang, Jingluan Tian, Ying Zhou, Yu Shen, Mingyuan Wang, Longhai Tang, Cuiping Liu, Xueguang Zhang, Fangrong Shen, Youguo Chen and Yanzheng Gu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
This study was supported by the Science and Technology Project of Suzhou (SYS2020094), Jiangsu Institute of Clinical Immunology, The First Affiliated Hospital of Soochow University, Suzhou, Jiangsu.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Science and Technology Project of Suzhou (SYS2020094), the Program of clinical medicine expert team of Suzhou, China (SZYJTD201824), the Health Science and Technology Program of Suzhou (No. GWZX 202004), and the Suzhou Medical Key Discipline Funding Project (No. SZXK202118).
Ethical Approval
The study was approved by the Ethics Committee of first affiliated hospital of Soochow university. All participants had signed the informed consent form. Ethics Number: 2022 Ethics Review (Declaration) Batch No. 031. All of the animals in the experiment have undergone ethical approval, and the ethical number: ECSU-2019000223 Soochow University, Suzhou, China.
Supplemental Material
Supplemental materials for this study have been uploaded.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
